Abstract
Laser therapy has been widely used to treat port-wine stains (PWS) and other cutaneous vascular lesions via selective photothermolysis. Animal models are a valuable tool for investigating thermal responses beneath the skin. However, in previous animal experiments, such as the dorsal skin chamber model, one side of the skin was removed, resulting in the loss of mechanical support for the target blood vessel. In this study, the optical clearing technique was applied to the dorsal skin, allowing direct observation of real thermal responses within the tissue without removing the covering skin. The target blood vessels were irradiated with a pulsed 1064 nm Nd: YAG laser. The corresponding thermal responses were recorded using a CCD camera. Additionally, variations in skin reflectance spectra were measured before and after laser irradiation. Due to the optical clearing and reflectance spectra measurement, vessel responses such as contraction, reperfusion, and full occlusion were correlated with specific variation patterns in reflectance spectral signals.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Laser thermal therapy based on selective photothermolysis (SP) is a standard treatment for skin vascular anomalies, such as port-wine stains (PWS) and other vessel-related dermatological disorders [1]. Currently, the pulsed dye laser (PDL) with a wavelength of 585–595 nm is the gold standard for PWS treatment in the clinic [2]. However, complete lesion clearance remains less than 20% [3]. Many patients demonstrate resistance to laser irradiation [4, 5], and recurrence occurs in the clinic [1]. Resistant PWS may be lightened using an alternative laser light with a longer wavelength that can penetrate deeper into the skin. Nd: YAG lasers with a 1,064 nm wavelength may treat deeply buried, resistant blood vessels due to their deep penetration (up to 5–6 mm) [6]. Clinical trials of vascular lesion treatment with Nd: YAG lasers seem promising [5, 7]. In our previous study, we showed that blood vessels are more prone to complete contraction and disappearance with a 1,064 nm laser rather than rupture induced by PDL [8]. However, side effects such as scarring have also been reported [5,6,7,8]. Further studies on the thermal responses of blood vessels to Nd: YAG lasers are needed.
Animal experimental studies using skin histological analysis [9,10,11,12], the chick chorioallantoic membrane model [13,14,15], and the dorsal skin chamber (DSC) model [16,17,18] are promising methods for investigating the thermal responses of blood vessels to laser irradiation. Among these, the DSC has been widely adopted in vivo because it can provide a relevant range of blood vessels (30–300 μm) in PWS lesions and enable serial imaging for a relatively long period. Based on our previous study, we identified that a strong thermal-mechanical interaction occurs within target blood vessels during and after laser irradiation [8]. However, in the DSC model, one side of the dorsal skin is removed to make the buried blood vessels observable. As a result, the uncovered blood vessels lose their mechanical support from the original skin covering. Therefore, the thermal response observed in the DSC model may differ from that in the real skin situation.
The quality of biomedical optical imaging can be enhanced using the optical clearing technique. By introducing hyperosmotic and high-refractive optical clearing agents (OCAs) into the dermis, scattering is reduced, and light penetration depth is increased by matching the refractive indexes of visible components. Intradermal injection of OCAs is a common and effective method for improving optical clearing in vivo [19].
Skin reflectance spectra can provide valuable quantitative information about blood vessel thermal responses under laser irradiation. In our previous study [20], we used a reflectance spectrometer to measure skin reflectance spectra before and after laser irradiation. The generation of thermal coagula and subsequent thrombus formation were accurately detected through reflectance spectra measurement. We also introduced a new repeated multi-pulse laser irradiation protocol to enhance vessel response to laser heating at low incident laser fluence.
In this study, a visualization experiment was conducted to investigate the thermal responses of blood vessels to laser irradiation. We applied the optical clearing technique to the dorsal skin without removing it, allowing direct observation of blood vessel thermal responses with skin mechanical support. An Nd: YAG laser was used as the power source in the experiments. After each laser irradiation, the immediate effects on the blood vessels were examined. The skin reflectance spectra were recorded before, during, and after laser irradiation to illustrate variations in vessel thermal responses.
