Abstract
Laser discectomy or nucleotomy is an increasingly important method for less invasive procedures of column, but the ideal kind of laser is still not established. As the wavelength is an important parameter for water absorption, this study was performed to investigate the action of the laser emission in the near infrared (808 to 1908 nm) region in the context of surgical procedures for percutaneous intervertebral disc decompression (nucleotomy). Forty intervertebral discs from pigs lumbar spines were irradiated with laser (λ = 808, 980, 1470 and 1908 nm), 1-s on/off time cycles, for 120 cycles and 10 W of power (808, 980, and 1470 nm) or 240 cycles and 5 W of power (1908 nm), with total power of 1200 J, and subjected to microscopic evaluation through hematoxylin-eosin (HE) staining in order to measure the ablation lesions and the residual thermal injury. Ten other discs were not irradiated and worked as controls. The ablation lesions were measured (in mm) at 1.08 ± 1.25, 1.70 ± 0.63, 2.23 ± 1.02, 1.37 ± 0.39, and 0.94 ± 0.41 (median ± SD) for the control, 808, 980, 1470, and 1908 nm groups, respectively. The difference between 1908 nm and all the other groups was statistically significant (p < 0.05). The residual thermal injury was less evident in 1908 nm laser and sharper in 980 nm laser wavelengths. The laser at a wavelength of 1908 nm was considered the most efficient for the vaporization of the nucleus pulposus, followed by the laser wavelengths of 1470, 808, and 980 nm, and proved to be useful for laser nucleotomy procedure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The incidence of disc lesions is increasing due to factors such as trauma, postural problems, physical inactivity, obesity, and populational ageing. The most common disc injury is the spinal disc herniation, especially in the lumbar region, followed by incidences of the same problem at the cervical region. Both types of injury lead to a poorer quality of life, productivity loss and need for increasing economic funding.
The most exerted method to correct herniated discs still consists in its withdrawal and in the settlement of the involved vertebrae through a procedure called arthrodesis, in an attempt to prevent intervertebral space collapse.
Other less aggressive methods (Table 1) [1], such as percutaneous interventions, improve the results. These procedures can be done through the application of high frequency (HF) or laser irradiation.
The advantages of these alternative methods over arthrodesis are: no need of hospitalization—they can be led as outpatient procedures—fast resolution, reduction in the recovery time, faster treatment and rehabilitation, lower social and economic costs. In addition, in open surgery, the access to the discs is attained through the disruption of the posterior longitudinal ligament and of the fibrous annulus, which increases the re-injury probability [2].
In the last 15 years, minimally invasive surgical procedures have been replacing the conventional open surgical ones in almost all areas of medicine. The percutaneous discectomy generally attends to a class of minimally invasive surgical procedures suggested for reducing intra disc pressure. One of the theories on the improvement brought by percutaneous discectomy suggests that the removal of disc material reduces the internal disc pressure, so as to lead the segment back to the core center. Another relevant mechanism is that the removal of disc material can prevent the release of chemical mediators which negatively affect the spinal nerve root [2–7].
The main purpose of percutaneous laser disc decompression, as a treatment for contained herniated (non-extruded) discs, is to selectively decrease the amount of the nucleus pulposus material, maintaining the adjacent tissues unchanged. Initially addressed only for lumbar discs treatment, the technique is also feasible for thoracic and cervical discs [2].
Laser discectomy does not mechanically remove disc material. This technique makes use of the fact that the intervertebral disc can be considered a closed hydraulic system to provoke a controlled reduction in intra disc pressure through the vaporization of the most appropriate nuclear section of the disc volume. This action leads to the retraction of the herniated part of the disc [2, 8, 9].
The open surgery has evolved to endoscopic procedures and to percutaneous techniques with discordant results [10]. Since the first percutaneous laser discectomy was performed by Choy and Ascher in 1986 [11], the search for best results has relied upon different types of laser and more appropriate parameters, in order to vaporize lesser amounts of liquid [12, 13]. The intervertebral discs are water-rich and contain few pigmented chromophores. For this reason, the ideal laser irradiation might present a high water absorption coefficient and a low tissue pervasion, therefore limiting the adjacent tissue thermal injury.
