Abstract
Objectives
This study aimed to investigate subclinical left ventricle (LV) systolic dysfunction in juvenile dermatomyositis (JDM) using two-dimensional speckle-tracking echocardiography (2DST). Possible associations between LV deformation impairment and disease activity/cumulative damage were also evaluated.
Methods
Thirty-five consecutive JDM patients without cardiac symptoms and 35 healthy volunteers were enrolled. Clinical data were collected from medical records, and echocardiograms were performed by a pediatric cardiologist, unaware of patients’ conditions.
Results
Patients and controls had similar age (12.6 ± 0.7 vs.12.5 ± 0.6; p = 0.97) and gender (11F:24M vs.11F:24M; p = 1.0). Median of JDM duration was 4.6 (0.04–17.6) years, and only 6/35 (17%) had active disease (disease activity score (DAS > 3)). Conventional echocardiogram revealed preserved LV ejection fraction (EF) (≥ 55%) in all individuals. In JDM, 2DST identified reduction of LV longitudinal [−22(−17.2 to −27.9) % vs. −23(−20.8 to −27.4) %; p = 0.028)] and circumferential −23.9 ± 2.8% vs. −26.7 ± 2.9%; p = 0.0002) strain. Lower longitudinal strain was associated with DAS >3 −19.9(−17.2 to −26.5)% vs. −22.1−18.9 to −27.9)%; p = 0.046], MDI extent > 0 [−19(−17.2 to −22.5)% vs. −22.1−19.2 to −27.9)%; p = 0.0008], MDI severity > 0 [−19(−17.2 to −22.1)% vs. −22.3(−20.3 to −27.9)%; p = 0.0001] and calcinosis[−20.6(−17.2 to −23)% vs. −22.3(−20.3 to −27.9)%; p = 0.03]. Lower circumferential strain was associated with MDI extent > 0 (−22.1 ± 3.87% vs. −24.4 ± 2.3%; p = 0.039), MDI severity > 0 (−21.7 ± 3% vs. 24.7 ± 2.3%; p = 0.004) and calcinosis (−22.5 ± 3.3% vs. −24.8 ± 2.1%; p = 0.02). There was a negative correlation between longitudinal strain and cumulative dose of prednisone (r = −0.44; p = 0.009) and methotrexate (r = −0.33; p = 0.0008).
Conclusions
LV 2DST detected early systolic myocardial compromise in asymptomatic pediatric JDM patients, with preserved EF. Longitudinal strain impairment was associated with disease activity and cumulative damage, whereas circumferential strain impairment was associated exclusively with cumulative damage.
Key Points: • Serious cardiac involvement is rare but has been associated with death in juvenile dermatomyositis. • Two-dimensional speckle tracking stands out for the identification of subclinical myocardial compromise in juvenile dermatomyositis. • Longitudinal strain impairment is associated with disease activity and cumulative damage, whereas circumferential strain impairment is associated exclusively with cumulative damage. |
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Juvenile dermatomyositis (JDM) is an autoimmune inflammatory muscle disorder and vasculopathy that affects children and adolescents younger than 18 years. Perivascular inflammation also affects the gastrointestinal tract, lungs, and heart. It is the most frequent childhood inflammatory myopathy, although it is less common in pediatric patients than in adults. It has an incidence of two to three cases/million children/year in the general population. Myocardial compromise is classically attributed to myocarditis, fibrosis, atherosclerosis, and vasculitis, mostly with a long subclinical course [1].
Early detection of myocardial involvement in JDM is an arduous task, as clinical signs and symptoms are often non-existent or non-specific. Myocardial impairment becomes clinically evident in advanced stages of the disease, usually with an unfavorable prognosis. Cardiovascular mortality ranges from 5 to 17%, most often caused by myocardial infarction and heart failure [2]. Most echocardiographic parameters routinely used to assess ventricular function lack sensitivity in a subclinical scenario, which certainly contributes to underestimate the prevalence of myocardial involvement in young JDM patients [3].
