Abstract
Objective
Idiopathic inflammatory myopathies (IIM) confer an increased risk of morbidity from atherosclerotic cardiovascular disease (ASCVD). While ASCVD risk has been studied in other countries, these results may not be applicable to patients with dermatomyositis (DM) and polymyositis (PM) in the United States. This retrospective analysis of a cohort of patients identified by ICD code from TriNetX investigated the incidence of ASCVD after International Classification of Disease (ICD) codes of DM, PM, dermatopolymyositis (DPM) or juvenile dermatomyositis (JDM).
Method
Patients were identified by entry of two ICD codes separated by at least 6 months, according to their first diagnosis code; ASCVD was defined as first ICD code for myocardial infarction, ischemic stroke, transient ischemic attack, or peripheral arterial disease. Cox proportional hazards regression modeled time from first IIM ICD code to ASCVD event.
Results
A total of 35,554 patients were identified with the mean age at first IIM code of 54 and 26.1% were male. The most common comorbidity for all groups except JDM was hyperlipidemia (39.9%) though 79.2% of patients were on no cholesterol lowering medication. ASCVD occurred in 30.4% of patients with PM, 24.3% of patients with DM and 0.9% of patients with JDM. Patients with PM had a median time to event of 9.7 years (95% Confidence interval (CI) 9.1, 10.7) and 14.3 years (95% CI 12.6, 14.8) for DM. This study demonstrates that ASCVD is a comorbidity occurring after a median of 12.5 years (95% CI 11.9, 13.6) in patients with IIM.
Conclusions
ASCVD appears to be a long-term complication for IIM patients occurring in nearly a quarter of US patients without prior ASCVD with at least two ICD codes for IIM, with a median time to event of 12.5 years. There appears to be a practice gap in the recognition and treatment of hyperlipidemia in these patients.
Key Points • Hyperlipidemia was a common comorbidity identified in patients with IIM though most patients were not on cholesterol lowering medication. • Development of ASCVD appears to be a long-term complication for patients with IIM in the United States. |
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Atherosclerotic cardiovascular disease (ASCVD) is an important cause of morbidity and mortality in patients with idiopathic inflammatory myopathies (IIM). A meta-analysis of cardiovascular disease defined as ischemic heart disease, cerebrovascular accident and/or venous thromboembolism in IIM patients compared to the general population found a relative risk (RR) of 2.4 (95% confidence interval (CI) 1.5, 3.7) among PM patients and 2.6 (95% CI 1.7, 3.9) for DM patients [2]. Several studies have identified higher risk of coronary artery disease in patients with IIM compared with sex and age matched controls [3,4,5,6,7]. While most studies have demonstrated an increased risk of MI in PM/DM patients, stroke risk has not always been significantly elevated, though this may be due to the infrequency of the event in study populations [3, 6, 8, 9].
Patients with IIM may be at increased risk of ASCVD due to a higher prevalence of traditional ASCVD risk factors compared to the general population. An Australian study found a higher prevalence of hypertension in patients with DM and PM compared with the general population (57%, 61%, and 9%, respectively). Similarly, 30% of patients with DM and 33% with PM had diabetes compared to only 4% in the general population [10]. A multicenter study in Denmark revealed similar results comparing patients with either PM or DM compared to age and sex matched healthy controls found a higher percentage of hypertension (71% vs 42%), diabetes (13% vs 0%), and obesity (33% vs 10%) [11]. Studies comparing patients with DM to healthy individuals have demonstrated a higher prevalence of hypertension (47.6% vs 18.1%) [12], (28.7% vs 22.6%) [6], diabetes mellitus (17.9% vs 1.0%) [12], (13% vs 9.7%) [6], and metabolic syndrome (41.7% vs 7.0%) [12].
