Abstract
This study aimed to investigate the effects and safety of 308 nm excimer laser (308 nm EL) and tacrolimus ointment (TO) in the treatment of facial vitiligo (FV). We searched Cochrane Library, PUBMED, EMBASE, CNKI, and WANGFANG from inception to June 1, 2023. Outcomes included overall response rate (ORR), total adverse reaction rate (TARR), recurrence rate at 3-month (RR-3) and recurrence rate at 6-month (RR-6). The outcome data were presented as odds ratios (OR) with 95% confidence intervals (CI). The risk of bias was assessed by Cochrane risk-of-bias tool and data analysis was performed by RevMan 5.4 software. This study included a total of 19 trials involving 2085 patients. When comparing 308 nm EL monotherapy with 308 nm EL plus TO, significant differences in the ORR (OR = 4.29, 95% CI [2.97, 6.19], I2 = 0%, P < 0.001), RR-3 (OR = 0.18, 95% CI [0.05, 0.69], I2 = 0%, P = 0.01), and RR-6 (OR = 0.38, 95% CI [0.14, 1.03], I2 = 39%, P = 0.06) were found between the two managements. When comparing TO monotherapy with TO plus 308 nm EL, its results showed significant differences in the ORR (OR = 4.21, 95% CI [2.90, 6.11], I2 = 0%, P < 0.001), TARR (OR = 0.42, 95% CI [0.22, 0.81], I2 = 4%, P = 0.009), and RR-3 (OR = 0.32, 95% CI [0.01, 8.03], P = 0.49) between the two modalities. The results of this study suggest that the combination of 308 nm EL and TO is more effective than either treatment alone for the treatment of FV.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Vitiligo is a chronic skin disorder that is characterized by the presence of depigmented patches on the skin [1, 2]. These patches are caused by the loss of melanocytes, which are responsible for producing melanin, in the skin and/or mucous membranes [3,4,5]. The exact mechanisms behind vitiligo are still not fully understood, but several risk factors have been identified, including an autoimmune association, heredity, and trigger events, such as stress, severe sunburn, and exposure to certain chemicals [6,7,8,9,10,11,12,13]. The prevalence of vitiligo varies among different populations, but it is estimated to affect approximately 0.5–1% of the global general population [14,15,16]. In some populations, this number can be as high as 2–3% [17, 18]. Vitiligo often presents in individuals below the age of 20 and typically develops before the age of 40 [3, 17, 18]. It affects both males and females equally and can have a significant negative impact on patients’ self-esteem and confidence, especially for those with facial vitiligo (FV) [1].
In the management of FV, several modalities have been utilized, including the 308 nm excimer laser (308 nm EL) and tacrolimus ointment (TO) [19,20,21,22,23,24,25,26]. The 308 nm EL is a type of phototherapy that delivers a specific wavelength of light, created by the dimerization of xenon and chlorine [27]. Research suggests that 308 nm EL monotherapy can benefit patients with vitiligo [28, 29], but its effects are often unsatisfactory and require a lengthy treatment course. Combining 308 nm EL with other therapies has been shown to produce better outcomes, highlighting the importance of combining modalities in the treatment of FV. TO, on the other hand, is a topical immunomodulatory agent that has shown promise as a treatment for vitiligo [30]. Studies have demonstrated that the combination of 308 nm EL and TO is more effective than either treatment alone [31, 32]. Previous clinical trials have examined the effects and safety of 308 nm EL and TO in individuals with FV [33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51]. However, there is a lack of systematic reviews and meta-analyses that comprehensively assess the efficacy and safety of this combined treatment approach. Therefore, the present study aimed to thoroughly and systematically evaluate the effects and safety of 308 nm EL and TO in the treatment of FV.
Materials and methods
Ethical approval statement
This study sought to gather secondary data from published articles and assessed their relevance to the research question. Therefore, no ethical approval is required in this study.
