Abstract
Mycoplasma genitalium is a widespread sexually transmitted infection (STI) with growing rate of antimicrobials resistance. In our study, 137 vaginal and 131 urethral M. genitalium–positive swabs were sequentially collected through the work of Reference Center for STI during 2019. For prevalence evaluation of macrolide-resistance mutations three commercially available kits were used: AmpliSens® M. genitalium-ML/FQ-Resist-FL (Central Research Institute of Epidemiology, Russia), ResistancePlus® MG (SpeeDx, Australia), and S-DiaMGRes™ (Diagenode, Belgium). Macrolide resistance mutations were detected in 16% (43 of 268) of samples. Diagnostic characteristics were evaluated against Sanger sequencing. For AmpliSens® M. genitalium-ML/FQ-Resist-FL specificity was shown to be 100% (CI 95%, 98.4–100), and sensitivity was 90.7% (CI 95%, 77.9–97.4). ResistancePlus® MG specificity was 100% (CI 95%, 98.3–100), and sensitivity was 92.1% (CI 95%, 78.6–98.3). S-DiaMGRes™ specificity was shown to be 88.6% (CI 95%, 83.9–92.4), and sensitivity was 100% (CI 95%, 84.4–100). Mutations of parC gene region were detected in 14.5% (38 of 268) using AmpliSens® M. genitalium-ML/FQ-Resist-FL with further validation by Sanger sequencing. Of studied samples, 6.3% (17 of 268) contained both antimicrobials of class resistance mutations. Prevalence of macrolide-resistant M. genitalium in Moscow was 21.7% (23 of 106) and of fluoroquinolone-resistant M. genitaliuim was 20.8% (22 of 106). In Moscow region, macrolide-resistant M. genitalium were 12.3% (20 of 162) and 9.9% (16 of 162) of fluoroquinolone-resistant M. genitalium. All three kits can be used both for epidemiological monitoring of M. genitalium presence and mutation prevalence estimation. In Moscow, macrolide- and fluoroquinolone-resistant mutant prevalence increased in 3.9 and 2.7 times in 3 years.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mycoplasma genitalium is the facultative anaerobic smallest known microorganism with genome consisting of approximately 580-kb pairs [1]. Firstly discovered in 1981 [2], now M. genitalium is a cause of widespread STI with prevalence ranging from 0.7 to 3.3% in general population, 0.9% in pregnant women, 3.2% in men who have sex with men (MSM), and 15.9% in female sex workers [3]. Mycoplasma genitalium infection can lead to urethritis in males and cervicitis, endometritis, pelvic inflammatory disease, possibly preterm birth, tubal factor infertility, and ectopic pregnancy in females [4]. It has been shown that M. genitalium may be linked to increased risk of HIV infection [5].
Antimicrobial resistance is a worldwide rapidly emerging problem [6]. It is extremely difficult to culture M. genitalium which strictly limits culture-based antimicrobial tests [7]. Macrolides and fluoroquinolones are the most commonly used antimicrobials in treatment of M. genitalium infections. According to European guidelines on Mycoplasma genitalium infection [8], only azithromycin, josamycin, moxifloxacin pristinamycin, and doxycycline are the antimicrobials of choice, and in accordance with the BASHH UK national guidelines, moxifloxacin, doxycycline, pristinamycin, and minocycline are used [9].
It is not known for M. genitalium to carry any extrachromosomal DNA [10]; therefore, all antimicrobial resistances are determined by chromosomal mutations. Macrolides interact with A2058 (Escherichia coli numeration) position of 23S rRNA by formation of hydrogen bond [11]. Macrolide resistance in M. genitalium is determined by mutations in 23S rRNA gene V region [12]. The most common mutations are A2071 and A2072 (A2058 and A2059 E. coli numeration) [13]. It was also shown that mutations A752, C2038, and Т2185 (E. coli numeration) may lead to macrolide resistance [14]; furthermore, it was shown previously that A2062 position leads to failure in josamycin treatment [15]. Fluoroquinolones inhibit DNA gyrase and topoisomerase IV encoded by gyrA and parC genes, respectively, which are essential for bacterial DNA synthesis [16]. It was previously shown that G205A/Ala(69)→Thr, A248T/Ser(83)→Ile, and G259T/Asp87(84)→Tyr (E. coli numeration) mutations in parC gene region for M. genitalium are connected with fluoroquinolone resistance [17,18,19].
