Abstract
Epidemiological data on primary progressive multiple sclerosis (PPMS) are scarce. This study was aimed to evaluate the burden of PPMS in Italy with healthcare resources utilisation and costs for Italian National Health System (INHS). A 2-year cross-sectional analysis of real-world data collected in the ARCO database, covering > 10 million Italian inhabitants, was performed. From a cohort of patients affected by MS in 2014, those supposedly affected by PPMS were defined by the concurrent matching of absence of disease-modifying treatments and use of rehabilitation services. Any other drug prescriptions, outpatient services and hospitalisations were analysed in 2015 for each subject. The average annual cost per patient was provided both for each expenditure item and by integrating these. Of 13,253,591 inhabitants, 18,453 resulted affected by MS (prevalence 139 × 100,000). Of these, 1849 agreed with additional criteria to identify PPMS (10% of MS population). The 26.8% of these experienced at least one admission in 1 year, 97.3% used at least one outpatient service and 94.3% received at least one reimbursed drug. In the perspective of INHS, PPMS generated an average annual cost of € 3783 per person: 49% for hospitalisations, 28% for outpatient services and 23% for drugs. This study provides a reliable estimation of the PPMS burden in Italy, in terms of healthcare utilisation and direct costs. These findings could be useful to estimate the changes in health expenditure following the incoming of new drugs to treat PPMS with increase of pharmaceutical cost and potential decrease of rehabilitation and hospitalisation costs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Multiple sclerosis (MS) is the most frequent demyelinating disease and one of the main cause for permanent disability in young adults. WHO estimated that, in 2013, this disease affected more than 2.3 million people worldwide, and 600,000 only in Europe: figures have increased in recent years [1].
In Italy, a country considered as high-risk area for MS, several epidemiological studies have been conducted at regional and local levels and with different methodologies, resulting in a wide range of prevalence estimations. A recent analysis of all these studies estimated that in Italy, in 2015, the average MS prevalence was 176 per 100,000, ranging from 122 to 232 cases per 100,000 [2]. However, in these studies, no data on the epidemiology of different disease courses were provided, due to the difficulty in identifying the subtypes of MS in different data sources investigated (e.g. in healthcare administrative database, there is no information on clinical course of disease).
Moreover, economic evaluations on MS generally are lacking for differential analyses according to the specific course, although economic analyses on the basis of disease severity (measured by EDSS—Expanded Disability Status Scale) showed an increase of the total cost with the augmentation of disability [3,4,5,6,7,8].
This issue is relevant due to MS is not a unique disease and it is characterised by variable courses. The majority of patients starts with relapsing-remitting (RR) form and approximately 50–60% of them progress to secondary progressive (SP) disease. Only 10–15% of patients develop a progressive disease course from the onset, termed primary progressive multiple sclerosis (PPMS) [9]. The course of this last subtype of MS differs from others since its progression consists mainly of gradual worsening of neurologic disability from symptom onset, although relapses may occur [10].
During the past years, several advances were reached through researches in MS field, especially for the treatment of RRMS; however, several clinical needs remain unmet [11]. The main unmet need, until recent years, was the treatment of progressive forms, in particular PPMS [12, 13]. After the failure of several trials in searching treatment for this form of disease [14,15,16], recently, new therapeutic options are incoming to treat PPMS [17].
In order to better forecast and manage the impact of the new therapeutic options for PPMS, it is essential to investigate the real-life population affected by this form of disease. This should allow to better allocate resources in prevision of an increase of pharmaceutical cost and a potential decrease of rehabilitation and hospitalisation expenditures.
In this scenario, the aim of this study was to evaluate the burden of PPMS in Italy and the related healthcare resources utilisation and costs for Italian National Health System (INHS), by using health administrative databases.
Methods
Study design
This was a 2-year cross-sectional study based on real-world data: each selected patient was identified in the accrual year (2014) and observed in the subsequent year (2015).
The analysis was conducted by using ARCO database, created with the collaboration of CINECA (Interuniversity Consortium).