Experimental materials and methods
In vivo animal model
The SD mice (initial bodyweight, 100–120 g) were obtained from the animal center of the medical school at Xi’an Jiaotong University and were housed in individual cages with free access to food and water. The dorsal skin chamber could provide the relevant range of blood vessels in PWS lesions. In this study, a total of six blood vessels were selected and treated. Their original diameters ranged from 90 μm to 100 μm, which is within the range of the typical diameter of real port-wine stain blood vessels.
Skin optical cleaning
Before conducting the experiment, the mice were intraperitoneally anesthetized with 10% chloral hydrate, and 8% NaS was applied to the mouse fur for 1–2 min to facilitate hair removal. The mouse’s back was cleaned with water and allowed to dry for 15 min after depilation. The optical agent was made using thiazone and PEG400. The thiazone (99% purity, Wuhan Hengwo Technology Co., Ltd., China) was heated and melted in a water bath at a temperature of 40 °C. The chemical reagent PEG400 (analytical reagent, Tianjin Kermel Chemical Reagent Co., Ltd., China) was then mixed with the melted thiazone at a volume ratio of 9:1. The optical agent was applied to the mouse’s back skin, left on for 10 min, and then removed to eliminate specular reflection. The reflectance spectra of the mouse’s back skin were measured before and after optical clearing to verify the effect of the optical clearing. All procedures related to animal experimentation were ethically approved by the Institutional Animal Care and Use Committee of Xi’an Jiaotong University (No. 2022 − 1152).
Laser and visualization system
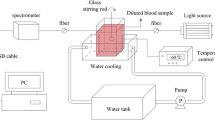
An Nd: YAG laser (WON-COSJET TR, WON Technology Co., Ltd.) was used to emit a 1064 nm laser, which was delivered via handpieces that projected a uniform circular beam with a 2 mm diameter. The energy fluence (35–57 J/cm²) and pulse duration (0.3 ms) were controlled. The thermal response of vessels was visualized using a CCD camera (DP25, Olympus Corp., Japan) or a high-speed camera system (Redlake HG-100-k; USA; resolution, 1504 × 1128; frame frequency, 25–2000 fps). The eyepiece objectives of the microscope included 10x magnifications, and the resolution of the CCD was 2560 × 1920 pixels. When the mouse was placed on the microscope platform after optical clearing, microcirculation could be observed.
Spectral reflectance measurement for dorsal skin
A spectrometer (HR400 CG–UV–NIR; measurement range, 400–900 nm), optical fiber, and light source (LS–1 tungsten halogen lamp) were used to record the spectral reflectance of the dorsal skin before and after laser irradiation.
Experimental results and discussion
Figure 1 shows the blood vessels before (a) and after (b) Nd: YAG laser irradiation with an incident energy of 35 J/cm², along with the corresponding measured reflectance spectra (c). As we can see from the figure, almost no thermal response is observed due to the relatively low incident energy of 35 J/cm² used. The corresponding reflectance spectra measured from the dorsal skin before and after Nd: YAG laser irradiation are also shown in Fig. 1c. The variations in reflectance spectra before and after laser irradiation were minimal, indicating that blood content remained essentially unchanged, and there was no alteration in blood vessel diameter. This corresponds to cases of ineffective treatment in clinical therapy.
Figure 2 shows the blood vessels before (a), immediately after (b), and 15 min after (c) Nd: YAG laser irradiation with an incident energy of 43 J/cm², along with the corresponding measured reflectance spectra (d). The blood vessel diameters were measured using Adobe Photoshop CC 2016. After laser irradiation, the major blood vessel undergoes partial contraction. The diameter decreases significantly, but complete occlusion of the vessel does not occur. Fifteen minutes after laser irradiation, a slight expansion of the irradiated blood vessel is observed. Compared to before irradiation, the vessel diameter increased from 95.8 μm to 105.6 μm, resulting in a radius increase of 10.2%. Figure 2d shows the reflectance spectra measured from the dorsal skin before and after Nd: YAG laser irradiation with an incident energy of 43 J/cm². The skin reflectance initially decreases, then increases, and ultimately stabilizes at a level close to that before irradiation.
