Abstract
Sepsis causes high mortality in intensive care units. Although there have been many studies on the gut microbiota in patients with sepsis, the impact of sepsis on the gut microbiota has not been directly determined because the treatment of sepsis also affects the gut microbiota. Therefore, we designed this animal experiment to explore gut microbiota alterations during sepsis. Mice were divided into two groups, mice that survived less than 3 days and mice that survived more than 3 days. Fecal samples collected on the day of cecal ligation and puncture (CLP), as well as on the 3rd and 7th days after CLP, were subjected to microbial community analysis and nontargeted metabolomics analysis. The results showed significantly lower bacterial diversity in fecal samples after CLP. At the genus level, the fecal samples obtained on the 3rd and 7th days after CLP exhibited significantly increased relative abundances of Bacteroides, Helicobacter, etc., and significantly decreased relative abundances of Alloprevotella, Prevotella, etc. Innate metabolite levels were significantly different in mice that survived less than 3 days and mice that survived more than 3 days. In conclusion, CLP-induced sepsis in mice changes the structure of the gut microbiome, and innate metabolites affect the prognosis of septic mice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sepsis is a life-threatening type of organ dysfunction caused by a dysregulated host response to an infection (Singer et al. 2016). Sepsis affects one-fifth of patients admitted to intensive care units (ICUs) in mainland China, with a 90-day mortality rate of 35.5% (Xie et al. 2020).
The gut microbiota is a complex ecosystem that includes a wide variety of bacteria, consisting of trillions of bacterial cells (Sender et al. 2016). Many studies have shown that these microbial communities are involved in many physiological activities, such as metabolism, control of the nervous system (Adak and Khan 2019), and regulation of the immune response (Rooks and Garrett 2016). In addition to the gut microbiota, the difference of metabolites caused by diets and lifestyles will also affect the immune status, leading to different outcomes of diseases (Leitner et al. 2023; Perler et al. 2023).
Although the pathogenesis of sepsis is multifactorial and not fully understood, an increasing amount of evidence shows that disruption of the gut microbiota predisposes patients to sepsis and negatively affects sepsis outcomes (Adelman et al. 2020). Changes in the gut microbiota do not only affect outcomes before the onset of sepsis; after the onset of sepsis, changes in the structure of the normal gut microbiota worsen and lead to worse outcomes.
There are many studies on the gut microbiota in patients with sepsis, but they have not directly determined the influence of sepsis, which is a single variable, on the gut microbiota because the gut microbiota is affected by sepsis treatments, such as antibiotics, opioids, parenteral nutrition, and proton pump inhibitors (Zaborin et al. 2014; Imhann et al. 2016; Dahlgren et al. 2019). Therefore, we designed this animal experiment. We found that 3 days after the operation was the peak time of death for septic mice. Therefore, we divided the mice into groups according to the postoperative survival time and explored the changes in the gut microbiota after sepsis and the effect of innate metabolites on the prognosis of mice.
Materials and methods
Animals
SPF-grade, 6- to 8-week-old male C57BL/6 J mice were purchased from Charles River. All animals were housed in a specific pathogen-free facility and given free access to food and water. The animal studies were approved by the Harbin Medical University Animal Care and Use Committee.
Experimental design
The mice were subjected to cecal ligation and puncture (CLP) surgery. The survival of the mice was recorded every day after the operation, and a survival curve was drawn (Suppl. Fig. 1). According to the postoperative survival time, the CLP mice were divided into two groups: mice that survived less than 3 days and mice that survived more than 3 days (8 mice in each group). We collected the feces from mice that survived more than 3 days on the day of surgery (preoperative), the 3rd day after surgery, and the 7th day after surgery, and the faces from the mice that survived less than 3 days on the day of surgery. Feces were collected in a sterile cryopreservation tube at 3 p.m. every day and stored in a -80 °C freezer after collection. Subsequently, the feces were used for microbial community analysis and untargeted metabolomics analysis.