Two-dimensional speckle tracking (2DST) is an angle-independent method for echocardiographic assessment of myocardial deformation (strain) in longitudinal, radial, and circumferential directions. This diagnostic tool has proven to have better accuracy in detecting subtle ventricular dysfunction in other pediatric rheumatologic diseases, even before ejection fraction (EF) compromise [4, 5].
In a recent publication, Guerra et al. have documented left ventricle (LV) longitudinal strain reduction in polymyositis and dermatomyositis, in patients with still preserved LV EF [6]. Nevertheless, those authors did not find any correlation between LV strain and disease activity or cumulative damage status. Besides, an eventual relationship between early myocardial damage and health-related quality of life parameters in inflammatory myopathies is yet to be studied.
Therefore, the aim of the present study was to investigate subclinical abnormalities in LV systolic function in JDM patients, using 2DST LV-derived longitudinal and circumferential strain. Possible associations between LV strain compromise, and clinical parameters were also interrogated.
Methods
From June 2017 to December 2018, 35 consecutive JDM patients were recruited during their routine outpatients’ visits to the Paediatric Rheumatology Unit of our University Hospital. All patients fulfilled the Bohan and Peter criteria for JMD [7], and none of them showed symptoms of heart failure (New York Heart Association class I) [8]. The control group comprised 35 healthy volunteers from primary care clinics. Congenital heart diseases had been excluded in all patients and controls by previous echocardiographic evaluations. The ethics committee of our institution approved this cross-sectional study, and written informed consent was obtained from all participants and their legal guardians.
Patients’ medical records were carefully reviewed for clinical, laboratory, and therapeutic data by the attendant physician. Disease Activity Score (DAS) > 3, Childhood Myositis Assessment Score (CMAS) < 48, and manual muscle strength test (MMT) < 78 were used as parameters of active disease. Myositis Damage Index Extent (MDI-extent) > 0 and Myositis Damage Index Severity (MDI-severity) > 0 were used as parameters of cumulative damage, as well as the presence of calcinosis [9,10,11]
Childhood Health Assessment Questionnaire (CHAQ) > 0 PedsQL TM (Pediatric Quality of Life inventory) reported by the patient < 78 and a PedsQL TM reported by parents < 78 were indicative of compromised functional abilities and health-related quality of life deterioration parameters, respectively [12,13,14].
Demographic information included age at diagnosis, current age, disease duration, and gender. Weight and height were obtained for each patient and control before the echocardiogram to calculate body surface area (BSA) by the Haycock formula [15].
Standard and 2DST echocardiograms were obtained by the same pediatric cardiologist (MFRD), blinded to disease activity and damage and health-related quality-of-life parameters.
Standard echocardiography
Standard transthoracic echocardiography was performed according to the recommendations of the American Society of Echocardiography and included M-mode, two-dimensional imaging, conventional, and tissue Doppler evaluation at the septal and lateral mitral annulus [16]. The equipment used was a Philips Affiniti 70 (Andover, MA 01810 USA), with multi-frequency transducers (S 5-1 and S 8-3 MHz).
Cardiac chamber dimensions were obtained using two-dimensional mode, and left ventricle ejection fraction (LV EF) was calculated by Simpson’s method [16]. LV mass (g) was estimated using Devereaux’s formula according to the Penn convention and indexed for height (m) raised to an exponential power of 2.7 [16].
Evaluation of LV diastolic function included conventional as well as tissue Doppler-based measurements: mitral E and A velocities, E/A ratio, and E/E’ ratio, with E’ being the average of values obtained by tissue Doppler at the septal and lateral annulus [16]. Left atrial volume was estimated using the biplane area-length method, and values were indexed to the BSA [16].
2DST echocardiography
The main principle of 2DST is that each segment of myocardial tissue displays a specific pattern of gray values in the ultrasound image, commonly referred to as a speckle pattern. Tracking this acoustic pattern during the cardiac cycle enables the observer to follow myocardial motion and to directly assess ventricular deformation [17].
