Abstract
Introduction
A high proportion of patients with low-risk community-acquired pneumonia (CAP) (classes I-III of the Pneumonia Severity Index) are hospitalized. The purpose of this study was to determine whether validated severity scales are used in clinical practice to make admission decisions, identify the variables that influence this decision, and evaluate the potential predictive value of these variables.
Materials and methods
A prospective, observational study of patients ≥ 18 years of age with a diagnosis of low-risk CAP hospitalized or referred from the Emergency Department to outpatient consultations. A multivariate logistic regression predictive model was built to predict the decision to hospitalize a patient.
Results
The study population was composed of 1,208 patients (806 inpatients and 402 outpatients). The severity of CAP was estimated in 250 patients (20.7%). The factors that determined hospitalization were “abnormal findings in complementary studies” (643/806: 79.8%; due to respiratory failure in 443 patients) and “signs of clinical deterioration” [64/806 (7.9%): hypotension (16/64, 25%); hemoptoic expectoration (12/64, 18.8%); tachypnea (10/64, 15.6%)]. In total, ambulatory management was not contraindicated in 24.7% of hospitalized patients (199). The predictive model built to decide about hospitalization had a good power of discrimination (AUC 0.876; 95%CI: 0.855–0.897).
Conclusions
Scales are rarely used to estimate the severity of CAP at the emergency department. The decision to hospitalize or not a patient largely depends on the clinical experience of the physician. Our predictive model showed a good power to discriminate the patients who required hospitalization. Further studies are warranted to validate these results.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Community-acquired pneumonia (CAP) is considered a public health problem owing to its high incidence and morbimortality, and the economic burden it represents for public health systems [1,2,3,4]. Determining whether a patient needs or not hospital care is one of the most complex and relevant decisions in clinical practice. The evidence available suggests that clinicians often overestimate patient risk, thereby resulting in many patients being unnecessarily hospitalized. This practice increases health spending. A range of prognostic scales have been developed for estimating CAP severity. These scales help clinicians estimate the severity of CAP objectively, thereby facilitating decision making [5, 6]. Based on these scales, clinical practice guidelines (CPG) recommend that low-risk patients are managed in ambulatory care and only the patients with a higher risk of mortality are hospitalized [7, 8]. However, the evidence available shows that patients with a low risk of mortality are frequently hospitalized [9, 10]. Some authors suggest that hospitalization decisions should also be based on clinician's experience [11,12,13], on the basis of a set of criteria that has not been clearly established. As a result, the decision is made on a case-by-case basis after the clinician has estimated the severity of CAP.
We posited that some factors are not considered or are underestimated in prognostic scales, which influences the decision to admit patients with low-risk CAP [Pneumonia Severity Index (PSI), classes I-III]5. The objectives of this study was: (i) to determine whether validated severity assessment scales are used in decision-making concerning the hospitalization of patients with CAP; (ii) to determine whether the decision to hospitalize a patient with low-risk CAP is influenced by any variables, regardless of them being considered or not in prognostic scales; and (iii) determine the value of some factors to predict hospitalization.
Materials and methods
A prospective, observational study was conducted involving patients older than 18 years who received a diagnosis of CAP between October 1st, 2016 and December 31st, 2021. The sample was composed of consecutive patients admitted to the Unit of Pulmonology for CAP, along with outpatients referred from the emergency department (ED) to the CAP Unit for follow-up. Informed consent was obtained.
Patients were included if they were ≥ 18 years; immunocompetent; diagnosed with CAP (based on consistent symptoms and the presence of a de novo pulmonary infiltrate on chest X-ray) [14] classified as PSI class I-III5 (low severity) that was not expected to require hospitalization [7, 8]. Pneumonias in institutionalized patients were also included, since the latest CPG consider them community-acquired pneumonias [8].
Patients were categorized into two groups: inpatients (defined as cases) and outpatients (defined as controls). Hospital-acquired pneumonias, tuberculosis, or CAPs in immunocompromised patients were excluded.
The reasons why ED clinicians and specialists on-call made the decision to hospitalize a patient were grouped into six categories, following the classification of previous studies [10]: [i) psychosocial factors; ii) comorbid conditions; iii) signs of clinical deterioration; iv) abnormal findings in clinical studies; v) readmissions to the ED in the last two weeks; and vi) other causes not included in other categories], each being divided into different subcategories. The physicians responsible for each patient during hospitalization reviewed admission criteria and divided patients into three groups based on the appropriateness of their admission, namely: Group 1, patients in whom outpatient care was contraindicated, independently of the severity of their CAP. Group 2, patients with identifiable risk factors that were not absolute indications for hospitalization nor were associated with a higher risk of mortality. Group 3, patients in whom outpatient care was not contraindicated and without any identifiable risk factor that warranted hospitalization.
