Abstract
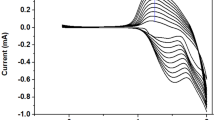
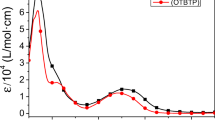
Two novel decyloxyphenylquinoxaline-based donor-acceptor (D-A) electroactive monomers bearing dialkoxythiophene as the donor unit are synthesized using Stille coupling reaction. The corresponding polymers, poly[2,3-bis(4-decyloxyphenyl)-5,8-bis(3,4-dimethoxylthiophen-2-yl)quinoxaline] (P1) and poly[2,3-bis(4-decyloxyphenyl)-5,8-bis(2,3-dihydrothieno[3,4-b][1,4]dioxin-5-yl)quinoxaline] (P2), are directly deposited onto the working electrode surface by electropolymerization. All materials were characterized by nuclear magnetic resonance (NMR), mass spectrometry (MS), scanning electron microscopy (SEM), cyclic voltammetry (CV), ultraviolet-visible absorption spectrometry (UV-Vis) and spectro-electrochemical measurements. Electrochemical studies demonstrate that both polymers are capable of showing both reasonable n- and p-doping processes, and advanced long-term switching stabilities. 3,4-Ethylenedioxythiophene substituted for 3,4-dimethoxythiophene as a donor unit, which enhances the conjugated double-bond character of the conducting polymer, thus leading to a lower electronic band-gap. Likewise, the neutral state color of the synthesized polymer tuned from blue to blue-green corresponding to the red shift of the maximum absorption wavelengths in the visible region. In addition, kinetics study of P1 revealed 42% (595 nm), 30% (839 nm) and 69% (1500 nm) transmittance changes (ΔT%), while P2 exhibited 32% (740 nm), 71% (2000 nm) at the dominant wavelengths. It was also observed that both films could switch quickly between the neutral state and oxidation state, with the response time less than 1 s both in visible and near infrared regions.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Garcia, A., Bakus, R.C., Zalar, P., Hoven, C.V.J., Brzezinski, Z. and Nguyen, T.Q., J. Am. Chem. Soc., 2011, 133: 2492
Beaujuge, P.M., Pisula, W., Tsao, H.N., Ellinger, S., Müllen, K. and Reynolds, J.R., J. Am. Chem. Soc., 2009, 131: 7514
Renuga, D., Udhayakumari, D., Suganya, S. and Velmathi, S., Tetrahedron Lett., 2012, 53: 5068
McQuade, D.T., Pullen, A.E. and Swager, T.M., Chem. Rev., 2000, 100: 2537
Nicho, M.E., Hailin, H., López-Mata, C. and Escalante, J., Sol. Energ. Mater. Sol. C., 2004, 82: 105
Sefer, E., Koyuncu, F.B., Oguzhan, E. and Koyuncu, S., J. Polym. Sci., Part A: Polym. Chem., 2010, 48: 4419
Zoombelt, A.P., Fonrodona, M., Turbiez, M.G.R., Wienk, M.M. and Janssen, R.A.J., J. Mater. Chem., 2009, 19: 5336
Kim, J., Cho, N., Ko, H.M., Kim, C., Lee, J.K. and Ko, J., Sol. Energ. Mater. Sol. C., 2012, 102: 159
Ramya, R., Sivasubramanian, R. and Sangaranarayanan, M.V., Electrochim. Acta, 2013, 101: 109
Walker, W., Veldman, B., Chiechi, R., Patil, S., Bendikov, M. and Wudl, F., Macromolecules., 2008, 41: 7278
Persson, N.K., Sun, M., Kjellberg, P., Pullerits, T. and Inganäs, O., J. Chem. Phys., 2005, 123: 204718
Sakthivel, P., Song, H.S., Chakravarthi, N., Lee, J.W., Gal, Y.S., Hwang, S. and Jin, S.H., Polymer, 2013, 54: 4883
Zoombelt, A.P., Leenen, M.A.M., Fonrodona, M., Nicolas, Y., Wienk, M.M. and Janssen, R.A.J., Polymer, 2009, 50: 4564