The comparative risk of hyperlipidemia for patients with PM or DM versus the general population is less clear. In the Danish study, mean levels of triglycerides were higher in patients than controls but there were no significant differences in serum total cholesterol, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), hypercholesterolemia, family history of CVD, or smoking habits [11]. A Taiwanese study showed a higher frequency of dyslipidemia (15% in DM, 17.4% in PM vs 5.9% in controls), but this finding was not statistically significant. Hyperlipidemia and a family history of premature CVD were significantly higher in patients with DM compared to controls in another study [12]. Even before disease-modifying antirheumatic drug (DMARD) therapy, patients with DM may be at increased risk for dyslipidemia. In a case control study at a single center in China, patients with DM prior to DMARD or steroid treatment, compared with age and sex matched controls, had significantly lower HDL-C, and higher very low density lipoprotein-cholesterol (VLDL-C) and triglycerides [13].
In addition to the greater ASCVD risk for patients with IIM, there is a significant association between mortality and cardiovascular disease in patients with IIM. In a retrospective Australian study, cardiovascular disease was the most common cause of death in patients with IIM (acute myocardial infarction caused 16/36 of cardiovascular deaths). In the same study, patients with IIM and ischemic heart disease had almost triple the risk for death (risk ratio = 3.0, 95% CI 1.6–5.3) compared to IIM patients without ischemic heart disease [14]. A large US study of hospitalized patients with DM found that about 20% of these hospitalizations were associated with ASCVD, that DM patients with ASCVD had double the odds of death during hospitalization (OR = 2, 95% CI 1.7–2.5), and that patients with both DM and cardiovascular disease were almost twice as likely to die as those with cardiovascular disease alone (OR = 2, 95% CI 1.6–2.5) [15].
Though there are several studies examining ASCVD risk in patients with PM or DM in other countries, there are limited data available regarding ASCVD risk in PM/DM patients in the United States (US). We hope to expand upon this work through the inclusion of patients with juvenile dermatomyositis (JDM) in a US population. The primary aim was to describe when ASCVD, as defined by ICD codes, occurs for patients with IIM without prior coding for ASCVD and to assess if there are differences between IIM subtypes as defined by initial ICD code. The secondary objective was to determine the utilization of cholesterol lowering medications.
Materials and methods
Study design and data source
This was a retrospective cohort analysis of TriNetX database, a health research network which incorporates de-identified data of electronic medical records (diagnoses, procedures, medications, laboratory values) from participating healthcare organizations in the United States. The data is de-identified based on the standard defined in Section §164.514(a) of the HIPAA Privacy Rule. The process by which datasets are de-identified is attested to through a formal determination by a qualified expert as defined in Section §164.514(b)(1) of the HIPAA Privacy Rule. TriNetX covers most regions with approximately 20% of patients from the Northeast, Midwest and West and 40% from the South. The data was acquired on March 21, 2022, with dates ranging from 01/09/2001 to 01/22/2020, the mean follow-up time including censored and time to ASCVD event was 4 years (standard deviation 2.7 years).
Patient selection
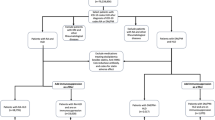
Only patients in the United States were included and were identified by entry of two ICD codes separated by at least 6 months apart, according to their first diagnosis code (i.e., DM, PM, DPM or JDM) (Fig. 1). Exposures included the first IIM ICD code which were noted as follows: DM (710.3, M33.1x), PM (710.4, M33.2x), DPM (M33.9x, M36.0x), or JDM (M33.0 × or anyone less than 18 years of age for 710.3 or M33.1x) (Supplementary Table 1). The use of ICD codes for DM or PM enrollment for database research has previously been described [16,17,18]. Patients with an ASCVD code that preceded the IIM diagnosis code were excluded in order to capture primary prevention (i.e., no prior history of ASCVD). Patients were censored at the time of last reporting of any diagnosis code. The mean length of time that patients were followed was 4 years (standard deviation of 2.7 years).
Primary outcome measure
The primary outcome was time from first IIM ICD code to ASCVD ICD code. We defined ASCVD as entry of an ICD code (Supplementary Table 1) based on the 2018 American Heart Association definition of ASCVD which includes myocardial infarction, ischemic stroke, transient ischemic attack or peripheral arterial disease [19].