The variables selected to assess the appropriateness of hospitalization are detailed in the Supplementary Material. The socio-familial situation of the patient was defined as good if a relative agreed to take care for the patient after discharge, regardless of them being cohabitants or not.
This study was approved by the Ethics Committee of Santiago-Lugo (code 2016/388).
Statistical analysis
Continuous variables were expressed as medians (25-75th percentiles), since most did not follow normal distribution. Qualitative variables were described as absolute frequencies and percentages. Chi-squared (qualitative variables), Cochran-Mantel–Haenszel Wilcoxon (ordinal) and Wilcoxon (continuous) test were used to assess the association between patient characteristics and decision about hospitalization or pneumonia severity.
A multivariate logistic regression model was built. The decision about hospitalization (yes/no) was the dependent variable for the patient. Independent variables included sex; age; medical history; previous vaccination (flu/pneumococcus); comorbilities; cardiovascular risk factors; Charlson index; radiological abnormalities and a range of laboratory variables. Variables with less than five patients were excluded.
The Akaike information criterion (AIC) was used to select the variables to be included in the model [15]. Based on the independent variables previously described, stepwise regression was used to sequentially identify and exclude the variables without any predictive value. Potential non-linear effects were examined using generalized additive models and spline models [16]. Results are expressed as Odds Ratio (OR) with their 95% confidence intervals (CI). Then, aspects related to performance were assessed, including calibration and discrimination. Discrimination was verified by the use of ROC curves and area under the curve. Calibration was performed using calibration plots and Brier score [17]. Data analysis was performed using R software, available at http://cran.r-project.org, with the mgcv, rms, vcdExtra y pROC packages. All analyses were carried out in accordance with TRIPOD standards [18].
Results
During the study period, a total of 1,208 patients with CAP PSI class I-III were included. Of them, 806 were admitted to the Unit of Pulmonology, whereas 402 were referred for outpatient follow-up. Figure 1 displays patient flow chart.
At the ED, CAP severity was only assessed (PSI class assessment) in 20.7% of patients [250/1208; inpatients, 181/806 (22.5%); outpatients, 69/402 (17.2%)]. In relation to hospitalized patients, if severity had not been assessed at the ED, assessment was performed within 24 h from admission. In outpatients, the risk of mortality was estimated in the first follow-up visit.
Hospitalized patients were significantly older (p < 0.001); reported a heavier alcohol use (p = 0.004); and had a higher number of chronic lung diseases (p < 0.001) and a higher Charlson index (p < 0.001), as compared to outpatients. The former also had a higher number of oxygen therapy (p = 0.007), CPAP (p = 0.011), and non-invasive mechanical ventilation (p = 0.020) machines at home. Moreover, a higher percentage of inpatients had a respiratory rate > 30 bpm and a heart rate > 120 bpm, as compared to outpatients (p < 0.001 and p = 0.008, respectively). Respiratory failure (PaO2 < 60 mmHg) was also more frequent among hospitalized patients [58% of inpatients (460/793) vs. 3.6% of outpatients (11/304)] (p < 0.001). Hospitalized patients more frequently had a BUN > 30 mg/dL (p = 0,002); lymphopenia (< 700 cell/mm3) (p < 0.001); multilobular/bilateral pulmonary infiltrates (p < 0.001 each); pulmonary cavitation (p = 0.019) and pleural effusion (p = 0.002). Factors influencing hospitalization included age; alcoholism; drug abuse; diagnosis of chronic lung disease; presence of comorbidities; home CPAP usage; respiratory rate > 30 BPM; or HR > 120 BPM; acute respiratory failure; and a variety of specific analytical [BUN > 30 mg/dL, sodium < 130 mE/L, lymphopenia (< 700 cells/mm3)] or radiological (multilobar/bilateral infiltrates, pulmonary cavitation or pleural effusion) abnormalities (see Table 1).