The choice for the best kind of laser for this procedure (Nd:YAG, Ho:YAG or diode laser) is controversial [1, 5, 8, 14–16], due to the differences among them. Most authors, however, believe that the diode laser is the most appropriate [9], followed by the Nd:YAG laser.
Recent improvements in diode laser engineering have engendered lasers with theoretically more appropriate wavelengths for these procedures. Therefore, this study aims to investigate the action of laser emission in the near infrared (808 to 1908 nm) region in tissues with high water content, however poorly pigmented, e.g., for percutaneous intervertebral discs (nucleotomy).
Material and methods
This research was approved by the Committee of Animal Rights (4530240314). In this analytical interventional double-blind experimental study, we have used 50 discs (L1 to L5) of intervertebral spinal columns from pigs. The column segments were mounted in a support, ensuring their immobility [1]. Eighteen-gauge needles were inserted in the intervertebral spaces and controlled through radioscopy [1, 4, 8, 9, 17, 18] (Fig. 1).
Once reached, the nucleus pulposus, a silica optical fiber with a core diameter of 400 μm (for 808, 908 and 1470 nm lasers) and 600 μm (for 1908 nm laser) was introduced, keeping the fiber tip 5 mm beyond the tip of the needle. Then the laser (n = 40) with wavelengths ranging from 808 to 1908 nm was fired, as shown in Table 2, released in pulses with 1 s of duration and intervals of 1 s between them, in a total of 120 and 240 pulses, respectively, for the vaporization of the nucleus pulposus on a single spot of application. The total energy applied to all the discs was 1200 J. In each column, one disc served as control, with no laser irradiation on it, in a total of ten discs.
Subsequently, the discs were frozen until their preparing for microscopic examination, in order to assess the changes generated by the different laser wavelengths.
We have rated the pervasion depth and the quality of the tissue injured by the laser shots in the remaining tissue and the kind of injury induced by this action.
Microscopic evaluation
All the surveyed discs were split apart, fixed in 10 % buffered formalin, decalcified, and submitted to histological processing. For descaling, the pieces were immersed in a solution of 112.6 g dibasic sodium phosphate, 11.12 g sodium phosphate monobasic, 2500 ml of distilled water, 175 g of EDTA and 125 ml of formaldehyde for 50 to 70 days. This solution was changed every 2 to 3 days. The histological sections (4 μm) were stained by hematoxylin-eosin (HE) and observed by optical microscopy to determine the lesion depth and the thermal damage. The extent of injuries was assessed by Image-J software. We have used the non-parametric test of Kruskal-Wallis to compare all the groups and the Mann-Whitney test (one-side) to compare the control group to laser groups (p < 0.05).
Results
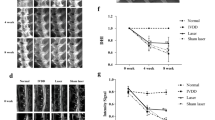
The effect of the different wavelengths lasers on nucleus pulposus tissue is summarized in Table 3. Lesion depths varied from 0.54 to 3.32 (control group), 0.74 to 2.30 (808 nm), 0.95 to 4.13 (980 nm), 1.12 to 2.12 (1470 nm), and from 0.52 to 1.58 (1908 nm) millimeters. Median and standard deviation were respectively 1.08 ± 1.25, 1.70 ± 0.63, 2.23 ± 1.02, 1.37 ± 0.39, and 0.94 ± 0.41.
These results are depicted in the chart below (Fig. 2):
From the microscopic point of view, we have found fewer injured tissues, with structural derangement in those discs irradiated with λ = 1908 nm, with increasing injuries in discs irradiated with wavelengths from 1470, 808, and 980 nm (Fig. 3).
Regarding the residual thermal injury, 1470 nm laser presented less derangement, but greater pervasion. The laser wavelength 1908 nm showed little pervasion and low residual lesion (Fig. 4).
Although the difference in terms of residual injuries was noteworthy, the extent of those injuries was undermined due to the deterioration of the tissues.
Discussion
Even today, the subject of herniated discs gives rise to discussions, from the very definition of hernia to the most appropriate surgical procedure to treat it. In a herniated disc, the rupture of the fibrous ring is assumed, while the protrusion is distinguished by the ring integrity.
