Abstract
We investigated the diagnostic and prognostic significance of metabolic parameters from 11C-methionine (MET) positron emission tomography (PET) in patients with malignant glioma. The MET-PET was examined in 42 patients who had been previously treated with adjuvant treatment for malignant glioma. Both ratios of maximal MET uptake of the tumors to those of the contralateral normal gray matter (T/N ratio) and metabolic tumor volume (MTV) were estimated in each lesion. The diagnostic performance for recurrence was investigated in all enrolled patients. A definitive diagnosis was done with pathologic confirmation or clinical follow-up. Among recurrent patients, we evaluated the prognostic value of metabolic parameters (T/N ratio and MTV) as well as clinical factors. Among 42 patients, 35 patients were revealed with recurrence. Both T/N ratios (p = 0.009) and MTV (p = 0.001) exhibited statistical significance to differentiate between recurrence and post-treatment radiation effect. A T/N ratio of 1.43 provided the best sensitivity and specificity for recurrence (91.4 and 100 %, respectively), and a MTV of 6.72 cm3 provided the best sensitivity and specificity (77.1 % and 100 %, respectively). To evaluate the prognostic impact, different cutoffs of MTV were examined in patients with recurrent tumor and a threshold of 60 cm3 was determined as a best cutoff value to separate the patients in two prognostic groups. Univariate analysis revealed improved overall survival (OS) for patients with Karnofsky performance scale (KPS) score ≥70 (p < 0.001) or MTV <60 cm3 (p = 0.049). Multivariate analysis showed that patients with KPS score ≥70 (p < 0.001; hazard ratio = 0.104; 95 % CI, 0.029–0.371) or MTV < 60 cm3 (p = 0.031; hazard ratio = 0.288; 95 % CI, 0.093–0.895) were significantly associated with a longer OS. However, T/N ratio was not correlated with patients’ outcome. Metabolic parameters had the diagnostic value to differentiate recurrence from post-treatment radiation effect. Compared with T/N ratio, MTV was an independent significant prognostic factor with KPS score in patients with recurrent tumor. Our study had a potential to manage these patients according to prognostic information using MET-PET.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cellular proliferation of tumors is associated with protein synthesis [1]. Amino acid transport and protein synthesis are increased in most tumors, compared to the normal brain. On 11C-methionine (MET) positron emission tomography (PET) images, areas that exhibit higher uptake than normal gray matter are considered potentially pathological. Before and after treatment of malignant glioma, MET-PET is often used to diagnose the tumor, detect recurrence, guide stereotactic biopsy and neurosurgical resection, monitor treatment, and delineate the target volume for radiotherapy [2].
For the analysis of MET-PET images, the ratio of tumor to normal background (T/N ratio) is generated by dividing the standardized uptake value (SUV) of the lesion by the SUV of corresponding contralateral gray matter or the contralateral frontal lobe. Generally, a threshold above 1.5–1.9 is used for the diagnosis of brain tumor or recurrence [3]. On the other hand, it remains controversial whether MET uptake before treatment can be used as a significant prognostic factor [4]. Maximum SUV (SUVmax) has a limitation to reflect the metabolic status of whole tumor. Furthermore, T/N ratios may vary considerably due to inappropriate region of interest (ROI) localization especially in large zones of hypermetabolic areas or hypometabolic necrosis in tumors.
To overcome the above limitation, MET-PET-derived three-dimensional volume parameter (metabolic tumor volume, MTV) has been used to evaluate tumor characteristics. However, there have been few reports to assess prognostic value of these metabolic parameters derived by MET-PET especially in patients with recurrent malignant gliomas. It is important to evaluate the post-treatment status of patients for the selection of a subsequent management plan. Therefore, we investigated the diagnostic (recurrence or post-treatment radiation effect) and prognostic values using metabolic parameters from post-treatment MET-PET in patients with malignant glioma.
