Abstract
Carbapenem-resistant Enterobacteriaceae represents a major public health issue. This study investigated the clonality and resistance mechanisms of 92 carbapenem-resistant E. coli (n = 21) and K. pneumoniae (n = 71) isolates collected consecutively from clinical specimens and patients at high risk of carriage between 2010 and 2012 in a healthcare region in Hong Kong. Combined disk tests (CDTs) and the Carba NP test were used for phenotypic detection of carbapenemases. PCR assays were used to detect carbapenemase genes. All isolates were intermediate or resistant to at least one carbapenem. Nine (9.8 %) isolates were genotypic carbapenemase producers and included six K. pneumoniae (one ST1306/bla IMP-4, one ST889/bla IMP-4, two ST11/bla KPC-2, one ST258/bla KPC-2, one ST483/bla NDM-1) and three E. coli (one ST131/bla IMP-4, two ST744/ bla NDM-1) isolates. All nine isolates carrying carbapenemase genes could be detected by the CDTs and the Carba NP test. PCR identified bla CTX-M and bla AmpC alone or in combination in 77.8 % (7/9) and 96.4 % (80/83) of the carbapenemase-producers and non-producers, respectively. Porin loss was detected in 22.2 % (2/9) and 59.0 % (49/83) of the carbapenemase-producers and non-producers, respectively. Overall, the E. coli clones were diverse (14 different STs), but 36.6 % (26/71) of the K. pneumoniae isolates belonged to ST11. In conclusion, the prevalence of carbapenemases among carbapenem-nonsusceptible E. coli and K. pneumoniae remained low in Hong Kong. Porin loss combined with AmpC and/or CTX-M type ESBL was the major mechanism of carbapenem resistance in the study population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Carbapenem-resistant Enterobacteriaceae (CRE) is a major public health challenge because therapeutic alternatives are severely limited and serious infections are associated with poor outcome [1]. Carbapenem resistance can be mediated by the production of carbapenemases (i.e. carbapenemase-producing Enterobacteriaceae, CPE) or by the combination of porin loss and production of AmpC or extended-spectrum β-lactamases (ESBLs) [2]. Of particular concern is the emergence of acquired carbapenemases among E. coli and K. pneumoniae because the two species are common causes of many human infections. Recent epidemiological studies have highlighted the potential of acquired carbapenemase producers to cause hospital outbreaks and to become endemic in healthcare settings [2, 3]. As a consequence, detecting the production of carbapenemases and distinguishing between carbapenemases and other mechanisms of carbapenem resistance has become an important task for diagnostic laboratories [4].
Globally, the major types of carbapenemases include KPC, NDM, IMP, VIM and OXA. Both epidemic clones and mobile genetic elements including plasmids, transposons and integrons are involved in their dissemination [1, 5]. The IMP and NDM type carbapenemases are most frequently reported in the Asia-Pacific region [5, 6]. Unlike KPC type carbapenemases, IMP and NDM producers may not exhibit high level resistance to the carbapenems, especially imipenem and meropenem [1, 5, 7]. False negative screening and phenotypic tests for IMP and NDM producing isolates have been described [4, 8]. In Hong Kong, the prevalence of carbapenem resistance among clinical isolates of E. coli and K. pneumoniae remains low [9]. Most reports concern sporadic isolates introduced by patients from high-prevalence areas [5, 7, 10]. The aim of this study was to investigate the clonality and resistance mechanisms of carbapenem-resistant E. coli and K. pneumoniae. The ability of two widely used phenotypic tests to distinguish between carbapenemase producers and non-producers were described.
