Abstract
To determine whether target concentration non-attainment can be anticipated in critically ill patients prior to initiating empiric β-lactam antibiotic therapy based on readily available clinical factors. Retrospective review of consecutive patients treated with piperacillin or meropenem and who underwent therapeutic drug monitoring (TDM) at St Vincent’s Hospital (Sydney, Australia) between January 2013 and December 2015 was performed. Predefined subgroups were patients who received continuous renal replacement therapy (CRRT) and those who did not (non-CRRT). Potential risk factors were evaluated by correlation with β-lactam antibiotic trough concentrations (Cmin) lower than or equal to targeted minimum inhibitory concentration (MIC). Only the first drug concentration after initiation of the antibiotic treatment was included to reflect empirical dose selection. A total of n = 249 patients (piperacillin, n = 169; meropenem, n = 80) were investigated. For non-CRRT patients (n = 210), multivariate analysis demonstrated the following: male gender (p = 0.006); younger age (p = 0.015); prescribed daily antibiotic dose less than 1.5 times the product information recommendations (p = 0.004); lack of positive microbiology (p = 0.006); lower overall illness severity (p = 0.005); and estimated glomerular filtration rate (eGFR) ≥ 90 mL/min/1.73 m2 (p < 0.001), to be associated with Cmin ≤ MIC. No predictor variable was found to be significantly associated with Cmin ≤ MIC for the CRRT cohort. Evaluating the risk of target concentration non-attainment using simple clinical factors is possible at the bedside for non-CRRT patients prior to empiric antibiotic initiation. Clinicians should be wary of selecting doses based on the product information especially when treating younger male patients with apparently ‘normal’ renal function.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
β-lactam antibiotics are routinely prescribed as empirical therapy for sepsis in the intensive care unit (ICU) [1]. Early and appropriate antibiotic initiation has been associated with improved clinical outcomes [2,3,4] yet consistent target concentration attainment in ICU patients remains an unresolved challenge [5]. This is critical given that empiric therapy within the first 48 h of sepsis significantly determines hospital mortality [6]. Therapeutic drug monitoring (TDM) has been proposed as a potential means of optimising β-lactam administration [7]; however, its application in clinical practice has thus far been limited [8].
Bactericidal activity of β-lactams depends on the time for which drug concentrations exceed the minimum inhibitory concentration (MIC) of the infective pathogen during a dosing period (T > MIC) [9]. Patients with T > MIC of 100% have been shown to have significantly higher rates of clinical cure and bacteriological eradication [10]. To achieve a pharmacodynamic target of 100% T > MIC, antibiotic trough concentrations (Cmin) must be kept above the MIC. At present, ~ 40% of critically ill patients fail to achieve the target of Cmin > MIC for 100% of the dosing interval [11].
This study aimed to determine whether target non-attainment can be anticipated at the bedside in ICU patients by evaluating the correlation between patient and clinical factors and the presence of sub-therapeutic β-lactam trough concentrations. Identifying at risk patients could prompt physicians to consider more aggressive empiric dosing regimens and regular monitoring of drug concentrations, where available. A retrospective cohort review of consecutive ICU patients treated with either of two commonly prescribed intravenous β-lactam agents, piperacillin and meropenem, and who underwent TDM was conducted.
Patients and methods
Study selection and data source
A β-lactam TDM program was implemented as part of routine care in a 20-bed ICU at a tertiary-level teaching hospital in Sydney. At our hospital, patients being treated for suspected or microbiologically confirmed sepsis are targeted for TDM, at the discretion of the treating clinician. β-lactam antibiotic concentrations are determined in EDTA plasma, using a previously validated high-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS) assay.
All TDM results between January 2013 to December 2015 were extracted from electronic records. Antibiotic trough concentrations were defined as those collected < 2 h prior to the subsequent dose, with the Cmin closest to the time of first ICU antibiotic dose considered to reflect empiric dose selection. Predefined subgroups were patients who received continuous renal replacement therapy (CRRT) and those who did not (non-CRRT). Dosing regimens were prescribed based on the product information (PI) for each antibiotic and adjusted for patient renal function. Baseline eGFR was calculated based on serum creatinine using the Chronic Kidney Disease Epidemiology Collaboration formula [12].
Inclusion and exclusion criteria
Patients were included if they were treated with intravenous piperacillin or meropenem during ICU admission within the study period and had at least one blood sample for TDM assay collected at Cmin as previously defined. There were no specified exclusion criteria.
Target concentration attainment
Laboratory derived MICs were used as target endpoints where available. Based on our laboratory protocol, positive cultures collected from non-sterile sites have no MIC determined. Hence, for these samples, a target MIC based on the relevant European Committee for Antimicrobial Susceptibility Testing (EUCAST) clinical breakpoint (http://www.eucast.org/clinical_breakpoints) was assigned. For patients with no positive culture, the EUCAST clinical breakpoints for Pseudomonas spp. (piperacillin = 16 mg/L and meropenem = 2 mg/L) were considered as a ‘worst-case’ empiric target MIC. Target concentration attainment (Cmin > MIC) and non-attainment (Cmin ≤ MIC) were conservatively defined.