CLP model
The mice were subjected to CLP, an established model to induce polymicrobial peritonitis. Mice were anesthetized before surgery with an animal anesthesia ventilator system. After opening the abdomen, the cecum was exteriorized and ligated 1 cm from the terminus of the cecum without obstructing intestinal continuity. The cecum was punctured once with a 23-G needle and squeezed to excrete a small amount of fecal material. The abdominal wall was closed in layers after placing the cecum back in the abdomen. After surgery, all animals were given a subcutaneous injection of 1 ml of physiological saline that had been prewarmed to 37 °C. To evaluate mortality, the mice were observed for 7 days after surgery. The animals were sacrificed 7 days after surgery.
Fecal collection
To ensure the sterility of the fecal collection environment as much as possible, a plastic box used exclusively for fecal collection was sterilized and desiccated, and the mice were placed in the plastic box. After the mice defecated, the feces were immediately collected into sterile cryopreservation tubes and placed in liquid nitrogen for quick freezing. The samples were then immediately placed at -80 °C.
Microbial community analyses
Microbial DNA was extracted from the fecal samples using the QIAamp® Fast DNA Stool Mini Kit according to the manufacturer’s protocols. The V3-V4 region of the bacterial 16S ribosomal RNA genes was amplified by PCR using primers 341F 5’-CCTACGGGRSGCAGCAG)-3’ and 806R5’-GGACTACVVGGGTATCTAATC-3’. Amplicons were extracted from 2% agarose gels, purified using the AxyPrep DNA Gel Extraction Kit (Axygen Biosciences, Union City, CA, U.S.) according to the manufacturer’s instructions and quantified using Qubit® 2.0 (Invitrogen, U.S.). After preparation of the library, the tags were sequenced on a MiSeq platform (Illumina, Inc., CA, USA) for paired-end reads of 250 bp, which overlapped on their 3 ends for concatenation into original longer tags.
Untargeted metabolomics GC‒MS analyses
The feces were collected in 2-mL EP tubes containing 500 µL of prechilled solution and 10 µL of ribitol, and then, the mixtures were ground and centrifuged for 15 min (12,000 rpm, 4 °C). A total of 400 µL of supernatant was collected, and 80 µL of each sample was mixed for quality control (QC) samples. The supernatant was dried in a vacuum centrifuge. After the addition of 30 µL methoxyamine salt reagent to the dried metabolites, the samples were incubated at 80 °C for 30 min. Then, 40 µL of BSTFA (containing 1% TMCS, v/v) was added to each sample and incubated at 70 °C for 1.5 h. After cooling to room temperature, 5 µL of FAMEs were added to the sample. Subsequently, an Agilent 7890 gas chromatography-time-of-flight mass spectrometer equipped with an Agilent DB-5MS capillary column (30 m × 250 μm × 0.25 μm, J&W Scientific, Folsom, CA, USA) was used for detection.
Statistical analyses
Operational taxonomic units (OTUs) were clustered with 97% similarity using UPARSE (http://drive5.com/uparse/), and chimeric sequences were identified and removed using Userach (version 7.0). Each representative tag was assigned to a taxon by RDP Classifier (http://rdp.cme.msu.edu/) according to the RDP database (http://rdp.cme.msu.edu/), using a confidence threshold of 0.8. An OTU profiling table was generated, and alpha/beta diversity analyses were performed with Python scripts in QIIME. ChromaTOF software (V 4.3x, LECO) was used to analyze the mass spectrum data, including analysis of peak extraction, baseline correction, deconvolution, peak integration, and peak alignment. In the qualitative analysis of the substances, the LECO-Fiehn Rtx5 database was used, including mass spectrometry matching and retention time index matching.
The data are presented as the means ± SEMs, and GraphPad Prism 7 was used for the statistical analyses. Data sets with 2 groups were analyzed using an unpaired t test. Data sets with more than 2 groups were analyzed by one-way ANOVA followed by Tukey’s multiple comparisons test. Survival was analyzed using the log-rank (Mantel‒Cox) test. A p value of < 0.05 indicated statistical significance for all statistical analyses.
Data availability
The data sets generated during the current study have been deposited at NCBI repository under the accession number PRJNA758244.