Figure 3 shows the blood vessels before (a), immediately after (b), 10 min after (c), and 15 min after (d) Nd: YAG laser irradiation with an incident energy of 52 J/cm², along with the corresponding measured reflectance spectra (e). One of the major blood vessels and capillaries on the right side of the image is subjected to laser irradiation, while the left side remains unaffected (Fig. 3a). After irradiation, the main blood vessel showed significant thrombus formation and strong contraction, leading to the occlusion of the vessel. The average diameter of the heated vessels decreased from 91.2 μm to 18.2 μm (Fig. 3b). Some parts of the vessel lumen constricted to almost disappear. Ten minutes after laser irradiation, part of the thrombus disappeared, and blood flow began to recover (Fig. 3c). Fifteen minutes after laser irradiation, part of the vessel lumen recovered due to blood circulation (Fig. 3d). Subsequently, reflectance spectra measurements were performed on the mouse skin, and the results are illustrated in Fig. 3e. The reflectance of the skin after laser irradiation initially decreased and then increased. In this set of cases, the blood vessels initially formed small thrombi, leading to vessel occlusion and blocking the flow of blood in the vessels. Blood accumulated at the two ends of the occlusion, causing an increase in blood content and a decrease in reflectance. Five minutes after irradiation, the reflectance reached its lowest value. Between 5 and 15 min, part of the thrombus disappeared, and the vessels re-perfused, leading to a continuous increase in reflectance.
Maintaining the beam diameter and pulse width constant, the incident energy was further increased to 57 J/cm². Figure 4 shows the blood vessels before (a), immediately after (b), 10 min after (c), and 15 min after (d) Nd: YAG laser irradiation with an incident energy of 57 J/cm², along with the corresponding measured reflectance spectra (e). After laser irradiation, the main blood vessel underwent significant contraction, while the two branching blood vessels remained unchanged. A segment of the blood vessel constricted to almost disappear. The average diameter decreased from 91.21 μm to 76.65 μm. A thrombus formed at one end of the occlusion, blocking the blood flow (Fig. 4b). Ten and fifteen minutes after laser irradiation, the irradiated portion of the main vessel remained fully contracted, leaving only the closed vessel wall (Fig. 4c and d). Figure 4e shows the reflectance spectra measured from the dorsal skin before and after Nd: YAG laser irradiation with an incident energy of 57 J/cm². The reflectance of the occluded blood vessel tissue continuously decreased. Due to the occlusion of the main blood vessel and continuous formation of the thrombus, there was an increase in blood absorption in the skin and a decrease in reflectance.
We summarized the variation in reflectance spectra at 577 nm before and after Nd: YAG laser irradiation with different incident energies in Fig. 5. As shown in the figure, some interesting results can be observed. With low incident energy, the target blood vessel shows almost no response, and its reflectance remains nearly unchanged. With higher incident energy, the target blood vessel shows obvious thermal responses but is not completely occluded; its reflectance decreases initially and then increases. The reflectance spectra of fully occluded blood vessels consistently decrease after laser irradiation. This is because thrombus formation and accumulation continue within the fully occluded blood vessels after effective laser heating. The thrombus strongly absorbs light, causing the reflectance spectra to decrease [19]. In cases of ineffective heating, the blood vessel re-perfuses and thrombus cannot accumulate, resulting in a slight increase in the corresponding reflectance spectra shortly after laser irradiation. In real skin without optical clearing, the reflectance spectra signal may be weaker than with optical clearing, but the specific variations will not change. Therefore, the trend presented in Fig. 5 can be used as a criterion to determine effective treatment using reflectance spectra measurement, assisting doctors in clinical practice.
In this paper, we focused on optical imaging and spectral measurement on the dorsal skin in vivo. The in vitro histological analysis was not performed, which is a limitation of the present study. The histological analysis would provide valuable information on the thermal responses within and surrounding the vessel lumen, including thrombus formation and potential vessel wall coagulation, which may further support the findings of this study.
