Abstract
The apparent diffusion coefficient (ADC) on diffusion-weighted imaging (DWI) plays an important role in diagnosing intracranial tumors and predicting the histopathological grade of the tumor. However, the differences in the ADC values between craniopharyngiomas and germ cell tumors (GCTs) have not been clarified. We therefore evaluated the DWI and ADC values at b = 1000 and b = 4000 s/mm2 on 3T magnetic resonance (MR) imaging and assessed the possibility of differentiating between craniopharyngiomas and GCTs. We retrospectively reviewed 19 patients with craniopharyngioma and 24 patients with GCT who underwent surgery and received a histopathological diagnosis. Thirty-four patients underwent DWI with b = 1000 and b = 4000 s/mm2 and nine patients underwent periodically rotated overlapping parallel lines with enhanced reconstruction (PROPELLER) DWI with b = 1000 s/mm2. The ADC was determined by manually placing regions of interests (ROIs) in the respective tumor regions on the ADC maps and is expressed as the minimum (ADCMIN), mean (ADCMEAN), and maximum (ADCMAX) absolute values. The craniopharyngiomas showed lower intensity on DWI at b = 1000 and b = 4000 s/mm2 than the GCTs. Furthermore, the craniopharyngiomas demonstrated significantly high ADC values (ADCMIN, ADCMEAN, and ADCMAX) in comparison with the GCTs on DWI at b = 1000 and b = 4000 s/mm2. The logistic discriminant analysis clarified the advantage of ADCMIN at b = 4000 s/mm2 in differentiating between craniopharyngiomas and GCTs compared with the other ADC values. DWI and the ADC values may help clinicians to differentiate between craniopharyngiomas and GCTs. The ADCMIN at b = 4000 s/mm2 is particularly useful for differentiation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Among primary brain tumors, craniopharyngiomas (2.5 %) and germ cell tumors (GCTs) (2.3 %) are common in the Japanese population [1]. The epidemiology, markers, and locations of the tumors are useful for diagnosing both craniopharyngiomas and GCTs. These lesions, however, are likely to occur in the suprasellar region in young patients and exhibit various findings on magnetic resonance (MR) imaging. The titer of human chorionic gonadotropin-beta (HCG-β) in the cerebral spinal fluid, which generally indicates the presence of GCTs, is often elevated in patients with craniopharyngiomas [2, 3]. On the other hand, there are many GCTs without elevation of the titer of HCG-β and alpha-fetoprotein [4, 5]. In such cases, accurately differentiating between craniopharyngiomas and GCTs preoperatively is very difficult. However, the treatment strategies for these tumors differ (craniopharyngiomas should be removed surgically and GCTs should be treated with chemotherapy); therefore, it is essential to accurately distinguish between craniopharyngiomas and GCTs preoperatively.
Diffusion-weighted imaging (DWI) and the apparent diffusion coefficient (ADC) are used to diagnose intracranial tumors and predict the histopathological grade of the tumor [6–9], and the advantages of high b value DWI have been reported [8, 10–13]. In the present study, we retrospectively evaluated the efficacy of DWI and ADC values at b = 1000 and b = 4000 s/mm2 on 3T MR imaging and assessed the usefulness of DWI and the ADC values for differentiating between craniopharyngiomas and GCTs.
Materials and methods
Patients
A total of 55 patients who underwent 3T MR imaging and DWI scans to detect tumors at Hiroshima University Hospital between September 2006 and December 2013 were included in this retrospective study. All 55 patients were treated surgically at our institution and received a histological diagnosis; however, we excluded 12 patients with cystic lesions from this study in order to prevent the partial volume effect on the measurement of regions of interest (ROIs). The median age of the 19 patients with craniopharyngioma and 24 patients with GCT at the time of surgery was 20 years (range 5 to 70; Table 1). The GCTs included several histopathological types: 16 patients with pure germinoma, 1 patient with mature teratoma, 1 patient with immature teratoma, 1 patient with choriocarcinoma, 2 patients with yolk sac tumors + immature teratomas, 1 patient with germinoma + immature teratoma, 1 patient with germinoma + mature teratoma, and 1 patient with germinoma + choriocarcinoma. The patients were categorized into two groups: those with pure germinoma (n = 16) and those with non-pure germinoma (n = 8). Calcification on computed tomography (CT) was founded in the craniopharyngiomas (n = 11) and GCTs (n = 17). The focal calcification of GCTs included the pineal grand calcification in a maximum of 13 patients.