Patients and methods
Clinical characteristics of 42 patients with malignant glioma
Between May 2009 and November 2013, 42 patients with malignant gliomas received MET-PET examinations for differential diagnosis between recurrence and post-treatment radiation effect. A retrospective review of medical records was performed on all patients. The mean age was 45.6 years (range, 13–75) and included both males (n = 23) and females (n = 19). The tumors were graded according to the World Health Organization (WHO) classification [5]. Pathological diagnosis revealed four anaplastic astrocytomas, one anaplastic oligoastrocytoma, seven anaplastic oligodendrogliomas, and 30 glioblastomas. The retrospective study has been approved by our institutional review board (IRB No: CNUHH-2015-100), and the need for written informed consent was waived. As the initial adjuvant treatment following operation, 25 patients received concomitant chemoradiotherapy (CCRT) with temozolomide (Temodal; Schering-Plough, Kenilworth, NJ, USA) [6]. Other 17 patients received radiotherapy with mean 56.6 Gy (range, 50.4–60.0). Following adjuvant treatment, 42 patients received a MET-PET examination to differentiate between recurrence and post-treatment radiation effect. The median interval between the end of initial adjuvant treatment and MET-PET exam was 22.0 months (range, 8.7–74.4). The patients were estimated by the Karnofsky performance scale (KPS) score which runs from 100 to 0, where 100 is “perfect” health and 0 is death [7]. Tumor recurrence according to the Macdonald criteria was determined retrospectively from the medical records [8]. During clinical and radiologic follow-up, one criterion used was the following: contrast enhancement or tumor enlargement ≥25 % in sum of the products of perpendicular diameters or any new contrast enhancing lesion or clinical deterioration.
11C-methionine positron emission tomography
MET-PET was performed using a Discovery ST PET/CT system (GE Medical Systems, Milwaukee, WI, USA) with a spatial resolution of 5.0 mm (full width at half maximum) and slice thickness of 3.27 mm. Patients were placed in the scanner such that slices parallel to the orbitomeatal line could be obtained. Patients were intravenously injected with11C-MET at 7 MBq/kg during a fasting period. For attenuation correction, we acquired a non-contrast-enhanced, low-dose CT scan and began the 10-min emission scan 20 min after the injection of 11C-MET. We reconstructed the images in the form of transaxial images of 256 × 256 × 98 anisotropic voxels (voxel size was 1.17 mm × 1.17 mm × 3.27 mm) with ordered subset expectation maximization (OSEM: iteration, 5; subset, 32) and used CT images for attenuation correction of the PET images.
Image analysis of 11C-methionine uptake
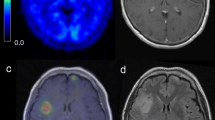
Two kinds of metabolic parameters (T/N ratio and MTV) from MET PET were measured. To achieve the reference value, we manually placed regions of interest (ROI) over the contralateral gray matter or, if impossible because of the tumor location, on intact brain regions in the axial plane (Fig. 1a). Next, a ROI was automatically placed along the tumoral margin set to ≥SUVmax of contralateral normal cortex (Fig. 1b). The T/N ratio was calculated by dividing the SUVmax of the tumor, which was calculated over the ROI of each targeted lesion, by the SUVmax of the contralateral normal gray matter. MTV was also measured by using the ROI over the threshold mentioned above (cm3).
Image analysis of 11C-methionine uptake. a The reference value was measured as maximal SUV from regions of interest (ROI) over the contralateral normal gray matter. b A ROI was automatically placed along the tumoral margin set to ≥SUVmax of contralateral normal gray matter and metabolic tumor volume was measured by using the ROI over the threshold mentioned above (cm3)
Statistical analysis
Descriptive statistics are presented as mean ± SD or as range. Student’s t test was used to compare the difference of T/N ratio or MTV between two groups (recurrence and post-treatment radiation effect). The diagnostic accuracy of T/N ratio and the MTV was evaluated using receiver operating characteristic (ROC) curve analysis by MedCalc software (version 11.6; Broekstraat, Mariakerke, Belgium). We defined the mean range as the follow-up length and determined single variable effects on overall survival (OS) via univariate and multivariate analyses. The single variables were age, KPS score, pathologic grade, T/N ratio, MTV (cm3), and adjuvant treatment. The prognostic impact of MET uptake and MTV was tested by an approximation procedure with different cutoffs to determine the cutoff value best separating the patients in two prognostic groups. OS was calculated from the dates of PET examination to death or last follow-up. Survival probability was calculated using the Kaplan-Meier method, and comparisons were performed with the log-rank test. We examined variables in the Cox proportional hazard analysis model to identify the independent predictors of survival. These variables which significance level was less than 0.25 from the univariate analyses were included in the multivariate analyses. All statistical analyses were performed with a significance level of p < 0.05 using SPSS 21.0 (SPSS, Chicago, IL).
