Abstract
Introduction/objectives
The associations between severity of knee osteoarthritis (KOA) and phase angle (PhA) and between PhA and quadriceps strength in patients with KOA are unclear. This study examined (1) whether the structural severity of KOA affects PhA and (2) whether PhA affects quadriceps strength in patients with KOA.
Method
Data of 1093 patients with KOA, obtained from Screening for People Suffering Sarcopenia in the Orthopedic cohort of Kobe study, were analyzed. PhA was determined by bioimpedance. Quadriceps strength was measured using a handheld dynamometer. Structural severity of KOA was determined using Kellgren-Lawrence radiographic grading scale. A series of general linear models were fitted to estimate the magnitude of differences in PhA by differences in KOA severity and quadriceps strength by differences in PhA.
Results
The mean age of the patients was 72.8 years, and 78% were women. Increasing KOA severity was associated with decreasing PhA, especially in men. In women, only grade 4 KOA was associated with a decrease in PhA (P for interaction = 0.048). PhA per leg was positively associated with quadriceps strength per leg, independent of age, sex, leg muscle mass, pain, and KOA severity (mean difference per 1° increase = 7.54 Nm, 95% confidence interval = 5.51–9.57 Nm). The association between PhA and quadriceps strength differed neither by sex nor by KOA severity (P for interaction = 0.133 and 0.185, respectively).
Conclusions
PhA decreased with increasing KOA severity, and increasing PhA was associated with increasing quadriceps strength. Clinicians should, therefore, evaluate PhA to assess quadriceps strength in patients with KOA.
Key Points • PhA gradually decreased with increasing severity of KOA, especially in men. • Increasing PhA was associated with increasing quadriceps strength. • Clinicians should focus on increasing muscle mass and PhA. |
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Quadriceps strength is a modifiable factor that determines functional movement regardless of the severity of knee osteoarthritis (KOA), as assessed by radiography [1]. It is associated with leg muscle mass in patients with KOA [2]. Recently, for the exertion of strength, attention has been paid not only to the volume of leg muscles but also to other indices related to muscle quality. As such, phase angle (PhA) has been investigated.
PhA can be calculated simultaneously with muscle mass via bioelectrical impedance using the following two measures: reactance (cell membrane-specific resistance, Xc) and resistance (intracellular and extracellular resistance, R). Theoretically, Xc is an index of the volume of cell membrane capacitance, and resistance to electrical current is determined by body fat, total body water, and extracellular water [3]. Therefore, PhA reflects electric tissue property and is considered a marker of cell membrane integrity [4]. Some researchers pointed out that disease or impairment in functional movement can disturb electrical properties of tissue and directly lower PhA [5]. Considering this possibility of disease-dependent change in PhA [5], severe KOA may theoretically reduce PhA. In support of this hypothesis, a previous study showed that PhA in patients with severe KOA was lower than that in patients with early KOA [6]. However, the study did not investigate the relationship between the grade of KOA and PhA, taking into consideration of other demographics such as age and sex. In addition, PhA has been associated with muscle strength among the general elderly population [7], but the association between PhA and quadriceps strength in patients with KOA remains unclear.
In this study, using data from the “Screening for People Suffering Sarcopenia in Orthopedic cohort of Kobe” (SPSS-OK) study—a large, single-center study—we aimed to investigate (1) whether KOA severity is associated with reduced PhA and (2) whether PhA is associated with quadriceps strength in patients with KOA. We hypothesize that there is an association between increasing severity of KOA and a decrease in PhA, and one between an increase in PhA and increasing quadriceps strength.
Materials and methods
Design, setting, and participants
This was a cross-sectional study. Patients were recruited consecutively from August 2016 to January 2019. The study center is located in the central part of Kobe, Japan. Only patients with KOA who were scheduled for their first knee replacement surgery were included, to avoid the potential interference of implants on bioimpedance analysis (BIA). The exclusion criteria were stroke, Parkinson’s disease, severe cognitive impairment, severe psychiatric impairment, and severe cardiac, pulmonary, or musculoskeletal disorders that limited walking and quadriceps strength. In addition, we excluded older adults with artificial implants such as cardiac pacemakers and joints, which did not permit the measurement of bioelectrical impedance. BIA data, quadriceps strength, radiographs of both knees, and other covariates were measured within 2 months before surgery. Based on the radiographs, a diagnosis of KOA was made by orthopedic surgeons, according to the American College of Rheumatology criteria [8]. The study was approved by the local institutional review board (no. 57, January 26, 2017) and the Research Ethics Committee of Fukushima Medical University School of Medicine (no. 2850, September 28, 2016). All patients provided written informed consent prior to participation. Because the study aimed to discover the association of KOA severity with PhA and that of PhA with quadriceps strength, sample size calculation was not pre-specified [9].