MR imaging
MR images were acquired on a 3T superconducting system (Signa Horizon; Medical Systems, Milwaukee, WI, USA). Axial DWI was performed with b values of 1000 and 4000 s/mm2, an effective gradient registered at 40 mT/m and a slew rate of 150 mT/m/ms. The parameters at b = 1000 and b = 4000 s/mm2 using an eight-channel-phased array head coil at repetition time (TR) 5000 ms with echo times (TEs) of 73.7 and 101.1 ms were as follows: field of view (FOV) = 22 × 22 cm, section thickness = 6 mm, intersection gap = 1.0 mm, number of slices = 20, and data acquisition matrix = 128 × 128. Tumors located in the intrasellar or suprasellar region were evaluated using periodically rotated overlapping parallel lines with enhanced reconstruction (PROPELLER) DWI to reduce susceptibility artifacts [7, 14]. For axial PROPELLER DWI, the b value was 1000 s/mm2 with the following settings: TR = 5000 ms, TE = 125 ms, FOV = 22 × 22 cm, section thickness = 4 mm, intersection gap = 0.3 mm, and data acquisition matrix = 128 × 128. The patients were subsequently divided into the following three categories for the evaluation of the signal intensity on DWI: (1) low intensity, (2) iso intensity, and (3) high intensity.
All ADC maps generated with the software program (Functool; GE Medical Systems) were obtained by calculating the signal intensity on DWI at two different b values (0 and 1000 s/mm2 for ADC map b = 1000, and 0 and 4000 s/mm2 for ADC map b = 4000 s/mm2 on a pixel-by-pixel basis). The ADC was determined by manually placing the ROIs in the respective tumor regions on ADC maps of b = 1000 s/mm2, and the procedure was repeated with an approximately similar size of ROI placed at the same site on the ADC maps of b = 4000 s/mm2. ROIs were purposely placed over the gadolinium (Gd)-enhanced components of the tumors in order to avoid volume averaging with cystic, hemorrhagic, or calcified regions that may influence the ADC values. The number of ROIs appropriated depended on the size of the enhanced lesion. Based on 1–3 ROIs on the ADC maps at b = 1000 and b = 4000 s/mm2, the ADC value for each tumor was calculated and expressed as the minimum (ADCMIN), mean (ADCMEAN), and maximum (ADCMAX) absolute values.
Statistical analysis
All statistical analyses were performed using the software package SPSS 16.0J for Windows (SPSS Inc., Chicago, IL, USA). The relationships between the average ADC values were evaluated using a logistic discriminant analysis with the model, Pr (Y = 1|x) = 1 / {1 + exp (−β0 − β1 x)} where Y denotes the binary variable having a value of 1 in cases of GCTs and 0 in case of craniopharyngiomas and x denotes the variable of the ADC measurement. The chi-square test and Fisher’s exact test were used for comparisons among the categories of signal intensity on DWI. The values are expressed as medians. Differences are represented as the t value of the magnitude of estimated coefficients β1 and were considered significant at a p value of <0.05.
Results
DWI
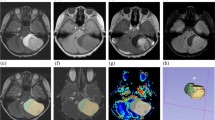
The craniopharyngiomas (Fig. 1) showed low intensity on DWI at b = 1000 s/mm2 (n = 14, 73.6 %), while the GCTs (Fig. 2) showed high intensity (n = 17, 70.8 %) (p = 0.0058). The differences were not influenced by the histopathological diagnosis of the adamantinomatous or papillary type or the presence/absence of pure germinoma. DWI at b = 4000 s/mm2 demonstrated similar findings; low intensity for craniopharyngiomas (n = 10, 83.3 %) and high intensity for GCTs (n = 18, 78.3 %) (p = 0.0009) (Fig. 3). The differences in intensity between the craniopharyngiomas and GCTs were clear on DWI at b = 4000 s/mm2.