Results
Alterations in the gut microbiota of septic mice
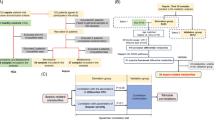
Principal coordinate analysis (PCoA) unweighted Adonis analysis revealed no significant differences in preoperative fecal samples between mice that survived less than 3 days and mice that survived more than 3 days (Fig. 1A), whereas the gut microbiota of the preoperative fecal samples and the postoperative fecal samples collected on the 3rd and 7th day from mice that survived more than 3 days were significantly separated (P < 0.001) (Fig. 1B). Principal component analysis (PCA) also showed that there was no significant difference in preoperative feces between the mice that survived less than 3 days and those that survived more than 3 days (Fig. 1C), while there were significant differences between preoperative and postoperative fecal samples for mice that survived more than 3 days (Fig. 1D).
OTU PCA and PCoA unweighted analysis of changes in the gut microbiota. SS (Survival Sepsis group) indicates that the mice survived less than 3 days; DS (Dead Sepsis group) indicates that the mice survived more than 3 days. (A) The PCoA analysis showed that the points between the two groups were aggregative, which means there were no significant differences in the gut microbiota between the two groups. (B) The gut microbiota was dispersed from point to point between preoperative and postoperative fecal samples from mice that survived more than 3 days. (C) OTU PCA showed that the points between the two groups were aggregative. (D) The points among the 3 groups were significantly separated. (Different colors in the figures represent different groups of samples, and the greater the distance between the two points, the greater the difference in microbial community between the two samples)
The Shannon index showed no significant difference in the alpha diversity of the microbial community between the feces of mice that survived less than 3 days and those of mice that survived longer than 3 days (Fig. 2A). However, in the feces of mice that survived for more than 3 days, there was a significant decrease in alpha diversity at 3 and 7 days postoperatively compared to the preoperative levels (Fig. 2B).
Alpha-diversity index difference analysis. (A) No significant difference in preoperative fecal samples was observed between mice that survived less than 3 days and mice that survived more than 3 days. (B) The preoperative fecal samples were significantly different from the samples collected on the 3rd and 7th days. **means p < 0.01, ***means p < 0.001
Effects of sepsis induced by the CLP model on variations in the gut microbiota composition of mouse feces
We selected the top 20 bacterial genera with high relative abundance. At the genus level, the fecal samples obtained on the 3rd day and 7th day after CLP exhibited significantly increased relative abundances of Bacteroides, Helicobacter, Clostridium sensu stricto, Clostridium XlVa, Mucispirillum, Roseburia, and Mycoplasma and significantly decreased relative abundances of Alloprevotella, Prevotella, Parabacteroides, Parasutterella, and Akkermansia (Fig. 3A). At the phylum level, the relative abundance of Bacteroidetes significantly decreased on the 3rd and 7th days after CLP, and the relative abundance of Proteobacteria significantly increased (Fig. 3B).
Species abundance analysis. The abscissa is the group name, and the ordinate is the relative abundance of species. Different colors correspond to different species names, and the length of the color block represents the relative abundance of the species represented by the color block. (A) At the genus level. (B) At the phylum level
Linear discriminant analysis effect size (LEfSe) analysis was used to analyze the gut microbiota of preoperative fecal samples and postoperative fecal samples on the 3rd and 7th days from mice that survived more than 3 days, and 72 OTUs with significant differences (LAD > 2.0) were found, including 7 at the phylum level, 8 at the class level, 10 at the order level, 18 at the family level, and 29 at the genus level. At the genus level, among the OTUs with significant differences, Alloprevotella and Prevotella were the two bacteria with the greatest influence on the difference in the preoperative fecal samples of mice. In the fecal samples on the 3rd day after CLP, Escherichia/Shigella and Clostridium XI were the top two most influential bacteria. Clostridium XlVa and Enterococcus were the two bacteria with the largest influence on abundance in the postoperative fecal samples on the 7th day (Fig. 4).