The pathogenesis of the lumbar pain lies in the rupture of the intervertebral disc annulus fibers, which leads to a cyclical process of inflammation, damage to the disc array and awareness of nociceptors [19]. Many neurosurgeons have reported that the minimally invasive technique would only be appropriate for the treatment of protrusions and hernias. The number of mini access procedures [20], however, has been growing, as has been evidenced in several research databases.
The gold standard for surgical treatment of a herniated disc is the laminectomy, established in 1829 by A. G. Smith, but it can lead to spinal arthrodesis. Despite the technological progress concerning surgical procedures in this matter, even today, there is a consensus about the advantages of minimally invasive surgery over conventional surgery. Adam et al. [21] compared patients undergoing classical laminectomy and coblation and found no significant variations related to pain over 1 year after the procedure. Iwatsuki et al.[19], in a clinical study with 11 patients with lumbar pain and follow-up from 1 day to 24 months after nucleotomy using a diode laser with λ = 805 nm, P = 20 W, and 1 s pulses with 1 s interval between pulses, found significant improvement, when assessed by the Oswestry Disability Index (ODI) and the Visual Analog Scale (VAS).
These data are significant whenever we consider the intervertebral disc as a closed system in which small amounts of removed tissue cause a negative pressure big enough to cause the retraction of the drive protrude [11, 21]. In addition, the laser also acts over the annular nociceptors, for its anti-inflammatory and proliferative action, already described by many authors. This action does not affect the laser focus—that vaporizes the tissue—but the peripheral edge, where the power is exponentially lower, since the power curve has a Gaussian shape. In this way, the surgical laser acts as a low-level intensity laser, modulating pro- and anti-inflammatory factors [22].
The nucleus pulposus volume reduction after vaporization is already established through studies with images based on mathematical models [23–25], either by high-frequency stream or by laser.
Ren et al. [25], in a retrospective study with 22 patients, showed that the nucleotomy by Nd:YAG laser (λ = 1064 nm) did not cause reduction of the intervertebral space, improved lumbar pain and decreased disc herniation, assessed by radiographic images and magnetic resonance.
Much about the discussion on nucleotomy is based on the amount of removed tissue required for creating the amount of negative pressure enough to cause the retraction of the disc protruding nose. It is estimated that the withdrawal of 10 to 15 % of the nucleus pulposus volume leads to a 90 % reduction of the intradiscal pressure. Choy and Diwan [26], in a study with cadaveric columns and four living patients, using Nd:YAG laser (λ = 1064 nm, E = 1000 J, P = 20 W, and 1 s pulses with 5 s intervals), obtained 50 % of the intradiscal pressure reduction. Min et al. [27], using Ho:YAG laser (λ = 2100 nm), have achieved average values of 0.556 ± 0.06 g (dry weight) and 1.89667 ± 0.162 g (physiological weight) of tissue taken from the nucleus pulposus in an ex vivo model.
From these data, we can infer that the best use of the technique under discussion lies within the achievement of a wavelength that vaporizes the nucleus pulposus tissue with minimal residual thermal injury. For its large amounts of water and little pigment, this tissue offers great absorption peaks around 980, 1064, 1470, and 1910 nm. The most commonly used devices to accomplish this goal are the Nd:YAG laser (λ = 1064 nm), for its specificity, and the diode laser (λ = 808 nm and λ = 980 nm), for its efficiency and easy shipping and handling. The Ho:YAG laser (λ ≅ 2.0 μm) would be the ideal choice in terms of absorption peak values. However, the power required for its action is too strong and generates an excessive amount of heat. Therefore, its use may be risky in respect to the injuries resulted from the increasing of temperature in the nerve roots and requires concomitant irrigation.
Diode lasers are semiconductor devices that shed light when stimulated by electric stream. The most widely employed diode lasers are the AlGaAs (gallium-aluminum arsenide) and the AlGaInP (aluminum indium gallium-phosphate). Some procedures may require an output of up to 25–30 W, which is perfectly accessible to these devices [28].
Recent advances in medical devices engineering made it possible to developing a kind of laser that operates in the range of 1908 nm, one of the water absorption peaks, and demands reduced power (5 W), without jeopardizing its efficiency.