It was reported that, in Europe, rates of M. genitalium macrolide resistance usually exceeding 50% with a trend is still growing (r2 = 0.531; p = 0.101) [20]. In European guidelines on Mycoplasma genitalium infections, it is strongly recommended to follow every positive M. genitalium test with macrolide resistance detecting assay [8], as well as in the BASHH UK national guidelines [9].
AmpliSens® M. genitalium–ML/FQ-Resist-FL (Central Research Institute of Epidemiology) kit detects mutations in 23S rRNA gene V region and mutations in parC gene. It is a unique method based on oligonucleotide probes complimentary to wild type (WT) of M. genitalium which allows detecting maximal spectrum of resistance-associated mutations in abovementioned regions. Detection is carried out simultaneously with no mutation differentiation within one region. ResistancePlus® MG (SpeeDx) kit simultaneously detects A2058G, A2059G, A2058T, A2058C, or A2059C mutations on one channel. S-DiaMGRes™ (Diagenode) simultaneously detects mutations in A2058 and A2059 positions using FAM (Ex./Em.: 494/520 nm) and YY (Ex./Em.: 530/549 nm), respectively, thus resulted obtained allow differentiating position of studied sample. All of the used reagent kits simultaneously detect M. genitalium DNA and resistance-associated mutations.
In our study, we evaluated prevalence of urogenital M. genitalium macrolide and fluoroquinolone resistance mutations in samples collected in Moscow during 2019 year through the work of Reference Center for STI of Central Research Institute of Epidemiology (Moscow, Russia) and compared the performance commercially available kits: newly developed real-time PCR-based AmpliSens® M. genitalium–ML/FQ-Resist-FL (Central Research Institute of Epidemiology), ResistancePlus® MG (SpeeDx), and S-DiaMGRes™ (Diagenode). As a reference method, Sanger sequencing was used.
Materials and methods
Clinical samples
All samples have been sequentially collected from February to June of 2019 year at the Center of Molecular Diagnostics (Central Research Institute of Epidemiology, Russia) through the work of Reference Center for STI. In our study, we included only M. genitalium–positive 137 female vaginal and 131 male urethral swabs determined previously with «AmpliSens® N. gonorrhoeae/C. trachomatis/M. genitalium/T. vaginalis-MULTIPRIME-FRT» (Central Research Institute of Epidemiology, Russia). Age median of patients was 29 ± 5.64 years. One-hundred six samples were from Moscow, and 162 were from Moscow region. Only data on age and sex of the patients were presented with no data obtained on risk profile or sexual behavior. All collected samples were stored at –70 °C before further processing.
DNA extraction
All samples were extracted using DNA-sorb-AM (AmpliSens, Russia) extraction kit. Briefly, all samples were vortexed and centrifuged for 5 s to remove excess drops on the lid. After, 100 μl of clinical sample was placed in the 1.5-ml Eppendorf tube and proceeded according to manufacturer’s instructions. IC included in AmpliSens® M. genitalium–ML/FQ-Resist-FL (Central Research Institute of Epidemiology, Russia), ResistancePlus® MG (SpeeDx, Australia), and S-DiaMGRes™ (Diagenode, Belgium) reagents kits were added at the stage of extraction. All samples were eluted in 100 μl.
Amplification
Extractions for each used qPCR kit were performed separately using appropriate IC according to manufacturer’s instructions. Both ResistancePlus® MG and S-DiaMGRes™ assays were performed on CFX96® Touch while M. genitalium–ML/FQ-Resist-FL (AmpliSens) assay was performed on Rotor-Gene® 6000.
Amplification data analysis
Data obtained using S-DiaMGRes™ (Diagenode) assay was analyzed using CFX Manager v 3.1 according to manufacturer’s results interpretation guidelines. For ResistancePlus® MG (SpeeDx) Ugen Tech FastFined v 3.5.8 software was used. For M. genitalium–ML/FQ-Resist-FL (AmpliSens) excel macro-AmpliSens® M. genitalium–ML/FQ-Resist (RUS) was used.
Sequencing
Sanger sequencing of 23S rRNA gene region and parC QRDR was used as a reference standard method to determine mutations of macrolide resistance. Sanger sequencing was performed with previously described primers [12, 21]. Sequencing was performed using 3500 × l Genetic Analyzer (Applied Biosystems, USA). Sequences were analyzed using FinchTV v 1.4.0 software. All studied samples were sequenced.
