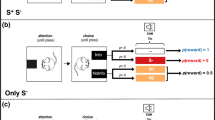
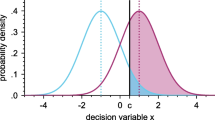
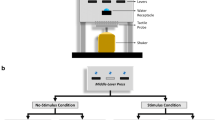
Abstract
Few studies have considered how signal detection parameters evolve during acquisition periods. We addressed this gap by training mice with differential prior experience in a conditional discrimination, auditory signal detection task. Naïve mice, mice given separate experience with each of the later correct choice options (Correct Choice Response Transfer, CCRT), and mice experienced in conditional discriminations (Conditional Discrimination Transfer, CDT) were trained to detect the presence or absence of a tone in white noise. We analyzed data assuming a two-period model of acquisition: a pre-solution and solution period (Heinemann EG (1983) in The Presolution period and the detection of statistical associations. In: Quantitative analyses of behavior: discrimination processes, vol. 4, pp. 21–36). Ballinger. http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.536.1978andrep=rep1andtype=pdf). The pre-solution period was characterized by a selective sampling of biased response strategies until adoption of a conditional responding strategy in the solution period. Correspondingly, discriminability remained low until the solution period; criterion took excursions reflecting response–strategy sampling. Prior experience affected the length and composition of the pre-solution period. Whereas CCRT and CDT mice had shorter pre-solution periods than naïve mice, CDT and Naïve mice developed substantial criterion biases and acquired asymptotic discriminability faster than CCRT mice. To explain these data, we propose a learning model in which mice selectively sample and test different response-strategies and corresponding task structures until they exit the pre-solution period. Upon exit, mice adopt the conditional responding strategy and task structure, with action values updated via inference and generalization from the other task structures. Simulations of representative mouse data illustrate the viability of this model.










Similar content being viewed by others
Availability of data and material
Supporting materials can be found at https://doi.org/10.17605/OSF.IO/FJZ87.
Code availability
Supporting materials can be found at https://doi.org/10.17605/OSF.IO/FJZ87.
Notes
We chose to not estimate parameters using maximum likelihood or Bayesian approaches precisely because of the two missing components: feedback function for updating P as a function of trial and the processes that inform π. Without knowing how to specify these components, misfits could be due to the inadequacy of our approximations rather than some in-principal failure of the model. Nevertheless, we took a principled approach in which we tried to minimize RSS by slowly adjusting parameters of each module and then asking the relative import of each module.
References
Alves-Pinto A, Sollini J, Sumner CJ (2012) Signal detection in animal psychoacoustics: analysis and simulation of sensory and decision-related influences. Neuroscience 220:215–227. https://doi.org/10.1016/j.neuroscience.2012.06.001
Anderson DR, Burnham KP, White GC (1998) Comparison of Akaike information criterion and consistent Akaike information criterion for model selection and statistical inference from capture-recapture studies. J Appl Stat 25(2):263–282. https://doi.org/10.1080/02664769823250