Magnetic resonance images of a pure germinoma in a 29-year-old male (a–c). A suprasellar mass was observed on a gadolinium-enhanced T1-weighted image (a), which showed slightly high intensity on DWI at b = 1000 s/mm2 (b) and high intensity at b = 4000 s/mm2 (c). The high intensity of the pure germinoma was emphasized on DWI at b = 4000 s/mm2
ADC values in the craniopharyngiomas and GCTs
The ADC values of the adamantinomatous- and papillary-type lesions are shown in Table 2. There were no significant differences in the ADC values between these tumors, although the papillary-type lesions demonstrated lower ADC values than the adamantinomatous-type lesions on all images. In addition, no significant differences were found in the ADC values between the pure germinoma and non-pure germinoma groups.
ADC values in the calcified and non-calcified tumors
The ADC values of the calcified and non-calcified craniopharyngiomas were shown in Table 3. There was no significant difference in the ADC values between the two types of craniopharyngiomas, although the calcified craniopharyngiomas demonstrated lower ADC values than the non-calcified craniopharyngiomas. In the ADC values between the calcified and non-calcified GCTs, no significant differences were also found.
Differences between the ADC values of the craniopharyngiomas and GCTs
The craniopharyngiomas demonstrated clearly high ADC values in comparison with the GCTs on all images (Table 4). The logistic discriminant analysis of 34 patients with ADC values at b = 1000 and b = 4000 s/mm2 showed the advantage of the ADC values at b = 4000 s/mm2, especially ADCMIN at b = 4000 s/mm2 (p = 0.00172, t = −3.14). Using ADCMIN at b = 4000 s/mm2, the sensitivity and specificity for the differential diagnosis of GCT were 90.9 and 91.7 %, respectively.
Discussion
DWI and the ADC are often used to diagnose intracranial tumors [7, 9, 11, 15, 16]. It has also been reported that the ADC values of craniopharyngiomas are significantly higher than the ADC values of pituitary adenomas and meningiomas [9], while the ADCMIN values of craniopharyngiomas are lower than those of Rathke’s cleft cysts [7]. The differences between craniopharyngiomas and GCTs have been examined in a small number of cases, with no significant differences having been founded [9]. Therefore, this is the first study to demonstrate the differences in DWI and ADC values between craniopharyngiomas and GCTs.
High-grade tumors in pediatric patients as well as high-grade gliomas and high-grade meningiomas have been reported to exhibit lower ADC values than low-grade tumors [8, 9, 17]. It has also been reported that the mechanisms underlying the restricted diffusion of high-grade tumors originate in the high cellularity of these lesions [11, 18, 19]. From the viewpoint of microscopic findings, it is reasonable that the ADC values of malignant GCTs are lower than those of benign craniopharyngiomas, as GCTs usually display a high cellular pattern, while craniopharyngiomas usually show a loose, low cellular pattern. Craniopharyngiomas are rich in the non-cellular components of cholesterin, keratin, and microcysts (adamantinomatous type) in addition to villous fibrovascular stroma (papillary type), all of which may demonstrate relatively high ADC values. On the other hand, germinomas, the common component of GCTs in this study, are composed of uniform cells with large nuclei and poor stroma exhibiting lymphocytic infiltration that may demonstrate low ADC values.
In the present study, the advantage of ADCMIN at b = 4000 s/mm2 for differentiating between craniopharyngiomas and GCTs was demonstrated. Furthermore, it has been reported that high b value DWI is useful for predicting the histopathological grade of cerebral gliomas [10, 12]. Moreover, the ADC at b = 4000 s/mm2 displays a better inverse correlation reflective of the degree of tumor cellularity than the ADC value at b = 1000 s/mm2 [11]. Our findings coincide well with these findings of past reports.
Craniopharyngiomas have pathologically two subtypes, while GCTs have five. In addition, the mixed type of GCT exhibits structural heterogeneity in the tumor. In the present study, there were no significant differences in the ADC values between the craniopharyngiomas of the adamantinomatous and papillary types and the GCTs in the pure germinoma and non-pure germinoma groups. Craniopharyngiomas of the papillary type, however, are likely to have lower ADC values than those of the adamantinomatous type. Unfortunately, the subtype of GCT could not be analyzed in this study due to the small number of these lesions. Further studies may help to clarify the differences in ADC values between the pathological subtypes in craniopharyngiomas and GCTs and aid in the preoperative diagnosis of each subtype in the future.