Materials and methods
Bacterial strains, identification and antimicrobial susceptibility testing
Seventy-one carbapenem-nonsusceptible Klebsiella pneumoniae and 21 E. coli isolates were included in the study. The isolates were recovered consecutively between September 2010 and December 2012 from inpatients treated in a healthcare region in Hong Kong comprising of a University-affiliated hospital with 1600 beds, three extended-care hospitals with a total of 1600 beds, and one paediatric hospital with 160 beds. The collection included 13 clinical isolates (from blood, wound, urine and other body fluids) and 79 stool isolates. In the hospital, carbapenem resistance among K. pneumoniae and E. coli clinical isolates were rare (<0.1 %) [9, 11]. As part of the healthcare region’s infection control policy, a combination of active surveillance cultures and “added tests” as described previously [9] were done to identify high risk patients carrying multiple drug-resistant organisms and to opportunistically identify faecal carriage of CRE among stool specimens sent for bacterial culture respectively. Admission screening was implemented for all inpatients with a history of hospitalization or surgical operation outside Hong Kong in the past 12 months [12]. Faecal samples or rectal swabs were plated on MacConkey plates supplemented with 1 μg/ml meropenem [12]. Colonies that grew on the plates were identified to species level by the VITEK GNI system (bioMerieux Vitek Inc., Hazelwood, MO, USA). Susceptibility testing of the isolates was performed by the disk diffusion method [11]. On each day of testing, standard strains (ATCC 25922 and 35218) were included as quality controls. Organisms identified as members of the Enterobacteriaceae were investigated further if they were found to be nonsusceptible to one or more of ertapenem (<22 mm, >0.5 μg/ml), imipenem (<23 mm, >1 μg/ml) and meropenem (<23 mm, >1 μg/ml) [11]. The 92 isolates were recovered from 88 patients, of which four patients had both K. pneumoniae and E. coli. All available isolates during the study period were included.
Phenotypic detection of carbapenemases
Two tests were used for phenotypic detection of carbapenemases. The combined disk test (CDT) was performed by using a previously described procedure [13]. Ethylene diamine tetra-acetic acid (EDTA) and 3-aminophenyl boronic acid (APBA) were obtained from Sigma. Two sets each of ertapenem (ETP, 10 μg), imipenem (IPM, 10 μg), meropenem (MEM, 10 μg) (all from Becton Dickinson) were placed onto Mueller-Hinton (MH) agar (EDTA-CDT) and MH agar containing 250 μg/ml cloxacillin agar (APBA-CDT) inoculated with the test isolate [13]. Immediately after the disks were placed onto the agar, 10 μl of a 29.2 mg/ml EDTA or a 30 mg/ml APBA solution was added to one of the two carbapenem disks in each set. The agar plates were incubated at 37 °C overnight. An increase of ≥5 mm in zone diameter around disks containing the β-lactamase inhibitor (APBA or EDTA), as compared with the carbapenem disk alone, was considered to be a positive result. EDTA-CDT positive and APBA-CDT positive results were used to indicate the possible presence of metallo-β-lactamase (MBL) and class A carbapenemase, respectively [13]. The chromogenic carbapenemase detection assay based on hydrolysis of imipenem, Carba NP test, was carried out and interpreted according to the Clinical and Laboratory Standards Institute (CLSI 2015, M100-S15) [11]. All strains were grown at 37 °C for 24 hours in Columbia agar (Becton Dickinson) supplemented with 5 % horse blood. Commercial protein extract reagent (B-PER-II, Thermo Scientific Pierce, IL, USA) was used for cell lysis. K. pneumoniae ATCC BAA-1705 and BAA-1706 were included as controls in each testing.
Molecular studies
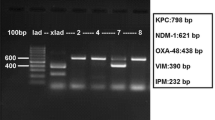
PCR assays were used to detect carbapenemase genes (class B families: bla IMP, bla NDM, bla SPM, bla SIM, bla GIM, bla VIM; class A families: bla KPC, bla GES, bla SME, bla IMI, bla SFO, bla IBC, bla PER, bla SPC; and class D family bla OXA-48), acquired bla AmpC genes (subgroups bla MOX, bla CIT, bla DHA, bla ACC, bla BEC, bla FOX), and bla CTX-M genes (subgroups bla CTX-M1G, bla CTX-M2G, bla CTX-M8G, bla CTX-M9G, bla CTX-M25G) [5, 10, 14]. The alleles of bla IMP, bla NDM and bla KPC were determined by PCR and sequencing of the full length of genes [5, 7, 10]. PCR assays were used to assign the E. coli isolates to phylogroups [15]. Multilocus sequence typing (MLST) of K. pneumoniae and E. coli isolates was carried out using the Pasteur Institute and the Warwick scheme, respectively [16, 17], and results were analysed by eBURST v3 [18]. Sequence types (STs) were assigned to clonal complexes (CCs) when there were at least three isolates. STs with one or two isolates were designated as singletons. CCs were generated by using the stringent group definition (6/7 shared alleles). STs within a CC were believed to be descended from the same founding genotype [18].