Andrzejewski ME, Holder N (2016) Chapter 11—amphetamine and signal detection. In: Preedy VR (Ed) Neuropathology of drug addictions and substance misuse (pp. 117–125). Academic Press. https://doi.org/10.1016/B978-0-12-800212-4.00011-X
Ashwood ZC, Roy NA, Stone IR, The International Brain Laboratory, Churchland AK, Pouget A, Pillow JW (2020)Mice alternate between discrete strategies during perceptual decision-making [Preprint]. Neuroscience. https://doi.org/10.1101/2020.10.19.346353
Aust U, Range F, Steurer M, Huber L (2008) Inferential reasoning by exclusion in pigeons, dogs, and humans. Anim Cogn 11(4):587–597. https://doi.org/10.1007/s10071-008-0149-0
Balcı F, Freestone D, Simen P, deSouza L, Cohen JD, Holmes P (2011) Optimal temporal risk assessment. Front Integrative Neurosci. https://doi.org/10.3389/fnint.2011.00056
Ball K, Sekuler R (1982) A specific and enduring improvement in visual motion discrimination. Science. https://www.science.org/doi/abs/https://doi.org/10.1126/science.7134968
Bathellier B, Tee SP, Hrovat C, Rumpel S (2013) A multiplicative reinforcement learning model capturing learning dynamics and interindividual variability in mice. Proc Natl Acad Sci 110(49):19950–19955. https://doi.org/10.1073/pnas.1312125110
Benovitski YB, Blamey PJ, Rathbone GD, Fallon JB (2014) An automated psychoacoustic testing apparatus for use in cats. Hear Res 309:1–7. https://doi.org/10.1016/j.heares.2013.11.002
Bieszczad KM, Weinberger NM (2010) Learning strategy trumps motivational level in determining learning-induced auditory cortical plasticity. Neurobiol Learn Mem 93(2):229–239. https://doi.org/10.1016/j.nlm.2009.10.003
Bouton ME (2004) Context and behavioral processes in extinction. Learn Mem 11(5):485–494. https://doi.org/10.1101/lm.78804
Bouton ME (2019) Extinction of instrumental (operant) learning: interference, varieties of context, and mechanisms of contextual control. Psychopharmacology 236(1):7–19. https://doi.org/10.1007/s00213-018-5076-4
Brackney RJ, Cheung THC, Neisewander JL, Sanabria F (2011) The isolation of motivational, motoric, and schedule effects on operant performance: a modeling approach. J Exp Anal Behav 96(1):17–38. https://doi.org/10.1901/jeab.2011.96-17
Brown SW, Newcomb DC, Kahrl KG (1995) Temporal-signal detection and individual differences in timing. Perception 24(5):525–538. https://doi.org/10.1068/p240525
Carandini M, Churchland AK (2013) Probing perceptual decisions in rodents. Nat Neurosci 16(7):824–831. https://doi.org/10.1038/nn.3410
Chen L, Wang X, Ge S, Xiong Q (2019) Medial geniculate body and primary auditory cortex differentially contribute to striatal sound representations. Nat Commun 10(1):1–10. https://doi.org/10.1038/s41467-019-08350-7
D’Amato MR, Jagoda H (1961) Analysis of the role of overlearning in discrimination reversal. J Exp Psychol 61(1):45–50. https://doi.org/10.1037/h0047757
Daniels CW, Sanabria F (2017a) Interval timing under a behavioral microscope: dissociating motivational and timing processes in fixed-interval performance. Learn Behav 45(1):29–48. https://doi.org/10.3758/s13420-016-0234-1
Daniels CW, Sanabria F (2017b) About bouts: a heterogeneous tandem schedule of reinforcement reveals dissociable components of operant behavior in Fischer rats. J Experimental Psychol 43(3):280–294. https://doi.org/10.1037/xan0000144
Daniels CW, Sanabria F (2018) An associability decay model of paradoxical choice. J Experimental Psychol 44(3):258–271. https://doi.org/10.1037/xan0000179
Daniels CW, Laude JR, Zentall TR (2014) Six-term transitive inference with pigeons: successive-pair training followed by mixed-pair training. J Exp Anal Behav 101(1):26–37. https://doi.org/10.1002/jeab.65