Predictor variables
Factors likely to contribute to target non-attainment at the time of antibiotic initiation were recorded and analysed for association. These included patient characteristics (age, gender, body mass index (BMI)), overall illness severity (Sequential Organ Failure Assessment (SOFA) score), antibiotic prescribed (meropenem or piperacillin), hypoalbuminemia (serum albumin < 34 g/L), prescribed: PI recommended daily dose (ratio) < 1.5, availability of a positive culture result and estimated glomerular filtration rate (eGFR).
Statistics
Statistical analyses were performed using Stata14 (StataCorp LP, College Station, TX, USA). Continuous variables are expressed as mean ± standard deviation (SD) for normally distributed data or median (interquartile range (IQR)) for non-parametric data. Categorical variables are presented as numbers (percentages). Factors affecting target non-attainment were analysed through a generalised linear model, with variables associated with a p < 0.1 in univariate analysis included. A p < 0.05 was considered statistically significant. The performance of eGFR as an independent risk factor for target non-attainment was assessed in the non-CRRT group using a receiver operating characteristic (ROC) curve. Simple logistical regression was used to predict the risk of target non-attainment with increasing eGFR.
Results
A total of n = 249 patients were included in the study (piperacillin, n = 169; meropenem, n = 80). Baseline patient characteristics have been summarised in Table 1. The median (IQR) time (h) to first drug assay was 45 (30–90) for piperacillin and 56 (31–86) for meropenem patients. Only 7% (n = 18) of patients were prescribed an antibiotic dosing schedule below the PI recommendations. Target non-attainment was more common amongst patients treated with piperacillin (n = 53; 31%) than those treated with meropenem (n = 12; 15%).
Infection was confirmed on the basis of microbiological cultures in 37% of study patients. A laboratory-derived MIC was available for 42% of all positive cultures. Considering those patients with positive cultures, the source of infection was frequently blood (36%) and respiratory (32%) sites. The most commonly identified pathogens were Pseudomonas aeruginosa (26%), Escherichia coli (19%) and Klebsiella pneumoniae (15%). Of all patients with laboratory confirmed infection, 85% (piperacillin, n = 45; meropenem, n = 32) survived to discharge from hospital.
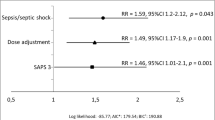
Antibiotic groups were combined and predictor variables for target non-attainment were investigated. For non-CRRT patients, in multivariate analysis (Table 2) of patient characteristics, male gender (p = 0.006) and younger age (p = 0.015) were found to be significantly correlated with target non-attainment. A daily prescribed antibiotic dose less than 1.5 times the PI recommendations (p = 0.004) and the lack of positive microbiology (p = 0.006) were also associated with target non-attainment. Target non-attainment was more frequently observed in patients with lower SOFA scores (p = 0.005).
Baseline eGFR ≥ 90 mL/min/1.73 m2 demonstrated statistically significant association (p < 0.001) with target non-attainment. Controlling for confounders, the odds of a critically ill patient with apparent ‘normal’ renal function (eGFR ≥ 90 mL/min/1.73 m2) attaining targets was 0.33 (95% CI, 0.10–0.51; p < 0.001). The performance of eGFR as an independent predictor for target non-attainment was assessed using a ROC curve, with an area under the curve equal to 0.76 (95% CI, 0.70–0.83; p < 0.001). The eGFR threshold value of ≥ 71.5 mL/min/1.73 m2 had a sensitivity and specificity of 77% and 65%, respectively. The predicted risk of target non-attainment was determined at laboratory reported ranges of eGFR (Fig. 1).
Patients receiving CRRT (n = 39) were analysed separately to non-CRRT patients. Rates of target non-attainment were slightly lower amongst CRRT (23%) compared to non-CRRT patients (27%), although this was not statistically relevant (p = 0.069). Multivariate analysis found no predictor variable to be significantly associated with target non-attainment for the CRRT cohort (Table S1, available as supplementary data).
Discussion
The findings of our study suggest that target non-attainment during β-lactam therapy is common in ICU but may be anticipated at the bedside prior to antibiotic initiation for non-CRRT patients. To the best of our knowledge, only one other large-scale study has attempted to quantify risk factors associated with target non-attainment [13]. Our results not only help validate their findings regarding patient demographics and overall illness severity but also investigate additional risk factors, notably ‘normal’ eGFR.