Statistical analysis
Statistical analysis was performed using Westgard QC (http://tools.westgard.com) and Epitools (https://epitools.ausvet.com.au) websites.
Results
M. genitalium detection
Presence of M. genitalium in all of the 268 collected samples was confirmed by real-time PCR amplification of M. genitalium gyrB gene region with «AmpliSens® M. genitalium–screen-titer-FL» (Central Research Institute of Epidemiology, Russia) qPCR kit after thawing and DNA extraction. ResistancePlus® MG (SpeeDx) detected M. genitalium in 263 of 268 samples with positive agreement 98.1% while two other kits detected M. genitalium in every tested sample with positive agreement of 100%. In all the samples, appropriate IC was detected by qPCR. There was no difference shown for all three used reagents kits in performance on female vaginal or male urethral swabs.
M. genitalium macrolide resistance mutation detection
It was shown that 16% (43 of 268) samples contained 23S rRNA gene mutations after sequencing assay was performed (Table 1). All chromatograms were analyzed on having double peaks. In case of A2062 mutations, double peaks were always observed which allows us to conclude that all 5 samples had mixed M. genitalium strains containing both 23S rRNA gene mutants and wild-type cells.
For macrolide resistance detection only S-DiaMGRes™ had sensitivity of 100%, while AmpliSens® M. genitalium–ML/FQ-Resist-FL and ResistancePlus® MG had 92.1% and 90.7%, respectively. Specificities for AmpliSens® M. genitalium–ML/FQ-Resist-FL, ResistancePlus® MG, and S-DiaMGRes™ were determined as 100%, 100%, and 88.6%, respectively. Both specificity and sensitivity were determined using Sanger sequencing reference method (Table 2).
M. genitalium fluoroquinolone resistance mutations detection
Mycoplasma genitalium–ML/FQ-Resist-FL (AmpliSens) kit can also determine fluoroquinolone resistance mutations in QRDR of parC gene. As a reference method, Sanger sequencing of parC gene region containing positions A246, T247, A248, G249, G259, and A260 was used (Table 1). Of all specimens 14.5% (38 of 268) contained QRDR mutation. Sample with T251C mutation was excluded from calculations for it was not described previously as determining fluoroquinolone resistance. Analysis of sequencing chromatographs showed no additional or double peaks. Sensitivity and specificity AmpliSens® M. genitalium–ML/FQ-Resist-FL were 100% and 100%, respectively (Table 3).
Prevalence of M. genitalium mutants with parC and 23S gene mutations
It is of great importance that 6.3% (17 of 268) of M. genitalium specimens contained both macrolide and fluoroquinolone resistance mutations which were determined using AmpliSens® M. genitalium–ML/FQ-Resist-FL and verified with reference method.
Prevalence of M. genitalium mutants in Moscow and Moscow region
In our study, overall macrolide resistance prevalence was 16% (43 of 268). Prevalence of macrolide-resistant M. genitalium in Moscow was shown to be 21.7% (23 of 106) and 12.3% (20 of 162) in Moscow region. Prevalence of macrolide-resistant mutants among female patients was 9.5% (13 of 137), and the mean age of the group was 26.8 ± 5.2 while, among male patients, there were 22.9% (30 of 131) with the mean age 30 ± 4.4.
Overall prevalence of fluoroquinolone-resistant M. genitalium was shown to be 14.5% (38 of 268). Prevalence of QRDR M. genitalium in Moscow was shown to be 20.8% (22 of 106) and 9.9% (16 of 162) in Moscow region. Among female patients, it was shown to be 14.6% (20 of 137), and the mean age of the group was 27.6 ± 5.3, and among male patients was 13.7% (18 of 131) with the mean age 31.9 ± 5.4.
It was also shown that the amount of M. genitalium containing both macrolide and fluoroquinolone mutations was 3.7%.
Discussion
Antimicrobial resistance is one of the major concerns, both in sense of health and economics. According to Aslam et al. [22], growing threat of antibiotic resistance by 2050 will lead to death of 444 million people with economic cost of eliminating the problem amounting about $120 trillion. In our study, we evaluated three commercially available assays on detection of 23S rRNA gene mutants M. genitalium.