Results
Metabolic parameters for the diagnosis of malignant glioma
Among 42 patients, 35 patients were revealed with recurrence. Recurrence was diagnosed with pathologic confirmation in 12 patients or clinical and radiologic follow-up in 23 patients. Depending on the pathologic subtypes, 24 out of 29 patients with glioblastomas showed recurrence. Ten patients were pathologically diagnosed after re-operation, and 14 patients were diagnosed with clinical and radiologic follow-up. Of 13 patients with anaplastic glioma, 11 patients showed recurrence. Two patients were pathologically diagnosed after operation, and 9 patients were diagnosed with clinical and radiologic follow-ups.
For all patients, MTV (p = 0.001) as well as T/N ratio (p = 0.009) were significantly higher in patients with recurrence (Table 1). The ROC curve analyses for the T/N ratio and the MTV were performed for all patients (Fig. 2). Although the area under curve (AUC) for T/N ratio was higher than that for MTV, there was no significance (0.953 vs. 0.906, p = 0.184). The optimal cutoff values for recurrent malignant gliomas were 1.43 in T/N ratio and 6.72 cm3 in MTV, respectively (Table 2).
Therapeutic management in 35 patients with recurrent tumor
The adjuvant treatment underwent in 33 out of 35 patients with recurrence. The mass was removed totally in two patients and subtotally in one. Chemotherapy was given to 30 patients. The regimens are as follows: temozolomide in 19; procarbazine, CCNU, and vincristine in 10; ACNU and cisplatin in four; and bevacizumab in 10. Re-radiotherapy was performed in one patient and gamma knife radiosurgery in two.
The prognostic impact according to different cutoffs of metabolic parameters
The prognostic impact of MTV and T/N ratio was tested with different cutoffs in 35 patients with recurrent tumor (Table 3). To determine the cutoff value best separating the patients in two prognostic groups, Kaplan-Meier analysis of MTV revealed a best cutoff value with the statistical significance at a threshold of 60 cm3. Using this threshold, patients with a MTV higher than 60 cm3 showed shorter OS (p = 0.049, log-rank test). On the T/N ratio results at different cutoff including the mean value, there was no statistical significance.
The prognostic significance related with the overall survival among 35 patients with recurrent tumor
The results are summarized in Table 4. The mean OS was 15.9 ± 2.7 months in all patients. Univariate analysis revealed improved OS for patients with KPS score ≥ 70 (p < 0.001) and MTV < 60 cm3 (p = 0.049). There was no statistical significance in age (p = 0.216), pathologic grade (p = 0.111), T/N ratio < 2.8 (p = 0.113), and adjuvant treatment (p = 0.098). Patients with KPS ≥ 70 (mean OS, 16.6 ± 2.0 months) showed improved OS compared to that in patients with KPS < 70 (mean OS, 5.8 ± 0.9 months). Patients with MTV < 60 cm3 (mean OS, 15.6 ± 1.9 months) had improved OS compared to that in patients with MTV ≥ 60 cm3 (mean OS, 7.9 ± 1.9 months) (Fig. 3).
Multivariate analysis showed the statistical significance on KPS and MTV. Patients with KPS ≥ 70 were significantly associated with a longer OS (p < 0.001; hazard ratio = 0.104; 95 % CI, 0.029–0.371) compared to that in patients with KPS < 70. Patients with MTV < 60 cm3 had a longer OS (p = 0.031; hazard ratio = 0.288; 95 % CI, 0.093–0.895) compared to that in patients with MTV ≥ 60 cm3.