Isolation and analysis of porins
Bacterial outer membrane porins were analysed by standard techniques [19]. In brief, isolates were grown in Luria broth, sonicated and centrifuged. Porins were separated by 12 % SDS-PAGE gel containing 6 M urea and stained with 0.125 % Coomassie brilliant blue. Two reference strains, K. pneumoniae ATCC 13883 and E. coli ATCC 25922 were included as controls in each run.
Results
Antimicrobial susceptibilities
Among the 92 isolates analysed, rates of nonsusceptibility to ertapenem, imipenem and meropenem were 98.9 % (91/92), 91.3 % (84/92) and 95.7 % (88/92), respectively (Online Resource, Table S1). Some discordant results to the three carbapenems (ertapenem, imipenem, meropenem) were observed: 81 (88.0 %) isolates were nonsusceptible to three carbapenems, nine (9.8 %) isolates were nonsusceptible to two carbapenems and two (2.1 %) were nonsusceptible to one carbapenem. Co-resistance to other β-lactams, aminoglycosides, ciprofloxacin and cotrimoxazole was common.
Phenotypic and PCR analyses for carbapenemases
In the CDTs, the proportions of isolates with positive phenotypic MBL (i.e. EDTA-CDT positive) and class A carbapenemase (i.e. APBA-CDT positive) results in at least one carbapenem disk were 6.5 % (6/92) and 4.3 % (4/92), respectively (Table 1). Valid results were obtained for all the isolates in the Carba NP test, including nine positives and 83 negatives. PCR and sequencing showed that the nine Carba NP-positive isolates were positive for bla NDM-1 (n = 3), bla IMP-4 (n = 3) or bla KPC-2 (n = 3) genes. No carbapenemase genes were detected in the 83 Carba NP negative isolates. Besides bla NDM, bla IMP and bla KPC, no other carbapenemase genes were detected in the isolates. Two bla IMP-4 isolates had false-negative results in the IPM EDTA-CDT (strain 1058) and ETP EDTA-CDT (strain 249), but positive results were obtained for the other carbapenem disks (Table 1). EDTA-CDT and APBA-CDT (IPM, MEM and ETP) gave true positive results for the remaining seven carbapenemase producers. One isolate (strain 15) had false-positive APBA-CDT (MEM and ETP) result (Table 1).
ESBL, AmpC and porin loss
At least one of the β–lactam resistance mechanisms (bla CTX-M, bla AmpC, porin loss) was detected in all except two K. pneumoniae isolates (Table 2). Overall, bla CTX-M, bla AmpC and porin loss were detected in 60.9 % (56/92), 73.9 % (68/92) and 55.4 % (51/92) of all isolates. bla CTX-M and bla AmpC alone or in combination was detected in 77.7 % (7/9) and 96.4 % (80/83) of the carbapenemase producers and non-producers, respectively. Porin loss was detected in 22.2 % (2/9) and 59.0 % (49/83) of the producers and non-producers, respectively.