Although previous literature has demonstrated the direct relationship between measured glomerular filtration rate and β-lactam clearance [14], this has limited clinical applicability. Measured glomerular filtration rate based on 24-h urine creatine clearance is not routinely undertaken in clinical practice [15]. When performed, results are unlikely to be available to clinicians during empiric antibiotic dose selection. Hence, the ability to rapidly predict the risk of target non-attainment using readily available eGFR retains substantial clinical value. As such, our novel finding that increasing eGFR is an independent marker for low trough β-lactam concentration without the need for additional diagnostic interventions is highly pertinent.
Our study corroborates previously published findings that younger age, male gender and lower overall illness severity increase the odds of target non-attainment in ICU patients [16,17,18]. These characteristics are thought to offer an underlying physiological reserve to critically ill patients [19] and contribute to target non-attainment by facilitating enhanced/augmented renal clearance (ARC). In our cohort, an eGFR ≥ 90 mL/min/1.73 m2 independently associated with age < 45 years (OR = 2.94, 95% CI 1.59–5.43; p < 0.001) and a SOFA score < 4 (OR = 3.13, 95% CI 1.74–5.65; p < 0.001)). In light of this association, it remains pertinent that predictions of ARC risk using eGFR still be made within clinical context and consideration of additional patient factors.
Our findings also pertain to the empiric prescription of β-lactams and the targeting of TDM services. With target non-attainment observed in > 25% of our study cohort, larger empiric doses could be warranted in ICU, particularly when microbial resistance rates are often higher [20]. We found a prescribed daily dose ≥ 1.5 times the PI recommendations was associated with better target attainment. Concerns about toxicity secondary to drug accumulation are valid but less pertinent given that toxicity thresholds are high for these agents [21]. In our cohort, only one patient treated with piperacillin exceeded these thresholds at the time of first drug assay. Additionally, identifying populations which may benefit from β-lactam TDM remains contentious as clinical practice guidelines are not yet established [22]. As such, if patients deemed to be at high-risk of target non-attainment could be identified based on risk factors, daily TDM with adaptive feedback may be selectively applied during the early stages of empiric therapy.
Our study has a number of limitations. First, it is a retrospective single-centre design which may hamper its generalisability to all critical care settings. Second, although common across most ICU populations and therefore realistic, actual MIC values were only available for less than half of the studied cohort. As true MIC is often substantially lower than epidemiological breakpoints this method may potentially inflate the frequency of target non-attainment [23]. To mitigate this, conservative pharmacokinetic/pharmacodynamic targets were adopted even though the efficacy of β-lactams is considered optimised when Cmin > 4 × MIC [24]. However, the ongoing effect of using epidemiologically derived breakpoints is evident in our finding that positive microbiology results were associated with better target attainment.
Conclusion
Evaluating the risk of target non-attainment is possible at the bedside for non-CRRT patients prior to antibiotic initiation through interpretation of simple patient factors and clinical findings. Although further large-scale and prospective studies are required to validate our results, our study suggests that current dosing guidelines should be revised with consideration of high-risk patients.
Change history
17 June 2021
A Correction to this paper has been published: https://doi.org/10.1007/s10096-021-04291-6
References
Dulhunty JM, Webb SA, Paterson DL, Bellomo R, Myburgh J, Roberts JA et al (2010) A survey of antibiotic prescribing practices in Australian and New Zealand intensive care units. Crit Care Resusc 12:162
Kollef MH, Sherman G, Ward S, Fraser VJ (1999) Inadequate antimicrobial treatment of infections: a risk factor for hospital mortality among critically ill patients. Chest 115:462–474
Alp E (2016) Right first time! Ann Transl Med 4:331
Ferrer R, Martin-Loeches I, Phillips G, Osborn TM, Townsend S, Dellinger RP et al (2014) Empiric antibiotic treatment reduces mortality in severe sepsis and septic shock from the first hour: results from a guideline-based performance improvement program. Crit Care Med 42:1749–1755
Taccone FS, Laterre PF, Dugernier T, Spapen H, Delattre I, Wittebole X et al (2010) Insufficient β-lactam concentrations in the early phase of severe sepsis and septic shock. Crit Care 14:R126
Liu VX, Fielding-Singh V, Greene JD, Baker JM, Iwashyna TJ, Bhattacharya J et al (2017) The timing of early antibiotics and hospital mortality in sepsis. Am J Respir Crit Care Med 196:856–863