Covariable assessment
Demographics in TriNetX include sex and age, but missing data precluded addition of race and ethnicity information. The following comorbidities, which could influence the risk of ASCVD, were also evaluated, with inclusion if the patient had two ICD codes separated by at least 90 days: hyperlipidemia, hypertension, diabetes, overweight, obesity, smoking, alcohol. Hyperlipidemia was assessed by ICD code not by measured lipid level. Steroid use was assessed by National Drug Code (NDC) which represents at least one dispense for the following: methylprednisolone, prednisone, prednisolone, dexamethasone (Supplementary Table 4).
Statistical analysis
Cox proportional hazards (CPH) regression was performed to model the time from initial IIM diagnosis code to ASCVD outcome allowing the generation of hazard ratios to compare ASCVD risk by ICD code (e.g., DM, DPM, PM or JDM). The CPH model was adjusted for age at first IIM diagnosis, sex, comorbidities and steroid use listed above, and use of 5-group generation from year of birth as strata to account for calendar effects. We assessed for evidence of effect modification between ICD IIM code and other covariables using interaction terms at 0.05 alpha level. We also assessed for evidence of non-proportional hazards using Schoenfeld residuals and used stratification, if evidence was found for any covariables.
Results
A total of 35,553 patients were identified, of whom 26.1% were male, and the mean age at first IIM diagnosis code was 54.4 years (Table 1). Patients were grouped into PM, DM, DPM or JDM based off the first ICD code entry. The most frequent group was PM, a small subset (70 patients) had a code for DM and PM on the same date although this made up 0.2% of the sample and was not included in the main statistical modeling (available in Supplementary Tables 2 and 3). Age at diagnosis was similar among DM, PM, and DPM, although PM had a slightly higher mean age (Table 1). The most common comorbidity overall was hyperlipidemia (39.9%), which was the most frequent among all groups except JDM, where the most common comorbidity was overweight and/or obese status (5.8%). Most patients (79.2%) were not on a cholesterol medication, and 20% had a prior dispensation for a statin medication.
In the overall sample, ASCVD occurred in 24.5% of patients with a median time from first ICD code to event of 12.5 years (95% CI 11.9, 13.6). Of the patients who had an ASCVD code, 589 (6.8%) had more than 1 ASCVD code on the same date (e.g., myocardial infarction and coronary arterial disease code). The ASCVD outcome was most frequent among patients with PM (30.4%), DM (24.3%), DPM (14.8%) and was infrequent among patients with JDM (< 1%). The median time to ASCVD event was 9.7 years (95% CI 9.1, 10.7) for PM, 14.3 (95% CI 12.6, 14.8) years for DM, and not estimable for JDM due to the low frequency of events (Fig. 2).
Kaplan–Meier ASCVD free survival curve with 95% confidence intervals. Legend: ASCVD free survival with number of participants at risk, participants were censored with a vertical line at the time of last reporting of any diagnosis code. Abbreviations: ASCVD: Atherosclerotic Cardiovascular Disease; JDM: Juvenile Dermatomyositis; DPM: Dermatopolymyositis; DM: Dermatomyositis; PM: Polymyositis
Due to the low frequency of events in the JDM group, all other IIM categories had a higher unadjusted hazard of ASCVD. Although when adjusting for other comorbidities, such as age, this was not statistically significant (Table 2). Patients with PM had approximately 30% increased hazards of ASCVD development compared to DPM (aHR 1.29, 95% CI 1.21, 1.39) and DM (aHR 1.27, 95% CI 1.21, 1.33) when adjusted for age at first diagnosis, birth strata, sex, overweight, obesity, hyperlipidemia, hypertension, diabetes, smoking, alcohol use and anytime use of steroids. We found evidence of effect measure modification for hyperlipidemia at 0.05 alpha level, thus we stratified the results by hyperlipidemia (Table 2). The aHR was similar although slightly higher among those with PM and no hyperlipidemia (PM vs DM without hyperlipidemia (aHR 1.34, 95% CI 1.24, 1.45) compared to those with hyperlipidemia (aHR 1.23, 95% CI 1.15, 1.30) although this effect was small as the confidence intervals overlap.
