Abstract
Objectives
The relationship between rheumatoid arthritis (RA) and the risk of leukemia was still controversial. This study aimed to assess the risk of leukemia in patients with rheumatoid arthritis by systematic review and meta-analysis.
Methods
Relevant studies were identified by searching PubMed, Embase, Cochrane Library, and SinoMed up to December 2019. Random effects model analysis was used to pool standardized incidence ratios (SIRs) and 95% confidence interval.
Results
A total of 15 relevant studies that met the criteria were included. Compared with the general population, patients with RA showed an increased risk of leukemia (SIR = 1.51, 95% CI: 1.34–1.70). The statistical heterogeneity was moderate with an I2 of 55.5%. In subgroup analysis, the source of heterogeneity may be due to differences in sample size. Publication bias was not found in the Begg funnel plot and the Egger test.
Conclusion
Our findings suggested that the risk of leukemia in RA was increased compared with the general population.
Key points • This is the first systematic review and meta-analysis to assess the risk of leukemia in RA. • Our study suggested that the risk of leukemia in RA was increased compared with the general population. • This study indicated that the risk of leukemia in RA was higher in non-Asian populations. |
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is the most common chronic systemic inflammatory disease. Musculoskeletal pain, swelling, and stiffness are common symptoms of RA, which can lead to joint deformity and the loss of function, thereby, it can significantly reduce patients’ quality of life and long-term prognosis of RA [1]. RA is a multifactorial disease that may be closely related to genetic, environmental factors, and autoimmune responses [2]. The disease has also many complications, among which malignant tumors are one of the most difficult to control and life-threatening complications.
The risk of cancer in patients with RA has received much attention. Many studies have investigated the relationship between RA and various cancers, and identified the relevant pathogenesis. The abnormal autoimmune mechanism may be a common cause of malignant tumors in RA [3]. In addition, related drugs for treating RA, such as biological agents and disease-modifying anti-rheumatic drug (DMARD), may increase the risk of malignant tumors [4,5,6].
In recent years, many cohort studies have shown that there was an increased risk of leukemia in RA patients. However, some other studies failed to observe the correlation between leukemia and RA. To date, there was no meta-analysis evaluating the risk of leukemia in RA. Therefore, we conducted a systematic review and meta-analysis to assess the risk of leukemia in RA.
Methods
Search strategy
We searched the literature published before December 2019 through the databases of PubMed, Embase, Cochrane Library, and SinoMed. In addition, the references cited in the above original documents if they meet the requirements were also manually searched. The search keywords included “rheumatoid arthritis,” “leukemia,” “cancer,” “tumor,” and “morbidity.” The selection criteria were as follows: (1) the study design conformed to the cohort study; (2) the article had clear data, such as SIR and confidence interval; (3) the sample size needed to be greater than 100. Exclusion criteria were as follows: (1) ambiguous data or lack of necessary data; (2) reviews, case reports, conference articles, unpublished studies, etc.
Data extraction and quality assessment
Two investigators (Xiao Luo and Mao Liu) independently extracted the original literature data. They resolved differences by reassessing, consulting relevant professionals, and discussing. The third investigator (Jie Chen) also participated in resolving differences. The extracted data included the following information: author, year of data collection, country, number of cases, length of follow-up, source of cohort, standardized incidence (SIR), and 95% CI-related data. The Newcastle-Ottawa Scale (NOS) was used to assess the quality of the study, where a score of 6 or less was classified as low quality.
Statistical analysis
In this meta-analysis, we examined the relationship between rheumatoid arthritis and leukemia based on SIR and 95% CI. The heterogeneity between the studies was evaluated using the Cochrane Q test and the Cochran I2 statistic. P < 0.1 indicated heterogeneity between the studies, and the random effects model was used [7]. Sensitivity analysis was used to exclude each of the included studies as a source of significant heterogeneity. Publication bias was assessed using Begg’s test and Egger’s test. All statistical analyses were performed using Stata 12.0.