The influence of mouse innate metabolites on the prognosis of sepsis caused by CLP
The preoperative feces of mice that survived less than 3 days and those of mice that survived more than 3 days were analyzed by metabonomics, and we found differences in metabolites between the two groups (Fig. 5A). 3-(3-Hydroxyphenyl)propionic acid, 3-methylamino-1,2-propanediol 2, and D-erythronolactone 2 levels were significantly higher in the preoperative feces of mice that survived less than 3 days; 1-hexadecanol, phenylalanine 1,4-aminobenzoic acid, formononetin, spermidine 2,6-hydroxynicotinic acid, phenylalanine 2, fluorene, 1,2,4-benzenetriol, 2-aminophenol 2,phenyl beta-D-glucopyranoside, 2'-deoxyadenosine, and 2-keto-isovaleric acid 1 levels were significantly higher in the preoperative feces of mice that survived more than 3 days (Fig. 5B). We analyzed the metabolic pathways for metabolic differences and used KEGG, SMPDB, PubChem and other databases to obtain the following metabolic pathways: beta-alanine metabolism, glutathione metabolism, and arginine and proline metabolism (Fig. 5C).
Difference analysis of metabolites. (A) Orthogonal partial least squares-discriminative analysis (OPLS-DA). The abscissa P1 represents the predicted principal component score of the first principal component, and the ordinate O1 represents the orthogonal principal component score. Different colors represent two groups of samples: mice that survived less than 3 days and mice that survived more than 3 days. (B) Boxplot of differential metabolites. The abundance of different metabolites in the two groups is intuitively displayed in a boxplot. (C) Bubble plot of metabolic pathways enriched in differential metabolites. Each bubble in the bubble plot represents a metabolic pathway. The larger bubbles represent a larger influence factor. The deeper the color, the smaller the p value, and the more significant the enrichment
Correlations between the gut microbiota and gut metabolites
Subsequently, to further investigate the relationships between the gut microbiota and metabolites, we performed a Spearman correlation analysis between 16 metabolites and 18 bacterial taxa (Fig. 6). The results showed that the low abundance of metabolites in the fecal samples of mice that survived more than 3 days, including D − erythronolactone 2,3 − methylamino − 1,2 − propanediol 2,3 − (3 − hydroxyphenyl)propionic acid, was positively correlated with Escherichia_Shigella and Blautia abundance at the genus level. The high abundance of metabolites in the fecal samples of mice that survived less than 3 days, including 1,2,4-benzenetriol, 4-aminobenzoic acid, phenylalanine 2, etc., was positively correlated with Clostridium sensu stricto, Alloprevotella and Desulfovibrio abundance at the genus level.
Correlations between the gut microbiota and gut metabolites. The horizontal coordinate shows the differential metabolites, and the ordinate is the differential species. The color depth indicates the correlation, blue indicates a negative correlation, and red indicates a positive correlation. The p value is the correlation test result; * means p < = 0.05, and ** means p < = 0.01
Discussion
First, preoperative fecal samples and postoperative fecal samples collected on the 3rd and 7th days from mice that survived more than 3 days were subjected to 16S rRNA analysis. Research has shown that sepsis can change the gut microbiota. After combining the Adonis analysis of beta diversity and the alpha diversity analysis, we concluded that the diversity of the gut microbiome of septic mice was reduced.
Clinical studies have shown that the diversity of the gut microbiome of septic patients is also reduced (Lankelma et al. 2017). In general, the microbial diversity of patients with sepsis declines rapidly on hospital admission, a finding that becomes more pronounced later in their hospitalization (McDonald et al. 2016). In addition, a handful of clinical studies have shown that low bacterial diversity is associated with poor outcomes. In previous studies, low microbial diversity has been linked to an increased risk of mortality (Shimizu et al. 2011; Taur et al. 2014). This may cause a vicious cycle in which sepsis reduces gut microbial diversity, and lower bacterial diversity aggravates sepsis.