Discussion
Diagnostic value of 11C-methionine positron emission tomography for malignant glioma
Due to the short half-life of 11C-labeled MET, approximately 20 min, the use of MET is relatively restricted to a few centers with a cyclotron unit. However, MET-PET is a non-invasive method to evaluate the regional metabolism of brain tumors. Studies report that MET-PET shows generally satisfactory sensitivity and specificity in distinguishing non-tumoral lesions from gliomas of 75–100 % [1, 9–11]. Especially, T/N ratio over SUV has been demonstrated for diagnosis of malignant potential or recurrence, due to the marked variation of MET accumulation in intact brain tissue among individuals. In our study, T/N ratio >1.43 showed high diagnostic accuracy for recurrence, which was in line with previous studies [3].
In addition to T/N ratio, MTV was also the significant metabolic parameter for recurrence in our study, which value >6.72 cm3 showed high diagnostic accuracy. However, the sensitivity of MTV (77.1 %) was suboptimal, compared to that of T/N ratio. Due to the limited spatial resolution of PET imaging, small brain lesions, less than 2 cm in diameter, could lead to an underestimation of the accumulation of radioisotope in tumors [12, 13]. By this partial-volume effect, MTV could be more affected than T/N ratio which was calculated using SUVmax.
On the other hand, these parameters were very specific (100 %) for recurrence, compared to previous studies [14]. Generally, MET uptake could increase in benign conditions, which include necrosis, leukoencephalitis, abscess, hematoma, ischemia, and demyelination. Especially, MET uptake may be increased due to the disruption of the blood-brain barrier and vascular proliferation after radiotherapy, which lead to false-positive results in the brain tumor [15, 16]. In addition, delayed radiation injury leads to an undefined zone of MET uptake, where it is difficult to differentiate between tumor recurrence and radiation effect [17, 18]. In our study, the proportion classified into patients with post-treatment radiation effect was low, which might be one of the reasons why metabolic parameter showed high specificity.
The prognostic significance of post-treatment metabolic tumor volume in patients with recurrent tumor
Clinical prognostic factors of malignant glioma, including young age, high KPS score, high mini-mental state examination score, and surgical resection of >98 % of tumor are indicative of higher OS [19]. We investigated whether metabolic parameters such as T/N ratio and MTV could give additional, important prognostic information among 35 patients with recurrent tumor.
The prognostic significance of MET uptake (or T/N ratio) remains controversial. One study showed that there was no statistical prognostic value of maximal MET uptake in low- and high-grade gliomas before treatment using a semiquantitative region of interest (ROI) [4]. Another study showed that high T/N ratio can be used as a prognostic factor for grades II and III gliomas, with high uptake being associated with poor survival [20, 21]. In this study, T/N ratio was not related with poor prognosis in patients with recurrent malignant glioma. These conflicting results might be due to the characteristics of T/N ratio, which could only reflect the maximal value of ROI, instead of whole tumor related to prognosis.
On the other hand, MET-PET can delineate the tumor extent more precisely, compared to MRI [22]. Furthermore, the distinction between recurrence and radiation effect after adjuvant treatment (especially concurrent chemoradiation therapy) remains unresolved, and the delineation of tumor burden is also difficult despite advanced imaging techniques [22, 23]. Therefore, MET-PET-derived three-dimensional metabolic parameter, MTV, could provide a better prognostic value by reflecting whole tumor effectively, compared with T/N ratio.
Galldiks et al. reported that pretreatment volumetry of MET uptake but not the T/N ratio is a useful prognostic marker in patients with malignant glioma [2]. However, there has been no previous report to investigate of the prognostic significance of MTV among patients who had recurrent tumor in spite of adjuvant treatment. In our knowledge, it is the first report to correlate MTV in recurrent malignant gliomas with prognosis and MTV was an independent prognostic factor with KPS in those patients.