This data source, covering more than 10 million of Italian inhabitants of different regions and local health units, was involved in published observational researches [18, 19]. ARCO is a patient-centred data-warehouse, where all INHS administrative databases are integrated with each other: reimbursed outpatient prescriptions database, hospital discharges database and outpatient services and visits database.
Based on this feature, ARCO can be searched to describe, for each subject, the precise healthcare pathway and related direct costs for INHS.
Patients
Patients with PPMS were identified among a population affected by MS in 2014 (accrual year).
Firstly, the population affected by MS (any type) was selected among the overall ARCO population, by the presence of at least one of the following information:
-
(i)
exemption code for MS (Italian exemption code: 046), or
-
(ii)
hospitalisation with a primary or secondary diagnosis of MS, codified by the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD9-CM): ICD9-CM code 340; or
-
(iii)
prescription of a disease-modifying drug (DMD) labelled for MS, identified by the Anatomical Therapeutic Chemical (ATC) codification: (interferon beta-1b L03AB08; interferon beta-1a L03AB07, glatiramer acetate L03AX13, teriflunomide L04AA31, dimethyl fumarate N07XX09, natalizumab L04AA23, fingolimod L04AA2 or alemtuzumab L04AA34).
This algorithm was already applied and validated by Bargagli and colleagues with the purpose to investigate the prevalence of MS in Lazio Region [20], and it is quite similar to that used by Bezzini and co-workers to estimate the prevalence of MS in Tuscany Region [21].
Among MS population, PPMS patients were identified by searching those subjects without immunomodulatory and immunosuppressive treatments and needing rehabilitation. Therefore, the following additional criteria that should be concurrently met in 2 years (2014 and 2015), were applied:
-
(i)
absence of any prescription of DMDs labelled for MS (listed above) and immunosuppressive drugs used for MS treatment (azathioprine ATC code L04Ax01, methotrexate L04AX03, cyclophosphamide L01AA01, mitoxantrone L01DB07 or mycophenolic acid L04AA06) over both years,
and
-
(ii)
at least one utilisation of ambulatory services for rehabilitation and visits during the 2 years.
Statistical analysis
At the accrual year (2014), the crude prevalence estimations (per 100,000 inhabitants) of MS (any type) and of PPMS were provided. Moreover, the rate of PPMS among overall MS population was calculated. Gender and age distribution of overall MS and PPMS patients was also depicted.
The healthcare resources utilisation and related direct costs were calculated for the 2015 population affected by PPMS. Hospital admissions were analysed in terms of primary diagnosis or procedure, by ICD9-CM codes. Reimbursed outpatient drug prescriptions were described in terms of therapeutic class, according to the IV level of ATC classification. Outpatient services and visits were analysed by using regional lists of all healthcare services reimbursed by INHS.
Healthcare costs were evaluated using real price paid by INHS and tariffs for in- and outpatient services (only direct costs). Cost analyses were performed both as single expenditure item (i.e. hospital discharges, drug prescriptions and ambulatory services and visits) and as integrated costs. The total annual cost and the average annual cost per patient, in the perspective of INHS, were calculated.
Results
Prevalence and socio-demographic characteristics of PPMS
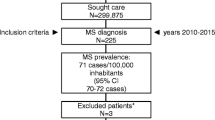
Starting from an overall population of 13,253,591 subjects, resident in 6 Italian Regions (Sardinia excluded) at 2014 and with complete data recorded in ARCO database, 18,453 patients resulted affected by MS (crude prevalence 139 per 100,000 inhabitants). Out of these, 1849 (10.0%) resulted without any pharmacological treatment and users of rehabilitation services or visits. Therefore, the 2014 prevalence of PPMS patients was 14 per 100,000 inhabitants (Fig. 1).
Females represented the 69.2% of PPMS group, with a F:M ratio similar to that retrieved in overall MS group (i.e. 2.3:1). The mean age of PPMS patients was 54.0 years old, higher than that recorded in overall MS population (47.4 years old) (Table 1).
Healthcare utilisation and direct costs of PPMS
In 1-year period, almost all PPMS population (99.3%) used at least one healthcare resource with a total cost for INHS of € 6,994,663, referring to an average annual cost per patient of € 3783.