Roberts JA, Norris R, Paterson DL, Martin JH (2012) Therapeutic drug monitoring of antimicrobials. Br J Clin Pharmacol 73:27–36
Muller AE, Huttner B, Huttner A (2018) Therapeutic drug monitoring of beta-lactams and other antibiotics in the intensive care unit: which agents, which patients and which infections? Drugs 23:1–13
Craig WA (1998) Pharmacokinetic/pharmacodynamic parameters: rationale for antibacterial dosing of mice and men. Clin Infect Dis 26:1–10
McKinnon PS, Paladino JA, Schentag JJ (2008) Evaluation of area under the inhibitory curve (AUIC) and time above the minimum inhibitory concentration (T > MIC) as predictors of outcome for cefepime and ceftazidime in serious bacterial infections. Int J Antimicrob Agents 31:345–351
Roberts JA, Paul SK, Akova M, Bassetti M, De Waele JJ, Dimopoulos G et al (2014) DALI: defining antibiotic levels in intensive care unit patients: are current β-lactam antibiotic doses sufficient for critically ill patients? Clin Infect Dis 58:1072–1083
Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, Feldman HI et al (2009) A new equation to estimate glomerular filtration rate. Ann Intern Med 150:604–612
De Waele JJ, Lipman J, Akova M, Bassetti M, Dimopoulos G, Kaukonen M et al (2014) Risk factors for target non-attainment during empirical treatment with β-lactam antibiotics in critically ill patients. Intensive Care Med 40:1340–1351
Conil JM, Georges B, Mimoz O, Dieye E, Ruiz S, Cougot P et al (2006) Influence of renal function on trough serum concentrations of piperacillin in intensive care unit patients. Intensive Care Med 32:2063–2066
Schaeffner E (2017) Determining the glomerular filtration rate—an overview. J Ren Nutr 27:375–380
Udy AA, Roberts JA, Shorr AF, Boots RJ, Lipman J (2013) Augmented renal clearance in septic and traumatized patients with normal plasma creatinine concentrations: identifying at-risk patients. Crit Care 17:R35
Claus BO, Hoste EA, Colpaert K, Robays H, Decruyenaere J, De Waele JJ (2013) Augmented renal clearance is a common finding with worse clinical outcome in critically ill patients receiving antimicrobial therapy. J Crit Care 28:695–700
Huttner A, Von Dach E, Renzoni A, Huttner BD, Affaticati M, Pagani L et al (2015) Augmented renal clearance, low β-lactam concentrations and clinical outcomes in the critically ill: an observational prospective cohort study. Int J Antimicrob Agents 45:385–392
Thomas DM, Coles GA, Williams JD (1994) What does the renal reserve mean? Kidney Int 45:411–416
Brusselaers N, Vogelaers D, Blot S (2011) The rising problem of antimicrobial resistance in the intensive care unit. Ann Intern Med 1:47
Imani S, Buscher H, Marriott D, Gentili S, Sandaradura I (2017) Too much of a good thing: a retrospective study of β-lactam concentration-toxicity relationships. J Antimicrob Chemother 72:2891–2897
Huttner A, Harbarth S, Hope WW, Lipman J, Roberts JA (2015) Therapeutic drug monitoring of the β-lactam antibiotics: what is the evidence and which patients should we be using it for? J Antimicrob Chemother 70:3178–3183
Woksepp H, Hällgren A, Borgström S, Kullberg F, Wimmerstedt A, Oscarsson A et al (2017) High target attainment for β-lactam antibiotics in intensive care unit patients when actual minimum inhibitory concentrations are applied. Eur J Clin Microbiol Infect Dis 36:553–563
Tam VH, McKinnon PS, Akins RL, Rybak MJ, Drusano GL (2002) Pharmacodynamics of cefepime in patients with gram-negative infections. J Antimicrob Chemother 50:425–428
Acknowledgements
The preliminary results of this study were presented at the 25th European Congress of Clinical Microbiology and Infectious Disease. We wish to thank the staff of the TDM Laboratory, SydPath (assaying of specimens) and Intensive Care (collection of specimens), St Vincent’s Hospital.
Funding
This work was supported by the University of Notre Dame Australia at the School of Medicine Sydney (NO333732 to SI) and an Australian Government Research Training Program (RTP) Scholarship to IS.
Author information
Authors and Affiliations
Contributions
SI collected data and prepared the manuscript. HB, DM, RD and RN assisted with study design and reviewed the manuscript. GJ assisted with study design, data collection and reviewed the manuscript. SG performed data analysis and reviewed the manuscript. IS conceived and designed the study, performed preliminary data analysis and reviewed the manuscript.
Corresponding author
Ethics declarations
Ethics
Ethical review and approval was obtained through St Vincent’s Hospital Sydney Human Research Ethics Committee (Reference: LNR/14/SVH/148, SSA: LNRSSA/14/SVH/226).
Competing interests
The authors declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOCX 15 kb)
Rights and permissions
About this article
Cite this article
Imani, S., Buscher, H., Day, R. et al. An evaluation of risk factors to predict target concentration non-attainment in critically ill patients prior to empiric β-lactam therapy. Eur J Clin Microbiol Infect Dis 37, 2171–2175 (2018). https://doi.org/10.1007/s10096-018-3357-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-018-3357-9