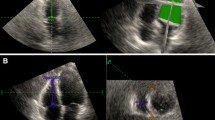
To evaluate global longitudinal LV systolic deformation, two-dimensional harmonic image cine-loop recordings of apical four-, three-, and two-chamber views, with good-quality electrocardiogram signals, were acquired and stored digitally for analysis. A sector scan angle of 30–60° and frame rates of 60–90 Hz were chosen. The endocardial and epicardial tracing was automatically generated by the computer algorithm and manually adjusted to cover the whole myocardium wall, when necessary (QLabTM software, Philips). Global LV peak longitudinal systolic strain (PLSS) was calculated, representing the average values of all the ventricular segments analyzed in the three views (Fig. 1a, b, and c). Parasternal short-axis views at the papillary muscle level were obtained to assess peak circumferential systolic strain (PCSS) (Fig. 1d) [18].
The extent of myocardial deformation (strain) in longitudinal and circumferential directions throughout the cardiac cycle was presented as percentages. Since those deformation parameters reflect myocardial shortening, they were expressed as negative values [18].
Whenever one or more ventricular segments were inadequately tracked due to poor image quality, the individual was excluded from 2DST evaluation.
Intra- and interobserver reproducibility was tested, regarding 2DST measurements. The first examiner (MFRD) repeated the analysis of 5 JDM patients and 5 healthy controls randomly selected, 12 months after having acquired images. A second observer (GNL), unaware of previous results, also performed off-line analysis of the same individuals.
Statistical analysis
Statistical analysis was performed using SPSS version 13.0 (SPSS Inc, Chicago IL, USA). Categorical data were reported as percentages and continuous data as mean ± standard deviation (sd) or median (range). Student’s t test was used to assess normally distributed continuous data and Mann-Whitney test to assess non-normally distributed continuous data. Fisher’s exact test was chosen to compare categorical data. Pearson’s correlation was used to investigate the relationship between global LV peak longitudinal systolic strain and parameters of disease activity, cumulative damage, health-related quality of life, and treatment. The level of significance was set at 5% (p < 0.05). Intra- and interobserver variability for strain measurements was assessed using intraclass correlation coefficient (ICC), with good correlation being defined as ICC > 0.8.
Results
JDM patients and controls had similar age (12.6 ± 0.7 vs. 12.5 ± 0.6; p = 0.97) and identical gender distribution (11F:24M vs. 11F:24M; p = 1.0). The other demographic parameters were also comparable (Table 1).
The median age at JDM diagnosis was 6.2 (2.1–13.6) years, and the median disease duration by the time of the echocardiogram was 4.6 (0.04–17.6) years. No patients nor controls had additional cardiovascular risk factors such as arterial hypertension or dyslipidemia, according to medical registers from outpatients’ clinics. Two patients (5.7%) had pulmonary involvement, two (5.7%) dysphagia and one (2.8%) dysphonia.
Disease activity indexes
Six (17%) patients had DAS > 3; 8 (22.8%) had CMAS < 48; and 3 (8.5%) had MMT < 78.
Cumulative damage indexes
Eight (22.8%) patients had MDI extension > 0; 9 (25.7%) had MDI severity > 0, and 14 (40%) exhibited calcinosis.
Functional abilities and health-related quality-of-life parameters
Eighteen (51.4%) patients had CHAQ > 0; 11 (31.4%) had PedsQL TM reported by the patient < 78, and 11 (31.4%) had PedsQL TM reported by parents < 78.
Laboratory
Median value of patients’ creatine phosphokinase (CPK) was 100 (36–1579) IU/L, lactic dehydrogenase (LDH) 200 (119–511) IU/L, erythrocyte sedimentation rate (ESR) 12 (3–47) mm/1st h, C-reactive protein (CRP) 1.0 (0–10) mg/ml, alanine transaminase (ALT) 17 (9–32), and aspartate transaminase (AST) 21 (11–45) U/L.