The 94.3% of subjects received during this period at least one reimbursed drug, with a total cost of € 1,583,612 and on average € 856 per patient. The most prescribed drug classes were “proton pump inhibitors” (39.2% of PPMS patients), followed by “vitamin D and analogues” (37.3%), “glucocorticoids” (27.0%) and “penicillins” (24.8%). Among the first 10, there were also therapies often used to treat symptoms of MS such as “muscle relaxants” (24.7%) and “drugs for neuropathic pain” (21.1%). This last therapeutic class generated the highest annual cost per patient (€ 103.7) among all reimbursed drugs.
The analysis of hospital admissions showed that 26.8% of the studied population was admitted at least one time in the 1-year period, with a yearly total cost of € 3,442,312 and on average € 1862 per patient. Hospitalisations for “multiple sclerosis” (as a primary diagnosis) occurred in 4.8% of patients with an average annual cost per patient of € 236.6. Other diagnoses or procedures recorded in more than 1% of the studied population were “administration of long-term high-risk medications” (3.1%) and “paraplegia” (1.6%).
Outpatient services or visits were used over 1 year by 97.3% of PPMS subjects generating a total cost of € 1,968,739 and an average annual cost per patient of € 1065. Visits were performed for 91.0% of patients, laboratory exams for 83.7% and rehabilitation services for 54.5%. This last service implicated the highest annual cost per patient (€ 352.9), among all outpatient services (Table 2).
The integrated cost analysis showed that, in the perspective of INHS, every subject affected by MS without any specific drug (i.e. PPMS) on average yearly accounted for € 3783. This expenditure was composed by 49% due to hospitalisations, 28% to outpatient services and the remaining 23% to drug prescriptions (Fig. 2).
Discussion
This study, investigating a specific subgroup of MS patient in Italy (i.e. PPMS), provided information on its burden of disease and related costs for INHS. This becomes essential in order to define the potential benefits in terms of healthcare expenditure of the incoming therapeutic strategies. Indeed, previous Italian epidemiological studies [20, 21] based on administrative databases, although recognising the value of this data source to investigate MS, did not perform any analysis on different disease courses. Moreover, previous Italian analyses of MS costs were based on questionnaires that stratified population according to EDSS and not to disease course, due to the difficulties in recognising by patient itself the specific disease subtype [7].
The study, by analysing real-world data, found a MS prevalence estimation in line with previous Italian studies [2] and it added the prevalence figures of PPMS, accounting for 10% of overall MS patient, as expected and reported in the literature [10, 22]. This was possible by applying specific selection criteria to administrative databases. One of these criteria was the absence of any prescription of drug aimed to reduce disease progression (i.e. immunomodulatory and immunosuppressive drugs); this could allow to argue that these subjects were affected by the only one subtype of disease without specific treatment in analysed years, i.e. PPMS. Nevertheless, also patients affected by other types of MS could receive no specific treatments for several reasons (e.g. intolerance, inefficacy or others). Therefore, the choice to add a second selection criterion based on the use of rehabilitation services allows us to reduce the risk of selection of those patients affected by relapsing forms and at early stage of the disease (i.e. they did not needing rehabilitations). Based on these considerations, we can reliably affirm that, in our analysis, the 10% of MS subjects meeting these two selection criteria suffered from PPMS. As expected, the mean age of this group of patients was higher than that recorded in overall MS population [10]. Concerning the F:M ratio, literature reports that it should be around 1:1 for PPMS [10], while our results found a ratio of 2.3:1. This discrepancy should be due to the choice to analyse prevalent cases and not incident ones.
In the light of these results, this study demonstrated that the lack of specific information on the MS course in the administrative databases could be overcome by applying specific inclusion criteria. These could be helpful to conduct epidemiologic and economic analyses on different MS courses by using this data sources. Indeed, the study showed that a patient with PPMS generated an annual average cost of € 3783 in the perspective of INHS. The main cost-diver for PPMS was hospital admissions, followed by outpatient services, and by drug prescriptions. This ranking is due to the absence of specific treatment for this group of MS patients. In view of incoming new strategies, it should be forecasted that the cost for drugs will increase but, at the same time, hospitalisations and outpatient services should be reduced. To these direct costs, should be added the indirect costs, that are huge for MS patients [6, 7], and should be reduced by new drugs.