We also found that the composition of the gut microbiome also changed after CLP. Clinical studies by McDonald D et al.(McDonald et al. 2016) and Li Q et al.(Li et al. 2015) confirmed that fecal samples from ICU patients and sepsis patients tend to have decreased Bacteroidetes and increased Proteobacteria abundance, which corresponds with our findings. The results of our study showed that Bacteroidetes levels in the fecal samples on the 3rd and 7th days were lower than those in postoperative fecal samples at the phylum level, and the decrease was the greatest on the 3rd day after surgery. Studies have shown that Bacteroides and Firmicutes are crucial producers of short-chain fatty acids (SCFAs), which can regulate gene expression in regulatory T cells and alter the microbiocidal abilities of macrophages (Arpaia et al. 2013; Schulthess et al. 2019). In another study (Hyoju et al. 2019), the microbiota of an experimental group of mice that experienced a combination of pressures, such as an obesogenic Western diet, antibiotic administration and eventual death from sepsis, was depleted of Bacteroidetes and enriched in antibiotic-resistant Proteobacteria. This is similar to our mouse microbiota on the 3rd day after CLP. This may explain why the peak time of death of the mice was on the 3rd day after surgery.
At the genus level, compared to that in preoperative fecal samples, an increased relative abundance of Bacteroides in the fecal samples was observed on the 3rd day and 7th day after CLP. Bacteroides is one of the predominant genera in the human gut and plays an important role in maintaining the stability of the healthy gut ecosystem. Interestingly, according to our hypothesis, these beneficial bacteria should have reduced abundance after sepsis, but this is contrary to our observations. Therefore, we speculated that the increased abundance of this bacterium might have protected the mice from sepsis.
16S rRNA analysis and metabonomics analysis of the fecal samples from the mice that survived less than 3 days and those that survived more than 3 days were performed. There was no significant difference in the composition of the gut microbiota, but the metabolites were quite different. Thus, we suspected that innate metabolites could determine both susceptibility to and severity of infection. The purpose of our experiment is to identify dominant metabolites to resist sepsis. The upregulated metabolites in the feces of mice that survived more than 3 days were considered beneficial to the prognosis of sepsis. Formononetin, an isoflavone extracted from Astragalus membranaceus, has exhibited multiple inhibitory effects on cancer (Ong et al. 2019), atherosclerosis (Ma et al. 2020), neuroinflammation (El-Bakoush and Olajide 2018), oxidative stress (Yi et al. 2020) and other diseases in different animal models. Formononetin also protects against bacterial and viral infections (Dai et al. 2019; Wang et al. 2020). Another metabolite, spermidine, is a polycation that is associated with DNA in most living organisms (Pietrocola et al. 2019). Spermidine has been found to be related to a decrease in cancer-related mortality (Pietrocola et al. 2019) and inhibition of oxidative stress and mitochondrial fragmentation (Chai et al. 2019). However, a study by Huang S et al. (Huang et al. 2019) suggested that high phenylalanine levels are associated with a higher mortality risk in cases of severe infection. This is contrary to our conjecture that the highly expressed products phenylalanine 1 and phenylalanine 2 are protective.
We consider that the significantly upregulated metabolites in the feces of the mice that survived less than 3 days are harmful and not conducive to a good prognosis after sepsis. 3-(3-Hydroxyphenyl)propionic acid is produced by the gut microflora through the breakdown of chlorogenic acid and is thereafter absorbed and oxidized in the liver before entering the circulation (Marin et al. 2015). This molecule has recently been characterized as bioactive in stimulating osteoblast activity and increasing bone mass in mice in a dose‐dependent manner (Chen et al. 2014). However, no studies have confirmed that this molecule is related to infection or immunity.
At present, the mechanism of action of intestinal microbial metabolites in the context of sepsis remains unclear. In future experiments, we can supplement protective metabolites before CLP in mice to verify the role and mechanism of these metabolites. If feasible, it can be used for patients with high risk factors of sepsis to improve the outcomes of sepsis, which provides a new idea for the prevention of sepsis.
Compared with clinical studies, animal experiments are not affected by interference effects of treatments, such as antibiotics. Indeed, work by our lab and others has demonstrated that the use of antibiotics can lead to changes in the intestinal microbiota and metabolite levels (Suez et al. 2018; Han et al. 2021). Therefore, our study better reflects the effect of sepsis as a single variable on intestinal flora changes. Moreover, we collected preoperative fecal samples and postoperative fecal samples from mice on the day of surgery, the 3rd and 7th days, which can dynamically reflect the changes in the gut microbiome after sepsis.