Results
Characteristics of included studies
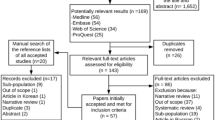
A total of 627 articles were searched according to the search criteria. After reading the title and reviewing the abstract, 326 articles were excluded. Through a full-text evaluation, a total of 15 studies met the inclusion criteria (Fig. 1). The main research characteristics of this meta-analysis are shown in Table 1. The number of patients in the included cohort studies from different countries between 1993 and 2016 was ranged from 862 to 84,475. The mean follow-up period of these studies was 3.95 to 17.4 years. The study of Hemminki et al. [13] and Chen et al. [10] reported the results of males and females respectively. We have calculated the combined SIR values and 95% CI. The study of Hashimoto et al. [20] and Cho et al. [21] was excluded because the data is not available.
Study quality
Among the 15 included articles, 7 articles were identified as high quality (NOS score > 6). In addition, three studies were overlapping cohort studies, which were all reported by Askling et al. [16]. The patients were divided into RA, early RA, and TNF antagonist RA. The subjects of the control group were the general population. These studies were eventually included in the meta-analysis and systematic review.
Meta-analysis results
Patients with rheumatoid arthritis have an increased risk of leukemia (SIR = 1.51, 95% CI 1.34–1.70) (Fig. 2). Due to the heterogeneity, the random effects model was adopted (I2 = 55.5%, P = 0.005). In order to explore the source of heterogeneity, a subgroup analysis was conducted by study quality, geographic region, publication time, type of control, and total number of rheumatoid arthritis. Studies with less than 10,000 patients showed a significantly higher risk of leukemia in RA patients (SIR = 3.18, 95% CI: 1.66–6.09, SIR = 1.47, 95% CI: 1.32–1.63) and heterogeneity was lower (I2 = 30.2%, I2 = 48.2%). The risk of non-Chinese is higher than that of Chinese (SIR = 1.61, 95% CI: 1.36–1.91, SIR = 1.34, 95% CI: 1.09–1.63, respectively). The studies published before 2005 suggested that RA patients had a higher risk of leukemia. (SIR = 1.65, 95% CI: 1.29–2.12, I2 = 45.9%, SIR = 1.44, 95% CI: 1.26–1.55, I2 = 52.9%). There was no significant change in the incidence of subgroup analysis by population and hospital. Studies with relatively higher quality according to NOS showed a higher risk of leukemia in RA (SIR = 1.75, 95% CI: 1.35–2.27, I2 = 73.6%), but the heterogeneity was obvious. The risk of leukemia was higher in patients with rheumatoid arthritis diagnosed after 1987 (SIR = 1.53, 95% CI: 1.33–1.76, SIR = 1.41, 95% CI: 1.03–1.92). 7 articles clearly indicated that the age of the subjects was at least 16 years old, excluding juvenile rheumatoid arthritis (JRA), which had a higher risk of leukemia compared with the overall rheumatoid arthritis (SIR = 1.58, 95% CI: 1.27–1.97, SIR = 1.45, 95% CI: 1.23–1.71). Subgroup analysis showed that heterogeneity might result from the sample size.
Publication bias
There was no significant publication bias based on Begg’s test and Egger’s test (Begg’s P = 0.322, Egger’s P = 0.495). Meanwhile, we conducted a sensitivity analysis for each group and excluded each of the included studies respectively to observe the stability of the meta-analysis. When two studies reported by Askling et al. [16] and Abasolo et al. [12] were removed respectively, the heterogeneity would be significantly reduced, but the combined SIR did not change much.
Discussion
Our study showed that there was a significantly increased risk of leukemia in patients with RA compared with the general population. The sample size of the study was negatively correlated with the risk of leukemia in patients with RA. It might be caused due to studies with a small sample size that would lead to a lower level of evidence and the results were highly biased.