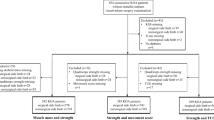
Conceptual framework
The conceptual framework is shown in Fig. 1. First, we evaluated the relationship between KOA severity as the exposure variable and PhA as the outcome variable, with sex as the effect modifier (analysis no. 1). Next, we assessed the relationship between PhA as the exposure variable and quadriceps strength as the outcome variable, with KOA severity as the effect modifier (analysis no. 2).
Conceptual framework used in the regression analyses. Outcome 1 was whether KOA severity would be associated with PhA and whether the associations differed by sex. Outcome 2 was whether PhA would be associated with quadriceps strength and whether the associations differed by KOA severity or by sex. KOA, knee osteoarthritis; PhA, phase angle
KOA severity
One trained observer (OW) graded KOA severity, using the patients’ radiographs, based on the Kellgren-Lawrence radiographic grading system [10]. Intra-rater reliability for radiographic severity was determined by the reassessment of 100 randomly selected radiographs 2 weeks later and was found to be excellent (κ = 0.89).
Bioelectrical impedance and PhA
A biometrical impedance system (MC-780A; TANITA CO, Ltd., Tokyo, Japan) was used to measure single-leg PhA and leg muscle mass. The monitor is a multifrequency device that measures bioimpedance at three frequencies between 5 and 250 kHz to directly measure the amount of extracellular and intracellular water in the body. The results were analyzed by an onboard body composition analyzer.
Single-leg PhA was calculated using the following formula: PhA (degrees) = arctangent (Xc/R) × (180/π). Single-leg resistance and reactance, measured at 50 kHz, were used to calculate the single-leg PhA [3]. Thus, leg PhA was derived for both legs, regardless of the side planned for surgery.
Quadriceps strength
Quadriceps strength was measured as the peak isometric knee extension torque (Nm) using a handheld dynamometer (μTas F1; Anima, Chofu, Japan). This method uses a belt to measure the strength of knee extension and is highly reliable [11]. Patients were seated with their hips flexed to 90°, and knees flexed to 75°. Following submaximal efforts, patients performed two maximal trials for 3 s while receiving verbal encouragement to facilitate maximal volitional force production. The distance between the attachment of the dynamometer and center of rotation of the knee joint was used as the lever arm length. The peak force (Newton) from two maximal contractions was multiplied by the lever arm length (m) and recorded as quadriceps strength.
Measurement of covariates
The examined covariates included sociodemographic characteristics, including age, fat mass measured by bioimpedance, type 2 diabetes, knee pain, and leg muscle mass. Type 2 diabetes was defined as glycosylated hemoglobin (HbA1c) level ≥ 6.5% [12], and the knee pain per leg was estimated using the 1-item new Knee Society Score (KSS) while walking on level ground; the score ranged from 0 = no pain to 10 = worst pain [13]. Muscle mass per leg was assessed as a proxy for quadriceps muscle mass. Quadriceps muscle mass accounts for approximately one quarter of the total single leg muscle volume, with the vastus lateralis, vastus medialis, vastus intermedius, and rectus femoris muscles contributing approximately 12%, 6%, 4%, and 4%, respectively [14]. Validation of the BIA method using dual-energy x-ray absorptiometry (DXA) (DPX-L, GE Healthcare, Madison, WI, USA) showed very high correlations between the two methods for both fat mass and appendicular skeletal muscle mass [2].
Statistical analysis
All statistical analyses were conducted with Stata/SE version 15 (Stata Corp., College Station, TX). Complete case analyses were performed because a small proportion of patients had missing data. Characteristics were described using summary statistics: mean and standard deviation for normally distributed continuous variables, median and interquartile range for skewed continuous variables, and number and proportion for categorical variables. The unit of analysis was limbs for both analyses no. 1 and no. 2 (i.e., PhA and quadriceps strength in analyses no. 1 and no. 2, respectively), as the outcome variable and some predictor variables (i.e., KOA severity in analysis no. 1 and PhA and leg muscle mass in no. 2) are limb-specific [15]. For this reason, we fitted general linear models with a cluster-robust variance to account for the correlation of paired outcome data among individuals [16]. For analysis no. 1, we estimated mean differences in PhA by differences in KOA severity (treated as categorical variable). The details of these analyses to examine sex-specific differences in the association between KOA severity and PhA are described in Supplementary Text 1. For analysis no. 2, we estimated mean differences in quadriceps strength by differences in PhA, with adjustment for age, sex, diabetes, fat mass, KSS, leg muscle mass, and KOA severity. The details of these analyses to examine KOA severity-specific or sex-specific differences in the association between PhA and quadriceps strength are described in Supplementary Text 1. Interactions were assessed with the Wald test. A p value < 0.05 was considered to indicate statistical significance.