In conclusion, CLP-induced sepsis in mice changes the structure of the gut microbiome, and innate metabolites affect the prognosis of septic mice.
Data availability
The datasets generated during the current study are available in the NCBI repository, https://www.ncbi.nlm.nih.gov/bioproject/PRJNA758244.
References
Adak A, Khan MR (2019) An insight into gut microbiota and its functionalities. Cell Mol Life Sci 76(3):473–493. https://doi.org/10.1007/s00018-018-2943-4
Adelman MW, Woodworth MH, Langelier C, Busch LM, Kempker JA, Kraft CS, Martin GS (2020) The gut microbiome’s role in the development, maintenance, and outcomes of sepsis. Crit Care 24(1):278. https://doi.org/10.1186/s13054-020-02989-1
Arpaia N, Campbell C, Fan X, Dikiy S, van der Veeken J, deRoos P, Liu H, Cross JR, Pfeffer K, Coffer PJ, Rudensky AY (2013) Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 504(7480):451–455. https://doi.org/10.1038/nature12726
Chai N, Zhang H, Li L, Yu X, Liu Y, Lin Y, Wang L, Yan J, Nikolaevna SE, Zhao Y (2019) Spermidine Prevents Heart Injury in Neonatal Rats Exposed to Intrauterine Hypoxia by Inhibiting Oxidative Stress and Mitochondrial Fragmentation. Oxid Med Cell Longev 2019:5406468. https://doi.org/10.1155/2019/5406468
Chen JR, Lazarenko OP, Zhang J, Blackburn ML, Ronis MJ, Badger TM (2014) Diet-derived phenolic acids regulate osteoblast and adipocyte lineage commitment and differentiation in young mice. J Bone Miner Res 29(5):1043–1053. https://doi.org/10.1002/jbmr.2034
Dahlgren AF, Pan A, Lam V, Gouthro KC, Simpson PM, Salzman NH, Nghiem-Rao TH (2019) Longitudinal changes in the gut microbiome of infants on total parenteral nutrition. Pediatr Res 86(1):107–114. https://doi.org/10.1038/s41390-019-0391-y
Dai W, Bi J, Li F, Wang S, Huang X, Meng X, Sun B, Wang D, Kong W, Jiang C, Su W (2019) Antiviral efficacy of flavonoids against enterovirus 71 infection in vitro and in newborn mice. Viruses 11(7):625. https://doi.org/10.3390/v11070625
El-Bakoush A, Olajide OA (2018) Formononetin inhibits neuroinflammation and increases estrogen receptor beta (ERbeta) protein expression in BV2 microglia. Int Immunopharmacol 61:325–337. https://doi.org/10.1016/j.intimp.2018.06.016
Han C, Guo N, Bu Y, Peng Y, Li X, Ma X, Yang M, Jia X, Zhang J, Liu X, Yu K, Wang C (2021) Intestinal microbiota and antibiotic-associated acute gastrointestinal injury in sepsis mice. Aging (albany NY) 13(7):10099–10111. https://doi.org/10.18632/aging.202768
Huang SS, Lin JY, Chen WS, Liu MH, Cheng CW, Cheng ML, Wang CH (2019) Phenylalanine- and leucine-defined metabolic types identify high mortality risk in patients with severe infection. Int J Infect Dis 85:143–149. https://doi.org/10.1016/j.ijid.2019.05.030
Hyoju SK, Zaborin A, Keskey R, Sharma A, Arnold W, van den Berg F, Kim SM, Gottel N, Bethel C, Charnot-Katsikas A, Jianxin P, Adriaansens C, Papazian E, Gilbert JA, Zaborina O, Alverdy JC (2019) Mice fed an obesogenic western diet, administered antibiotics, and subjected to a sterile surgical procedure develop lethal septicemia with multidrug-resistant pathobionts. mBio 10(4). https://doi.org/10.1128/mBio.00903-19
Imhann F, Bonder MJ, Vich Vila A, Fu J, Mujagic Z, Vork L, Tigchelaar EF, Jankipersadsing SA, Cenit MC, Harmsen HJ, Dijkstra G, Franke L, Xavier RJ, Jonkers D, Wijmenga C, Weersma RK, Zhernakova A (2016) Proton pump inhibitors affect the gut microbiome. Gut 65(5):740–748. https://doi.org/10.1136/gutjnl-2015-310376