In our study, we employ an ex vivo model, similar to those previously addressed in literature [23, 26, 27], with some adjustments. Cselik et al. [29] have compared two wavelengths (980 and 1470 nm) of a diode laser (P = 5 W and E = 500 J) and showed that the injuries with 980 nm were restrained to the application site, but were larger, whereas the 1470 nm laser has produced smaller local lesions, but with other tissue variations around the nucleus, suggesting that the λ = 1470 nm had greater interaction with the tissue. Choy et al. [30], comparing the continuous and pulsed CO2 laser (λ = 10,600 nm), the erbium:YAG laser (Er:YAG), the Nd:YAG 1064 nm, the argon laser 1318 nm, the Ho:YAG and the Excimer laser efficiency, have showed best results with pulsed CO2 and the lowest efficiency with argon laser. For establishing it as the second best and because the CO2 laser is not driven by optical fiber, they have opted for Nd:YAG as a choice for nucleotomy.
The ideal laser for each specific application is not determined just by the power, however. Regarding vaporization, we must take into account the time of application, the thickness of the tissue and its absorption coefficient, which depends on each chromophore concentration. The laser interacts with the tissue according to Beer’s Law, which states that the injury of tissue at the site of absorption decreases exponentially as we move away from the edge of the crater [31]. In other words, if power is strong enough and time is shorter than the thermal relaxation time, each laser pulse will vaporize a critical volume of tissue and no thermal damage will occur beyond the point of impact. Literature presents quite variable parameters employed by different authors. We have chosen the nearest average energy value (around 1200 J) to act in great absorption coefficients for the wavelengths that we have studied.
Given the time elapsed between the sample collection and the irradiation procedure, it was necessary to keep the spinal columns frozen, for preservation. Although the tissues remained apparently intact after this process, the water contained in them may have been modified by temperature. Furthermore, the pieces were irradiated at room temperature, and the thawing process may have affected the amount of water not solidified within the tissues. In discs to which the laser was not applied, the samples deterioration was higher, hence the smaller number of measurements achieved. By modifying the tissue structure through water evaporation and by forming a charred tissue with lower aqueous content, the laser application lessens tissue degradation due to defrost. This fact justifies some measures of greater injury depth in the control group if compared to the laser group. These measures may be caused by tissues deterioration, which increases the lesions size, but without leading to any residual damage.
Cselik et al. [29] have used oxen columns in their study and have showed the changes generated by lasers λ = 980 nm and λ = 1470 nm. They call attention to the fact that the experiment was done at room temperature, and that the changes found in the MRI scan represent the water volume variation. They also justify the findings difference in favor of the 1470 nm wavelength by the increased free fraction of water results, since this wavelength is 40 times more absorbed by water than the 980 nm.
Since the wavelength of 1908 nm is even more water-absorbed than the 1470 nm (Fig. 1), a slight variation could result in great absorption differences. The fact that the values have presented little variation within each group could point to the fact that the lesion produced could have been provoked precisely by the laser action. The wide range of measures within the control group could imply that the injury caused by the needle/fiber was due only to their interaction with the tissue, since there is no thermal injury involved in the process.
The fact that the 980 nm laser, widely used for this kind of procedures and taken as standard along with 1064 nm, has presented a worse result than that of 808 nm has caught our attention . On the one hand, we know that the heat inside the tissues generated by laser absorption is determined by the parameters of the tissues and their optical properties. On the other hand, the heat transport is due only to the tissue thermal characteristics, such as specific heat, thermal conductivity, and density. Laser thermal effects are determined by temperature and tissue type. Above 100 °C, with the evaporation of all water molecules, the adjacent tissue charring occurs. As the charred tissue thermal absorption coefficient is higher than the water coefficient, carbonization raises laser effects and leads to more damage [32].
In this way, water is the first element for laser absorption, followed by the absorption by the carbonized tissue. The higher the affinity for water, the lower the forming of carbonization zone, which therefore results in less injury. We expect that the 980 nm laser, which has a higher affinity with water than the 808 nm laser, can beget better results. However, we observe that the 980 nm laser has caused bigger injury and more relevant carbonized tissue formation under the same physical conditions than the 808 nm laser.