The diagnostic criteria, treatment methods, and evaluation methods have acknowledged quite a bit of transition due to the large span of research years. Three of the articles [8, 9, 12] fulfilled the diagnostic criteria of the 1987 American College of Rheumatology classification (ACR) in our meta-analysis. Other research objects are according to the International Classification of Diseases (ICD) and did not indicate which diagnostic criteria were fulfilled. We classified the articles according to the year the subjects were diagnosed, which fluctuated between 1965 and 2010. Rheumatoid arthritis was diagnosed in 2010 fulfilled by the 1987ACR diagnostic criteria [8]. Three articles were diagnosed pre-1987 [18, 19, 22]. The results presumably inferred that newer diagnostic time is slightly higher in the risk of leukemia. Therefore, diagnostic criteria have no significant impact on our study. We need to include the 2010 American College of Rheumatology/European League Against Rheumatism (ACR/EULAR) classification criteria for RA to update our conclusions in the future.
Subgroup analysis (Table 2) indicated that the risk of developing leukemia in RA was higher in non-Chinese populations. One study on the epidemiological patterns of leukemia showed that the incidence of leukemia in North America, Oceania, and certain European countries was markedly higher than in Asia [23]. Although there is probably a role for aetiological factors, including gene-environment interactions, the reduced incidence of leukemia in low and middle human development index countries such as Asia may be due to the inadequate diagnostic capacity. Chronic lymphocytic leukemia (CLL) is the most common type of leukemia in western countries [23,24,25], while the Asian population is more susceptible to acute lymphoblastic leukemia (ALL) [24], which has a high risk in childhood and cure rate of up to 90% [26]. Middle-aged and elderly people have increased access to health care or advanced diagnostic techniques, which may lead to a higher diagnostic rate of chronic blood diseases. RA is more common in middle-aged women [1], thus, the age of disease may affect the diagnostic rate of leukemia in a patient with RA. Our subgroup analysis showed that subjects who did not include JRA had a slightly higher risk of leukemia compared with the overall rheumatoid arthritis, but significantly higher than the general population, so the inclusion of the JRA or not had little impact on the final conclusion. However, there is no relevant evidence to prove this risk factor, and further research is needed. Other subgroup analyses have moderate heterogeneity, and the analysis has limitations that may be related to different sources of data.
Studies have shown that the mechanism of the malignant tumorigenesis may be due to that RA leads to lymphocyte activation under chronic immune stimulation, and B cell proliferation is not controlled [27]. It may also be the cause of leukemia in RA, but there has been no research to prove this yet; the above mechanism still needs to be continuously verified. Due to the long-term severity or treatment of the disease, there could also be an increased risk of immune system deterioration [6, 28]. Some drugs inhibit the function of the immune system and cause these immune-impaired patients to become more susceptible to cancer. For example, the use of steroid hormones, cytotoxic drugs, or immunosuppressive agents may inhibit immune defense against malignant cells and lead to tumorigenesis [29]. Biologics are a new type of drug that has been widely used in rheumatic diseases in recent years. Studies have shown that patients with long-term exposure to TNF inhibitors have a higher risk of developing T cell lymphoblastic leukemia (T-LGL), which is consistent with the results of Askling et al. [21]. However, studies have shown that the use of bDMARD may not affect the risk of hematological malignancies [16]. Immunosuppressive drugs such as methotrexate may be also not involved in the pathogenesis of lymphoproliferative tumors [28]. In an animal study, it has been shown that methotrexate may be even used to treat T-type leukemia/lymphoma [30]. In recent years, high-dose methotrexate (Hd-MTX) has been used in the treatment of childhood ALL [31]. Sakura et al. [32] demonstrated that Hd-MTX can effectively and safely treat adult ALL; however, other studies have shown that methotrexate and azathioprine may increase the risk of chronic myeloid tumors [33, 34]. Some steroid hormones, such as glucocorticoids, play an important role in the treatment of leukemia due to their pro-apoptotic action, especially ALL, which is given in high doses as a standard induction therapy. Steroids are also used to treat CLL, chronic myeloid leukemia (CML), and acute myeloid leukemia (AML) [35]. Therefore, the treatment of RA includes immunosuppressive and steroids, which may cause leukemia to be hidden. But these drugs do not cure leukemia, and symptoms can still appear and be diagnosed some time later. Other immunosuppressive drugs such as phenylbutanone and d-penicillamine may be involved in the promotion of hematologic malignancies [28]. Therefore, the impact of drugs on leukemia needs to be investigated further.