The association between time to ASCVD development and IIM category (assessed by first TriNetX ICD code) displayed evidence of non-proportional hazards for age and presence of hypertension (Table 3). The effect of these two covariables (age and hypertension) varied over time. Thus, the analysis was stratified by hypertension and age group ≥ 65 and aHR were shown by these strata. While PM compared to DM had increased hazards of 42% (aHR 1.42, 95% CI 1.29, 1.56) for patients without hypertension and less than age 65 — this was larger than the hazard for PM compared to DM for patients with hypertension and age 65 or older (aHR 1.18, 95% CI 1.08, 1.29).
Discussion
The risk of ASCVD in US patients with IIM is understudied, and the primary aim was to describe when ASCVD occurs a population of patients with IIM without prior ASCVD. We show that a quarter of IIM patients develop new onset ASCVD with a median of 12.5 years from diagnosis, appearing sooner for PM, than DM. Furthermore, PM patients seem to be at uniquely elevated risk. Our data define a window of increased risk for the development of ASCVD in patients without preceding ASCVD and highlight the surprising underutilization of lipid-lowering therapy in a patient population with elevated risk. This previously unrecognized gap represents an opportunity for enhanced risk management in IIM patients. Assessing long-term ASCVD risk may be key: in a Swedish population based study of patients with IIM, the incidence rate of deaths from diseases of the circulatory system was persistently elevated compared to the general population but highest after 10 years from IIM diagnosis (28.5 deaths/1000 person years compared to 12.8 deaths/1000 person years in the general population) [20]. Studying the time preceding an event is critical as this represents an ideal target window for risk reduction. The increased risk of ASCVD faced by IIM patients may not be captured by traditional risk calculators. From a single center in Czech Republic, there was a discordance for patients with IIM between Systemic Coronary Risk Evaluation (SCORE), a widely used European cardiovascular disease risk calculator, and imaging of subclinical atherosclerosis, resulting in approximately one third of patients getting reclassified to a higher CV risk level after imaging [21].
Patients at high risk of ASCVD may have suboptimal management from concern for a potential association between statin use and dermatomyositis development [22,23,24] or concern for triggering a myositis flare or muscle symptoms [25, 26]. In support of this point, although 40% of patients with IIM in our study had a diagnosis of hyperlipidemia, four out of five (79.2%) were not on any cholesterol medication. Use of statins for primary prevention in IIM is understudied: a single retrospective cohort of IIM patients found 32 of 214 patients reported statin use, of whom, most were used (21/32) for primary prevention. Within this cohort, the mean ASCVD Pooled Cohort Equation of 10-year ASCVD risk was 11.8% for patients not on statins this was not significantly different from those on statins, however this highlights a potential gap in care as a score above 7.5% would indicate statin treatment. This study found no significant difference in disease activity between those on statins and not on statins and for a subgroup started on statins (n = 7), no increase in disease activity measures [27]. A UK population-based assessment of patients with a systemic autoimmune rheumatic disease, including IIM, who initiated statin use had reduced all-cause mortality when compared to a group not on statins although there was no subgroup analysis by primary or secondary prevention and the IIM subgroup was too small to evaluate on its own [28]. These studies highlight the need for assessment of primary prevention and statin use for IIM patients.