Cihaner, A. and Algi, F., Adv. Funct. Mater., 2008, 18: 3583
Xu, Z., Wang, M., Zhao, J.S., Cui, C.S., Fan, W.Y. and Liu, J.F., Electrochim. Acta, 2014, 125: 241
Xu, Z., Wang, M., Fan, W.Y., Zhao, J.S. and Wang, H.S., Electrochim. Acta, 2015, 160: 271
Hou, Y.F., Xu, G.Q., Zhao, J.S., Kong, Y. and Yang, C., Acta. Chim. Sinica, 2014, 72: 1238
Groenendaal, L., Zotti, G., Aubert, P.H., Waybright, S.M. and Reynolds, J.R., Adv. Mater., 2003, 15: 855
Kvarnstrom, C., Neugebauer, H., Blomquist, S., Ahonen, H.J., Kankare, J. and Ivaska, A., Electrochim. Acta, 1999, 44: 2739
Gunbas, G.E., Durmus, A. and Toppare, L., Adv. Funct. Mater., 2008, 18: 2026
Sonmez, G., Schwendeman, I., Schottland, P., Zong, K. and Reynolds, J.R., Macromolecules, 2003, 36: 639
Heywang, G. and Jonas, F., Adv. Mater., 1992, 4: 116
Hanna, R. and Leclerc, M., Chem. Mater., 1996, 8: 1512
Sonmez, G., Sonmez, H.B., Shen, C.K.F., Jost, R.W., Rubin, Y. and Wudl, F., Macromolecules, 2005, 38: 669
Li, M., Sheynin, Y., Patra, A. and Bendikov, M., Chem. Mater., 2009, 21: 2482
Tsubata, Y., Suzuki, T., Miyashi, T. and Yamashita, Y., J. Org. Chem., 1992, 57: 6749
Zhao, H., Wei, Y.Y., Zhao, J.S. and Wang, M., Electrochim. Acta, 2014, 146: 231
Zhu, S.S. and Swager, T.M., J. Am. Chem. Soc., 1997, 119: 12568
Mohr, B., Enkelmann, V. and Wegner, G., J. Org. Chem., 1994, 59: 635
Berlin, A., Zotti, G., Zecchin, S., Schiavon, G., Vercelli, B. and Zanelli, A., Chem. Mater., 2004, 16: 3667
DuBois, C.J., Abboud, K.A. and Reynolds, J.R., J. Phys. Chem. B., 2004, 108: 8550
DuBois, C.J. and Reynolds, J.R., Adv. Mater., 2002, 14: 1844
Ozkut, M.I., Atak, S., Onal, A.M. and Cihaner, A., J. Mater. Chem., 2011, 21: 5268
Jespersen, K.G., Beenken, W.J.D., Zaushitsyn, Y., Yartsev, A., Andersson, M.R., Pullerits, T. and Sundstrom, V., J. Chem. Phys., 2004, 121: 12613
Zhao, H., Tang, D.D., Zhao, J.S., Wang, M. and Dou, J.M., RSC Adv., 2014, 4: 61537
Deepa, M., Awadhia, A. and Bhandari, S., Phys. Chem. Chem. Phys., 2009, 11: 5674
Bechinger, C., Burdis, M.S. and Zhang, J.G., Solid State Commun., 1997, 101: 753
McDonagh, A.M., Bayly, S.R., Riley, D.J., Ward, M.D., McCleverty, J.A., Cowin, M.A., Morgan, C.N., Varrazza, R., Penty, R.V. and White, I.H., Chem. Mater., 2000, 12: 2523
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was financially supported by the National Natural Science Foundation of China (Nos. 51473074 and 31400044), the General and Special Program of the Postdoctoral Science Foundation China (Nos. 2013M530397 and 2014T70861), the Postgraduate Innovation Project of China University of Petroleum (East China) (No. YCX2015022) and the Fundamental Research Funds for the Central Universities (No. 15CX06049A).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Xu, Z., Kong, Lq., Zhao, Js. et al. Decyloxyphenyl-substituted quinoxaline-embedded conjugated electrochromic polymers with high switching stability and fast response speed. Chin J Polym Sci 34, 407–419 (2016). https://doi.org/10.1007/s10118-016-1759-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-016-1759-7