Eligibility criteria
To ensure the appropriateness of the included studies, certain eligibility criteria were established and applied. Firstly, patients diagnosed with FV, irrespective of their age or gender, were considered eligible for inclusion. In addition, only randomized controlled trials (RCTs) focusing on the treatment of FV using 308 nm EL or TO were included in the study. Furthermore, the treatment group consisted of patients receiving both 308 nm EL and TO, while the control group included patients receiving either 308 nm EL or TO.
On the other hand, certain studies were excluded based on the established exclusion criteria. Studies that did not meet the requirements of being RCTs, such as animal studies, case reports, reviews, conferences, non-clinical studies, and non-RCTs, were excluded. Additionally, studies that did not specifically investigate the use of 308 nm EL or TO for the treatment of FV were also excluded. Finally, studies that provided insufficient information, had inaccurate comparisons, involved combined therapies, or duplication were excluded to maintain the integrity of the data.
Search strategy for eligible records
To identify relevant and eligible studies, a comprehensive search strategy was employed. Electronic databases including Cochrane Library, PUBMED, EMBASE, CNKI, and WANGFANG were searched from their inception to June 1, 2023. Additional literature sources such as dissertations and reference lists of relevant reviews were also identified and assessed. The search strategy involved using specific retrieval terms related to topics such as 308 nm excimer laser, laser therapy, tacrolimus ointment, vitiligo, topical applications, and facial treatments.
Outcome measurement
The outcomes assessed in this study are multiple and comprehensive, including the overall response rate (ORR), the total adverse reaction rate (TARR), the recurrence rate at 3 months (RR-3), and the recurrence rate at 6 months (RR-6). These outcome measures were chosen to provide a comprehensive evaluation of the effects and safety of the intervention being studied.
Study selection, data collection, and risk of bias evaluation
Study selection, data collection, and risk of bias evaluation to ensure the reliability and accuracy of the study findings, a rigorous and systematic approach was employed. Two independent authors conducted the study selection, data collection, and risk of bias evaluation. Any disagreements or discrepancies were resolved through discussions between the two authors, and if necessary, a third author was consulted to reach a consensus. This meticulous process aimed to minimize any potential bias or errors in the study. Data collection was carried out using a pre-designed data collection form, which allowed for consistent and organized data recording. This ensured that all relevant information from the eligible trials was captured accurately and comprehensively. To evaluate the risk of bias in each eligible study, the Cochrane risk-of-bias tool was employed. This tool is a widely recognized and established tool for assessing the methodological quality and risk of bias in RCTs.
Statistical analysis
For data analysis, this study utilized RevMan 5.4 software, a commonly used statistical software package for conducting systematic reviews and meta-analyses. All outcome data were calculated as odds ratios (OR) and 95% confidence intervals (CI). The OR provides a measure of the association between the intervention and the outcomes of interest, while the CI indicates the range of values within which the true effect size is likely to fall. To assess heterogeneity across the included RCTs, the I2 index was used. This index quantifies the proportion of total variation across studies that is due to heterogeneity rather than chance. Finally, a fixed-effects model was employed to pool the outcome data, providing a summary measure of the intervention’s effectiveness across the included studies. This statistical approach ensured a robust and reliable analysis of the available evidence.
Results
Study selection
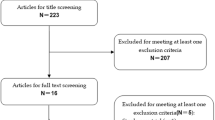
The study initially identified 127 records from electronic databases and other sources. After removing duplicates and irrelevant records, 33 full-text articles were assessed for eligibility. Finally, a total of 19 RCTs involving 2085 patients with FV were included for analysis (Fig. 1) [33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51].
Study characteristics
All eligible trials were conducted in China, except for one trial in France (Table 1). The sample sizes of the included RCTs ranged from 20 to 138 in the treatment group and from 20 to 131 in the control group. In all studies, the treatment group utilized 308 nm EL plus TO, while the control group used either 308 nm EL or TO. The follow-up visits for the patients ranged from seven to 24 weeks (Table 1).