Treatment
By the time of echocardiogram, 18 (51%) patients were in use of hydroxychloroquine, 8 (23%) prednisone, 4 (11%) azathioprine, 16 (45%) methotrexate, 3 (8%) mycophenolate mofetil, 1 (3%) cyclosporine, and 12 (34.3%) were not receiving any medications.
Thirteen (37%) patients had received intravenous immunoglobulin and 10 (28.5%) azathioprine at some point of their previous follow-up.
Standard echocardiogram
LV ejection fraction was preserved (> 55%) in all individuals, although lower in patients than in controls (67.5 ± 4% vs. 69.5 ± 3.9%; p = 0.04) (Table 1). Although patients showed a significant reduction of e’ velocity at septal and lateral mitral annulus, all values obtained were still within normal range [19]. E/e’ and atrial volume were similar comparing patients and controls, reassuring normal LV end diastolic pressure (Table 1).
2DST echocardiogram
Satisfactory images were obtained from all JDM patients and controls, and no individuals were excluded from myocardial deformation analysis. Patients exhibited lower LV global PLSS, as well as lower PCSS than controls (Table 1).
Lower longitudinal strain was associated with DAS > 3, MDI extent > 0, MDI severity > 0, and calcinosis (Table 2). Lower LV PCSS was associated with MDI extent > 0, MDI severity > 0, and calcinosis (Table 2).
There was a negative correlation between LV global PLSS and the number of abnormal JDM clinical parameters per patient, including disease activity (DAS > 3, CMAS < 48, MMT < 78), cumulative damage (MDI-extent > 0, MDI-severity > 0, the presence of calcinosis), and functional abilities and health-related quality of life parameters (CHAQ > 0, PedsQLTM reported by the patient < 78, PedsQLTM reported by parents < 78) (Fig. 2a).
a: correlation between LV Global PLSS (absolute values) and the number of abnormal JDM clinical parameters per patient, including disease activity (DAS > 3, CMAS < 48, MMT < 78), cumulative damage (MDI-extent > 0, MDI-severity > 0, presence of calcinosis) and quality of life (CHAQ > 0, PedsQL TM reported by the patient < 78 and PedsQL TM reported by parents < 78) indexes b: correlation between LV Global PLSS (absolute values) and cumulative prednisone dose (g) c: correlation between LV Global PLSS (absolute values) and cumulative methotrexate dose (g). JDM: juvenile dermatomyositis; LV Global PLSS: left ventricle global peak longitudinal systolic strain; DAS: Disease Activity Score; CMAS: Childhood Myositis Assessment Score; MMT: Manual Muscle Strength Test; MDI: Myositis Damage Index; CHAQ: Childhood Health Assessment Questionnaire; PedsQL TM: Pediatric Quality of Life inventory
There was a negative correlation between LV global PLSS and cumulative dose of prednisone and methotrexate (Fig. 2b and c). The median value of PLSS in patients’ group was −22%; the frequency of individuals with past/present use of azathioprine among patients with lower values of LV global PLSS (< −22%) was greater than the frequency identified among patients with higher values of LV global PLSS (≥ −22%): 8/17 (47%) vs. 2/18 (11%); p = 0.027.
There was no correlation between serum muscles enzymes (CPK, LDH, AST, ALT) or inflammatory markers (CRP, ESR) and LV myocardial deformation parameters (global PLSS and PCSS) (p > 0.05).
Strain measures reproducibility
Regarding LV global PLSS, the ICC obtained was 0.81 (95% confidence interval: 0.78– 0.95; p = 0.0042) for interobserver variability and 0.94 (95% confidence interval: 0.77–0.98; p < 0.0001) for intraobserver variability. Regarding LV PCSS, the ICC obtained was 0.87 (95% confidence interval 0.6–0.96; p = 0.001) for interobserver variability and 0.95 (95% confidence interval 0.80–0.98; p < 0.0001) for intraobserver variability.