This study found that individuals with coding for PM experience higher hazards of ASCVD, and these events appear sooner than peers with DM or DPM. Our results stand in contrast to a meta-analysis which defined cardiovascular events as ischemic heart disease, stroke and/or venous thromboembolism which demonstrated a slightly higher relative risk for patients with DM compared to PM (2.55, 95% CI 1.66, 3.93 and 2.35, 95% CI 1.51, 3.66, respectively) [2]. A South Korean population based study found lower hazards for cardiovascular disease for PM patients [29]. While some studies have noted a higher relative risk of coronary disease for patients with PM compared to DM [3, 4], many have reported pooled results compared to the general population [5, 7]. The inconsistency between DM and PM likely reflects the heterogeneity of PM. There has been greater understanding and molecular phenotyping of IIM with the addition of myositis-specific autoantibodies. This heterogeneity is not reflected in the current ICD codes which lack terms for immune-mediated necrotizing myopathy (IMNM) and anti-synthetase syndrome. Patients with PM likely represent a heterogenous collection of patients with IMNM, anti-synthetase syndrome, and other IIM entities, which may confer different risks. Patients with IMNM may be at increased risk compared to other subtypes from a single center assessment of atherosclerosis, 20 patients with IMNM had a trend towards the highest measures of subclinical atherosclerosis, when compared to other 70 patients with different IIM subtypes [21]. The PM group in this study may include patients with IMNM and patients with anti-HMG CoA reductase antibody who should not be on statin medications potentially making ASCVD risk management more challenging.
A strength of this study is the evaluation of a large number of patients evaluated in a real-world dataset. IIM is a rare disease with an estimated prevalence of 14 to 17.4 per 100,000 making an infrequent event (ASCVD) challenging to study at a single center [30]. This study has several limitations, the first of which is the reliance on ICD coding to identify cases of IIM and the lack of ability to perform chart review to ensure appropriate patient inclusion or assess disease activity. There is misclassification bias associated with use of ICD codes for case detection — we anticipate that there would be inclusion of more false positive cases (patients with the ICD code without disease) than false negative cases. By requiring at least two codes six months apart — we also may be introducing bias by not be capturing sicker patients who do not have full six months of data. Another limitation is that this dataset has a high amount of missing race/ethnicity data and is confined to patients who have health insurance. Age is an important confounder, and we did find evidence that this covariable’s effect on ASCVD varied with time (e.g., non-proportional hazards). However, we were able to stratify by age greater than or equal to 65 compared to less than 65 and overall, the effects were similar with a slight increase in hazards for patients who have the diagnosis code of PM compared to DM or DPM. Another limitation is the lack of an age or sex-matched population comparator in this study.
This study highlights that in a group of US patients with ICD codes for IIM without pre-existing ASCVD, ASCVD occurred in approximately a quarter of the patients with a median time to event of 12.5 years. Individuals with an initial ICD code for PM had higher hazards of ASCVD compared to individuals with DM or DPM code. Despite nearly 40% of patients with the diagnosis of hyperlipidemia, most patients (79.2%) were not on any cholesterol lowering medications which may reflect a need for more accurate risk stratification and treatment or concern that cholesterol lowering medications may trigger muscle symptoms or disease activity. This may represent a significant practice gap. Additional study is needed to evaluate how ASCVD develops in patients with IIM, how patients should be risk stratified and how cholesterol lowering medications are used in this population.