Risk of bias assessment
The risk of bias analysis of the 19 included RCTs revealed that all studies provided sufficient information on random sequence generation, incomplete outcome data, selective reporting, and other forms of bias (Fig. 2) [33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51]. However, all the included studies failed to clearly report information on allocation concealment or blinding of participants, personnel, and outcome assessment (Fig. 2) [33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51].
nm EL plus TO vs. 308 nm EL
Meta-analysis of ORR
The meta-analysis included 12 studies with 1246 patients, and the results showed a statistically significant difference in ORR between the two groups (OR = 4.29, 95% CI [2.97, 6.19], I2 = 0%, P < 0.001; Fig. 3) [33,34,35, 38,39,40,41,42,43, 45, 47, 51].
Meta-analysis of TARR
Six RCTs involving 672 patients assessed the TARR. The meta-analysis results did not show a statistically significant difference between the two groups (OR = 0.68, 95% CI [0.40, 1.16], I2 = 0%, P = 0.16; Fig. 4A) [33,34,35, 39, 43, 47].
Meta-analysis of RR-3
Two trials involving 161 patients evaluated RR-3. The meta-analysis results showed a statistically significant difference between the two groups (OR = 0.18, 95% CI [0.05, 0.69], I2 = 0%, P = 0.01; Fig. 4B) [38, 41].
Meta-analysis of RR-6
Two studies involving 131 patients examined the RR-6. The meta-analysis results also showed a statistically significant difference between the two groups (OR = 0.38, 95% CI [0.14, 1.03], I2 = 39%, P = 0.06; Fig. 4C) [38, 41].
nm EL plus TO vs. TO
Meta-analysis of ORR
Nine studies involving 948 patients evaluated ORR. The meta-analysis results showed a statistically significant difference between the two groups (OR = 4.21, 95% CI [2.90, 6.11], I2 = 0%, P < 0.001; Fig. 5A) [36, 37, 42,43,44, 46, 48,49,50].
Meta-analysis of TARR
Four trials involving 465 patients assessed the TARR. The meta-analysis results demonstrated a significant difference between the two groups (OR = 0.42, 95% CI [0.22, 0.81], I2 = 4%, P = 0.009; Fig. 5B) [43, 46, 49, 50].
Meta-analysis of RR-3
One study involving 83 patients investigated RR-3. However, no statistically significant difference was identified between the two groups (OR = 0.32, 95% CI [0.01, 8.03], P = 0.49; Fig. 5C) [43].
Discussion
FV is a common skin disorder characterized by the loss of pigment in certain areas of the skin. It affects approximately 0.5–3% of the population, making it a relatively prevalent condition. If left untreated, FV can have significant socio-psychological consequences, such as low self-esteem, depression, and anxiety, which can greatly impact the quality of life for those affected.
The current treatments for FV mainly include local therapy, phototherapy, and surgical treatment [52, 53]. Local therapy options such as topical medications, corticosteroids, and immunomodulators (e.g., TO) are commonly employed. Phototherapy typically involves ultraviolet B (UVB) phototherapy (e.g., 308 nm EL) and psoralen plus ultraviolet A therapy, which stimulate the activity of skin pigment cells through light exposure, thereby promoting pigment deposition. Surgical options such as skin grafting and pigment cell transplantation may be considered to enhance pigment deposition in affected areas. Among these modalities, 308 nm EL and TO are frequently utilized for FV.
The 308 nm EL enables precise targeting of vitiligo-affected areas on the face, delivering controlled doses of UVB radiation specifically to the depigmented patches [54]. A typical 308 nm EL treatment protocol often involves applying the laser with a dosage of 50 mJ/cm2 on affected areas twice weekly for at least 12 weeks. This focused UVB light stimulates melanocyte activity, promoting repigmentation in characteristic vitiligo depigmented patches [54].
In the case of TO, it offers a localized treatment approach for FV. Direct application to the depigmented patches allows for targeted therapy, minimizing exposure to unaffected areas of the skin [55]. A typical TO treatment protocol involves applying topical 0.1% TO twice daily on the target lesion for a total of at least 12 weeks. Tacrolimus functions by inhibiting the activity of specific immune cells, namely T lymphocytes, which contribute to the inflammatory response associated with vitiligo [55].