Daniels CW, Overby PF, Sanabria F (2018) Between-session memory degradation accounts for within-session changes in fixed-interval performance. Behav Proc 153:31–39. https://doi.org/10.1016/j.beproc.2018.05.004
David SV, Fritz JB, Shamma SA (2012) Task reward structure shapes rapid receptive field plasticity in auditory cortex. Proc Natl Acad Sci 109(6):2144–2149. https://doi.org/10.1073/pnas.1117717109
Dayan P, Daw ND (2008) Decision theory, reinforcement learning, and the brain. Cogn Affect Behav Neurosci 8(4):429–453. https://doi.org/10.3758/CABN.8.4.429
DeVito LM, Lykken C, Kanter BR, Eichenbaum H (2010) Prefrontal cortex: role in acquisition of overlapping associations and transitive inference. Learn Mem 17(3):161–167. https://doi.org/10.1101/lm.1685710
Eck KO, Thomas DR (1970) Discrimination learning as a function of prior discrimination and nondifferential training: a replication. J Exp Psychol 83(1):511–513. https://doi.org/10.1037/h0028874
Fay RR (1988) Comparative psychoacoustics. Hear Res 34(3):295–305. https://doi.org/10.1016/0378-5955(88)90009-3
Frey PW, Sears RJ (1979) Model of conditioning incorporating the Rescorla-Wagner associative axiom, a dynamic attention process, and a catastrophe rule. Psychol Rev 85(4):321. https://doi.org/10.1037/0033-295X.85.4.321
Gallistel CR, Fairhurst S, Balsam P (2004) The learning curve: implications of a quantitative analysis. Proc Natl Acad Sci 101(36):13124–13131. https://doi.org/10.1073/pnas.0404965101
Gallun FJ, Kampel SD (2016) Using detection theory to analyze human decision making and sensory processing. Proc Meetings Acoust 26(1):050004. https://doi.org/10.1121/2.0000379
Gershman SJ (2015) A unifying probabilistic view of associative learning. PLOS Comput Biol 11(11):e1004567. https://doi.org/10.1371/journal.pcbi.1004567
Gershman SJ, Jones CE, Norman KA, Monfils M.-H, Niv Y (2013) Gradual extinction prevents the return of fear: implications for the discovery of state.Front Behav Neurosci. https://doi.org/10.3389/fnbeh.2013.00164
Gold JM, Sekuler AB, Bennett PJ (2004) Characterizing perceptual learning with external noise. Cogn Sci 28(2):167–207. https://doi.org/10.1207/s15516709cog2802_3
Green DM (1960) Psychoacoustics and detection theory. J Acoust Soc Am 32(10):1189–1203. https://doi.org/10.1121/1.1907882
Guez D, Stevenson G (2011) Is reasoning in rats really unreasonable? Revisiting recent associative accounts. Front Psychol 2:277. https://doi.org/10.3389/fpsyg.2011.00277
Hangya B, Ranade SP, Lorenc M, Kepecs A (2015) Central cholinergic neurons are rapidly recruited by reinforcement feedback. Cell 162(5):1155–1168. https://doi.org/10.1016/j.cell.2015.07.057
Hanks TD, Summerfield C (2017) Perceptual decision making in rodents, monkeys, and humans. Neuron 93(1):15–31. https://doi.org/10.1016/j.neuron.2016.12.003
Heffner HE, Heffner RS (2007) Hearing ranges of laboratory animals. J Am Assoc Lab Animal Sci 46(1):20–22 (PMID: 17203911)
Heinemann EG (1983) The Presolution period and the detection of statistical associations. In: Quantitative analyses of behavior: discrimination processes (Vol. 4, pp. 21–36). Ballinger. http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.536.1978andrep=rep1andtype=pdf. Accessed 31 Dec 2021
Heinemann EG, Chase S (1970) Conditional stimulus control. J Exp Psychol 84(2):187. https://doi.org/10.1037/h0029152