Discussion
The present study stands out for the identification of subclinical myocardial compromise in JDM using 2DST echocardiography, even in a population with low disease activity (DAS > 3 in only 17%) and otherwise normal standard echocardiograms. Moreover, it was possible to demonstrate significant correlation between LV myocardial deformation parameters and JDM activity, cumulative damage, and functional abilities and health-related quality-of-life parameters, an innovative accomplishment.
Similarly to our results, Guerra et al. found comparable standard parameters of systolic and diastolic function among dermatomyositis patients and controls, corroborating that conventional echocardiogram is not a sensitive tool to detect myocardial involvement in asymptomatic individuals [6]. Conversely, Schwartz et al. identified LV diastolic dysfunction in JDM patients by standard echocardiogram, with abnormal E/e’ ratio in 22% of individuals. It must be pointed out, however, that their population included older individuals with longer disease duration than ours. Some of their patients had also arterial hypertension, known to contribute to LV diastolic dysfunction [20].
Although Guerra et al. had also demonstrated subclinical LV systolic impairment in dermatomyositis patients using speckle-tracking echocardiogram, they did not find any association between LV longitudinal strain and disease activity, damage, or treatment [6]. The present study not only demonstrated association between reduced strain parameters and disease activity/damage indexes but also documented negative correlations between PLSS and cumulative doses of prednisone and methotrexate in JDM patients, probably a reflection of disease severity and duration. In fact, the use of azathioprine was also associated with reduced values of LV longitudinal deformation, suggesting greater myocardial injury in more severe JDM presentations.
It is interesting to notice that only LV global PLSS impairment was associated with both JDM disease activity and damage scores. This can be justified by the fact that subendocardial myocardial layers, mainly responsible for LV longitudinal deformation, are the first to be injured in an inflammatory scenario [21]. Due to its early and persistent impairment in JDM, LV global PLSS may represent a more sensitive tool than standard echocardiography for subclinical myocardial injury detection.
Subepicardial myocardial layers, responsible for LV circumferential deformation, are compromised later in disease progression and are known to sustain preserved LV ejection fraction until symptomatic stages [22]. Not surprisingly, PCSS impairment was associated exclusively with JDM cumulative damage indexes. Paralleling our results, a previous cardiac magnetic resonance (CMR) studies had documented late gadolinium enhancement predominantly in subepicardial myocardial layers, indicating fibrosis of circumferential fibers in JDM patients with inactive disease but proven cumulative damage [23, 24]. It is important to notice that there was no correlation between muscles enzymes or acute-phase reactants and myocardial deformation, corroborating the hypothesis that subtle myocardial injury may be missed in a scenario of low serum biomarkers [25, 26].
Finally, we have demonstrated that LV global PLSS impairment was negatively correlated with the number of abnormal JDM clinical indexes per patients, including functional abilities and health-related quality of life parameters in the analysis for the first time in literature. This result certainly broadens LV global PLSS contribution to overall assessment of JDM patients throughout their development.
2DST echocardiography feasibility and reproducibility were both clearly demonstrated by the present study, favoring its incorporation in JDM patient’s follow-up routine.
Limitations
Possible limitations include the small number of patients enrolled and the single center character of the study, which may preclude generalizations of conclusions to larger populations.
In addition, all the 35 patients were diagnosed with juvenile dermatomyositis in the previous period to the publication of the EULAR/ACR classification criteria (First published November 14, 2017) [27]. That is why we have used Peter and Bohan criteria.
Unfortunately, we have not included electrocardiograms or serum levels of Troponin and BNP in the present study, since they are not routinely ordered by the physicians at our outpatients’ clinics. Those exams could have helped to detect subtle myocardial impairment associated to myocardial deformation compromise.