The main strength of this analysis is that it describes a real-world setting concerning a large population. However, this strength is also the well-known limitation of observational researches in relation to the features of data source used for the analysis [23, 24].
In conclusion, the results of this study could be helpful for policy decision-maker for an appropriate allocation of the resource in the management of patients affected by PPMS, especially in view of the changes of therapeutic strategies that are incoming for this group of patients until now without specific treatment.
References
Browne P, Chandraratna D, Angood C, Tremlett H, Baker C, Taylor BV, Thompson AJ (2014) Atlas of multiple sclerosis 2013: a growing global problem with widespread inequity. Neurology 83(11):1022–1024. https://doi.org/10.1212/wnl.0000000000000768
Battaglia MA, Bezzini D (2017) Estimated prevalence of multiple sclerosis in Italy in 2015. Neurol Sci 38(3):473–479. https://doi.org/10.1007/s10072-016-2801-9
Amato MP, Battaglia MA, Caputo D, Fattore G, Gerzeli S, Pitaro M, Reggio A, Trojano M (2002) The costs of multiple sclerosis: a cross-sectional, multicenter cost-of-illness study in Italy. J Neurol 249(2):152–163
Kobelt G, Berg J, Lindgren P, Battaglia M, Lucioni C, Uccelli A (2006) Costs and quality of life of multiple sclerosis in Italy. Eur J Health Econ 7(Suppl 2):S45–S54. https://doi.org/10.1007/s10198-006-0385-7
Karampampa K, Gustavsson A, Miltenburger C, Teruzzi C, Fattore G (2012) Treatment experience, burden and unmet needs (TRIBUNE) in MS study: results from Italy. Mult Scler 18(2 Suppl):29–34. https://doi.org/10.1177/1352458512441566c
Ponzio M, Gerzeli S, Brichetto G, Bezzini D, Mancardi GL, Zaratin P, Battaglia MA (2015) Economic impact of multiple sclerosis in Italy: focus on rehabilitation costs. Neurol Sci 36(2):227–234. https://doi.org/10.1007/s10072-014-1925-z
Battaglia M, Kobelt G, Ponzio M, Berg J, Capsa D, Dalen J (2017) New insights into the burden and costs of multiple sclerosis in Europe: results for Italy. Mult Scler 23(2_suppl):104–116. https://doi.org/10.1177/1352458517708176
Olesen J, Gustavsson A, Svensson M, Wittchen HU, Jonsson B (2012) The economic cost of brain disorders in Europe. Eur J Neurol 19(1):155–162. https://doi.org/10.1111/j.1468-1331.2011.03590.x
Antel J, Antel S, Caramanos Z, Arnold DL, Kuhlmann T (2012) Primary progressive multiple sclerosis: part of the MS disease spectrum or separate disease entity? Acta Neuropathol 123(5):627–638. https://doi.org/10.1007/s00401-012-0953-0
Miller DH, Leary SM (2007) Primary-progressive multiple sclerosis. Lancet Neurol 6(10):903–912. https://doi.org/10.1016/s1474-4422(07)70243-0
Reich DS, Lucchinetti CF, Calabresi PA (2018) Multiple sclerosis. N Engl J Med 378(2):169–180. https://doi.org/10.1056/NEJMra1401483
Mehr SR, Zimmerman MP (2015) Reviewing the unmet needs of patients with multiple sclerosis. Am Health Drug Benefits 8(8):426–431
Holland NJ, Schneider DM, Rapp R, Kalb RC (2011) Meeting the needs of people with primary progressive multiple sclerosis, their families, and the health-care community. Int J MS Care 13(2):65–74. https://doi.org/10.7224/1537-2073-13.2.65