The rate of tissue vaporization, e.g., the amount of tissue removed within one unit of time, increases with the level of generated heat and changes with more carbonized tissue formation. The absorptive heat generated (W/cm3), proportional to the absorption product coefficient (cm−1) and irradiance (W/cm2), suffers diffusion and leads to a change of temperature in the area close to the vaporization region, with corresponding tissue change. Under the conditions of this study, apparently the 980 nm laser has vaporized the water faster and has carbonized tissue more intensively, two factors which increase both the lesion ablation (vaporization rate) and the residual thermal injury (carbonization). The carbonized tissue has an absorption coefficient which decreases exponentially with respect to the increase of wavelength [33], as shown in the table below (Table 4):
Once the maintained parameters for all the irradiated segments and the assessment made double-blind (researcher and pathologist), the results obtained reflect the occurrence of variables that must be established and taken into account in experimental and clinical works regarding laser nucleotomy.
Device engineering should consider that tissue conditions vary during the procedure and require a dynamic reconfiguration, in order to recalculate the parameters as the process evolves. It is important to notice that the laser at a wavelength of 1908 nm proved to be more efficient than the others.
Conclusion
The laser discectomy (nucleotomy) at a wavelength of 1908 nm, power of 5 W, and 1200 J dose proved to be more efficient, causing minor injury and tissue breakdown than the laser wavelength 808, 980, and 1470 nm.
References
Choy DSJ (2004) Percutaneous laser disc decompression: a 17-year experience. Photomed Laser Surg 22:407
Choy DSJ, Hellinger J, Hellinger S, Tassi GP, Lee SH (2009) 23rd anniversary of percutaneous laser disc decompression (PLDD). Photomed Laser Surg 27(4):535–8
Choy DSJ, Michelsen J, Getrajdman D (1992) Percutaneous laser disc decompression: an update—Spring 1992. J Clin Laser Med Surg 10:177–84
Choy D (2003) Percutaneous laser disc decompression: a practical guide. Springer, New York
Gupta AK, Bodhey NK, Jayasree RS, Kapilamoorthy T, Kesavadas C, Krishnamoorthy T (2006) Percutaneous laser disc decompression: Clinical experience at SCTIMST and long term follow up. Neurol India 54:164–7
Schenk B, Brouwer PA, Peul WC, van Buchem MA (2006) Percutaneous laser disc decompression: a review of the literature. Am J Neuroradiol 27:232
Schenk B, Brouwer PA, Peul WC, van Buchem MA (2006) Experimental basis of percutaneous laser disc decompression (PLDD): a review of literature. Lasers Med Sci 21:245
Morelet A, Boyer F, Vitry F, Ackah-Miezan S, Berquet R, Langlois S (2007) Efficacy of percutaneous laser disc decompression for lumbar disc hernia due to radiculalgia (149 patients). Presse Med 36(11 Pt 1):1527–35
Menchetti P, Bini W, Canero G, Menotti F (2008) Diode laser Percutaneous Discectomy: Multicenter study at 4 years follow up. Int J Minim Invasive Spinal Technol Suppl I-to IJMIST [serial on the Internet] 1 (2)
Siebert W (1999) Percutaneous nucleotomy procedures in lumbar intervertebral disk displacement. Curr Status Orthop 28(7):598–608
Choy D, Case R, Fielding W, Hughes J, Liebler WPA (1987) Percutaneous laser lumbar disks nucleolysis. N Engl J Med 317:771–2
Nachemson A (1961) The influence of spinal movements on the lumbar intradiscal pressure and on the tensile stresses in the annulus annulus fibrosus. Acta Orthop Scand 33:183–207
Nachemson A (1965) In vivo discometry in lumbar discs with irregular nucleograms. Some differences in stress distribution between normal and moderately degenerated discs. Acta Orthop Scand 36(4):418–34
Siebert W (1993) Percutaneous laser disc decompression: the European experience. Spine State Arts Rev 7:37–42
Casper GD, Hartman VL, Mullins LL (1996) Results of a clinical trial of the holmium: YAG laser in disc decompression utilizing the side-firing fiber: a two-year follow-up. Lasers Surg Med 19(1):90–6
Jayasree RS, Gupta AK, Bodhey NK, Mohanty M (2009) Effect of 980-nm Diode laser and 1064-nm Nd: YAG laser on the intervertebral disc in vitro and in vivo studies. Photomed Laser Surg 27(4):547–52
Gevargez A, Groenemeyer D, Czerwinsky F (2000) CT-guided percutaneous laser disc decompression with Ceralas D, the diode laser with 980-nm wavelength and 200-μm fiber optics. Eur Radiol 10:1239
Iwatsuki K, Yoshimine, TI, Awazu, K (2007) Percutaneous laser disc decompression for lumbar disc hernia: indications based on Lasegue’s Sign. Photomed Laser Surg 25 (40).