In addition to drug relation, another explanation is the effect of genetic mutations on malignant tumors of immune system diseases. Molecular aberrations such as PI3K [36], AKT, mTOR, and p53 [37] were reported to be involved in the development of malignant tumors. MiRNAs may also be associated with autoimmune diseases in hematological malignancies [38]. Carcinoma-associated fibroblasts (CAFs) [39] and interleukin-6 (IL-6) also play a significant role in tumorigenesis and inflammatory diseases including RA [39, 40]. Recently, Cuenca et al. [41] studied that CD84 were associated with cancer and autoimmune disorders, including CLL.
It was found that the risk of hematological tumors in RA might be the result of interactions between genetic mutations and environmental factors [42], but there was currently no research on the mechanism of leukemia in RA.
The advantage of this meta-analysis is that our study included a large number of subjects, which significantly improve the statistical effectiveness. Our analysis included only cohort studies that might minimize selection bias or recall bias. The data, analysis, and quality assessment were independently extracted by two independent reviewers, and the third review confirmed the reliability, which led to more reliable conclusions. However, the limitations of the current meta-analysis cannot be ignored. First, the moderate heterogeneity may come from the sample and the data itself. Since the data is derived from the national database, the statistical data is prone to deviations, and no controlled trials are more convincing. Second, the confounding factors inherent in the study may affect the risk estimates. Most of the included studies are adjusted for age, gender, and follow-up years. Other important confounding factors are not standardized. Third, we exclude 2 articles that are not available for data, which may affect the outcome judgment. In that study, Sweden was the largest country included, which may have an impact on the results. It might need to be further included in a multi-country cohort study to correct the results of meta-analysis [24].
In conclusion, this meta-analysis showed that the risk of leukemia in patients with rheumatoid arthritis was increased, especially in non-Asian populations. Our study provides up-to-date information and might improve the early diagnosis strategy to optimize treatment and improve survival.
Data availability
All data generated or analyzed during this study are included in this published article.
References
Sparks JA (2019) Rheumatoid arthritis. Ann Intern Med 170(1):ITC1–ITC16
Aslani S, Mahmoudi M, Karami J, Jamshidi AR, Malekshahi Z, Nicknam MH (2016) Epigenetic alterations underlying autoimmune diseases. Autoimmunity 49(2):69–83
Hemminki K, Liu X, Försti A, Ji J, Sundquist J, Sundquist K (2013) Subsequent leukaemia in autoimmune disease patients. Br J Haematol 161(5):677–687
Love T, Solomon DH (2008) The relationship between cancer and rheumatoid arthritis: still a large research agenda. Arthritis Res Ther 10(3):109
Cush JJ, Dao KH (2012) Malignancy risks with biologic therapies. Rheum Dis Clin N Am 38(4):761–770
Simon TA, Thompson A, Gandhi K, Hochberg MC, Suissa S (2015) Incidence of malignancy in adult patients with rheumatoid arthritis: a meta-analysis. Arthritis Res Ther 17(1):212