The limitation of this study
There are several limitations in this study. First, the results could be affected by selection bias because of the retrospective analysis. Especially, the proportion between recurrence and post-treatment radiation effect affected the optimal cutoff value of metabolic parameters in this study although only patients with recurrence were included to evaluate prognosis. Second, the prognosis could be different according to subsequent therapeutic regimens among patients with recurrent tumors. Even if there was no statistical significance, variable therapeutic regimens could affect the patient’s outcome. Third, the measurement of MTV can also be different depending on the level of threshold, because boundary threshold of MTV was set to over SUVmax of the contralateral normal cortex. Further studies are needed to improve the reproducibility of the level of threshold.
Conclusion
Metabolic parameters (T/N ratio and MTV) had the diagnostic value to differentiate recurrence from post-treatment radiation effect. Compared with T/N ratio, MTV was an independent significant prognostic factor with KPS in patients with recurrent tumor. Although further investigations should be performed to assess prognosis according to pathologic subtype or subsequent specific therapeutic regimens, our study had a clinical implication about how to manage patients with recurrent tumor according to additional prognostic information using MET-PET.
References
Glaudemans AW, Enting RH, Heesters MA, Dierckx RA, van Rheenen RW, Walenkamp AM, Slart RH (2013) Value of 11C-methionine PET in imaging brain tumours and metastases. Eur J Nucl Med Mol Imaging 40:615–635
Galldiks N, Dunkl V, Kracht LW, Vollmar S, Jacobs AH, Fink GR, Schroeter M (2012) Volumetry of [(1)(1)C]-methionine positron emission tomographic uptake as a prognostic marker before treatment of patients with malignant glioma. Mol Imaging 11:516–527
Stober B, Tanase U, Herz M, Seidl C, Schwaiger M, Senekowitsch-Schmidtke R (2006) Differentiation of tumour and inflammation: characterisation of [methyl-3H]methionine (MET) and O-(2-[18F]fluoroethyl)-L-tyrosine (FET) uptake in human tumour and inflammatory cells. Eur J Nucl Med Mol Imaging 33:932–939
Ceyssens S, Van Laere K, de Groot T, Goffin J, Bormans G, Mortelmans L (2006) [11C]methionine PET, histopathology, and survival in primary brain tumors and recurrence. AJNR. Am J Neuroradiol 27:1432–1437
Reifenberger GKJBP, Louis DN, Collins VP (2000) World health organization classification of tumours. Pathology and genetics of tumours of the nervous system. In: Kleihues P, Cavenee WK (eds) Astrocytic and oligodendroglioma. IARC Press, Lyon, France, pp 56–67
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for R, Treatment of Cancer Brain T, Radiotherapy G, National Cancer Institute of Canada Clinical Trials G (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Karnofsky DA BJ (1949) The Clinical Evaluation of Chemotherapeutic Agents in Cancer. In: MacLeod CM (Ed), Evaluation of Chemotherapeutic Agents. Columbia Univ Press. .196
Macdonald DR, Cascino TL, Schold SC Jr, Cairncross JG (1990) Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol 8:1277–1280
Kato T, Shinoda J, Nakayama N, Miwa K, Okumura A, Yano H, Yoshimura S, Maruyama T, Muragaki Y, Iwama T (2008) Metabolic assessment of gliomas using 11C-methionine, [18F] fluorodeoxyglucose, and 11C-choline positron-emission tomography. AJNR Am J Neuroradiol 29:1176–1182
Kato T, Shinoda J, Oka N, Miwa K, Nakayama N, Yano H, Maruyama T, Muragaki Y, Iwama T (2008) Analysis of 11C-methionine uptake in low-grade gliomas and correlation with proliferative activity. AJNR Am J Neuroradiol 29:1867–1871