The bla CTX-M gene was more frequently detected in E. coli (85.7 %, 18/21) than in K. pneumoniae (53.5 %, 38/71) isolates (Table 2). Among E. coli and K. pneumoniae, bla CTX-M1G alone, bla CTX-M9G alone and both bla CTX-M1G and bla CTX-M9G were found in 42.9 % (9/21) and 15.5 % (11/71), 28.6 % (6/21) and 14.1 % (10/71), and 14.3 % (3/21) and 23.9 % (17/71), respectively.
bla AmpC genes were detected in 33.3 % (7/21) of E. coli and 85.9 % (61/71) of K. pneumoniae isolates (Table 2). The acquired bla AmpC genes in E. coli included five (23.8 %) bla CIT and two (9.5 %) bla DHA. In K. pneumoniae, bla DHA alone, bla CIT alone and both bla DHA and bla CIT were detected in 57 (80.3 %), three (4.2 %) and one (1.4 %) isolates, respectively. Among E. coli and K. pneumoniae, porin loss was detected in 85.7 % (18/21) and 46.5 % (33/71) of isolates, respectively (Table 2). No bla MOX, bla ACC, bla BEC and bla FOX genes were detected.
Among E. coli isolates, 16 (76.2 %) displayed both OmpC and OmpF loss and two (9.5 %) displayed OmpC loss (Table 2). Among K. pneumoniae isolates, 24 (33.8 %) displayed OmpK36 loss, one (1.4 %) displayed OmpK35 loss, and eight (11.3 %) displayed both OmpK35 and OmpK36 loss.
Clonal structure
MLST analyses revealed 14 and 29 different STs among the E. coli and K. pneumoniae isolates, respectively (Fig. 1). The E. coli isolates could be grouped into three clonal complexes (CCs), including CC10/phylogroups A or B1 (52.4 %, 11/21), CC131/phylogroup B2 (14.3 %, 3/21) and CC405/phylogroup D (14.3 %, 3/21) and four singletons. In contrast, the majority (77.5 %, 55/71) of the K. pneumoniae isolates could be grouped to CC37, among which ST11 (36.6 %, 26/71) was the predominant ST. No association was found between the types of bla CTX-M, bla AmpC and porin loss, and the clones of E. coli and K. pneumoniae. The bla DHA gene was found in 25 different STs of K. pneumoniae. The STs and carbapenemase gene combinations of the nine CPE isolates were as follows: K. pneumoniae (one ST1306/bla IMP-4, one ST889/bla IMP-4, two ST11/bla KPC-2, one ST258/bla KPC-2, one ST483/bla NDM-1); E. coli (one ST131/bla IMP-4, two ST744/ bla NDM-1).
Clonal structure of 21 E. coli and 71 K. pneumoniae isolates non-susceptible to carbapenems. The population snapshots were generated by eBURST v3. The STs detected in this study were labelled with arrows pointing to the circle. When more than one isolate in the indicated ST was found, the number of isolates was indicated inside the bracket. (a) all strains from the E. coli MLST database at the University of Warwick (http://mlst.warwick.ac.uk/mlst/dbs/Ecoli, last accessed on 5 Nov 2015) belonging to CC10, CC131 and CC405 are depicted, together with the singleton strains found in this study. (b) All strains from the K. pneumoniae MLST database at Pasteur institute (http://bigsdb.web.pasteur.fr/, last accessed on 5 Nov 2015) belonging to CC23, together with the singleton strains detected here or related, are shown
Discussion
This study investigated the antimicrobial susceptibilities and molecular characteristics of 13 clinical and 79 stool carbapenem-nonsusceptible E. coli and K. pneumoniae isolates. Nine carbapenemase producers including one from a clinical specimen and eight from screening specimens were identified, giving a carbapenemase prevalence of 9.8 % (9/92) among the carbapenem nonsusceptible isolates. In Hong Kong, carbapenem nonsusceptibility among clinical E. coli and K. pneumoniae isolates remains rare and limited to sporadic occurrence [20, 21]. All the carbapenemase-producing strains in this study were identified in patients who had a history of hospitalization in mainland China or the United States. The importance of medical tourism as a significant risk factor for acquisition of CPE has been reported in many previous studies [2, 5]. In areas where the prevalence of CPE remains low, active surveillance culture of all patients with a history of travel should continue to be emphasized [20].