Results
Patient flow and descriptive statistics
Initially, 1217 patients with KOA who met the inclusion criteria were identified. Of these, 69 patients without the new KSS data, KOA severity category data, or diabetes were excluded. After further excluding patients with missing exposure and outcome variables for analyses, 1093 patients were included in the analyses (Fig. 2). Table 1 summarizes the baseline characteristics of the study population. The mean age was 72.8 years (standard deviation, 8.0 years), and more than three quarters (78%) of the patients were women.
KOA severity and PhA per leg
Table 2 shows the association between KOA severity and PhA. Evidence of a sex-specific difference in the association between KOA severity and PhA was found (P = 0.073 for interaction and 0.048 for sex- and multivariable-adjusted models, respectively). We found statistically significant relationships between increasing KOA severity and decreasing PhA in men, as shown in Table 2 (grade 4 KOA, mean difference = − 0.26°, 95% confidence interval (CI) = − 0.46 to − 0.07° and grade 3 KOA, mean difference = − 0.37°, 95% CI = − 0.55 to − 0.20°). In women, only KOA grade 4 was significantly associated with a decrease in PhA, and its magnitude is smaller than that in men (Table 2: mean difference = − 0.18°, 95% CI = − 0.27 to − 0.09°). Women also had smaller PhAs than men (Table 2: mean difference = − 0.71°, 95% CI = − 0.89 to − 0.53°). Figure 3 shows the adjusted mean PhA values by KOA severity for each sex; those with grade 4 disease had the lowest PhA values in both sexes (PhA at grade 4 was 4.29° (95% CI = 4.19 to 4.39°) and 3.77° (95% CI = 3.72 to 3.81°) in men and women, respectively).
Predicted phase angles by KOA severity per leg for each sex. Using the multivariable-adjusted general linear model including age, sex, fat mass, KOA severity, and interaction terms of sex and KOA severity, adjusted mean phase angles by KOA severity were predicted for each sex. Solid blue circles indicate point estimates for men. Solid red circles indicate point estimates for women. Error bars indicate 95% confidence intervals. KOA, knee osteoarthritis
PhA and quadriceps strength per leg
Table 3 shows the associations between PhA and quadriceps strength. Evidence of a difference in the association between PhA and quadriceps strength was not found for KOA severity and sex (P for interaction = 0.185 and 0.133 for KOA severity and sex, respectively). Therefore, analyses were performed without considering the interaction terms. PhA per leg was positively associated with quadriceps strength per leg independent of age, sex, leg muscle mass, pain, and KOA severity (Table 3: mean difference per 1° increase = 7.54 Nm (95% CI = 5.51 to 9.57 Nm). Figure 4 shows the adjusted mean quadriceps strength by PhA per leg. For example, patients who had a PhA of 2° per leg had a quadriceps strength of 35.9 Nm on average (95% CI = 30.0 to 41.9 Nm), whereas those who had a PhA of 7° per leg had a quadriceps strength of 73.6 Nm on average (95% CI = 69.2 to 78.1 Nm).
Predicted quadriceps strength by phase angle per leg. Using the multivariable-adjusted general linear model including age, sex, diabetes, fat mass, pain score, leg muscle mass, x-ray grade, and interaction pairs (the product terms of leg muscle mass and x-ray grade), adjusted mean quadriceps strength by phase angle per leg was predicted. The left vertical axis shows adjusted mean quadriceps strength. The solid line indicates point estimates of quadriceps strength. The dotted lines indicate 95% confidence intervals. Gray bars indicate frequency at which the phase angle values were measured. The right vertical axis shows frequency of each gray bar
Discussion
Our results showed that PhA gradually decreased with increasing KOA severity, and this association was similar in both sexes. In addition, PhA was positively associated with quadriceps strength regardless of KOA severity. These findings may encourage clinicians to pay more attention to PhA to improve quadriceps function in patients with KOA.
Several explanations are possible for the association of lower PhA with KOA progression. First, it can be explained in part by the decrease in muscle mass as KOA progresses [17]. PhA in part reflects the amount and quality of soft tissue, such as muscle and fat, and a previous study has shown that decreases in PhA were associated with loss of muscle mass, whereas increases in PhA were associated with increased fat mass [5]. Second, low PhA in patients with KOA can be explained by alteration in the cells and molecules of the quadriceps muscle. A previous study has shown that the quadriceps muscle in patients with KOA is characterized by excessive extracellular matrix, fewer satellite cells, and greater collagen content than in patients without KOA [18].