Commercial kits of analytical characteristics
It is important to highlight that we only included M. genitalium–positive samples in our study. Thus, sensitivity and specificity on M. genitalium detection could not be estimated. We only evaluated positive agreement with «AmpliSens® M. genitalium–screen-titer-FL» and further Sanger sequencing.
Diagnostic and analytical characteristics of both S-DiaMGRes™ and ResistancePlus® MG assays were previously evaluated in other studies. It is important to note that, in present study, no M. genitalium–negative samples were used. For ResistancePlus® MG, specificity was evaluated in our study as 100% (CI 95% 98.3−100) which is accordant to previous studies, and sensitivity was evaluated as 92.1% which is lower than sensitivity in other studies; however, this value is in the boundaries of CI 95% measured previously [23, 24]. For S-DiaMGRes™, specificity was 88.5% that differs from another study [25], while it has the highest sensitivity of 100%.
Discordant results
Of all the commercial kits, only S-DiaMGRes™ (Diagenode) allows to determine not only that M. genitalium has 23S mutations but also whether it is in A2058 or A2059 positions. In our study, using S-DiaMGRes™ 76.9% (30 of 39) 23S rRNA gene mutants were determined in accordance with Sanger sequencing. Among the discordant results, 63% (17 of 27) were false-positive results on WT samples, and 37% (10 of 27) were the discordant result on the type of mutation. Authors suggest that, even if correct type of mutation detection is important for research purposes, it is not of great importance for clinical use because both A2058 and A2059 mutations cause macrolide resistance. For M. genitalium–ML/FQ-Resist-FL (AmpliSens), the only discordant results obtained were in samples containing both WT and 23S A2062C mutants. Authors imply that having a mixed mutant A2062C and WT sample may have an impact on mutant detection for both types of amplicons are synthetized during multiplex qPCR, but further research on that is required. The only discordant results for ResistancePlus® MG (SpeeDx) were false-negative results on M. genitalium DNA detection in samples.
Prevalence of M. genitalium mutants in Moscow and Moscow region
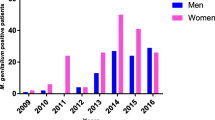
Two studies presented on antimicrobial-resistant M. genitalium mutant rate evaluation in Moscow and Moscow region. According to study performed on samples from dermatovenereological clinic in Moscow from 2014 to 2018, increment of macrolide-resistant mutants was 6.0% [13]. It was previously shown by Shipitsyna et al. [26] that prevalence of macrolide-resistant mutants in Moscow was 5.6% in samples collected 2013–2016.
Overall prevalence of fluoroquinolone-resistant M. genitalium was shown to be 14.5% (38 of 268). Prevalence of QRDR M. genitalium in Moscow was shown to be 20.8% (22 of 106) and 9.9% (16 of 162) in Moscow region. Among female patients, it was shown to be 14.6% (20 of 137), and the mean age of the group was 27.6 ± 5.3, and among male patients was 13.7% (18 of 131) with the mean age 31.9 ± 5.4. Samples obtained from dermatovenereological patients in Moscow from 2014 to 2018 had 7.1% fluoroquinolone-resistant M. genitalium mutants [13], and in Shipitsyna et al. study [26], prevalence of fluoroquinolone-resistant M. genitalium in Moscow was 7.6%.
Shipitsyna et al. [26] study was also performed using samples obtained from the Center of Molecular Diagnostics of Central Research Institute of Epidemiology in Moscow. Thus, authors imply that prevalence of antimicrobials resistance in Moscow increased at an alarming rate with macrolide resistance increasing in 3.9 times and of fluoroquinolone resistance—in 2.7 times in 3 years.
It was also shown that the amounts of M. genitalium containing both macrolide and fluoroquinolone mutations were dramatically increased in Moscow from 1.3% [26] to 3.7%, in 2.7 times.
Also, it is of interest that one of the QRDR mutants contained mutation T251C that was not described previously. We excluded it from statistical analysis for data on phenotypical susceptibility of fluoroquinolones was not presented. Further studies on the matter are required.
Conclusion
In our study, we evaluated the performance of three commercially available assays on macrolide resistance. AmpliSens® M. genitalium–ML/FQ-Resist-FL (Central Research Institute of Epidemiology, Russia), ResistancePlus® MG (SpeeDx, Australia), and S-DiaMGRes™ (Diagenode, Belgium) are able to determine 23S rRNA gene mutations. Thus, all of them can be used both for epidemiological monitoring of M. genitalium and mutation prevalence estimation. It was shown that prevalences of M. genitalium containing macrolide, fluoroquinolone, and both mutations in Moscow in 3 years from 2016 to 2019 were dramatically increased in 3.9, 2.7, and 2.8 times, respectively. Authors heavily imply that resistance surveillance on M. genitalium resistance mutations is of high importance and has to be included in clinical guidelines of the Russian Federation.
