Abstract
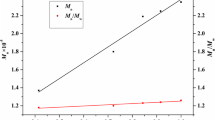
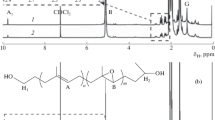
Herein, we demonstrate the synthesis of a well-defined diblock copolymer consisting of isotactic polystyrene (iPS) and linear polyethylene, isotactic polystyrene-block-polyethylene (iPS-b-PE), by the combination of sequential monomer addition and hydrogenation. Isospecific living polymerization of styrene and living trans-1,4-polymerization of 1,3-butadiene were catalyzed by 1,4-dithiabutandiyl-2,2′-bis(6-cumenyl-4-methylphenoxy) titanium dichloride (complex 1) activated by triisobutyl aluminum modified methylaluminoxane (MMAO) at room temperature to provide highly isotactic polystyrene (iPS) and 1,4-trans-polybutadiene (1,4-trans-PBD) with narrow molecular weight distribution. Furthermore, the iPS-b-1,4-trans-PBD was synthesized via sequential monomer addition in the presence of complex 1 and MMAO. The hydrogenation of the 1,4-trans-PBD block was promoted by RuCl2(PPh3)3 used as a catalyst to produce iPS-b-PE.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Park, C., Yoon, J. and Thomas, E.L., Polymer, 2003, 44: 6725
Jin, K.K., Yang, S.Y., Lee, Y. and Kim, Y., Prog. Polym. Sci., 2010, 35: 1325
Kim, H.J., Kim, J.H., Ryu, J.H., Kim, Y., Kang, H., Lee, W.B., Kim, T.S. and Kim, B.J., ACS Nano, 2014, 8: 10461
Pandav, G. and Ganesan, V., Macromolecules, 2015, 73: 16
Fujita, H., Michinobu, T., Fukuta, S., Koganezawa, T. and Higashihara, T., ACS Appl. Mater. Interfaces, 2016, 8: 5484
Wu, M., Wu, Z., Wang, K., Zhang, Q. and Fu, Q., Polymer, 2014, 55: 6409
Wu, H., Jiang, T., Zhu, J., Cui, K., Zhao, Q. and Ma, Z., J. Polym. Sci., Part A: Polym. Chem., 2015, 8: 341
Chen, L., Zhang, C., Du, Z., Li, H., Zhang, L. and Wei, Z., RSC Adv., 2015, 5: 65184
Raynaud, J., Liu, N., Gnanou, Y. and Taton, D., Polym. Chem., 2011, 2: 1706
Wu, Z.Q., Chen, Y., Wang, Y., He, X.Y., Ding, Y.S. and Liu, N., Chem. Commun., 2013, 49: 8069
Yang, J.X., Long, Y.Y., Pan, L., Men, Y.F. and Li, Y.S., ACS Appl. Mater. Interfaces, 2016, 8: 12445
Wu, J., Jiang, H., Zhang, L., Cheng, Z. and Zhu, X., Polym. Chem., 2016, 7: 2486
Tanaka, S., Goseki, R., Ishizone, T. and Hirao, A., Macromolecules, 2014, 47: 934
Ren, Y., Wei, Z., Leng, X., Wang, Y. and Li, Y., Polymer, 2015, 78: 51
Todd, A.D. and Bielawski, C.W., ACS Macro Lett., 2015, 4: 1254
Anastasaki, A., Waldron, C., Wilson, P., Boyer, C., Zetterlund, P.B., Whittaker, M.R. and Haddleton, D., ACS Macro Lett., 2013, 2: 896
Goseki, R., Onuki, S., Tanaka, S., Ishizone, T. and Hirao, A., Macromolecules, 2015, 48: 3230
Leone, G., Polym. Chem., 2012, 3: 1987
Kima, D.G., Bell, A. and Register, R.A., ACS Macro Lett., 2015, 4: 327
Capacchione, C., Saviello, D., Ricciardi, R. and Proto, A., Macromolecules, 2011, 44: 7940
Kobayashi, E., Hayashi, N., Aoshima, S. and Furukawa, J., J. Polym. Sci., Part. A: Polym. Chem., 1998, 36: 241
Kaita, S., Hou, Z. and Wakatsuki, Y., Macromolecules, 2001, 34: 1539
Monteil, V., Spitz, R. and Boisson, C., Polym. Int., 2004, 53: 576
Ventura, A., Chenal, T., Bria, M., Bonnet, F., Zinck, P., Ravache, Y.N., Balanzat, E. and Visseaux, M., Eur. Polym. J., 2013, 49: 4130
Jian, Z., Tang, S. and Cui, D., Chem. Eur. J., 2010, 16: 14007
Proto, A., Avagliano, A., Saviello, D., Ricciardi, R. and Capacchione, C., Macromolecules, 2010, 43: 5919
Capacchione, C., Proto, A., Ebeling, H., Mulhaupt, R., Moller, K., Spaniol, T.P. and Okuda, J., J. Am. Chem. Soc., 2003, 125: 4964
Beckerle, K., Manivannan, R., Spaniol, T.P. and Okuda, J., Organometallics, 2006, 25: 3019