References
Allenzara A, Alvarez C, Nelson A, Foulke G (2023) Frequency of Atherosclerotic Cardiovascular Disease Following a Diagnosis of Idiopathic Inflammatory Myopathy: Data from a Large National Registry. Arthritis Rheumatol 74 (suppl 9) abstract #2577
Xiong A, Hu Z, Zhou S et al (2022) Cardiovascular events in adult polymyositis and dermatomyositis: a meta-analysis of observational studies. Rheumatol Oxf Engl 61:2728–2739. https://doi.org/10.1093/rheumatology/keab851
Rai SK, Choi HK, Sayre EC, Aviña-Zubieta JA (2016) Risk of myocardial infarction and ischaemic stroke in adults with polymyositis and dermatomyositis: a general population-based study. Rheumatol Oxf Engl 55:461–469. https://doi.org/10.1093/rheumatology/kev336
Weng M-Y, Lai EC-C, Kao Yang Y-H (2019) Increased risk of coronary heart disease among patients with idiopathic inflammatory myositis: a nationwide population study in Taiwan. Rheumatol Oxf Engl 58:1935–1941. https://doi.org/10.1093/rheumatology/kez076
Lin Y-N, Lin C-L, Chang K-C, Kao C-H (2015) Increased subsequent risk of acute coronary syndrome for patients with dermatomyositis/polymyositis: a nationwide population-based retrospective cohort study. Scand J Rheumatol 44:42–47. https://doi.org/10.3109/03009742.2014.918652
Lai Y-T, Dai Y-S, Yen M-F et al (2013) Dermatomyositis is associated with an increased risk of cardiovascular and cerebrovascular events: a Taiwanese population-based longitudinal follow-up study. Br J Dermatol 168:1054–1059. https://doi.org/10.1111/bjd.12245
Leclair V, Svensson J, Lundberg IE, Holmqvist M (2019) Acute Coronary Syndrome in idiopathic inflammatory myopathies: a population-based study. J Rheumatol 46:1509–1514. https://doi.org/10.3899/jrheum.181248
Moshtaghi-Svensson J, Lundberg IE, Von Euler M et al (2019) The risk of ischemic and hemorrhagic stroke in patients with idiopathic inflammatory myopathies: a Swedish population-based cohort study. Arthritis Care Res 71:970–976. https://doi.org/10.1002/acr.23702
Ungprasert P, Cheungpasitporn W, Wijarnpreecha K et al (2015) Risk of ischemic stroke in patients with polymyositis and dermatomyositis: a systematic review and meta-analysis. Rheumatol Int 35:905–909. https://doi.org/10.1007/s00296-014-3166-0
Limaye VS, Lester S, Blumbergs P, Roberts-Thomson PJ (2010) Idiopathic inflammatory myositis is associated with a high incidence of hypertension and diabetes mellitus. Int J Rheum Dis 13:132–137. https://doi.org/10.1111/j.1756-185X.2010.01470.x
Diederichsen LP, Diederichsen ACP, Simonsen JA et al (2015) Traditional cardiovascular risk factors and coronary artery calcification in adults with polymyositis and dermatomyositis: a Danish multicenter study. Arthritis Care Res 67:848–854. https://doi.org/10.1002/acr.22520
de Moraes MT, de Souza FHC, de Barros TBM, Shinjo SK (2013) Analysis of metabolic syndrome in adult dermatomyositis with a focus on cardiovascular disease. Arthritis Care Res 65:793–799. https://doi.org/10.1002/acr.21879
Wang H, Tang J, Chen X et al (2013) Lipid profiles in untreated patients with dermatomyositis. J Eur Acad Dermatol Venereol JEADV 27:175–179. https://doi.org/10.1111/j.1468-3083.2011.04437.x
Limaye V, Hakendorf P, Woodman RJ et al (2012) Mortality and its predominant causes in a large cohort of patients with biopsy-determined inflammatory myositis. Intern Med J 42:191–198. https://doi.org/10.1111/j.1445-5994.2010.02406.x
Linos E, Fiorentino D, Lingala B et al (2013) Atherosclerotic cardiovascular disease and dermatomyositis: an analysis of the Nationwide Inpatient Sample survey. Arthritis Res Ther 15:R7. https://doi.org/10.1186/ar4135
Kwa MC, Ardalan K, Laumann AE et al (2017) Validation of international classification of diseases codes for the epidemiologic study of dermatomyositis. Arthritis Care Res 69:753–757. https://doi.org/10.1002/acr.23010
Svensson J, Arkema EV, Lundberg IE, Holmqvist M (2017) Incidence and prevalence of idiopathic inflammatory myopathies in Sweden: a nationwide population-based study. Rheumatology 56:802–810. https://doi.org/10.1093/rheumatology/kew503