Horga G, Abi-Dargham A (2019) An integrative framework for perceptual disturbances in psychosis. Nat Rev Neurosci 20(12):763–778. https://doi.org/10.1038/s41583-019-0234-1
Jaramillo S, Zador AM (2014) Mice and rats achieve similar levels of performance in an adaptive decision-making task. Front Syst Neurosci. https://doi.org/10.3389/fnsys.2014.00173
Jarosz AF, Wiley J (2014) What are the odds? A practical guide to computing and reporting bayes factors. J Problem Solving. https://doi.org/10.7771/1932-6246.1167
JASP Team (2019) JASP (Version 0.11.1)[Computer software]. https://jasp-stats.org/. Accessed 31 Dec 2021
Jensen G, Alkan Y, Ferrera VP, Terrace HS (2019) Reward associations do not explain transitive inference performance in monkeys. Sci Adv 5(7):eaaw2089. https://doi.org/10.1126/sciadv.aaw2089
Jones M, Canas F (2010) Integrating reinforcement learning with models of representation learning. Proceedings of the Annual Meeting of the Cognitive Science Society, 32(32). https://escholarship.org/uc/item/88x6f84q. Accessed 31 Dec 2021
Karim M, Harris JA, Morley JW, Breakspear M (2012) Prior and present evidence: how prior experience interacts with present information in a perceptual decision making task. PLoS ONE 7(5):e37580. https://doi.org/10.1371/journal.pone.0037580
Killeen P (1981) Quantitative analysis of behavior. In: Learning as causal inference (Vol. 1, pp. 321–340). Ballinger. https://www.academia.edu/21780437/Learning_as_causal_inference. Accessed 31 Dec 2021
Killeen PR (2016) Signal detection theory. In: The SAGE Encyclopedia of Theory in Psychology. SAGE Publications, Inc. https://doi.org/10.4135/9781483346274.n292
Killeen PR, Sanabria F, Dolgov I (2009) The dynamics of conditioning and extinction. J Exp Psychol Anim Behav Process 35(4):447–472. https://doi.org/10.1037/a0015626
Killeen PR, Taylor TJ, Treviño M (2018) Subjects adjust criterion on errors in perceptual decision tasks. Psychol Rev 125(1):117–130. https://doi.org/10.1037/rev0000056
Kim Y-H, Kang D-W, Kim D, Kim H-J, Sasaki Y, Watanabe T (2015) Real-time strategy video game experience and visual perceptual learning. J Neurosci 35(29):10485–10492. https://doi.org/10.1523/JNEUROSCI.3340-14.2015
Klink KB, Bendig G, Klump GM (2006) Operant methods for mouse psychoacoustics. Behav Res Methods 38(1):1–7. https://doi.org/10.3758/BF03192744
Kornell N (2009) Metacognition in Humans and Animals. Curr Dir Psychol Sci 18(1):11–15. https://doi.org/10.1111/j.1467-8721.2009.01597.x
Krakauer JW, Ghazanfar AA, Gomez-Marin A, MacIver MA, Poeppel D (2017) Neuroscience needs behavior: correcting a reductionist bias. Neuron 93(3):480–490. https://doi.org/10.1016/j.neuron.2016.12.041
Kuchibhotla KV, Sten TH, Papadoyannis ES, Elnozahy S, Fogelson KA, Kumar R, Boubenec Y, Holland PC, Ostojic S, Froemke RC (2019) Dissociating task acquisition from expression during learning reveals latent knowledge. Nat Commun 10(1):1–13. https://doi.org/10.1038/s41467-019-10089-0
Kurt S, Ehret G (2010) Auditory discrimination learning and knowledge transfer in mice depends on task difficulty. Proc Natl Acad Sci 107(18):8481–8485. https://doi.org/10.1073/pnas.0912357107
Kurylo DD, Waxman R, Kidron R, Silverstein SM (2017) Visual training improves perceptual grouping based on basic stimulus features. Atten Percept Psychophys 79(7):2098–2107. https://doi.org/10.3758/s13414-017-1368-8
Lany J, Gómez RL, Gerken LA (2007) The role of prior experience in language acquisition. Cogn Sci 31(3):481–507. https://doi.org/10.1080/15326900701326584