In relation to inpatients, the median age and percentage of males increased significantly as PSI severity increased, whereas the percentage of smokers and abuse drug users decreased. A higher PSI class was associated with a higher frequency of the following variables: multiple comorbidities; Charlson index; need for home oxygen therapy; respiratory rate > 30/minute; acute respiratory failure; BUN > 30 mg/dL; and a longer mean length of hospital stay. On another note, the percentage of patients with pulmonary infiltrates and pulmonary cavitation decreased significantly as CAP severity increased. In outpatients, the severity of pneumonia increased significantly with age, percentage of males, presence of some comorbidities, Charlson index, and percentage of patients with acute respiratory failure, glucose > 250 mg/dL and lymphopenia (lymphocytes < 700 cell/mm3). In contrast, the higher the severity of pneumonia, the lower the number of smokers (see Table 1S and Table 2S in supplementary material).
The group in which the most income was included was "abnormal findings in complementary studies" (79.8%), followed by "signs of clinical deterioration" (7.9%). In the first group, the most frequent reason was respiratory failure (443 patients). The higher the severity of pneumonia was, the higher the percentage of patients who were admitted specifically for respiratory failure, and more generally for abnormal findings in complementary studies. Similarly, the more severe pneumonia was, the longer the mean length of hospital stay (p < 0.001) (see Table 2).
In total, 61.5% (496/806) of hospitalized patients were classified as group 1 (outpatient care was contraindicated); 13.8% (110 patients) as group 2 (presence of risk factors for admission but without absolute indication for admission); and 24.7% (199 patients) as group 3 (no contraindication for outpatient care and absence of any identifiable risk factor for admission) (Table 3).
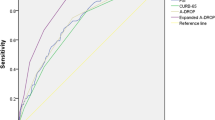
The results of the multivariate analysis of baseline clinical and epidemiological characteristics that influenced hospitalization decision are shown in Table 4. Figure 2A displays the ROC curve of the predictive model of hospitalization [AUC 0.876; 95%CI: 0.855–0.897]. Figure 2B describes the calibration of the prediction model.
To estimate the individual risk of being admitted for low-grade CAP, the total score of all the variables included in the model must be taken into accounnt. For example, a 70-year-old patient (21 points), smoker (18 points), with a chronic respiratory disease (14 points), respiratory rate 32 BPM (53 points), systolic blood pressure 110 mm Hg (0 points), heart rate 90 bpm (0 points), SaO2 95% (0 points), BUN 40 mg/dL (42 points), lymphocytes 1,000 cells/mm3 (0 points), without multilobar involvement (0 points), pulmonary cavitation (0 points) or pleural effusion (0 points), has a total score of 148 points, which is equivalent to a risk of admission greater than 98% (Table 5). The optimal cutpoint will depend on whether priority is given to the maximization of sensitivity; maximization of specificity with a minimum sensitivity value; equilibrium between sensitivity and specificity; maximization of sensitivity with a minimum specificity value; applying Youden index (a score higher than the sum of sensitivity plus specificity); or maximization of specificity (Table 5) [19]. To facilitate decision-making for clinicians, an Excel spreadsheet (HospitalizationClassificationRule) is attached in the Supplementary Material, which will allow them to easily calculate the probability of admission for each patient.
Discussion
The two most relevant findings of this study are that clinicians make the decision to hospitalize patients with CAP based on their clinical experience, rather than on the severity of the disease, as assessed using mortality risk assessment scales. In addition, the predictive model accurately identified the patients who were hospitalized.
Following recommendations, patients with severe CAP (PSI class IV-V) are generally admitted. However, there is a tendency to admit patients with low-risk CAP (classes I-III) that could theoretically be treated in the ambulatory setting. The latter account for 30–65% of cases in each series [5, 9, 10, 20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45] (Table 6). Otherwise said, in clinical practice, a low risk of mortality does not mean that a patient can be treated safely in outpatient care [46]. It is at this point that physician's discretion emerges as a factor to be considered, as established in guidelines [7, 8, 11,12,13]. In our series, the most relevant factors considered by the physicians who made the admission decision included respiratory failure and, to a lesser extent, presence of clinical signs of deterioration, i.e. heart rate > 120 bpm and respiratory rate > 30 BPM. The fact that heart and respiratory rate are considered less frequently may be due to their low incidence in low-risk CAP. Although all these factors are considered in the PSI scale, physicians assess them separately in clinical practice. However, these medical criteria are not defined nor are there any recommendations in this regard. As a result, assessment of CAP severity is not free of subjectivity. In addition, the reported reason for admission does not necessarily mean that admission is warranted. This situation results in hardly acceptable variability across clinicians and institutions.