As a preliminary study, there are several other limitations. The differences in thermal responses between various laser parameters, such as wavelength, pulse duration, spot size, and repetition rate, have not been carefully examined. The experiment was only conducted on optically cleared dorsal skin. Clinical measurements on PWS patients have not been conducted and compared with animal experiments. Only spectral reflectance measurements were conducted. Other methods, such as infrared imaging, can be applied simultaneously to provide more information on blood vessel thermal responses. All of the above concerns will be evaluated in our further study.
Conclusion
In this study, multiple animal experiments were conducted at different incident energy levels after skin optical clearing. Under lower incident energy conditions (35 J/cm²), there were no significant changes in the target blood vessels. As the incident energy increased, three phenomena occurred: (1) blood vessel contraction followed by recovery (43 J/cm²); (2) vessel fully contracted followed by recovery (52 J/cm²); (3) vessel fully occluded after thrombus formation (57 J/cm²). In all cases, the reflectance spectra of the skin significantly decreased immediately after laser irradiation, followed by an increase if the vessel lumen was not fully occluded. Only when the vessel lumen was fully occluded and no reperfusion occurred, which is the desired vessel response in the clinic, did the reflectance spectra continue to decrease even several minutes after laser irradiation.
References
Kelly KM, Choi B, McFarlane S et al (2005) Description and analysis of treatments for port-wine stain birthmarks. Arch Facial Plast Surg 7(5):287–294. https://doi.org/10.1001/archfaci.7.5.287
Brightman LA, Geronemus RG, Reddy KK (2015) Laser treatment of port-wine stains. Clin Cosmet Investig Dermatol 8:27–23. https://doi.org/10.2147%2FCCID.S53118
Tierney EP, Hanke CW (2011) Alexandrite laser for the treatment of port wine stains refractory to pulsed dye laser. Dermatol Surg 37(9):1268–1278. https://doi.org/10.1111/j.1524-4725.2011.02079.x
Fölster-Holst R, Shukla R, Kassir M, Galadari H, Lotti T, Wollina U, Grabbe S, Goldust M (2021) Treatment Update of Port-Wine Stain: A Narrative Review. J Drugs Dermatol. ;20(5):515–518. https://doi.org/10.36849/JDD.5005. PMID: 33938700
Yang MU, Yaroslavsky AN, Farinelli WA, Flotte TJ, Rius-Diaz F, Tsao SS, Anderson RR (2005) Long-pulsed neodymium: yttrium-aluminum-garnet laser treatment for port-wine stains. J Am Acad Dermatol 52:480–490. https://doi.org/10.1016/j.jaad.2004.10.876
Dover JS (2000) New approaches to the laser treatment of vascular lesions. Australas J Dermatol 41:14–18. https://doi.org/10.1046/j.1440-0960.2000.00382.x
Groot D, Rao J, Johnston P, Nakatsui T (2003) Algorithm for using a long-pulsed nd:YAG laser in the treatment of deep cutaneous vascular lesions. Dermatol Surg 29:35–42. https://doi.org/10.1046/J.1524-4725.2003.29016.X
Li D, Farshidi D, Wang GX, He YL, Kelly KM, Wu WJ, Ying ZX (2014) A comparison of microvascular responses to visible and near-infrared lasers. Lasers Surg Med 46:479–487. https://doi.org/10.1002/lsm.22250
Tan OT, Morrison P, Kurban AK (1990) 585-nm for the treatment of port-wine stains. Plast Reconstr Surg 86(6):1112–1117. https://doi.org/10.1097/00006534-199012000-00011
Dai T, Pikkula BM, Tunnell JW, Chang DW, Anvari B (2003) Thermal response of human skin epidermis in different skin types to 595-nm laser irradiation and cryogen spray cooling: An ex-vivo study. Lasers in Surgery: Advanced Characterization, Therapeutics, and Systems XIII, SPIE Proceedings 4949:1–9. https://doi.org/10.1117/12.476134