Lawrence DH (1950a) Acquired distinctiveness of cues: I. Transfer between discriminations on the basis of familiarity with the stimulus. J Experimental Psychol 39(6):770. https://doi.org/10.1037/h0058097
Lawrence DH (1950b) Acquired distinctiveness of cues: II. Selective association in a constant stimulus situation. J Experimental Psychol 40(2):175. https://doi.org/10.1037/h0063217
Leeuwen EJCV, Cronin KA, Schütte S, Call J, Haun DBM (2013) Chimpanzees (Pan troglodytes) Flexibly Adjust Their Behaviour in Order to Maximize Payoffs, Not to Conform to Majorities. PLoS ONE 8(11):e80945. https://doi.org/10.1371/journal.pone.0080945
Limpert E, Stahel WA, Abbt M (2001) Log-normal distributions across the sciences: keys and clues. Bioscience 51(5):341. https://doi.org/10.1641/0006-3568(2001)051[0341:LNDATS]2.0.CO;2
Love J, Selker R, Marsman M, Jamil T, Dropmann D, Verhagen J, Ly A, Gronau QF, Smíra M, Epskamp S, Matzke D, Wild A, Knight P, Rouder JN, Morey RD, Wagenmakers E-J (2019) JASP: graphical statistical software for common statistical designs. J Statistical Softw. https://doi.org/10.1863/jss.v088.i02
Luksys G, Gerstner W, Sandi C (2009) Stress, genotype and norepinephrine in the prediction of mouse behavior using reinforcement learning. Nat Neurosci 12(9):10
MacMillan NA (2002) Signal detection theory. In: Stevens’ handbook of experimental psychology. American Cancer Society. https://doi.org/10.1002/0471214426.pas0402
Marshall AT, Kirkpatrick K (2017) Reinforcement learning models of risky choice and the promotion of risk-taking by losses disguised as wins in rats. J Exp Psychol 43(3):262. https://doi.org/10.1037/xan0000141
Mccarthy D, Davison M (1981) Towards a behavioral theory of bias in signal detection. Percept Psychophys 29(4):371–382. https://doi.org/10.3758/BF03207347
McCormack T, Simms V, McGourty J, Beckers T (2013) Encouraging children to think counterfactually enhances blocking in a causal learning task. Q J Exp Psychol 66(10):1910–1926. https://doi.org/10.1080/17470218.2013.767847
McNamara JM, Trimmer PC (2019) Sequential choices using signal detection theory can reverse classical predictions. Behav Ecol 30(1):16–19. https://doi.org/10.1093/beheco/ary132
Meier GW, McGee RK (1959) A re-evaluation of the effect of early perceptual experience in discrimination performance during adulthood. J Comp Physiol Psychol 52(4):390–395. https://doi.org/10.1037/h0045596
Mill RW, Alves-Pinto A, Sumner CJ (2014) Decision criterion dynamics in animals performing an auditory detection task. PLoS ONE 9(12):e114076. https://doi.org/10.1371/journal.pone.0114076
Mistry P, Skewes JC, Lee M (2018) An adaptive signal detection model applied to perceptual learning. Cog Sci
Mueller ST, Weidemann CT (2008) Decision noise: an explanation for observed violations of signal detection theory. Psychon Bull Rev 15(3):465–494. https://doi.org/10.3758/PBR.15.3.465
Niv Y (2019) Learning task-state representations. Nat Neurosci 22(10):1544–1553. https://doi.org/10.1038/s41593-019-0470-8
Odic D, Hock H, Halberda J (2014) Hysteresis affects approximate number discrimination in young children. J Exp Psychol Gen 143(1):255–265. https://doi.org/10.1037/a0030825
Pineño O, Miller RR (2007) Comparing associative, statistical, and inferential reasoning accounts of human contingency learning. Q J Exp Psychol 60(3):310–329. https://doi.org/10.1080/17470210601000680
Rabin MD (1988) Experience facilitates olfactory quality discrimination. Percept Psychophys 44(6):532–540. https://doi.org/10.3758/BF03207487