Calcification is one of the characteristics in craniopharyngiomas. We tried to avoid the dense calcified lesions; however, the contamination of diffuse calcification might affect the ADC values of the craniopharyngiomas. Calcification itself has no signal on MR imaging, and the contamination of calcified lesions would make ADC values lower than original ADC values of the tumors. In other words, there is a possibility that the calcified craniopharyngiomas demonstrate lower ADC values than the non-calcified craniopharyngiomas. We compared the difference between the calcified and non-calcified tumors in our series, and we could not find a statistically significant difference between these two types of tumors. Our results implied that the influence of calcification could be reduced by carefully placing the ROIs on the non-calcified areas. In the end, the craniopharyngiomas with or without calcification showed markedly higher ADC values than the GCTs in the present study.
There are some limitations associated with our study. Sellar and parasellar lesions have high sensitivity to susceptibility artifacts due to nasal sinus air. Although we conducted PROPELLER MR imaging in patients with sellar and parasellar lesions, we could not completely exclude the susceptibility artifacts. In addition, craniopharyngiomas and GCTs include small cysts, microscopic calcification, and areas of microbleeding, all of which may affect ADC measurements. We compared CT and T1- and T2-weighted images on MR and carefully placed the ROIs in order to prevent the partial volume effect. Furthermore, 12 patients were excluded from the present study, because the effects of the cysts could not be avoided. Hence, our results should be fairly evaluated based on the effects of small cysts, calcification, and microbleeding, although we made every effort to remove these effects.
Conclusions
In this study, the craniopharyngiomas demonstrated lower intensity on DWI and higher ADC values at b = 1000 and b = 4000 s/mm2 on 3T MR imaging in comparison with the GCTs, especially with ADCMIN at b = 4000 s/mm2. DWI and the ADC values may help clinicians to differentiate between craniopharyngiomas and GCTs preoperatively.
References
The committee of the brain tumor registry of Japan (2014) Report of brain tumor registry of Japan (2001–2004) 13th edition. Neurol Med Chir (Tokyo) 54:9–102
Honegger J, Mann K, Thierauf P, Zrinzo A, Fahlbusch R (1995) Human chorionic gonadotrophin immunoactivity in cystic intracranial tumours. Clin Endocrinol (Oxf) 42:235–241
Kinoshita Y, Tominaga A, Usui S, Kurisu K (2014) A craniopharyngioma with spontaneous involution of a gadolinium-enhanced region on magnetic resonance imaging. Surg Neurol Int 5:128
Matsutani M, Sano K, Takakura K, Fujimaki T, Nakamura O, Funata N, Seto T (1997) Primary intracranial germ cell tumors: a clinical analysis of 153 histologically verified cases. J Neurosurg 86:446–455
Qaddoumi I, Sane M, Li S, Kocak M, Pai-Panandiker A, Harreld J, Klimo P, Wright K, Broniscer A, Gajjar A (2012) Diagnostic utility and correlation of tumor markers in the serum and cerebrospinal fluid of children with intracranial germ cell tumors. Childs Nerv Syst 28:1017–1024
Kunii N, Abe T, Kawamo M, Tanioka D, Izumiyama H, Moritani T (2007) Rathke’s cleft cysts: differentiation from other cystic lesions in the pituitary fossa by use of single-shot fast spin-echo diffusion-weighted MR imaging. Acta Neurochir (Wien) 149:759–769
Mahmoud OM, Tominaga A, Amatya VJ, Ohtaki M, Sugiyama K, Saito T, Sakoguchi T, Kinoshita Y, Shrestha P, Abe N, Akiyama Y, Takeshima Y, Arita K, Kurisu K, Yamasaki F (2010) Role of PROPELLER diffusion weighted imaging and apparent diffusion coefficient in the diagnosis of sellar and parasellar lesions. Eur J Radiol 74:420–427
Watanabe Y, Yamasaki F, Kajiwara Y, Takayasu T, Nosaka R, Akiyama Y, Sugiyama K, Kurisu K (2013) Preoperative histological grading of meningiomas using apparent diffusion coefficient at 3T MRI. Eur J Radiol 82:658–663
Yamasaki F, Kurisu K, Satoh K, Arita K, Sugiyama K, Ohtaki M, Takaba J, Tominaga A, Hanaya R, Yoshioka H, Hama S, Ito Y, Kajiwara Y, Yahara K, Saito T, Thohar MA (2005) Apparent diffusion coefficient of human brain tumors at MR imaging. Radiology 235:985–991