Buwalda, S.J., Amgoune, A. and Bourissou, D., J. Polym. Sci., Part. A: Polym. Chem., 2015, 54: 1222
Reuther, J.F., Bhatt, M.P., Tian, G., Batchelor, B.L., Campos, R. and Novak, B.M., Macromolecules, 2014, 47: 4587
Chen, Y.J., Wu, B.J., Wang, F.S., Chi, M.H., Chen, J.T. and Peng, C.H., Macromolecules, 2015, 48: 6832
Yang, Y. and Knauss, D.M., Macromolecules, 2015, 48: 4471
Stalmach, U., Boer, B.D., Videlot, C., Hutten, P.F.V. and Hadziioannou, G., J. Am. Chem. Soc., 2000, 122: 5464
Piunova, V.A., Horn, H.W., Jones, G.O., Rice, J.E. and Miller, R.D., J. Polym. Sci., Part A: Polym. Chem., 2016, 54: 563
Zhang, Z., Altaher, M., Zhang, H., Wang, D. and Hadjichristidis, N., Macromolecules, 2016, 49: 2630
Pottier, C., Morandi, G., Dulong, V., Souguir, Z., Picton, L. and Cerf, D.L., J. Polym. Sci., Part A: Polym. Chem., 2015, 53: 2606
Hu, Y., Darcos, V., Monge, S. and Li, S., Int. J. Pharmaceut., 2015, 491: 152
Alkayal, N., Polym. Chem., 2016, 7: 2986
Bellas, V. and Rehahn, M., Macromol. Chem. Phys., 2009, 210: 320
Hoyle, C.E. and Bowman, C.N., Angew. Chem. Int. Ed., 2010, 49: 1540
Tsuchiya, K., Ando, K., Shimomura, T. and Ogino, K., Polymer, 2016, 92: 125
Park, J., Lee, K.S., Choi, C., Kwak, J., Moon, H.C. and Kim, J.K., Macromolecules, 2016, 49: 3647
Pessoni, L., Winter, J.D., Surin, M., Hergué, N., Delbosc, N., Lazzaroni, R., Dubois, P., Gerbaux, P. and Coulembier, O., Macromolecules, 2016, 49: 3001
Guillaume, G., Roberts, D.A., Thomas, M. and Sébastien, P., J. Am. Chem. Soc., 2016, 138: 4061
Xu, J., Ye, J. and Liu, S., Macromolecules, 2007, 40: 203
Soeriyadi, A.H., Boyer, C., Nyström, F., Zetterlund, P.B. and Whittaker, M.R., J. Am. Chem. Soc., 2011, 133: 11128
Hansell, C.F., Espeel, P., Stamenovi, M.M., Barker, I.A., Dove, A.P., Prez, F.E.D. and O’Reilly, R.K., J. Am. Chem. Soc., 2011, 133: 13828
Li, T., Wang, W.J., Liu, R., Liang, W.H., Zhao, G.F., Li, Z.Y., Wu, Q. and Zhu, F.M., Macromolecules, 2009, 42: 3804
Li, Z.Y., Liu, R., Mai, B.Y., Feng, S., Wu, Q., Liang, G.D., Gao, H.Y. and Zhu, F.M., Polym. Chem., 2013, 4: 954
Ferrer, M.D.P. and Calle, J.A.B., 1996, U.S. Pat., 5,583,185
Chamberlain, L.R. and Gibler, C.J., 1991, U.S. Pat., 5,039,755
Gibler, C.J. and Wilson, S.E., 1993, U.S. Pat., 5,244,980
Mohammadi, N.A. and Rempel, G.L., J. Mol. Catal., 1989, 50: 259
Guo, X. and Rempel, G.L., J. Mol. Catal., 1990, 63: 279
Guo, X., Scott, P.J. and Rempel, G.L., J. Mol. Catal., 1992, 72: 193
Hahn, S.F., J. Polym. Sci., Part A: Polym. Chem., 1992, 30: 397
Petzetakis, N., Stone, G.M. and Balsara, N.P., Macromolecules, 2014, 47: 4151
Caporaso, L., Izzo, L., Sisti, I. and Oliva, L., Macromolecules, 2002, 35: 4866
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was financially supported by the National Natural Science Foundation of China (Nos. 21174167 and 51573212) and the Natural Science Foundation of Guangdong Province (Nos. S2013030013474 and 2014A030313178).
Electronic supplementary material
10118_2017_1933_MOESM1_ESM.pdf
Synthesis of Isotactic Polystyrene-block-Polyethylene by the Combination of Sequential Monomer Addition and Hydrogenation of 1,4-Trans-polybutadiene Block
Rights and permissions
About this article
Cite this article
Liang, Hq., Zhou, Qh., Long, Yj. et al. Synthesis of isotactic polystyrene-block-polyethylene by the combination of sequential monomer addition and hydrogenation of 1,4-trans-polybutadiene block. Chin J Polym Sci 35, 866–873 (2017). https://doi.org/10.1007/s10118-017-1933-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-017-1933-6