PhA was higher among male patients with KOA than among their female counterparts, which is in accordance with the results of previous studies [3, 7]. However, the association between PhA and KOA severity differed with sex. The reason for the sex-specific difference in the association is not known; however, it can be attributed to the differing amount and quality of muscle and fat and alteration in the cells and molecules of the quadriceps muscle in both sexes.
We believe that the demonstrated association of increasing PhA with increasing quadriceps strength has several implications for researchers and clinicians. First, the observed association is independent of lower limb muscle mass. Quadriceps strength may be decreased by alterations in cellular and molecular properties, which, in turn, may be reflected by the decreased PhA value, as PhA is generally reflective of cell membrane integrity and cell function [4, 5, 19]. Second, PhA may be modified through short-term resistance training [20, 21]. Thus, resistance training may be effective in improving PhA among patients with less severe KOA. Conversely, in patients with advanced KOA, for whom resistance training may be unfeasible because of knee pain, suspension training may promote PhA increase [22] by modulating exercise intensity, thereby controlling knee pain during weight-bearing exercise. Further research is required to determine whether these interventions are effective in improving PhA in patients with KOA.
Our study has several strengths. First, we analyzed both limbs in each patient using a suitable statistical method. Accordingly, our findings can be applied to both limbs in a single patient with KOA, given that osteoarthritis often affects both knees; this is supported by our finding that 95% of the non-surgical sides had grade 3/4 KOA. Second, PhA values are expected to be similar regardless of the BIA instruments used or their manufacturers: this is because PhA is directly derived from reactance and resistance values via a uniform, simple mathematical transformation.
Several limitations are also acknowledged in this study. First, the cross-sectional design did not allow investigation of the causal relationships between PhA and KOA severity and quadriceps strength. Second, the participants in this study were not representative of patients with KOA because most of them had severe KOA and were awaiting knee replacement surgery. However, we believe that the association between PhA and KOA severity and quadriceps strength is a valuable finding that should be validated in patients with KOA who do not require surgical treatment.
In conclusion, our study showed that PhA gradually decreased with increasing KOA severity, especially in men. In addition, the association of increasing PhA with increasing quadriceps strength was found regardless of KOA severity. To create a better program for improving quadriceps function in patients with KOA, clinicians should focus on increasing both muscle mass and PhA.
References
Chun SW, Kim KE, Jang SN, Kim KI, Paik NJ, Kim KW, Jang HC, Lim JY (2013) Muscle strength is the main associated factor of physical performance in older adults with knee osteoarthritis regardless of radiographic severity. Arch Gerontol Geriatr 56:377–382
Wada O, Kurita N, Kamitani T, Nakano N, Mizuno K (2019) Influence of the severity of knee osteoarthritis on the association between leg muscle mass and quadriceps strength: the SPSS-OK study. Clin Rheumatol 38:719–725
Kumar S, Dutt A, Hemraj S, Bhat S, Manipadybhima B (2012) Phase angle measurement in healthy human subjects through bio-impedance analysis. Iranian J Basic Med Sci 15:1180–1184
Lukaski HC, Kyle UG, Kondrup J (2017) Assessment of adult malnutrition and prognosis with bioelectrical impedance analysis: phase angle and impedance ratio. Curr Opin Clin Nutr Metabol Care 20:330–339
Norman K, Stobaus N, Pirlich M, Bosy-Westphal A (2012) Bioelectrical phase angle and impedance vector analysis—clinical relevance and applicability of impedance parameters. Clin Nutr 31:854–861
Erturk C, Altay MA, Sert C, Levent A, Yapti M, Yuce K (2015) The body composition of patients with knee osteoarthritis: relationship with clinical parameters and radiographic severity. Aging Clin Exp Res 27:673–679
Yamada M, Kimura Y, Ishiyama D et al (2019) Phase angle is a useful indicator for muscle function in older adults. J Nutr Health Aging 23:251–255
Altman R, Asch E, Bloch D, Bole G, Borenstein D, Brandt K, Christy W, Cooke TD, Greenwald R, Hochberg M (1986) Development of criteria for the classification and reporting of osteoarthritis. classification of osteoarthritis of the knee. Diagnostic and Therapeutic Criteria Committee of the American Rheumatism Association. Arthritis Rheum 29:1039–1049