Previous studies have explored the use of 308 nm EL and TO therapy for the treatment of FV. These treatment modalities have shown some promising results in repigmenting the affected areas. However, there is limited evidence available to support the effectiveness and safety of combining 308 nm EL with TO therapy compared to using either therapy alone. To address this gap in knowledge, this study conducted a systematic investigation to evaluate the effects and safety of combining 308 nm EL with TO therapy in the treatment of FV. The researchers aimed to assess the repigmentation outcomes, evaluate the side effects and adverse events associated with the combined therapy, and provide evidence-based recommendations for clinicians and patients considering this treatment approach. By thoroughly examining the effects and safety of combining 308 nm EL with TO therapy, this study aimed to contribute to the existing body of knowledge on FV treatment options and potentially provide a more effective and safe treatment approach for individuals with this skin disorder.
The analysis included 19 RCTs with a total of 2085 patients diagnosed with FV. The meta-analysis results showed that when comparing the combination therapy of 308 nm EL with TO to using only 308 nm EL, there were significant differences in the ORR. The combination therapy demonstrated a higher ORR than using 308 nm EL alone. Additionally, there were significant differences observed in the RR-3 and RR-6. However, there was no significant difference in the TARR between the two treatment modalities. These findings suggest that combining 308 nm EL with TO is more effective in treating FV compared to using 308 nm EL alone, while maintaining a similar safety profile for the patients. Similarly, when comparing the combination therapy of 308 nm EL with TO to using only TO, significant differences were observed in the ORR, TARR, and RR-3. This indicates that the combination therapy yields better treatment outcomes for FV patients compared to using TO alone. However, it is important to note that the safety profile of TO monotherapy was found to be better than that of combining TO with 308 nm EL. Therefore, while the combination therapy shows superior treatment effects, clinicians should consider the potential risks associated with this combination therapy and carefully assess the individual patient’s condition before proceeding with the treatment.
The results of this study highlight the potential of this combined approach in achieving better repigmentation outcomes for patients with FV. By combining the precise and targeted action of the 308 nm EL with the occlusion provided by topical application, a synergistic effect is observed. The EL delivers focused ultraviolet light to the depigmented areas of the skin, stimulating melanocyte proliferation and repigmentation. Concurrently, topical occlusion helps enhance the penetration of medications and increases the absorption of ultraviolet radiation, further promoting the therapeutic effects of the laser. However, it is important to note that while the results are promising, further research needs to be conducted to validate these findings. Future systematic reviews should involve RCTs with larger sample sizes to provide a more robust analysis of the combined therapy’s effectiveness. Additionally, a more standardized approach across different research studies would help ensure consistency and comparability of the results. Overall, this study provides valuable insights into the potential benefits of combining 308 nm EL therapy with topical occlusion for the treatment of FV. Further research and validation are necessary to solidify these findings and establish the combined therapy as a preferred and widely adopted treatment option for FV.
It is crucial to recognize and highlight the limitations of this retrospective study. Firstly, the small number of eligible RCTs available for analysis can significantly impact the overall validity and generalizability of the study findings. A limited sample size may compromise the statistical power and reliability of the results, making it challenging to draw definitive conclusions. Furthermore, the variations in the implementation of the included studies can introduce potential biases into the analysis. Each study might have adopted different methodologies, inclusion criteria, and interventions, leading to heterogeneity in the data. This heterogeneity can potentially obscure the true effect of the treatments being evaluated. In addition, differences in the treatments and controls used across the included studies can contribute to the complexity of interpreting the results. These variations might involve variations in dosages, administration protocols, or even the choice of comparators. Such differences can introduce confounding factors, making it difficult to isolate the effects of the specific interventions being investigated. Moreover, the variance in follow-up duration among the included studies may have limited the ability to establish concrete and robust conclusions. Different studies might have utilized varying follow-up durations, which can impact the assessment of long-term outcomes and the reliability of the observed effects. The lack of consistency in follow-up duration increases the potential for uncertainty and may hinder the comprehensive understanding of the intervention’s efficacy or safety. In conclusion, while this retrospective study provides valuable insights, it is important to be cautious in interpreting the results due to the limitations inherent in the study design.