Data availability
All used reagent kits are commercially available.
References
Taylor-Robinson D, Jensen JS (2011) Mycoplasma genitalium: from chrysalis to multicolored butterfly. Clin Microbiol Rev 24:498–514. https://doi.org/10.1128/CMR.00006-11
Tully J, Cole R, Taylor-Robinson D, Rose D (1981) A newly discovered mycoplasma in the human urogenital tract. Lancet 317:1288–1291. https://doi.org/10.1016/S0140-6736(81)92461-2
Baumann L, Cina M, Egli-Gany D, Goutaki M, Halbeisen FS, Lohrer G-R et al (2018) Prevalence of Mycoplasma genitalium in different population groups: systematic review andmeta-analysis. Sex Transm Infect 94:255–262. https://doi.org/10.1136/sextrans-2017-053384
Lis R, Rowhani-Rahbar A, Manhart LE (2015) Mycoplasma genitalium infection and female reproductive tract disease: a meta-analysis. Clin Infect Dis 61:418–426. https://doi.org/10.1093/cid/civ312
Napierala Mavedzenge S, Weiss HA (2009) Association of Mycoplasma genitalium and HIV infection: a systematic review and meta-analysis. AIDS 23:611–620. https://doi.org/10.1097/QAD.0b013e328323da3e
Doyle M, Vodstrcil LA, Plummer EL, Aguirre I, Fairley CK, Bradshaw CS (2020) Nonquinolone Options for the treatment of Mycoplasma genitalium in the era of increased resistance. Open Forum Infect Dis 1:7. https://doi.org/10.1093/ofid/ofaa291
Gaydos CA (2017) Mycoplasma genitalium: accurate diagnosis is necessary for adequate treatment. J Infect Dis 216:S406–S411. https://doi.org/10.1093/infdis/jix104
Jensen JS, Cusini M, Gomberg M, Moi H (2016) 2016 European guideline on Mycoplasma genitalium infections. J Eur Acad Dermatol Venereol 30:1650–1656. https://doi.org/10.1111/jdv.13849
Soni S, Horner PJ (2019) Launch of the BASHH guideline for the management of M. genitalium in adults. Sex Transm Infect 95:237–237. https://doi.org/10.1136/sextrans-2018-053831
McGowin CL, Totten PA (2017) The unique microbiology and molecular pathogenesis of Mycoplasma genitalium. J Infect Dis 216:S382–S388. https://doi.org/10.1093/infdis/jix172
Dinos GP (2017) The macrolide antibiotic renaissance. Br J Pharmacol 174:2967–2983. https://doi.org/10.1111/bph.13936
Jensen JS, Bradshaw CS, Tabrizi SN, Fairley CK, Hamasuna R (2008) Azithromycin treatment failure in Mycoplasma genitalium–positive patients with nongonococcal urethritis is associated with Induced macrolide resistance. Clin Infect Dis 47:1546–1553. https://doi.org/10.1086/593188
Romanova IV, Kisina VI, Khayrullina GA, Frigo NV, Zhukova OV, Gushchin AE (2020) The prevalence and type of mutations of in dermatovenereological patients from the Moscow region for 2014–2018. Klin dermatologiya i Venerol 19:7. https://doi.org/10.17116/klinderma2020190117
Fyfe C, Grossman TH, Kerstein K, Sutcliffe J (2016) Resistance to macrolide antibiotics in public health pathogens. Cold Spring Harb Perspect Med 6:a025395. https://doi.org/10.1101/cshperspect.a025395
Guschin A, Ryzhikh P, Rumyantseva T, Gomberg M, Unemo M (2015) Treatment efficacy, treatment failures and selection of macrolide resistance in patients with high load of Mycoplasma genitalium during treatment of male urethritis with josamycin. BMC Infect Dis 15:40. https://doi.org/10.1186/s12879-015-0781-7
Blondeau JM (2004) Fluoroquinolones: mechanism of action, classification, and development of resistance. Surv Ophthalmol 49:S73–S78. https://doi.org/10.1016/j.survophthal.2004.01.005
Murray GL, Bodiyabadu K, Danielewski J, Garland SM, Machalek DA, Fairley CK et al (2019) The parC mutation G248T (S83I), and concurrent gyrA mutations, are associated with moxifloxacin and sitafloxacin treatment failure for Mycoplasma genitalium. J Infect Dis. https://doi.org/10.1093/infdis/jiz550