Some authors classify reasons for the hospitalization of patients with low-risk CAP into three large groups [22]. The first group includes patients in whom ambulatory care is contraindicated for medical or psychosocial reasons. This includes socio-economic reasons [25, 27, 28, 47], severe dementia or mental illness, inability to take oral antibiotics, severe neuromuscular diseases or acute comorbid conditions [25, 28]. Thus, authors agree that some patients with a low risk of mortality may require hospitalization [21]. The second group is composed of patients with identifiable risk factors that are not absolute indications of hospitalization, nor are they associated with a higher risk of mortality. These include patients with an advanced age [24], stable comorbidities [24, 28, 47] (especially when the comorbidity is COPD [48]), previous treatment with corticosteroids [47], and radiological abnormalities [24], to name a few. In these cases, the site of care is unclear. Finally, group 3, which includes 20% of patients with low-risk CAP. This group is composed of patients in whom ambulatory care is not contraindicated. These patients may not have any identifiable risk factors for hospitalization or a low risk for adverse events, therefore, ambulatory management is indicated. In this group, reasons for hospitalization include "pressure" exerted by the patient or their family, or reluctance of physicians to send home patients who could later develop complications. In our series, 199 patients were included in group 3, which is consistent with the percentage reported in previous studies (24.7%)[22].
Validated scales should be routinely used at Emergency departments to estimate CAP severity and identify low-risk patients. Additionally, presuming that the use of this tool alone may be inappropriate, the clinical judgment of the physician should also be considered. This way, the number of patients that can be treated safely in the ambulatory setting would increase, since the number of patients in the third group would be minimized.
Our prognostic model for predicting the patients with CAP who will be hospitalized is based on the baseline characteristics of patients (Table 4) and includes variables that provide a good power of discrimination (AUC 0.876; 95%CI: 0.855–0.897). This model is useful, as it can be used in most clinical settings, since the variables assessed are routinely recorded in clinical practice. The risk score obtained would enable physicians to estimate the probability of admission and facilitate admission decision making.
This study has some limitations. It is a single-center study, which limits the external validity of the results to other populations, geographical regions or health systems. The results were not reproduced in a validation series. In most cases, the decision about admission was made without using any severity assessment scale, and an objective severity score was not available. It is possible that, if a severity score had been calculated, some patients would not have been admitted. However, this does not reflect the reality of clinical practice. Finally, although a strict definition of pneumonia was used, its clinical and radiological characteristics may mimic those of other diseases (congestive heart failure, cryptogenic pneumonitis obliterans, etc.), which may lead to misdiagnosis. However, this situation in the diagnosis and management of a case of pneumonia may occur in real clinical practice.
To conclude, the results of this study demonstrate that CAP severity is rarely assessed at the ED using validated scales. Thus, the decision about site of care largely relies on the clinical experience of the physician. This subjectivity results in large variability in decision making. Severity assessment scales should be routinely applied to all patients with CAP. However, admission decisions should not only be based on scales, but also on the clinical judgment of the physician, determined through the use of a predictive model with a good power of discrimination. These results require confirmation. Clinical practice in EDs should be standardized to improve the quality of care provided to CAP patients. Further studies are needed to validate these results.
Data availability
Data available on request fro the authors.