Ungprasert P, Srivali N, Kittanamongkolchai W (2017) Risk of incident atrial fibrillation in patients with rheumatoid arthritis: a systematic review and meta-analysis. Int J Rheum Dis 20(4):434–441
Yu KH, Kuo CF, Huang LH, Huang WK, See LC (2016) Cancer risk in patients with inflammatory systemic autoimmune rheumatic diseases: a nationwide population-based dynamic cohort study in Taiwan. Medicine (Baltimore) 95(18):e3540
Huang WK, Chiou MJ, Kuo CF, Lin YC, Yu KH, See LC (2014) No overall increased risk of cancer in patients with rheumatoid arthritis: a nationwide dynamic cohort study in Taiwan. Rheumatol Int 34(10):1379–1386
Chen YJ, Chang YT, Wang CB, Wu CY (2011) The risk of cancer in patients with rheumatoid arthritis: a nationwide cohort study in Taiwan. Arthritis Rheum 63(2):352–358
Parikh-Patel A, White RH, Allen M, Cress R (2009) Risk of cancer among rheumatoid arthritis patients in California. Cancer Causes Control 20(6):1001–1010
Abásolo L, Júdez E, Descalzo MÁ, González-Álvaro I, Jover JA, Carmona L (2008) Cancer in rheumatoid arthritis: occurrence, mortality, and associated factors in a south European population. Semin Arthritis Rheum 37(6):388–397
Hemminki K, Li X, Sundquist K, Sundquist J (2008) Cancer risk in hospitalized rheumatoid arthritis patients. Rheumatology (Oxford) 47(5):698–701
Wolfe F, Michaud K (2007) Biologic treatment of rheumatoid arthritis and the risk of malignancy: analyses from a large US observational study. Arthritis Rheum 56(9):2886–2895
Setoguchi S, Solomon DH, Weinblatt ME, Katz JN, Avorn J, Glynn RJ, Cook EF, Carney G, Schneeweiss S (2006) Tumor necrosis factor alpha antagonist use and cancer in patients with rheumatoid arthritis. Arthritis Rheum 54(9):2757–2764
Askling J, Fored CM, Baecklund E, Brandt L, Backlin C, Ekbom A, Sundström C, Bertilsson L, Cöster L, Geborek P, Jacobsson LT, Lindblad S, Lysholm J, Rantapää-Dahlqvist S, Saxne T, Klareskog L, Feltelius N (2005) Haematopoietic malignancies in rheumatoid arthritis: lymphoma risk and characteristics after exposure to tumour necrosis factor antagonists. Ann Rheum Dis 64(10):1414–1420
Thomas E, Brewster DH, Black RJ, Macfarlane GJ (2000) Risk of malignancy among patients with rheumatic conditions. Int J Cancer 88(3):497–502
Mellemkjær L, Linet MS, Gridley G, Frisch M, Møller H, Olsen JH (1996) Rheumatoid arthritis and cancer risk. Eur J Cancer 32(10):1753–1757
Gridley G, McLaughlin JK, Ekbom A, Klareskog L, Adami HO, Hacker DG, Hoover R, Fraumeni JF (1993) Incidence of cancer among patients with rheumatoid arthritis. J Natl Cancer Inst 85(4):307–311
Hashimoto A, Chiba N, Tsuno H, Komiya A, Furukawa H, Matsui T, Nishino J, Tohma S (2015) Incidence of malignancy and the risk of lymphoma in Japanese patients with rheumatoid arthritis compared to the general population. J Rheumatol 42(4):564–571
Cho SK, Lee J, Han M, Bae SC, Sung YK (2017) The risk of malignancy and its incidence in early rheumatoid arthritis patients treated with biologic DMARDs. Arthritis Res Ther 19(1):277
Cibere J, Sibley J, Haga M (1997) Rheumatoid arthritis and the risk of malignancy. Arthritis Rheum 40(9):1580–1586
Miranda-Filho A, Piñeros M, Ferlay J, Soerjomataram I, Monnereau A, Bray F (2018) Epidemiological patterns of leukaemia in 184 countries: a population-based study. Lancet Haematol 5(1):e14–e24
Oliveira PD (2018) Leukaemia prevalence worldwide: raising aetiology questions. Lancet Haematol 5(1):e2–e3