Lankelma JM, van Vught LA, Belzer C, Schultz MJ, van der Poll T, de Vos WM, Wiersinga WJ (2017) Critically ill patients demonstrate large interpersonal variation in intestinal microbiota dysregulation: a pilot study. Intensive Care Med 43(1):59–68. https://doi.org/10.1007/s00134-016-4613-z
Leitner DF, Siu Y, Korman A, Lin Z, Kanshin E, Friedman D, Devore S, Ueberheide B, Tsirigos A, Jones DR, Wisniewski T, Devinsky O (2023) Metabolomic, proteomic, and transcriptomic changes in adults with epilepsy on modified Atkins diet. Epilepsia. https://doi.org/10.1111/epi.17540
Li Q, Wang C, Tang C, He Q, Zhao X, Li N, Li J (2015) Successful treatment of severe sepsis and diarrhea after vagotomy utilizing fecal microbiota transplantation: a case report. Crit Care 19:37. https://doi.org/10.1186/s13054-015-0738-7
Ma C, Xia R, Yang S, Liu L, Zhang J, Feng K, Shang Y, Qu J, Li L, Chen N, Xu S, Zhang W, Mao J, Han J, Chen Y, Yang X, Duan Y, Fan G (2020) Formononetin attenuates atherosclerosis via regulating interaction between KLF4 and SRA in apoE(-/-) mice. Theranostics 10(3):1090–1106. https://doi.org/10.7150/thno.38115
Marin L, Miguelez EM, Villar CJ, Lombo F (2015) Bioavailability of dietary polyphenols and gut microbiota metabolism: antimicrobial properties. Biomed Res Int 2015:905215. https://doi.org/10.1155/2015/905215
McDonald D, Ackermann G, Khailova L, Baird C, Heyland D, Kozar R, Lemieux M, Derenski K, King J, Vis-Kampen C, Knight R, Wischmeyer PE (2016) Extreme Dysbiosis of the Microbiome in Critical Illness. mSphere 1(4). https://doi.org/10.1128/mSphere.00199-16
Ong SKL, Shanmugam MK, Fan L, Fraser SE, Arfuso F, Ahn KS, Sethi G, Bishayee A (2019) Focus on formononetin: anticancer potential and molecular targets. Cancers (Basel) 11(5). https://doi.org/10.3390/cancers11050611
Perler BK, Friedman ES, Wu GD (2023) The Role of the Gut Microbiota in the Relationship Between Diet and Human Health. Annu Rev Physiol 85:449–468. https://doi.org/10.1146/annurev-physiol-031522-092054
Pietrocola F, Castoldi F, Kepp O, Carmona-Gutierrez D, Madeo F, Kroemer G (2019) Spermidine reduces cancer-related mortality in humans. Autophagy 15(2):362–365. https://doi.org/10.1080/15548627.2018.1539592
Rooks MG, Garrett WS (2016) Gut microbiota, metabolites and host immunity. Nat Rev Immunol 16(6):341–352. https://doi.org/10.1038/nri.2016.42
Schulthess J, Pandey S, Capitani M, Rue-Albrecht KC, Arnold I, Franchini F, Chomka A, Ilott NE, Johnston DGW, Pires E, McCullagh J, Sansom SN, Arancibia-Carcamo CV, Uhlig HH, Powrie F (2019) The Short Chain Fatty Acid Butyrate Imprints an Antimicrobial Program in Macrophages. Immunity 50(2):432-445 e437. https://doi.org/10.1016/j.immuni.2018.12.018
Sender R, Fuchs S, Milo R (2016) Are We Really Vastly Outnumbered? Revisiting the Ratio of Bacterial to Host Cells in Humans. Cell 164(3):337–340. https://doi.org/10.1016/j.cell.2016.01.013
Shimizu K, Ogura H, Hamasaki T, Goto M, Tasaki O, Asahara T, Nomoto K, Morotomi M, Matsushima A, Kuwagata Y, Sugimoto H (2011) Altered gut flora are associated with septic complications and death in critically ill patients with systemic inflammatory response syndrome. Dig Dis Sci 56(4):1171–1177. https://doi.org/10.1007/s10620-010-1418-8