Shinozaki N, Uchino Y, Yoshikawa K, Matsutani T, Hasegawa A, Saeki N, Iwadate Y (2011) Discrimination between low-grade oligodendrogliomas and diffuse astrocytoma with the aid of 11C-methionine positron emission tomography. J Neurosurg 114:1640–1647
Okamoto S, Shiga T, Hattori N, Kubo N, Takei T, Katoh N, Sawamura Y, Nishijima K, Kuge Y, Tamaki N (2011) Semiquantitative analysis of C-11 methionine PET may distinguish brain tumor recurrence from radiation necrosis even in small lesions. Ann Nucl Med 25:213–220
Soret M, Bacharach SL, Buvat I (2007) Partial-volume effect in PET tumor imaging. J Nucl Med 48:932–945
Nihashi T, Dahabreh IJ, Terasawa T (2013) Diagnostic accuracy of PET for recurrent glioma diagnosis: a meta-analysis. AJNR Am J Neuroradiol 34(944–950):S941–911
Kracht LW, Friese M, Herholz K, Schroeder R, Bauer B, Jacobs A, Heiss WD (2003) Methyl-[11C]- l-methionine uptake as measured by positron emission tomography correlates to microvessel density in patients with glioma. Eur J Nucl Med Mol Imaging 30:868–873
Tsuyuguchi N, Sunada I, Iwai Y, Yamanaka K, Tanaka K, Takami T, Otsuka Y, Sakamoto S, Ohata K, Goto T, Hara M (2003) Methionine positron emission tomography of recurrent metastatic brain tumor and radiation necrosis after stereotactic radiosurgery: is a differential diagnosis possible? J Neurosurg 98:1056–1064
Torii K, Tsuyuguchi N, Kawabe J, Sunada I, Hara M, Shiomi S (2005) Correlation of amino-acid uptake using methionine PET and histological classifications in various gliomas. Ann Nucl Med 19:677–683
Tsuyuguchi N, Takami T, Sunada I, Iwai Y, Yamanaka K, Tanaka K, Nishikawa M, Ohata K, Torii K, Morino M, Nishio A, Hara M (2004) Methionine positron emission tomography for differentiation of recurrent brain tumor and radiation necrosis after stereotactic radiosurgery—in malignant glioma. Ann Nucl Med 18:291–296
Adamson C, Kanu OO, Mehta AI, Di C, Lin N, Mattox AK, Bigner DD (2009) Glioblastoma multiforme: a review of where we have been and where we are going. Expert Opin Investig Drugs 18:1061–1083
De Witte O, Goldberg I, Wikler D, Rorive S, Damhaut P, Monclus M, Salmon I, Brotchi J, Goldman S (2001) Positron emission tomography with injection of methionine as a prognostic factor in glioma. J Neurosurg 95:746–750
Nariai T, Tanaka Y, Wakimoto H, Aoyagi M, Tamaki M, Ishiwata K, Senda M, Ishii K, Hirakawa K, Ohno K (2005) Usefulness of L-[methyl-11C] methionine-positron emission tomography as a biological monitoring tool in the treatment of glioma. J Neurosurg 103:498–507
Chu HH, Choi SH, Ryoo I, Kim SC, Yeom JA, Shin H, Jung SC, Lee AL, Yoon TJ, Kim TM, Lee SH, Park CK, Kim JH, Sohn CH, Park SH, Kim IH (2013) Differentiation of true progression from pseudoprogression in glioblastoma treated with radiation therapy and concomitant temozolomide: comparison study of standard and high-b-value diffusion-weighted imaging. Radiology 269:831–840
Tran DK, Jensen RL (2013) Treatment-related brain tumor imaging changes: so-called “pseudoprogression” vs. tumor progression: review and future research opportunities. Surg Neurol Int 4:S129–S135
Acknowledgments
This study was supported by a grant (CRI13022-1) from the Chonnam National University Hospital Research Institute of Clinical Medicine.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The retrospective study has been approved by our institutional review board (IRB No: CNUHH-2015-100), and the need for written informed consent was waived.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Jung, TY., Min, JJ., Bom, HS. et al. Prognostic value of post-treatment metabolic tumor volume from 11C-methionine PET/CT in recurrent malignant glioma. Neurosurg Rev 40, 223–229 (2017). https://doi.org/10.1007/s10143-016-0748-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-016-0748-1