Alvarez-Linera J, Benito-Leon J, Escribano J, Rey G (2008) Predicting the histopathological grade of cerebral gliomas using high b value MR DW imaging at 3-tesla. J Neuroimaging 18:276–281
Doskaliyev A, Yamasaki F, Ohtaki M, Kajiwara Y, Takeshima Y, Watanabe Y, Takayasu T, Amatya VJ, Akiyama Y, Sugiyama K, Kurisu K (2012) Lymphomas and glioblastomas: differences in the apparent diffusion coefficient evaluated with high b-value diffusion-weighted magnetic resonance imaging at 3 T. Eur J Radiol 81:339–344
Seo HS, Chang KH, Na DG, Kwon BJ, Lee DH (2008) High b-value diffusion (b = 3000 s/mm2) MR imaging in cerebral gliomas at 3T: visual and quantitative comparisons with b = 1000 s/mm2. Am J Neuroradiol 29:458–463
Yamasaki F, Kurisu K, Aoki T, Yamanaka M, Kajiwara Y, Watanabe Y, Takayasu T, Akiyama Y, Sugiyama K (2012) Advantages of high b-value diffusion-weighted imaging to diagnose pseudo-responses in patients with recurrent glioma after bevacizumab treatment. Eur J Radiol 81:2805–2810
Forbes KP, Pipe JG, Karis JP, Heiserman JE (2002) Improved image quality and detection of acute cerebral infarction with PROPELLER diffusion-weighted MR imaging. Radiology 225:551–555
Bull JG, Saunders DE, Clark CA (2012) Discrimination of paediatric brain tumours using apparent diffusion coefficient histograms. Eur Radiol 22:447–457
Dumrongpisutikul N, Intrapiromkul J, Yousem DM (2012) Distinguishing between germinomas and pineal cell tumors on MR imaging. Am J Neuroradiol 33:550–555
Porto L, Jurcoane A, Schwabe D, Kieslich M, Hattingen E (2013) Differentiation between high and low grade tumours in paediatric patients by using apparent diffusion coefficients. Eur J Paediatr Neurol 17:302–307
Guo AC, Cummings TJ, Dash RC, Provenzale JM (2002) Lymphomas and high-grade astrocytomas: comparison of water diffusibility and histologic characteristics. Radiology 224:177–183
Sugahara T, Korogi Y, Kochi M, Ikushima I, Shigematu Y, Hirai T, Okuda T, Liang LX, Ge YL, Komohara Y, Ushio Y, Takahashi M (1999) Usefulness of diffusion-weighted MRI with echo-planar technique in the evaluation of cellularity in gliomas. J Magn Reson Imaging 9:53–60
Acknowledgments
This study was partially supported by a grant-in-aid from the Japan Society for the Promotion of Science Grant-in Aid for Scientific Research (C) No. 25462262.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Masahiko Tosaka, Gunma, JAPAN
Diffusion-weighted MR imaging (DWI) is becoming a commonly performed preoperative evaluation to investigate tumor cell density. The authors used periodically rotated overlapping parallel lines with enhanced reconstruction (PROPELLER) DWI as a special MR imaging method to reduce susceptibility artifacts for the preoperative qualitative evaluation of pituitary adenoma and other parasellar tumors in the previous reports. This unique study is an important investigation of the use of PROPELLER DWI for the differential diagnosis of craniopharyngiomas and germ cell tumors (GCTs). GCTs are enhanced well after gadolinium administration and sometimes show cyst formation, rarely with large multilobulated cysts, and sometimes involve calcification. Suprasellar GCTs are rarely misdiagnosed as craniopharyngioma. Papillary craniopharyngiomas including a relatively large solid component and GCTs share some imaging similarities in adult patients. However, craniopharyngioma and GCTs have quite different treatment strategies. The present study indicates that PROPELLER DWI evaluation of tumor cell density may offer important preoperative information for the differentiation of these tumors. Differentiation of pathological subtypes in craniopharyngiomas and GCTs might be the next target of using this special MR imaging method
Rights and permissions
About this article
Cite this article
Kinoshita, Y., Yamasaki, F., Tominaga, A. et al. Diffusion-weighted imaging and the apparent diffusion coefficient on 3T MR imaging in the differentiation of craniopharyngiomas and germ cell tumors. Neurosurg Rev 39, 207–213 (2016). https://doi.org/10.1007/s10143-015-0660-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-015-0660-0