Hamasuna R, Le PT, Kutsuna S, Furubayashi K, Matsumoto M, Ohmagari N et al (2018) Mutations in ParC and GyrA of moxifloxacin-resistant and susceptible Mycoplasma genitalium strains. PLoS One 13:e0198355. https://doi.org/10.1371/journal.pone.0198355
Ke W, Li D, Tso LS, Wei R, Lan Y, Chen Z et al (2020) Macrolide and fluoroquinolone associated mutations in Mycoplasma genitalium in a retrospective study of male and female patients seeking care at a STI Clinic in Guangzhou, China, 2016-2018. BMC Infect Dis 20:950. https://doi.org/10.1186/s12879-020-05659-3
Fernández-Huerta M, Vall M, Fernández-Naval C, Barberá M-J, Arando M, López L et al (2020) Mycoplasma genitalium macrolide resistance update: Rate among a 2016–2017 cohort of patients in Barcelona. Spain Enferm Infecc Microbiol Clin 38:99–104. https://doi.org/10.1016/j.eimc.2019.06.008
Deguchi T (2001) Analysis of the gyrA and parC genes ofMycoplasma genitaliumdetected in first-pass urine of men with non-gonococcal urethritis before and after fluoroquinolone treatment. J Antimicrob Chemother 48:742–744. https://doi.org/10.1093/jac/48.5.742
Aslam B, Wang W, Arshad MI, Khurshid M, Muzammil S, Rasool MH et al (2018) Antibiotic resistance: a rundown of a global crisis. Infect Drug Resist 11:1645–1658. https://doi.org/10.2147/IDR.S173867
Tabrizi SN, Su J, Bradshaw CS, Fairley CK, Walker S, Tan LY et al (2017) Prospective evaluation of ResistancePlus MG, a new multiplex quantitative PCR assay for detection of Mycoplasma genitalium and macrolide resistance. J Clin Microbiol 55:1915–1919. https://doi.org/10.1128/JCM.02312-16
Su JP, Tan LY, Garland SM, Tabrizi SN, Mokany E, Walker S et al (2017) Evaluation of the SpeeDx ResistancePlus MG diagnostic test for Mycoplasma genitalium on the Applied Biosystems 7500 Fast Quantitative PCR Platform. J Clin Microbiol 1:56. https://doi.org/10.1128/JCM.01245-17
Le Roy C, Bébéar C, Pereyre S (2019) Clinical evaluation of three commercial PCR assays for the detection of macrolide resistance in Mycoplasma genitalium. J Clin Microbiol 4:58. https://doi.org/10.1128/JCM.01478-19
Shipitsyna E, Rumyantseva T, Golparian D, Khayrullina G, Lagos AC, Edelstein I et al (2017) Prevalence of macrolide and fluoroquinolone resistance-mediating mutations in Mycoplasma genitalium in five cities in Russia and Estonia. PLoS One 12:e0175763. https://doi.org/10.1371/journal.pone.0175763
Code availability
Not applicable.
Author information
Authors and Affiliations
Contributions
Study concept and design—Shedko E.D., Khayrullina G.A., Goloveshkina E.N.
Collection and processing of material—Shedko E.D., Khayrullina G.A.
Statistical analysis—Shedko E. D.
Writing—Shedko E.D.
Editing—Goloveshkina E.N., Akimkin V.G.
Approval of article final version—all co-authors.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Retrospective analysis of previously obtained samples and cross-sectional examination does not require ethical review.
Consent for publication
Not applicable.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shedko, E.D., Khayrullina, G.A., Goloveshkina, E.N. et al. Clinical evaluation of commercial PCR assays for antimicrobal resistance in Mycoplasma genitalium and estimation of resistance-mediated mutation prevalence in Moscow and Moscow region. Eur J Clin Microbiol Infect Dis 40, 1413–1418 (2021). https://doi.org/10.1007/s10096-021-04170-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-021-04170-0