Dai T, Diagaradjane P, Yaseen MA, Pikkula BM, Thomsen S, Anvari B (2005) Laser-induced thermal injury to dermal blood vessels: analysis of wavelength (585 nm vs. 595 nm), cryogen spray cooling, and wound healing effects. Lasers Surg Med 37(3):210–218. https://doi.org/10.1002/lsm.20217
Borges da Costa J, Boixeda P, Moreno C, Santiago J (2009) Treatment of resistant port-wine stains with a pulsed dual wavelength 595 and 1064 nm laser: a histochemical evaluation of the vessel wall destruction and selectivity. Photomed Laser Surg 27(4):599–605. https://doi.org/10.1111/j.1524-4725.2007.33197.x
Kimel S, Svaasand LO, Wilson MH, Schell MJ, Milner TE, Neslon JS, Berns MW (1994) Differential vascular response to laser photothermolysis. J Invest Dermatol 103(5):693–700. https://doi.org/10.1111/1523-1747.ep12398548
Kimel S, Svaasand LO, Cao D et al (2002) Vascular response to laser photothermolysis as a function of pulse duration, vessel type, and diameter: implications for port wine stain laser therapy. Lasers Surg Med 30(2):160–169. https://doi.org/10.1002/lsm.10016
Kimel S, Svaasand LO, Hammer-Wilson MJ, Nelson JS (2003) Influence of wavelength on response to laser photothermolysis of blood vessels: implications for port wine stain laser therapy. Lasers Surg Med 33(5):288–295. https://doi.org/10.1002/lsm.10224
Barton JK, Welch AJ, Izatt JA (1998) Investigating pulsed dye laser-blood vessel interaction with color Doppler optical coherence tomography. Opt Exp 3(6):251–256. https://doi.org/10.1364/OE.3.000251
Barton JK, Frangineas G, Pummer H et al (2001) Cooperative phenomena in two-pulse, two-color laser photocoagulation of cutaneous blood vessels. Photochem Photobiol 73(6):642–650. https://doi.org/10.1562/0031-8655(2001)0730642CPITPT2.0.CO2
Babilas P, Shafirstein G, Bäumler W, Baier J, Landthaler M, Szeimies R, Abels C (2005) Selective photothermolysis of blood vessels following flashlamp-pumped pulsed dye laser irradiation: in vivo results and mathematical modelling are in agreement. J Invest Dermatol 20(5):343–352. https://doi.org/10.1111/j.0022-202x.2005.23773.x
Liu X, Chen B (2019) In vivo experimental study on the enhancement of Optical Clearing Effect by Laser Irradiation in Conjunction with a chemical penetration enhancer. Appl Sci 9:3:542. https://doi.org/10.3390/app9030542
Li D, Wu W, Chen B et al (2020) Thermal coagulum formation and hemostasis during repeated multipulse nd:YAG laser treatment of cutaneous vascular lesions: animal experiment study. Lasers Med Sci 35:1589–1597. https://doi.org/10.1007/s10103-020-03007-0
Acknowledgements
Hong Zhang: animal experiments and manuscript writing; Yanjie Bian: skin reflectance spectra measurement; Wei Yin: animal experiments and data processing; Dong Li: Supervision the entire project. Zhaoxia Ying: Recommendation the typical clinic laser parameters used in clinic. Guarantee the clinic relevant of our study.
Funding
This study was funded by the National Natural Science Foundation of China (No. 52276084).
Author information
Authors and Affiliations
Contributions
Hong Zhang: animal experiments and manuscript writing; Yanjie Bian: skin reflectance spectra measurement; Wei Yin: animal experiments and data processing; Dong Li: Supervision the entire project. Zhaoxia Ying: Recommendation the typical clinic laser parameters used in clinic. Guarantee the clinic relevant of our study.
Corresponding authors
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Not applicable.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, H., Bian, Y., Yin, W. et al. A feasible way to explore real blood vessels thermal responses to laser irradiation by combing optical clearing and the reflectance spectra measurements: animal experiment study. Lasers Med Sci 39, 193 (2024). https://doi.org/10.1007/s10103-024-04142-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10103-024-04142-8