deAndrés-Galiana EJ, Fernández-Martínez JL, Luaces O, del Coz JJ, Huergo-Zapico L, Acebes-Huerta A, González S, González-Rodríguez AP (2016) Analysis of clinical prognostic variables for chronic lymphocytic leukemia decision-making problems. J Biomed Inform 60:342–351
Paul S, Kantarjian H, Jabbour EJ (2016) Adult acute lymphoblastic leukemia. Mayo Clin Proc 91(11):1645–1666
Hemminki K, Liu X, Ji J, Försti A (2016) Origin of B-cell neoplasms in autoimmune disease. PLoS One 11(6):e0158360
Symmons DP (1985) Neoplasms of the immune system in rheumatoid arthritis. Am J Med 78(1):22–28
Shen K, Xu G, Wu Q, Zhou D, Li J (2014) Risk of multiple myeloma in rheumatoid arthritis: a meta-analysis of case-control and cohort studies. PLoS One 9(3):e91461
Pawlak A, Kutkowska J, Obmińska-Mrukowicz B, Rapak A (2017) Methotrexate induces high level of apoptosis in canine lymphoma/leukemia cell lines. Res Vet Sci 114:518–523
Gervasini G, Mota-Zamorano S (2019) Clinical implications of methotrexate pharmacogenetics in childhood acute lymphoblastic leukaemia. Curr Drug Metab 20(4):313–330
Sakura T, Hayakawa F, Sugiura I et al (2018) High-dose methotrexate therapy significantly improved survival of adult acute lymphoblastic leukemia: a phase III study by JALSG. Leukemia 32(3):626–632
Lee H (2019) The risk of malignancy in Korean patients with rheumatoid arthritis. Yonsei Med J 60(2):223–229
Ertz-Archambault N, Kosiorek H, Taylor GE, Kelemen K, Dueck A, Castro J, Marino R, Gauthier S, Finn L, Sproat LZ, Palmer J, Mesa RA, al-Kali A, Foran J, Tibes R (2017) Association of therapy for autoimmune disease with myelodysplastic syndromes and acute myeloid leukemia. JAMA Oncol 3(7):936–943
Strehl C, Ehlers L, Gaber T, Buttgereit F (2019) Glucocorticoids-all-rounders tackling the versatile players of the immune system. Front Immunol 10:1744
Wang D, Zhou W, Chen J, Wei W (2019) Upstream regulators of phosphoinositide 3-kinase and their role in diseases. J Cell Physiol 234(9):14460–14472
Kumar N, Chugh H, Tomar R, Tomar V, Singh VK, Chandra R (2018) Exploring the interplay between autoimmunity and cancer to find the target therapeutic hotspots. Artif Cells Nanomed Biotechnol 46(4):658–668
Di Marco M, Ramassone A, Pagotto S, Anastasiadou E, Veronese A, Visone R (2018) MicroRNAs in autoimmunity and hematological malignancies. Int J Mol Sci 19(10):3139
Tian G, Liang JN, Wang ZY, Zhou D (2014) Breast cancer risk in rheumatoid arthritis: an update meta-analysis. Biomed Res Int 2014:453012
Yao X, Huang J, Zhong H, Shen N, Faggioni R, Fung M, Yao Y (2014) Targeting interleukin-6 in inflammatory autoimmune diseases and cancers. Pharmacol Ther 141(2):125–139
Cuenca M, Sintes J, Lányi Á, Engel P (2018) CD84 cell surface signaling molecule: an emerging biomarker and target for cancer and autoimmune disorders. Clin Immunol 204:43–49
Piletič K, Kunej T (2016) MicroRNA epigenetic signatures in human disease. Arch Toxicol 90(10):2405–2419
Funding
This study was supported by the project of the Affiliated Hospital of Southwest Medical University Doctoral Fund (18048), the project of the Sichuan Education Department (18ZB0640), and the project of the Health Department in Sichuan Province (150078).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Luo, X., He, Y., Xu, W. et al. The risk of leukemia in patients with rheumatoid arthritis: a systematic review and meta-analysis. Clin Rheumatol 40, 1283–1289 (2021). https://doi.org/10.1007/s10067-020-05396-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05396-7