References
WHO. The top 10 causes of death [Internet]. WHO. [Citado el 19 de julio de 2017.] Disponible en: http://www.who.int/mediacentre/factsheets/fs310/en/
GBD (2015) Mortality and Causes of Death Collaborators. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Lond Engl 388:1459–544
Welte T, Torres A, Nathwani D (2012) Clinical and economic burden of community-acquired pneumonia among adults in Europe. Thorax 67:71–79
Sato R, Gomez Rey G, Nelson S et al (2013) Community-acquired pneumonia episode costs by age and risk in commercially insured US adults aged ≥50 years. Appl Health Econ Health Policy 11:251–258
Fine MJ, Auble TE, Yealy DM et al (1997) A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med 336:243–250
Lim WS, van der Eerden MM, Laing R et al (2003) Defining community acquired pneumonia severity on presentation to hospital: An international derivation and validation study. Thorax 58:377–382
Menéndez R, Cilloniz C, España PP et al (2020) Community-Acquired Pneumonia. Spanish Society of Pulmonology and Thoracic Surgery (SEPAR) Guidelines. 2020 Update. Arch Bronconeumol 56(S1):1–10
Metlay JP, Waterer GW, Long AC et al (2019) Diagnosis and treatment of adults with community-acquired pneumonia. An oficial clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med 200:e45-67
Jain S, Williams DJ, Arnold SR et al (2015) Community-acquired pneumonia requiring hospitalization among U.S. children. N Engl J Med 372:835–45
Marcos PJ, Restrepo MI, González-Barcala FJ et al (2018) Discordance of physician clinical judgment vs. pneumonia severity index (PSI) score to admit patients with low risk community-acquired pneumonia: a prospective multicenter study. J Thorac Dis 9:1538–46
Bartlett JG, Dowell SF, Mandell LA et al (2000) Practice guidelines for the management of community acquired pneumonia in adults. Infectious Diseases Society of America. Clin Infect Dis 31:347–82
Niederman MS, Mandell LA, Anzueto A et al (2001) Guidelines for the management of adults with community-acquired pneumonia: American Thoracic Society. Am J Respir Crit Care Med 163:1730–1754
Hoe L, Keang L (1998) Hospitalized low-risk community-acquired pneumonia: outcome and potential for cost savings. Respirology 4:307–309
Musher DM, Thorner AR (2014) Community-acquired pneumonia. N Engl J Med 371:1619–1628
Van Buuren S (2018) Flexible Imputation of Missing Data, 2nd edn. Chapman & Hall/CRC Interdisciplinary Statistics, Boca Raton
Regression HF, Strategies M (2015) With Applications to Linear Models, Logistic and Ordinal Regression, and Survival Analysis. Springer, New York
Heymans MW, van Buuren S, Knol DL, van Mechelen W, de Vet HCW (2007) Variable selection under multiple imputation using the bootstrap in a prognostic study. BMC Med Res Methodol 7:33
Steyerberg EW (2009) Clinical Prediction Models. A Practical Approach to Development, Validation, and Updating. Springer, New York. https://doi.org/10.1007/978-3-030-16399-0
López-Ratón M, Rodríguez-Álvarez MX, Cadarso-Suárez C et al (2014) Optimal cutpoints: An R package for selecting optimal cutpoints in diagnostic tests. J Statistic Software 61:1–36
Marrie TJ, Lau CY, Wheeler SL et al (2000) A controlled trial of a critical pathway for treatment of community acquired pneumonia. CAPITAL Study Investigators. JAMA 283:749–755
Carratala J, Fernandez-Sabe N, Ortega L et al (2005) Outpatient care compared with hospitalization for community-acquired pneumonia: a randomized trial in low-risk patients. Ann Intern Med 142:165–172
Labarere J, Stone RA, Obrosky DS, et al (2006) Factors associated with the hospitalization of low-risk patients with community-acquired pneumonia in a cluster-randomized trial. J Gen Intern Med 745–52
Aujesky D, McCausland JB, Whittle J et al (2009) Reasons Why Emergency Department Providers do not Rely On the Pneumonia Severity ndex to determine the initial site of treatment for patients with pneumonia. Clin Infect Dis 49:e100–e108
Halm EA, Atlas SJ, Borowsky LH et al (2000) Understanding physician adherence with a pneumonia practice guideline. Arch Intern Med 160:98–104
Rosón B, Carratalà J, Dorca J et al (2001) Etiology, reasons for hospitalization, risk classes, and outcomes of community-acquired pneumonia in patients hospitalized on the basis of conventional admission criteria. CID 33:158–165
España PP, Capelastegui A, Quintana JM et al (2003) A prediction rule to identify allocation of inpatient care in community-acquired pneumonia. Eur Respir J 21:695–701
Goss CH, Rubenfeld GD, Park DR et al (2003) Cost and incidence of social comorbidities in low-risk patients with community-acquired pneumonia admitted to a public hospital. Chest 124:2148–2155
Arnold FW, Ramirez JA, McDonald LC et al (2003) Hospitalization for Community-Acquired Pneumonia. The Pneumonia Severity Index vs Clinical Judgment. Chest 124:121–4