MLST revealed that our strains carrying bla IMP-4 or bla NDM-1 were multi-clonal, as these resistance determinants mainly spread among bacterial populations by mobile genetic elements [1]. On the other hand, all three K. pneumoniae isolates carrying bla KPC-2 were members of the widespread ST11 or ST258 clones [22]. Besides carbapenemases, carriage of ESBL and/or AmpC in combination with porin loss or drug efflux has been reported to contribute to carbapenem resistance in Enterobacteriaceae [2, 23]. In the present study, CTX-M type ESBL, AmpC and porin loss were highly prevalent among the carbapenem-nonsusceptible isolates. Among the strains negative for all carbapenemases genes, the mechanism of carbapenem resistance in about half of them could be attributed to presence of CTX-M and/or AmpC in combination with porin loss. As revealed by the MLST analysis, ST11 is also an important clonal group among K. pneumoniae isolates not producing carbapenemases [8].
This study used two previously validated methods for phenotypic detection of carbapenemases [13, 24, 25]. Some variations in the ability of ertapenem, imipenem and meropenem as substrates for potentiation by APBA and EDTA in CDTs were observed. Inclusion of more than one substrate in the CDT was required for phenotypic detection of all the carbapenemase producers in the collection. In contrast, the Carba NP test based upon hydrolysis of imipenem was able to detect all the carbapenemase producers and no false-positive results were observed. In diagnostic laboratories, workflow for the detection of CPE is evolving as new approaches and improvement in tests for their detection continue to be reported [13, 26]. The short turnaround time (2 hours) of Carba NP test is an important advantage. Given the greater epidemiological and clinical importance of CPE, this can allow the infection control team to make better use of isolation facilities for reducing the risk of nosocomial transmission [9]. However, false negative or equivocal Carba NP test results may occur for mucoid colonies because of difficulty in protein extraction and for enzyme types with low carbapenemase activity, especially the OXA group of enzymes [13, 27, 28]. Carba NP test may also fail to detect less common carbapenemases (SME, GES, IMI) [28–30]. Currently, the CLSI (2015, M100-S25) only recommends the Carba NP test for epidemiological or infection control purposes [11]. Its routine use in diagnostic laboratories is not recommended.
This study is limited by the small number of carbapenemase producers. We restricted the investigation of carbapenem resistance mechanisms to the most common types [1, 2]. Besides those that were investigated, drug efflux, other porins (PhoE, lamB) and other ESBLs (e.g. SHV, TEM) have also been described to contribute to carbapenem resistance [31, 32].
In conclusion, the prevalence of KPC, IMP and NDM types of carbapenemases among carbapenem-nonsusceptible E. coli and K. pneumoniae remained low in Hong Kong. Porin loss combined with AmpC and/or CTX-M type ESBL was the major mechanism of carbapenem resistance in the study population.
References
Tzouvelekis LS, Markogiannakis A, Psichogiou M, Tassios PT, Daikos GL (2012) Carbapenemases in Klebsiella pneumoniae and other Enterobacteriaceae: an evolving crisis of global dimensions. Clin Microbiol Rev 25(4):682–707
Grundmann H, Livermore DM, Giske CG, Canton R, Rossolini GM, Campos J, Vatopoulos A, Gniadkowski M, Toth A, Pfeifer Y, Jarlier V, Carmeli Y (2010) Carbapenem-non-susceptible Enterobacteriaceae in Europe: conclusions from a meeting of national experts. Euro Surveill 15(46):1–13