Wolinsky JS, Narayana PA, O'Connor P, Coyle PK, Ford C, Johnson K, Miller A, Pardo L, Kadosh S, Ladkani D (2007) Glatiramer acetate in primary progressive multiple sclerosis: results of a multinational, multicenter, double-blind, placebo-controlled trial. Ann Neurol 61(1):14–24. https://doi.org/10.1002/ana.21079
Hawker K, O’Connor P, Freedman MS, Calabresi PA, Antel J, Simon J, Hauser S, Waubant E, Vollmer T, Panitch H, Zhang J, Chin P, Smith CH (2009) Rituximab in patients with primary progressive multiple sclerosis: results of a randomized double-blind placebo-controlled multicenter trial. Ann Neurol 66(4):460–471. https://doi.org/10.1002/ana.21867
Lublin F, Miller DH, Freedman MS, Cree BAC, Wolinsky JS, Weiner H, Lubetzki C, Hartung HP, Montalban X, Uitdehaag BMJ, Merschhemke M, Li B, Putzki N, Liu FC, Haring DA, Kappos L (2016) Oral fingolimod in primary progressive multiple sclerosis (INFORMS): a phase 3, randomised, double-blind, placebo-controlled trial. Lancet 387(10023):1075–1084. https://doi.org/10.1016/s0140-6736(15)01314-8
Lyon J (2018) New inroads against multiple sclerosis. JAMA 319(1):9–11. https://doi.org/10.1001/jama.2017.14033
Calabria S, Cinconze E, Rossini M, Rossi E, Maggioni AP, Pedrini A, De Rosa M (2016) Adherence to alendronic or risedronic acid treatment, combined or not to calcium and vitamin D, and related determinants in Italian patients with osteoporosis. Patient Prefer Adherence 10:523–530. https://doi.org/10.2147/ppa.s95634
Maggioni AP, Orso F, Calabria S, Rossi E, Cinconze E, Baldasseroni S, Martini N (2016) The real-world evidence of heart failure: findings from 41 413 patients of the ARNO database. Eur J Heart Fail 18(4):402–410. https://doi.org/10.1002/ejhf.471
Bargagli AM, Colais P, Agabiti N, Mayer F, Buttari F, Centonze D, Di Folco M, Filippini G, Francia A, Galgani S, Gasperini C, Giuliani M, Mirabella M, Nociti V, Pozzilli C, Davoli M (2016) Prevalence of multiple sclerosis in the Lazio region, Italy: use of an algorithm based on health information systems. J Neurol 263(4):751–759. https://doi.org/10.1007/s00415-016-8049-8
Bezzini D, Policardo L, Meucci G, Ulivelli M, Bartalini S, Profili F, Battaglia MA, Francesconi P (2016) Prevalence of multiple sclerosis in Tuscany (Central Italy): a study based on validated administrative data. Neuroepidemiology 46(1):37–42. https://doi.org/10.1159/000441567
Koch M, Kingwell E, Rieckmann P, Tremlett H (2009) The natural history of primary progressive multiple sclerosis. Neurology 73(23):1996–2002. https://doi.org/10.1212/WNL.0b013e3181c5b47f
van Walraven C, Austin P (2012) Administrative database research has unique characteristics that can risk biased results. J Clin Epidemiol 65(2):126–131. https://doi.org/10.1016/j.jclinepi.2011.08.002
Gini R, Schuemie MJ, Pasqua A, Carlini E, Profili F, Cricelli I, Dazzi P, Barletta V, Francesconi P, Lapi F, Donatini A, Dal Co G, Visca M, Bellentani M, Sturkenboom M, Klazinga N (2017) Monitoring compliance with standards of care for chronic diseases using healthcare administrative databases in Italy: strengths and limitations. PLoS One 12(12):e0188377. https://doi.org/10.1371/journal.pone.0188377
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Piccinni, C., Ronconi, G., Calabria, S. et al. Healthcare resources utilisation in primary progressive multiple sclerosis. Neurol Sci 39, 1169–1174 (2018). https://doi.org/10.1007/s10072-018-3404-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-018-3404-4