Besides, it would have been interesting to investigate possible correlations between myocardial deformation measures obtained by 2DST echocardiography and contrast-enhanced CMR imaging, considered the gold standard method to identify myocardial inflammation and fibrosis. Nevertheless, it must be emphasized that CMR is not currently available at all healthcare facilities and frequently implies anesthetic procedures in pediatric population.
Conclusions
2DST echocardiogram can detect subclinical myocardial impairment in JDM patients, even before standard echocardiogram. Myocardial deformation parameters correlated with clinical disease indexes and emerge as promising diagnostic tools for overall assessment of JDM patients throughout development. Future studies must be held in order to stablish if early myocardial injury detection by 2DST can impact on therapeutic decisions and future cardiovascular outcomes in JDM patients.
References
Rider LG, Lindsley CB, Miller FW (2016) Juvenile dermatomyositis. In: Petty RE, Laxer RM, Lindsley CB, Wedderburn LR (eds) Textbook of Pediatric Rheumatology, 7th edn. Elsevier, Philadelphia, pp 351–383
Schwartz T, Sanner H, Gjesdal O, Flato B, Sjaastad I (2014) In juvenile dermatomyositis, cardiac systolic dysfunction is present after long-term follow-up and is predicted by sustained early skin activity. Ann Rheum Dis 73:1805–1810. https://doi.org/10.1136/annrheumdis-2013-203279
Caforio ALP, Adler Y, Agostini C, Allanore Y, Anastasakis A, Arad M, Böhm M, Charron P, Elliott PM, Eriksson U, Felix SB, Garcia-Pavia P, Hachulla E, Heymans S, Imazio M, Klingel K, Marcolongo R, Matucci Cerinic M, Pantazis A, Plein S, Poli V, Rigopoulos A, Seferovic P, Shoenfeld Y, Zamorano JL, Linhart A (2017) Diagnosis and management of myocardial involvement in systemic immune-mediated diseases: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Disease. Eur Heart J 38(35):2649–2662. https://doi.org/10.1093/eurheartj/ehx321
Leal GN, Silva KF, Lianza AC, Giacomin MF, Andrade JL, Kozu K, Bonfá E, Silva CA (2016) Subclinical left ventricular dysfunction in childhood-onset systemic lupus erythematosus: a two-dimensional speckle-tracking echocardiographic study. Scand J Rheumatol 45:202–229. https://doi.org/10.3109/03009742.2015.1063686
Leal GN, Silva KF, França CM, Lianza AC, Andrade JL, Campos LM, Bonfá E, Silva CA (2015) Subclinical right ventricle systolic dysfunction in childhood-onset systemic lupus erythematosus: insights from two-dimensional speckle-tracking echocardiography. Lupus 24:613–620. https://doi.org/10.1177/0961203314563135
Guerra F, Gelardi C, Capucci A, Gabrielli A, Danieli MG (2017) Subclinical Cardiac Dysfunction in polymyositis and dermatomyositis: a speckle-tracking case-control study. J Rheumatol 44:815–821. https://doi.org/10.3899/jrheum.161311
Bohan A, Peter JB (1975) Polymyositis and dermatomyositis. N Engl J Med 13:344–347. https://doi.org/10.1056/NEJM197502132920706
Dulcan M (1994) Nomenclature and criteria for diagnosis of diseases of the heart and great vessels, 9th edn. Little, Brown & Co, Boston, pp 253–256
Huber AM, Feldman BM, Rennebohm RM, Hicks JE, Lindsley CB, Perez MD et al (2004) Validation and clinical significance of the Childhood Myositis Assessment Scale for assessment of muscle function in the juvenile idiopathic inflammatory myopathies. Arthritis Rheum 50:1595–1603. https://doi.org/10.1002/art.20179