Muscarella LF (2014) Risk of transmission of carbapenem-resistant Enterobacteriaceae and related "superbugs" during gastrointestinal endoscopy. World J Gastrointest Endosc 6(10):457–474
Hrabak J, Chudackova E, Papagiannitsis CC (2014) Detection of carbapenemases in Enterobacteriaceae: a challenge for diagnostic microbiological laboratories. Clin Microbiol Infect 20(9):839–853
Ho PL, Li Z, Lo WU, Cheung YY, Lin CH, Sham PC, Chi-Chung C, V, Ng TK, Que TL, Chow KH (2012) Identification and characterization of a novel incompatibility group X3 plasmid carrying bla NDM-1 in Enterobacteriaceae isolates with epidemiological links to multiple geographical areas in China. Emerg Microbes Infect 1(11): e39
Espedido BA, Partridge SR, Iredell JR (2008) bla IMP-4 in different genetic contexts in Enterobacteriaceae isolates from Australia. Antimicrob Agents Chemother 52(8):2984–2987
Ho PL, Lo WU, Chan J, Cheung YY, Chow KH, Yam WC, Lin CH, Que TL (2014) pIMP-PH114 carrying bla IMP-4 in a Klebsiella pneumoniae strain is closely related to other multidrug-resistant IncA/C2 plasmids. Curr Microbiol 68(2):227–232
Wang JT, Wu UI, Lauderdale TL, Chen MC, Li SY, Hsu LY, Chang SC (2015) Carbapenem-nonsusceptible Enterobacteriaceae in Taiwan. PLoS One 10(3):e0121668
Cheng VC, Chan JF, Wong SC, Chen JH, Tai JW, Yan MK, Kwan GS, Tse H, To KK, Ho PL, Yuen KY (2013) Proactive infection control measures to prevent nosocomial transmission of carbapenem-resistant Enterobacteriaceae in a non-endemic area. Chin Med J (Engl ) 126(23):4504–4509
Ho PL, Cheung YY, Lo WU, Li Z, Chow KH, Lin CH, Chan JF, Cheng VC (2013) Molecular characterization of an atypical IncX3 plasmid pKPC-NY79 carrying bla KPC-2 in a Klebsiella pneumoniae. Curr Microbiol 67(4):493–498
CLSI (2015) Performance standards for antimicrobial susceptibility testing: twenty-fifth informational supplement M100-S25. CLSI, Wayne, PA, USA
Ho PL, Lo WU, Yeung MK, Lin CH, Chow KH, Ang I, Tong AH, Bao JY, Lok S, Lo JY (2011) Complete sequencing of pNDM-HK encoding NDM-1 carbapenemase from a multidrug-resistant Escherichia coli strain isolated in Hong Kong. PLoS One 6(3):e17989
Maurer FP, Castelberg C, Quiblier C, Bloemberg GV, Hombach M (2015) Evaluation of carbapenemase screening and confirmation tests with Enterobacteriaceae and development of a practical diagnostic algorithm. J Clin Microbiol 53(1):95–104
Perez-Perez FJ, Hanson ND (2002) Detection of plasmid-mediated AmpC beta-lactamase genes in clinical isolates by using multiplex PCR. J Clin Microbiol 40(6):2153–2162
Clermont O, Christenson JK, Denamur E, Gordon DM (2013) The Clermont Escherichia coli phylo-typing method revisited: improvement of specificity and detection of new phylo-groups. Environ Microbiol Rep 5(1):58–65
Diancourt L, Passet V, Verhoef J, Grimont PA, Brisse S (2005) Multilocus sequence typing of Klebsiella pneumoniae nosocomial isolates. J Clin Microbiol 43(8):4178–4182
Wirth T, Falush D, Lan R, Colles F, Mensa P, Wieler LH, Karch H, Reeves PR, Maiden MC, Ochman H, Achtman M (2006) Sex and virulence in Escherichia coli: an evolutionary perspective. Mol Microbiol 60(5):1136–1151
Spratt BG, Hanage WP, Li B, Aanensen DM, Feil EJ (2004) Displaying the relatedness among isolates of bacterial species -- the eBURST approach. FEMS Microbiol Lett 241(2):129–134
Hernandez-Alles S, Alberti S, Alvarez D, Domenech-Sanchez A, Martinez-Martinez L, Gil J, Tomas JM, Benedi VJ (1999) Porin expression in clinical isolates of Klebsiella pneumoniae. Microbiology 145(Pt 3):673–679
Cheng VC, Wong SC, Ho PL, Yuen KY (2015) Strategic measures for the control of surging antimicrobial resistance in Hong Kong and mainland of China. Emerg Microbes Infect 4(2):e8