Iwatsuki K, Yoshimine TI, Umegaki M, Yoshimura K, Ohnishi Y, Ishihara M, Moriwaki T (2011) Percutaneous diode laser irradiation for lumbar discogenic pain: a clinical study. Photomed Laser Surg 29(7):459–63
Zhao XL, Fu ZJ, Xu YG, Zhao XJ, Song WG, Zheng H (2012) Treatment of lumbar intervertebral disc herniation using C-Arm fluoroscopy guided percutaneous laser disc decopmpression. Photomed Laser Surg 30(2):92–5
Adam D, Pevzner, Gepstein A (2013) Comparison of percutaneous nucleoplasty and open discectomy in patients with lumbar disc protrusions. Chirurgia (Bucur) 108(1):94–8
Fukuda TY, Tanji MM, Sb SR, Sato MN, Plapler H (2013) Low-level infrared diode laser on inflammatory process modulation in mice: pro-and anti-inflammatory cytokines. Lasers med sci 28(5):1305–13. doi:10.1007/s10103-012-1231-z
Kasch R, Mensel B, Florian S, Ruetten S, Barz T, Froehlich S et al (2012) Percutaneous nucleoplasty volume reduction with disc in an animal model. PLoS ONE [Internet] 7(11):e50211
Kasch R, Mensel B, Schmidt F, Drescher W, Pfuhl R, Ruetten S et al (2012) Percutaneous disc decompression with nucleoplasty-volumetry of the nucleus pulposus using ultrahigh-field MRI. PLoS ONE [Internet] 7(7):e41497
Ren L, Guo B, Zhang T, Bai Q, Wang X-h, Zhang L et al (2013) Medium-term follow-up findings in imaging after percutaneous laser disc decompression manifestation. Photomed Laser Surg 31(6):247–51
Choy D, Diwan S (1992) In vitro and in vivo fall of intradiscal pressure with laser disc decompression. J Clin Laser Med Surg 10:435–7
Min K, Read H, Parties K (1996) Quantitative determination of radiofrequency ablation in weight of lumbar intervertebral discs with holmium: YAG laser. Lasers Surg Med 18:187–90
Manni J (1992) Surgical diode lasers. J Clin Laser Med Surg 10(5):377–80
Cselik Z, Aradi M, von Jako RA, Lelovics Z, Juhasz I, Egyhazi Z et al (2012) Impact of infrared laser light at different wavelengths-induced ablation on intervertebral disc bovine ex vivo: evaluation with magnetic resonance imaging and histology. Lasers Surg Med 44:406–12
Choy D, Altman P, Trokel S (1995) Efficiency of disc radiofrequency ablation with lasers of various wavelengths. J Clin Laser Med Surg 13(3):153–6
Goldman MP, Fitzpatrick RE (1994) Cutaneous laser surgery: the art and science of selective photothermolysis, 1st edn. Mosby, San Diego, 336 p
Niemz M (2007) Thermal interaction. Laser-tissue interactions: Fundamentals and applications, 3rd edn. Springer, Berlin, p 79
Jacques N (1998) Continuous laser ablation of carbonized tissue: simple rules Oregon Medical Laser Center [cited 2013 10/20/2013]. Available from: http://omlc.ogi.edu/news/may98/ablation/index.html
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This research was approved by the Committee of Animal Rights of the Federal University of São Paulo (CEUA—#4530240314)
Funding
This research has received a grant from São Paulo State Research Fund (FAPESP), process number 2010/12607-9.
Rights and permissions
About this article
Cite this article
Plapler, H., Mancini, M.W., Sella, V.R.G. et al. Evaluation of different laser wavelengths on ablation lesion and residual thermal injury in intervertebral discs of the lumbar spine. Lasers Med Sci 31, 421–428 (2016). https://doi.org/10.1007/s10103-016-1865-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-016-1865-3