Conclusion
In conclusion, this study demonstrates that combining 308 nm EL with TO is a superior treatment option for FV compared to using either therapy alone.
Abbreviations
- FV:
-
Facial vitiligo
- 308 nm EL:
-
308Nm excimer laser
- TO:
-
Tacrolimus ointment
- RCTs:
-
Randomized controlled trials
- ORR:
-
Overall response rate
- TARR:
-
Total adverse reaction rate
- RR-3:
-
Recurrence rate at 3-month
- RR-6:
-
Recurrence rate at 6-month
- OR:
-
Odds ratios
- CI:
-
Confidence intervals
References
Bergqvist C, Ezzedine K (2020) Vitiligo: a review. Dermatology 236(6):571–592
van Geel N, Speeckaert R (2017) Segmental vitiligo. Dermatol Clin 35(2):145–150
Ezzedine K, Eleftheriadou V, Whitton M, van Geel N (2015) Vitiligo. Lancet 386(9988):74–84
Picardo M, Dell’Anna ML, Ezzedine K, Hamzavi I, Harris JE, Parsad D, Taieb A (2015) Vitiligo. Nat Rev Dis Primers 1:15011
Lotti T, D’Erme AM (2014) Vitiligo as a systemic disease. Clin Dermatol 32(3):430–434
Mohammed GF, Gomaa AH, Al-Dhubaibi MS (2015) Highlights in pathogenesis of vitiligo. World J Clin Cases 3(3):221–230
Halder RM, Chappell JL (2009) Vitiligo update. Semin Cutan Med Surg 28(2):86–92
Spritz RA (2008) The genetics of generalized vitiligo. Curr Dir Autoimmun 10:244–257
Le Poole IC, Luiten RM (2008) Autoimmune etiology of generalized vitiligo. Curr Dir Autoimmun 10:227–243
Sitek JC (2006) Vitiligo–loss of cutaneous pigmentation. Tidsskr Nor Laegeforen 126(18):2370–2372
Sehgal VN, Srivastava G (2006) Vitiligo: auto-immunity and immune responses. Int J Dermatol 45(5):583–590
Huggins RH, Schwartz RA, Janniger C (2005) Vitiligo. Acta Dermatovenerol Alp Pannonica Adriat 14(4):137–142
Boissy RE, Manga P (2004) On the etiology of contact/occupational vitiligo. Pigment Cell Res 17(3):208–214
Hann SK, Park YK, Chun WH (1997) Clinical features of vitiligo. Clin Dermatol 15(6):891–897
Handa S, Kaur I (1999) Vitiligo: clinical findings in 1436 patients. J Dermatol 26(10):653–657
Abdel-Malek ZA, Jordan C, Ho T, Upadhyay PR, Fleischer A, Hamzavi I (2020) The enigma and challenges of vitiligo pathophysiology and treatment. Pigment Cell Melanoma Res 33(6):778–787
Krüger C, Schallreuter KU (2012) A review of the worldwide prevalence of vitiligo in children/adolescents and adults. Int J Dermatol 51(10):1206–1212
Whitton M, Pinart M, Batchelor JM, Leonardi-Bee J, Gonzalez U, Jiyad Z et al (2016) Evidence-based management of vitiligo: summary of a Cochrane systematic review. Br J Dermatol 174(5):962–969
Nahhas AF, Braunberger TL, Hamzavi IH (2019) Update on the Management of Vitiligo. Skin Therapy Lett 24(3):1–6
Deng Y, Li J, Yang G (2020) 308-nm excimer laser plus platelet-rich plasma for treatment of stable vitiligo: a prospective, randomized case-control study. Clin Cosmet Investig Dermatol 13:461–467
Bae JM, Kwon SH, Ju HJ, Kang HY (2020) Suberythemic and erythemic doses of a 308-nm excimer laser treatment of stable vitiligo in combination with topical tacrolimus: A randomized controlled trial. J Am Acad Dermatol 83(5):1463–1464
Shah S, Sakhiya J, Deshpande P, Sakhiya D, Inamadar AC (2020) Safety and efficacy of the combination of 308-nm monochromatic excimer light and topical 0.1% tacrolimus ointment in segmental vitiligo: an open-label study. J Clin Aesthet Dermatol 13(2):E69–E75