Ratcliff R, Voskuilen C, McKoon G (2017) Internal and external sources of variability in perceptual decision-making. Psychol Rev 125(1):33. https://doi.org/10.1037/rev0000080
Reader SM (2015) Causes of individual differences in animal exploration and search. Top Cogn Sci 7(3):451–468. https://doi.org/10.1111/tops.12148
Redish AD, Jensen S, Johnson A, Kurth-Nelson Z (2007) Reconciling reinforcement learning models with behavioral extinction and renewal: Implications for addiction, relapse, and problem gambling. Psychol Rev 114(3):784–805. https://doi.org/10.1037/0033-295X.114.3.784
Ritchie JB, Carlson TA (2016) Neural decoding and “Inner” psychophysics: a distance-to-bound approach for linking Mind, brain, and behavior. Front Neurosci. https://doi.org/10.3389/fnins.2016.00190
Roy NA, Bak JH, Akrami A, Brody CD, Pillow JW (2021) Extracting the dynamics of behavior in sensory decision-making experiments. Neuron 109(4):597-610.e6. https://doi.org/10.1016/j.neuron.2020.12.004
Saksida LM (1999) Effects of similarity and experience on discrimination learning: a nonassociative connectionist model of perceptual learning. J Exp Psychol Anim Behav Process 25(3):308–323. https://doi.org/10.1037/0097-7403.25.3.308
Santos C, Sanabria F (2020) Past outcomes and time flexibly exert joint control over midsession reversal performance in the rat. Behav Processes 171:104028. https://doi.org/10.1016/j.beproc.2019.104028
Sewell DK, Jach HK, Boag RJ, Van Heer CA (2019) Combining error-driven models of associative learning with evidence accumulation models of decision-making. Psychon Bull Rev 26(3):868–893. https://doi.org/10.3758/s13423-019-01570-4
Shepard KN, Kilgard MP, Liu RC (2013) Experience-dependent plasticity and auditory cortex. In: Cohen YE, Popper AN, Fay RR (Eds) Neural correlates of auditory cognition (Vol. 45, pp. 293–327). Springer New York. https://doi.org/10.1007/978-1-4614-2350-8_10
Shettleworth SJ (2012) Do animals have insight, and what is insight anyway? Can J Exp Psychol/revue Canadienne De Psychologie Expérimentale 66(4):217. https://doi.org/10.1037/a0030674
St. Claire-Smith R,MacLaren D (1983) Response preconditioning effects. J Exp Psychol 9(1):41–48. https://doi.org/10.1037/0097-7403.9.1.41
Stanislaw H, Todorov N (1999) Calculation of signal detection theory measures. Behav Res Methods Instrum Comput 31(1):137–149. https://doi.org/10.3758/BF03207704
Stüttgen MC, Yildiz A, Güntürkün O (2011) Adaptive Criterion Setting in Perceptual Decision Making. J Exp Anal Behav 96(2):155–176. https://doi.org/10.1901/jeab.2011.96-155
Sutton RS, Barto AG (2018) Reinforcement learning, second edition: an introduction. MIT Press
Teichert T, Ferrera V (2010) Suboptimal integration of reward magnitude and prior reward likelihood in categorical decisions by monkeys. Front Neurosci 4:186. https://doi.org/10.3389/fnins.2010.00186
Trachtenberg JT, Chen BE, Knott GW, Feng G, Sanes JR, Welker E, Svoboda K (2002) Long-term in vivo imaging of experience-dependent synaptic plasticity in adult cortex. Nature 420(6917):788–794. https://doi.org/10.1038/nature01273
Turner BM, Van Zandt T, Brown S (2011) A dynamic stimulus-driven model of signal detection. Psychol Rev 118(4):583. https://doi.org/10.1037/a0025191
Wagenmakers E-J, Love J, Marsman M, Jamil T, Ly A, Verhagen J, Selker R, Gronau QF, Dropmann D, Boutin B, Meerhoff F, Knight P, Raj A, van Kesteren E-J, van Doorn J, Šmíra M, Epskamp S, Etz A, Matzke D, Morey RD (2018a) Bayesian inference for psychology. Part II: Example applications with JASP. Psychonomic Bull Rev 25(1):58–76. https://doi.org/10.3758/s13423-017-1323-7