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche JD, Coopersmith CM, Hotchkiss RS, Levy MM, Marshall JC, Martin GS, Opal SM, Rubenfeld GD, van der Poll T, Vincent JL, Angus DC (2016) The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 315(8):801–810. https://doi.org/10.1001/jama.2016.0287
Suez J, Zmora N, Zilberman-Schapira G, Mor U, Dori-Bachash M, Bashiardes S, Zur M, Regev-Lehavi D, Ben-Zeev Brik R, Federici S, Horn M, Cohen Y, Moor AE, Zeevi D, Korem T, Kotler E, Harmelin A, Itzkovitz S, Maharshak N, Shibolet O, Pevsner-Fischer M, Shapiro H, Sharon I, Halpern Z, Segal E, Elinav E (2018) Post-Antibiotic Gut Mucosal Microbiome Reconstitution Is Impaired by Probiotics and Improved by Autologous FMT. Cell 174(6):1406-1423 e1416. https://doi.org/10.1016/j.cell.2018.08.047
Taur Y, Jenq RR, Perales MA, Littmann ER, Morjaria S, Ling L, No D, Gobourne A, Viale A, Dahi PB, Ponce DM, Barker JN, Giralt S, van den Brink M, Pamer EG (2014) The effects of intestinal tract bacterial diversity on mortality following allogeneic hematopoietic stem cell transplantation. Blood 124(7):1174–1182. https://doi.org/10.1182/blood-2014-02-554725
Wang G, Liu H, Liu Y, Li H, Li Z, Shao G, Lv X (2020) Formononetin alleviates Streptococcus suis infection by targeting suilysin. Microb Pathog 147:104388. https://doi.org/10.1016/j.micpath.2020.104388
Xie J, Wang H, Kang Y, Zhou L, Liu Z, Qin B, Ma X, Cao X, Chen D, Lu W, Yao C, Yu K, Yao X, Shang H, Qiu H, Yang Y, Investigators C.H.E.S.o.S.S (2020) The Epidemiology of Sepsis in Chinese ICUs: A National Cross-Sectional Survey. Crit Care Med 48(3):e209–e218. https://doi.org/10.1097/CCM.0000000000004155
Yi, Cui J, Wang W, Tang W, Teng F, Zhu X, Qin J, Wuniqiemu T, Sun J, Wei Y, Dong J (2020) Formononetin Attenuates Airway In fl ammation and Oxidative Stress in Murine Allergic Asthma. Front Pharmacol 11:533841. https://doi.org/10.3389/fphar.2020.533841
Zaborin A, Smith D, Garfield K, Quensen J, Shakhsheer B, Kade M, Tirrell M, Tiedje J, Gilbert JA, Zaborina O, Alverdy JC (2014) Membership and behavior of ultra-low-diversity pathogen communities present in the gut of humans during prolonged critical illness. mBio 5(5):e01361-01314. https://doi.org/10.1128/mBio.01361-14
Funding
This work was supported by the National Natural Science Foundation of China (No. 81770276, U20A20366).
Author information
Authors and Affiliations
Contributions
All authors participated in the design and interpretation of the studies, the analysis of the data and the review of the manuscript. Kaijiang Yu, Changsong Wang and Xiaohui Ma designed the experiments. Xiaohui Ma, Xiaonan Jia, Yahui Peng and Xueting Li conducted the experiments. Xiaohui Ma analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Ethical statement
Experiments were performed under a project license (NO. KY2018-02) granted by regional ethics board of Harbin Medical University Cancer Hospital on December 2018, in compliance with chinese guidelines for the care and use of animals. The chairperson of the ethics committee is Changhong Zhao.
Competing interests
The authors declare no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ma, X., Jia, X., Peng, Y. et al. Gut microbiota disruption during sepsis and the influence of innate metabolites on sepsis prognosis. Int Microbiol 26, 929–938 (2023). https://doi.org/10.1007/s10123-023-00349-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10123-023-00349-x