Bernatsky S, Linehan T, Hanly JG (2011) The accuracy of administrative data diagnoses of systemic autoimmune rheumatic diseases. J Rheumatol 38:1612–1616. https://doi.org/10.3899/jrheum.101149
Grundy SM, Stone NJ, Bailey AL et al (2019) 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the management of blood cholesterol: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 73:3168–3209. https://doi.org/10.1016/j.jacc.2018.11.002
Dobloug GC, Svensson J, Lundberg IE, Holmqvist M (2018) Mortality in idiopathic inflammatory myopathy: results from a Swedish nationwide population-based cohort study. Ann Rheum Dis 77:40–47. https://doi.org/10.1136/annrheumdis-2017-211402
Oreska S, Storkanova H, Kudlicka J et al (2023) Cardiovascular risk in myositis patients compared to the general population. Rheumatol Oxf Engl kead271. https://doi.org/10.1093/rheumatology/kead271
Visconti MJ, Bashyam A, Jorizzo JL (2020) Statin-induced dermatomyositis for the practicing dermatologist: a review of the literature. Int J Dermatol 59(3):383–7. https://doi.org/10.1111/ijd.14751
Spiro J, Butts M (2018) Atorvastatin-induced dermatomyositis: resolution with change in statin? J Clin Rheumatol Pract Rep Rheum Musculoskelet Dis 24:406–409. https://doi.org/10.1097/RHU.0000000000000709
Oztas M, Ugurlu S, Aydin O (2017) Atorvastatin-induced dermatomyositis. Rheumatol Int 37:1217–1219. https://doi.org/10.1007/s00296-017-3658-9
Caughey GE, Gabb GM, Ronson S et al (2018) Association of statin exposure with histologically confirmed idiopathic inflammatory myositis in an Australian population. JAMA Intern Med 178:1224–1230. https://doi.org/10.1001/jamainternmed.2018.2859
Borges IBP, Silva MG, Misse RG, Shinjo SK (2018) Lipid-lowering agent-triggered dermatomyositis and polymyositis: a case series and literature review. Rheumatol Int 38:293–301. https://doi.org/10.1007/s00296-017-3821-3
Bae SS, Oganesian B, Golub I, Charles-Schoeman C (2020) Statin use in patients with non-HMGCR idiopathic inflammatory myopathies: a retrospective study. Clin Cardiol 43:732–742. https://doi.org/10.1002/clc.23375
Jorge AM, Lu N, Keller SF et al (2018) The effect of statin use on mortality in systemic autoimmune rheumatic diseases. J Rheumatol 45:1689–1695. https://doi.org/10.3899/jrheum.171389
Jung KH, Kim HJ, Park W et al (2020) Incidence, survival, and risk of cardiovascular events in adult inflammatory myopathies in South Korea: a nationwide population-based study. Scand J Rheumatol 49:323–331. https://doi.org/10.1080/03009742.2019.1707281
Furst DE, Amato AA, Iorga ŞR et al (2012) Epidemiology of adult idiopathic inflammatory myopathies in a U.S. managed care plan. Muscle Nerve 45:676–683. https://doi.org/10.1002/mus.23302
Acknowledgements
The authors would like to thank Dr. Fred Miller for his insightful feedback and edits.
Funding
Funding for this study was provided in part by: NIH/NIAMS P30AR072580 (Alvarez, Nelson), NIH/NIAMS K24AR081368 (Nelson), and a Dermatology Foundation Medical Dermatology Career Development Award (Foulke). The funding sources had no role in study design, collection or interpretation of data, or the decision to submit.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Galen Foulke consulted with Astra Zeneca (on use of anifrolumab for cutaneous lupus erythematosus) who had no role in study design, collection or interpretation of data, or the decision to submit. Astia Allenzara’s spouse is employed by LatigoBio who had no role in study design, collection or interpretation of data, or the decision to submit. The other authors have disclosed no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This has been presented at American College of Rheumatology Convergence 2023 – see citation below in footnote [1].
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Allenzara, A., Jicha, K., Álvarez, C. et al. Atherosclerotic cardiovascular disease following a diagnosis of idiopathic inflammatory myopathy: analysis from a retrospective cohort in the TriNetX registry. Clin Rheumatol (2024). https://doi.org/10.1007/s10067-024-07109-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10067-024-07109-w