Wu Y, Sun Y, Qiu L, Wang H, Zhang Y, Chen K et al (2019) A multicentre, randomized, split face and/or neck comparison of 308-nm excimer laser and 01% tacrolimus ointment for stable vitiligo plus intramuscular slow-releasing betamethasone for active vitiligo. Br J Dermatol 181(1):210–211
Wang LM, Lu WJ, Yuan JT, Zeng BB, Li D, Zhang F, Li JJ (2018) Utility of dermoscopy for evaluating the therapeutic efficacy of tacrolimus ointment plus 308-nm excimer laser combination therapy in localized vitiligo patients. Exp Ther Med 15(4):3981–3988
Du J, Wang XY, Ding XL, Xu QX, Chen Z, Chang JM, Zhang JZ (2013) Long-term efficacy and safety of tacrolimus ointment in the treatment of vitiligo. J Dermatol 40(11):935–936
Wang X, Liu GY (2017) Progress of 308 nm excimer laser combined with other methods in the treatment of vitiligo. Chin J Dermatol 50(9):695–697
Baltás E, Nagy P (2001) Repigmentation of localized vitiligo with the xenon chloride laser. Br J Dermatol 144(6):1266–1267
Spencer JM, Nossa R, Ajmeri J (2002) Treatment of vitiligo with the 308 nm excimer laser: a pilot study. J Am Acad Dermatol 46:727–731
Mehraban S, Feily A (2014) 308 nm excimer laser in dermatology. J Lasers Med Sci 5:8–12
Travis LB, Weinberg JM, Silverberg NB (2003) Successful treatment of vitiligo with 0.1% tacrolimus ointment. Arch Dermatol 139(5):571–574
Matin M, Latifi S, Zoufan N, Koushki D, MirjafariDaryasari SA, Rahdari F (2014) The effectiveness of excimer laser on vitiligo treatment in comparison with a combination therapy of Excimer laser and tacrolimus in an Iranian population. J Cosmet Laser Ther 16(5):241–245
Park OJ, Park GH, Choi JR, Jung HJ, Oh ES, Choi JH, Lee MW, Chang SE (2016) A combination of excimer laser treatment and topical tacrolimus is more effective in treating vitiligo than either therapy alone for the initial 6 months, but not thereafter. Clin Exp Dermatol 41(3):236–241
Chen J, Li YL, Peng SC et al (2021) The efficacy of 308 nm Excimer laser and tacrolimus in the treatment of adult vitiligo on the face. Chin Contemp Med 28(3):38–40
Guan JY, Ge Y, Lai YL et al (2022) The efficacy of tacrolimus ointment combined with 308 nm Excimer laser in the treatment of facial vitiligo in children. J Dermatol Venereol 29(2):130–134
Li ZM, Xie PY, Zhou XY (2017) Clinical efficacy of tacrolimus ointment combined with 308 nm excimer light in the treatment of vitiligo. Chin foreign Med Res 15(24):114–115
Li RZ, Wei L, Wei S et al (2021) The efficacy of 308 nm Excimer laser combined with tacrolimus ointment in the treatment of facial vitiligo and its influence on patients’ psychological status. Psychol Monthly 16(19):49–51
Li HH, Xu WC, Zeng QR (2022) Clinical study of 308 nm Excimer laser combined with 0.1% tacrolimus ointment in the treatment of vitiligo of the face and neck. Primary Care Forum 26(10):11–3
Liu JJ, Yang HF, Li YJ et al (2018) Efficacy of 308 nm Excimer laser combined with drugs in the treatment of adult facial vitilig. Shaanxi Med J 47(8):1017–1019
Passeron T, Ostovari N, Zakaria W et al (2004) Topical tacrolimus and the 308-nm excimer laser: a synergistic combination for the treatment of vitiligo. Arch Dermatol 140(9):1065–1069
Pei Y (2020) Tacrolimus combined with 308 nm Excimer laser for the treatment of facial vitiligo. Chin Continuing Med Education 12(13):131–133
Pu Y (2020) Efficacy of 308 nm Excimer laser combined with drugs in the treatment of adult facial vitiligo. Med Aesthetic Cosmetology 29(1):13–14