Lazarevic D, Pistorio A, Palmisani E, Miettunen P, Ravelli A, Pilkington C et al (2013) The PRINTO criteria for clinically inactive disease in juvenile dermatomyositis. Ann Rheum Dis 72:686–693. https://doi.org/10.1136/annrheumdis-2012-201483
Rider LG, Werth VP, Huber AM, Alexanderson H, Rao AP, Ruperto N, Herbelin L, Barohn R, Isenberg D, Miller FW (2011) Measures of adult and juvenile dermatomyositis, polymyositis, and inclusion body myositis: Physician and Patient/Parent Global Activity, Manual Muscle Testing (MMT), Health Assessment Questionnaire (HAQ)/Childhood Health Assessment Questionnaire (C-HAQ), Childhood Myositis Assessment Scale (CMAS), Myositis Disease Activity Assessment Tool (MDAAT), Disease Activity Score (DAS), Short Form 36 (SF-36), Child Health Questionnaire (CHQ), physician global damage, Myositis Damage Index (MDI), Quantitative Muscle Testing (QMT), Myositis Functional Index-2 (FI-2), Myositis Activities Profile (MAP), Inclusion Body Myositis Functional Rating Scale (IBMFRS), Cutaneous Dermatomyositis Disease Area and Severity Index (CDASI), Cutaneous Assessment Tool (CAT), Dermatomyositis Skin Severity Index (DSSI), Skindex, and Dermatology Life Quality Index (DLQI). Arthritis Care Res 63(Suppl 11):S118–S157. https://doi.org/10.1002/acr.20532
Apaz MT, Saad-Magalhães C, Pistorio A, Ravelli A, Sato JO, Marcantoni MB, Meiorin S, Filocamo G, Pilkington C, Maillard S, Al-Mayouf S, Prahalad S, Fasth A, Joos R, Schikler K, Mozolova D, Landgraf JM, Martini A, Ruperto N, Paediatric Rheumatology International Trials Organisation (2009) Health-related quality of life of patients with juvenile dermatomyositis: results from the paediatric rheumatology international trials organisation multinational quality of life cohort study. Arthritis Rheum 6:509–517. https://doi.org/10.1002/art.24343
Klatchoian DA, Len CA, Terreri MT, Silva M, Itamoto C, Ciconelli RM, Varni JW, Hilário MO (2008) Quality of life of children and adolescents from São Paulo: reliability and validity of the Brazilian version of the Pediatric Quality of Life Inventory version 4.0 Generic Core Scales. J Pediatr 84:308–315. https://doi.org/10.2223/JPED.1788
Kountz-Edwards S, Aoki C, Gannon C, Gomez R, Cordova M, Packman W (2017) The family impact of caring for a child with juvenile dermatomyositis. Chronic Illn 13:262–274. https://doi.org/10.1177/1742395317690034
Haycock GB, Schwartz GJ, Wisotsky DH (1978) Geometric method for measuring body surface area: a height-weight formula validated in infants, children, and adults. J Pediatr 93:62–66. https://doi.org/10.1016/s0022-3476(78)80601-5
Lopez L, Colan SD, Frommelt PC, Ensing GJ, Kendall K, Younoszai AK, Lai WW, Geva T (2010) Recommendations for quantification methods during the performance of a pediatric echocardiogram: a report from the pediatric measurements writing group of the American society of echocardiography pediatric and congenital heart disease council. J Am Soc Echocardiogr 23:465–495. https://doi.org/10.1016/j.echo.2010.03.019
Mor-Avi V, Mor-Avi V, Lang RM, Badano LP, Belohlavek M, Cardim NM, Derumeaux G, Galderisi M, Marwick T, Nagueh SF, Sengupta PP, Sicari R, Smiseth OA, Smulevitz B, Takeuchi M, Thomas JD, Vannan M, Voigt JU, Zamorano JL (2011) Current and Evolving Echocardiographic Techniques for the Quantitative Evaluation of Cardiac Mechanics: ASE/EAE Consensus Statement on Methodology and Indications Endorsed by the Japanese Society of Echocardiography. J Am Soc Echocardiogr 24:277–313. https://doi.org/10.1016/j.echo.2011.01.015