Wagenmakers E-J, Marsman M, Jamil T, Ly A, Verhagen J, Love J, Selker R, Gronau QF, Šmíra M, Epskamp S, Matzke D, Rouder JN, Morey RD (2018b) Bayesian inference for psychology. Part I: Theoretical advantages and practical ramifications. Psychonomic Bull Rev 25(1):35–57. https://doi.org/10.3758/s13423-017-1343-3
Wickens TD (2002) Elementary Signal Detection Theory. Oxford University Press
Witt JK, Taylor JET, Sugovic M, Wixted JT (2015) Signal detection measures cannot distinguish perceptual biases from response biases. Perception 44(3):289–300. https://doi.org/10.1068/p7908
Wixted JT (2007) Dual-process theory and signal-detection theory of recognition memory. Psychol Rev 114(1):152. https://doi.org/10.1037/0033-295X.114.1.152
Wixted JT (2019) The forgotten history of signal detection theory. J Exp Psychol Learn Mem Cogn 46(2):201. https://doi.org/10.1037/xlm0000732
Xiong Q, Znamenskiy P, Zador AM (2015) Selective corticostriatal plasticity during acquisition of an auditory discrimination task. Nature 521(7552):348–351. https://doi.org/10.1038/nature14225
Yanagihara S, Yazaki-Sugiyama Y (2016) Auditory experience-dependent cortical circuit shaping for memory formation in bird song learning. Nat Commun 7(1):11946. https://doi.org/10.1038/ncomms11946
Zeng F-G, Fu Q-J, Morse R (2000) Human hearing enhanced by noise11Published on the World Wide Web on 23 May 2000. Brain Res 869(1):251–255. https://doi.org/10.1016/S0006-8993(00)02475-6
Znamenskiy P, Zador AM (2013) Corticostriatal neurons in auditory cortex drive decisions during auditory discrimination. Nature 497(7450):482–485. https://doi.org/10.1038/nature12077
Acknowledgements
We would like to thank Charles Gallistel, David Freestone, Başak Akdoğan, and Jorge Mallea for helpful conversations that clarified analysis, interpretation, and presentation of data. We would also like to thank Kimberly Bowman for help with data collection. This work was conducted while Carter W. Daniels was a T32 post-doctoral fellow at Columbia University; he is now a Quantitative Scientist at Prevail Therapeutics—a wholly owned subsidiary of Eli Lilly and Co. Correspondence should be addressed to Carter W. Daniels at carter.wa.daniels@gmail.com.
Funding
Carter W. Daniels was supported by a Fellowship in Schizophrenia Research (5T32MH018870) during this study; this study was supported by National Institutes of Mental Health Grant R01MH068073 to Peter Balsam.
Author information
Authors and Affiliations
Contributions
CD and PB conceived and designed the study. CD collected data, conducted data analysis, and wrote the paper. PB provided feedback throughout the study and helped write the paper.
Corresponding author
Ethics declarations
Conflict of interest
We have no conflicts of or competing interests to report.
Ethical approval
This study was approved by both the Columbia University and New York State Psychiatric Institute Institutional Animal Care and Use Committees under protocols entitled Cognition and Motivation in the Mouse: Behavior and Neurobiology with protocol numbers of AC-AAAW4459 and 1507, respectively.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Daniels, C.W., Balsam, P.D. Prior experience modifies acquisition trajectories via response–strategy sampling. Anim Cogn 26, 1217–1239 (2023). https://doi.org/10.1007/s10071-023-01769-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10071-023-01769-y