Ho PL, Chu YP, Lo WU, Chow KH, Law PY, Tse CW, Ng TK, Cheng VC, Que TL (2015) High prevalence of Escherichia coli sequence type 131 among antimicrobial-resistant E. coli isolates from geriatric patients. J Med Microbiol 64(Pt 3):243–247
Mathers AJ, Peirano G, Pitout JD (2015) The role of epidemic resistance plasmids and international high-risk clones in the spread of multidrug-resistant Enterobacteriaceae. Clin Microbiol Rev 28(3):565–591
Pfeifer Y, Cullik A, Witte W (2010) Resistance to cephalosporins and carbapenems in Gram-negative bacterial pathogens. Int J Med Microbiol 300(6):371–379
Gallagher LC, Roundtree SS, Lancaster DP, Rudin SD, Bard JD, Roberts AL, Marshall SH, Bonomo RA, Sullivan KV (2015) Performance of the CLSI Carba NP and the Rosco Carb Screen Assays Using North American Carbapenemase-Producing Enterobacteriaceae and Pseudomonas aeruginosa Isolates. J Clin Microbiol 53(10):3370–3373
Dortet L, Brechard L, Cuzon G, Poirel L, Nordmann P (2014) Strategy for rapid detection of carbapenemase-producing Enterobacteriaceae. Antimicrob Agents Chemother 58(4):2441–2445
Osei SJ, Govinden U, Essack SY (2015) Review of established and innovative detection methods for carbapenemase-producing Gram-negative bacteria. J Appl Microbiol 119(5):1219–1233
Chong PM, McCorrister SJ, Unger MS, Boyd DA, Mulvey MR, Westmacott GR (2015) MALDI-TOF MS detection of carbapenemase activity in clinical isolates of Enterobacteriaceae spp., Pseudomonas aeruginosa, and Acinetobacter baumannii compared against the Carba-NP assay. J Microbiol Methods 111:21–23
Tijet N, Boyd D, Patel SN, Mulvey MR, Melano RG (2013) Evaluation of the Carba NP test for rapid detection of carbapenemase-producing Enterobacteriaceae and Pseudomonas aeruginosa. Antimicrob Agents Chemother 57(9):4578–4580
Osterblad M, Hakanen AJ, Jalava J (2014) Evaluation of the Carba NP test for carbapenemase detection. Antimicrob Agents Chemother 58(12):7553–7556
Papagiannitsis CC, Studentova V, Izdebski R, Oikonomou O, Pfeifer Y, Petinaki E, Hrabak J (2015) Matrix-assisted laser desorption ionization-time of flight mass spectrometry meropenem hydrolysis assay with NH4HCO3, a reliable tool for direct detection of carbapenemase activity. J Clin Microbiol 53(5):1731–1735
Garcia-Sureda L, Juan C, Domenech-Sanchez A, Alberti S (2011) Role of Klebsiella pneumoniae LamB Porin in antimicrobial resistance. Antimicrob Agents Chemother 55(4):1803–1805
Kaczmarek FM, Dib-Hajj F, Shang W, Gootz TD (2006) High-level carbapenem resistance in a Klebsiella pneumoniae clinical isolate is due to the combination of bla ACT-1 beta-lactamase production, porin OmpK35/36 insertional inactivation, and down-regulation of the phosphate transport porin phoe. Antimicrob Agents Chemother 50(10):3396–3406
Acknowledgments
This work was supported by grants from the Health and Medical Research Fund of the Food and Health Bureau of the Government of the HKSAR and the RGC Collaborative Research Fund Project on Syndromic Surveillance and Modelling for Infectious Diseases (CityU8/CRF/12G).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Transparency declaration
Nothing to declare
Conflict of interest statement
The authors declares that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
(PDF 110 kb)
Rights and permissions
About this article
Cite this article
Ho, P.L., Cheung, Y.Y., Wang, Y. et al. Characterization of carbapenem-resistant Escherichia coli and Klebsiella pneumoniae from a healthcare region in Hong Kong. Eur J Clin Microbiol Infect Dis 35, 379–385 (2016). https://doi.org/10.1007/s10096-015-2550-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-015-2550-3