Marrie TJ (2004) Deaths in risk classes I-III: a measure of quality of care in patients hospitalised with CAP? Eur Respir J 23:103–105
Calbo E, Ochoa de Echagüen A, Rodríguez-Carballeira M et al (2004) Hospital admission, duration of stay and mortality in community-acquired pneumonia in an acute care hospital. Correlation between a pneumonia prognosis index and conventional clinical criteria for assessing severity. Enferm Infecc Microbiol Clin 22:64–9
Marrie TJ, Huang JQ (2005) Low-risk patients admitted with community-acquired pneumonia. Am J Med 118:1357–1363
Buising KL, Thursky KA, Black JF et al (2006) A prospective comparison of severity scores for identifying patients with severe community acquired pneumonia: reconsidering what is meant by severe pneumonia. Thorax 61:419–424
Johnstone J, Eurich DT, Majumdar SR et al (2008) Long-term morbidity and mortality after hospitalization with community-acquired pneumonia. A population-based cohort study. Medicine 87:329–334
Singanayagam A, Chalmers JD, Hill AT (2009) Admission hypoglycaemia is associated with adverse outcome in community-acquired pneumonia. Eur Respir J 34:932–939
Chang CL, Mills GD, Karalus NC et al (2013) Biomarkers of cardiac dysfunction and mortality from community-acquired pneumonia in adults. PLoS One 8:e62612
Viasus D, Garcia-Vidal C, Simonetti A et al (2013) Prognostic value of serum albumin levels in hospitalized adults with community-acquired pneumonia. J Infect 66:415–423
Li J, Ye H, Zhao L (2015) B-type natriuretic peptide in predicting the severity of community-acquired pneumonia. World J Emerg Med 6:131–136
Violi F, Cangemi R, Falcone M et al (2017) Cardiovascular Complications and Short-term Mortality Risk in Community-Acquired Pneumonia. Clin Infect Dis 64:1486–1493
Ramirez JA, Wiemken TL, Peyrani P et al (2017) Adults hospitalized with pneumonia in the United States: Incidence, epidemiology, and mortality. Clin Infect Dis 65:1806–1812
Bramley AM, Reed C, Finelli L et al (2017) Relationship Between Body mass index and outcomes among hospitalized patients with community-acquired pneumonia. J Infect Dis 215:1873–1882
Çilli A, Sayner A, Çelenk B et al (2018) Antibiotic treatment outcomes in community-acquired pneumonia. Turk J Med Sci 48:730–736
Samaniego FA, Ghelfi AM, Rodríguez MS (2019) Neumonía adquirida en la comunidad de bajo riesgo: consecuencias evolutivas de los determinantes de internación no contemplados por los scores. Semergen 45:516–522
Peyrani P, Arnold FW, Bordon J et al (2020) Incidence and mortality of adults hospitalized with community-acquired pneumonia according to clinical course. Chest 157:34–41
Alonso R, Santillán-Barletta M, Rodríguez CL et al (2021) Community acquired pneumonia in patients requiring hospitalization. Medicina (B Aires) 81:37–46
Dwyer R, Kalin M (2021) Significance of the physician’s and the patient’s sex in hospitalized patients with community-acquired pneumonia. Infect Dis (Lond) 53:538–545
Chalmers JD, Rutherford J (2012) Can we use severity assessment tolos to increase outpatient management of community-acquired pneumonia? Eur J Intern Med 23:398–406
Stauble SP, Reichlin S, Dieterle T et al (2001) Community-acquired pneumonia: which patients are hospitalised? Swiss Med Wkly 131:188–192
Restrepo MI, Mortensen EM, Pugh JA et al (2006) COPD is associated with increased mortality in patients with community-acquired pneumonia. Eur Respir J 28:346–351
Funding
None.
Author information
Authors and Affiliations
Contributions
Adriana Lama Author and drafting, Conception and design, Approval of the final manuscript, Francisco Gude Author and drafting, Conception and design, Approval of the final manuscript, María Elena Toubes Co-author, Approval of the final manuscript, Ana Casal Co-author, Approval of the final manuscript, Jorge Ricoy Co-author, Approval of the final manuscript, Carlos Rábade. Co-author. Approval of the final manuscript, Nuria Rodríguez Núñez Co-author, Approval of the final manuscript, Ana Cao-Ríos Co-author, Approval of the final manuscript, Uxío Calvo Co-autor, Approval of the final manuscript, Luis Valdés Author and drafting, Conception and design, Approval of the final manuscript.
Corresponding author
Ethics declarations
Competing interest
We declare no conflicts of interest associated with this publication. This project did not receive financial support that could have influenced its outcome.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lama, A., Gude, F., Toubes, M.E. et al. Usefulness of a predictive model to hospitalize patients with low-risk community-acquired pneumonia. Eur J Clin Microbiol Infect Dis 43, 61–71 (2024). https://doi.org/10.1007/s10096-023-04683-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-023-04683-w