Levy PT, Machefsky A, Sanchez AA, Patel MD, Rogal S, Fowler S, Yaeger L, Hardi A, Holland MR, Hamvas A, Singh GK (2016) Reference ranges of left ventricular strain measures by two-dimensional speckle-tracking echocardiography in children: a systematic review and meta-analysis. J Am Soc Echocardiogr 29:209–225. https://doi.org/10.1016/j.echo.2015.11.016
Eidem BW, McMahon CJ, Cohen RR, Wu J, Finkelshteyn I, Kovalchin JP, Ayres NA, Bezold LI, O'Brian Smith E, Pignatelli RH (2004) Impact of cardiac growth on Doppler tissue imaging velocities: a study in healthy children. J Am Soc Echocardiogr 17:212–221. https://doi.org/10.1016/j.echo.2003.12.005
Schawartz T, Sanner H, Husebye T, Flato B, Sjaastad I (2011) Cardiac dysfunction in juvenile dermatomyositis: a case-control study. Ann Rheum Dis 70:766–771. https://doi.org/10.1136/ard.2010.137968
Gupta R, Wayangankar SA, Targoff IN, Hennebry TA (2011) Clinical cardiac involvement in idiopathic inflammatory myopathies: a systematic review. Int J Cardiol 148:261–270. https://doi.org/10.1016/j.ijcard.2010.08.013
Leal GN (2019) Applications of the Myocardial Strain Study using Two-Dimensional Speckle Tracking in Pediatrics. Arq Bras Cardiol: Imagem Cardiovasc 32:29–33. https://doi.org/10.5935/2318-8219.20190008
Mavrogeni S, Douskou M, Manoussakis MN (2011) Contrast-Enhanced CMR Imaging Reveals Myocardial Involvement in Idiopathic Inflammatory Myopathy Without Cardiac Manifestations. JACC Cardiovasc Imaging 4:1324–1325. https://doi.org/10.1016/j.jcmg.2011.05.009
Schwartz T, Diederichsen LP, Lundberg IE, Sjaastad I, Sanner H (2016) Cardiac involvement in adult and juvenile idiopathic inflammatory myopathies. RMD Open 2016:e000291. https://doi.org/10.1136/rmdopen-2016-000291
Wienke J, Deakin CT, Wedderburn LR, van Wijk F, van Royen-Kerkhof A (2018) Systemic and Tissue Inflammation in Juvenile Dermatomyositis: From Pathogenesis to the Quest for Monitoring Tools. Front Immunol 9:2951. https://doi.org/10.3389/fimmu.2018.02951
Rosa Neto NS, Goldenstein-Schainberg C (2010) Juvenile dermatomyositis: review and update of the pathogenesis and treatment. Rev Bras Reumatol 50:299–312
Bottai M, Tjärnlund A, Santoni G, The International Myositis Classification Criteria Project consortium, the Euromyositis register and the Juvenile Dermatomyositis Cohort Biomarker Study and Repository (JDRG) (UK and Ireland) et al (2017) EULAR/ACR classification criteria for adult and juvenile idiopathic inflammatory myopathies and their major subgroups: a methodology report. RMD Open 3:e000507. https://doi.org/10.1136/rmdopen-2017-000507
Funding
This work was supported by grant from Fundação do Amparo a Pesquisa do Estado de São Paulo (FAPESP) (#2015/03756-4 to CAS), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPQ #303422/2015-7 to CAS), and by Núcleo de Apoio à Pesquisa “ Saúde da Criança e do Adolescente” da USP (NAP-CriAd) to CAS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Diniz, M.d.R., Kozu, K.T., Elias, A.M. et al. Echocardiographic study of juvenile dermatomyositis patients: new insights from speckle-tracking-derived strain. Clin Rheumatol 40, 1497–1505 (2021). https://doi.org/10.1007/s10067-020-05418-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05418-4