Suo DF, Zeng SW (2020) Efficacy of 308 nm Excimer laser combined with tacrolimus ointment in the treatment of vitiligo of face and neck. Med Inform 33(11):116–117
Tang HB, Liu X, Chen XY et al (2013) 308 nm excimer light combined with 0.03% tacrolimus ointment in the treatment of facial vitiligo. J Pract Dermatol 6(6):343–5
Wang J (2016) Clinical analysis of 308 nm Excimer laser combined with tacrolimus ointment in the treatment of vitiligo. Chin Cosmetic Med 25(10):83–84
Wang SL (2017) The efficacy of 308 nm Excimer laser combined with tacrolimus in the treatment of facial vitiligo. J Clin Rational Drug Use 10(10):85–86
Wang X (2018) Clinical study of tacrolimus ointment combined with 308 nm Excimer laser in the treatment of facial vitiligo. Modern Drug Use Chin 12(1):116–117
Wen XY, Tian WW, Li G et al (2021) Effect of tacrolimus ointment combined with 308 nm excimer light on adult vitiligo and quality of life. Chin Med Innovation 18(24):134–137
Wen XY, Liu JH, Ding M et al (2022) Clinical study of tacrolimus ointment combined with 308 nm excimer laser photocoagulation in the treatment of adult facial vitiligo. Chin Cosmetic Med 31(6):13–16
Zhang HY (2016) 308 nm high energy excimer light combined with tacrolimus cream in the treatment of facial vitiligo. World Newest Abstracts Med Information 16(53):56–61
Zhang QW, Wang YH (2016) The efficacy of 308 nm Excimer laser combined with tacrolimus in the treatment of stable vitiligo of the head and face. Shandong Med 56(18):107
Zhu Q (2018) The efficacy of 308 nm Excimer laser combined with tacrolimus in the treatment of facial vitiligo was evaluated. Electronic J Clin Med Literature 5(1):117–119
Liu CH, Wahefu A, Lu XY, Abdulla R, Dou J, Zhao HQ et al (2022) Chemical Profiling of Kaliziri Injection and Quantification of Six Caffeoyl Quinic Acids in Beagle Plasma by LC-MS/MS. Pharmaceuticals (Basel) 15(6):663
BikashRanjanKar RR, Singh BSTP, Agrawal I (2022) Treating Vitiligo at the angle of lips: a narrative review. J Cutan Aesthet Surg 15(4):355–362
Birlea SA, Goldstein NB, Norris DA (2017) Repigmentation through melanocyte regeneration in vitiligo. Dermatol Clin 35(2):205–218
Rico MJ, Lawrence I (2002) Tacrolimus ointment for the treatment of atopic dermatitis: clinical and pharmacologic effects. Allergy Asthma Proc 23(3):191–197
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Concept and design: Suo DF, Zeng SW, and Meng LH; Data curation: Suo DF, Zeng SW, and Meng LH; Formal analysis: Suo DF and Zeng SW; Funding acquisition: Not applicable; Investigation: Zeng SW; Methodology: Suo DF, Zeng SW; Project administration: Suo DF, Zeng SW; Resources: Suo DF, Zeng SW, and Meng LH. Software: Suo DF and Meng LH; Supervision: Zeng SW; Validation: All authors; Visualization: All authors.
Corresponding author
Ethics declarations
Informed consent
Not applicable.
Competing interest
All authors declare that they have no completing interests in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Suo, Df., Zeng, Sw. & Meng, Lh. 308 nm excimer laser and tacrolimus ointment in the treatment of facial vitiligo: a systematic review and meta-analysis. Lasers Med Sci 39, 90 (2024). https://doi.org/10.1007/s10103-024-04033-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10103-024-04033-y