Noordzij M, Dekker FW, Zoccali C, Jager KJ (2011) Sample size calculations. Nephron Clin Pract 118:c319–c323
Kellgren JH, Lawrence JS (1957) Radiological assessment of osteo-arthrosis. Ann Rheum Dis 16:494–502
Katoh M, Yamasaki H (2009) Comparison of reliability of isometric leg muscle strength measurements made using a hand-held dynamometer with and without a restraining belt. J Phys Ther Sci 21:37–42
Ito C, Maeda R, Ishida S, Sasaki H, Harada H (2000) Correlation among fasting plasma glucose, two-hour plasma glucose levels in OGTT and HbA1c. Diabetes Res Clin Pract 50:225–230
Scuderi GR, Bourne RB, Noble PC, Benjamin JB, Lonner JH, Scott WN (2012) The new knee society knee scoring system. Clin Ortho Rel Res 470:3–19
Handsfield GG, Meyer CH, Hart JM, Abel MF, Blemker SS (2014) Relationships of 35 lower limb muscles to height and body mass quantified using MRI. J Biomech 47:631–638
Sutton AJ, Muir KR, Jones AC (1997) Two knees or one person: data analysis strategies for paired joints or organs. Ann Rheum Dis 56:401–402
Cameron AC, Miller DL (2015) A practitioner’s guide to cluster-robust inference. J Hum Resour 50:317–372
Taniguchi M, Fukumoto Y, Kobayashi M, Kawasaki T, Maegawa S, Ibuki S, Ichihashi N (2015) Quantity and quality of the lower extremity muscles in women with knee osteoarthritis. Ultrasound Med Biol 41:2567–2574
Noehren B, Kosmac K, Walton RG et al (2018) Alterations in quadriceps muscle cellular and molecular properties in adults with moderate knee osteoarthritis. Osteoarthr Cartilage 26:1359–1368
Gonzalez MC, Barbosa-Silva TG, Bielemann RM, Gallagher D, Heymsfield SB (2016) Phase angle and its determinants in healthy subjects: influence of body composition. Am J Clin Nutr 103:712–716
Dos Santos L, Cyrino ES, Antunes M, Santos DA, Sardinha LB (2016) Changes in phase angle and body composition induced by resistance training in older women. Eur J Clin Nutr 70:1408–1413
Souza MF, Tomeleri CM, Ribeiro AS et al (2017) Effect of resistance training on phase angle in older women: a randomized controlled trial. Scand J Med Sci Sport 27:1308–1316
Campa F, Silva AM, Toselli S (2018) Changes in phase angle and handgrip strength induced by suspension training in older women. Int J Sport Med 39:442–449
Acknowledgments
The authors greatly thank the following research assistants and medical staff members for their assistance in collecting the clinical information used in this study: Takehiro Kaga, Tomohiro Oka, Yoriko Tamura, Hiroshi Nishi, Yuichi Isaji, Yutaka Sato, Tomohiro Takagi, Kaho Shibata, Maho Wakai, Chisato Shindoh, Kenta Hirose, Takuma Ota, Tatsuya Arita, Yuuki Ikawa, Tsuyoshi Fukui, Riuji Nakagawa, Taisuke Hayashida, Shuto Fujii, Keisuke Yoneya, Kazuaki Mori (Anshin Hospital, Kobe), and Lisa Shimokawa (Fukushima Medical University Hospital, Fukushima city, Fukushima).
Availability of data and material
The datasets generated during and/or analyzed during the present study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Funding
This study was supported by JSPS KAKENHI (Grant Number: JP15K16518). The JSPS had no role in this study except for funding.
Author information
Authors and Affiliations
Contributions
Research idea and study design: OW, NK; data acquisition: OW, KM; data analysis and interpretation: OW, NK, MY, KM; statistical analysis: NK; supervision or mentorship: NK, MY, KM. Each author contributed important intellectual content during article drafting or revision and accepts accountability for the overall work by ensuring that questions pertaining to the accuracy or integrity of any portion of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Disclosures
None.
Ethical approval
The study was approved by the local institutional review board (no. 57, January 26, 2017) and by the Research Ethics Committee of Fukushima Medical University School of Medicine (no. 2850, September 28, 2016).
Consent to participate
Informed consent for the present study was not mandatory according to the “ethical guidelines for medical and health research involving human subjects” in Japan.
Consent for publication
Not applicable
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM1
(DOCX 52.5 kb)
Rights and permissions
About this article
Cite this article
Wada, O., Kurita, N., Yamada, M. et al. Structural severity, phase angle, and quadriceps strength among patients with knee osteoarthritis: the SPSS-OK study. Clin Rheumatol 39, 3049–3056 (2020). https://doi.org/10.1007/s10067-020-05056-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05056-w