Abstract
Objective
Previous studies of the diagnostic accuracy of ultrasound (US) and dual-energy computed tomography (DECT) in patients with gout have reported different results. The aim of this study is to compare the diagnostic value of US and DECT in patients with different stages of acute gouty arthritis.
Methods
Based on the presence of monosodium urate (MSU) crystals in the synovial fluid, patients (n = 37) were divided into three groups according to gout duration: early stage (within 1 year, n = 15), middle stage (1 to 3 years, n = 12), and late stage (more than 3 years, n = 10). All the affected joints were examined by US and DECT.
Results
In the early-stage group, the sensitivity of US was significantly higher than DECT in identifying MSU deposition (66.7% vs 26.6%, p < 0.05), while in the middle- and late-stage groups, the sensitivity of US and DECT was similar. In the early-stage group, the US results in nine joints were positive (four with double contour sign, four with snowstorm sign, and one with both double contour sign and snowstorm sign), while DECT did not show any urate crystal deposits.
Conclusion
These findings indicate that US should be the first choice for acute gouty arthritis, especially in patients with early-stage disease.
Key Points • Previous studies have compared DECT with US to evaluate the reliability of each method in diagnosing gout but have reported different results, which may be partly due to different gout duration. • In our study, the sensitivity of US was significantly higher than DECT in identifying MSU deposition in the early-stage group, while in the middle- and late-stage groups, the sensitivity of US and DECT was similar. • US should be the first choice for the diagnosis of acute gouty arthritis, especially in patients with early-stage disease. |
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Gout is the most common inflammatory arthropathy, reported to affect 2.13% of the population of the USA in 2009 [1]. The detection of monosodium urate (MSU) crystals in the synovial fluid by compensated polarized light microscopy is recognized as the gold standard for the diagnosis of gouty arthritis [2]. However, synovial fluid aspiration is invasive and usually is not feasible. Recently, new imaging techniques, such as ultrasonography (US) and dual-energy computed tomography (DECT), have been used more widely [3,4,5,6]. Both techniques are included in the 2015 gout classification criteria developed by the American College of Rheumatology in collaboration with the European League Against Rheumatism [7].
Several studies have compared DECT with US to evaluate the reliability of each method in diagnosing gout. Gruber et al. found that the sensitivity of DECT and US in diagnosing gout was comparable [8]. Another study by Huppertz et al. found that DECT was less sensitive than US [9], whereas Zhu et al. found that the sensitivity of DECT was higher than US in identifying MSU deposition [10]. This discrepancy may be due to different durations of disease, different joint regions, and whether onset is recent or not.
The aim of our study is to compare US with DECT for the diagnosis of acute gouty arthritis in patients with different durations of gout.
Patients and methods
Patients
In this cross-sectional study, we recruited consecutive patients with a clinical suspicion of acute gouty arthritis seen at the First Affiliated Hospital of Jiaxing University from July 2017 to July 2019. Patients had to have at least one swollen joint that could be aspirated. Patients with gouty tophi were excluded. Based on the results of our reference test for the presence of MSU crystals, patients were divided into three groups according to the duration of gout: early stage (within 1 year), middle stage (1 to 3 years), and late stage (more than 3 years).
The study protocol was approved by the First Affiliated Hospital of Jiaxing University Ethics Committee. Written informed consent was obtained from each patient. The study was conducted in accordance with the principles of the Declaration of Helsinki.
MSU crystals analysis
Synovial fluid aspiration was conducted under US guidance. If patients were referred for aspiration of more than one joint, the most prominently involved joint clinically was chosen as the index joint. The aspirated synovial fluid was viewed under a polarizing microscope within minutes after aspiration by a rheumatologist with 5 years of experience in microscopic examinations. When synovial fluid contained MSU crystals, they appear as bright yellow, needle-like, negatively birefringent crystals.
US examination
US scanning of the symptomatic joints or areas was performed after crystals analysis on the same day. All US scanning was conducted by an experienced ultrasonic expert with more than 5 years of experience in maneuvering musculoskeletal US. The US examiner was blind to the results of the synovial fluid analysis. The presences of MSU crystal deposits and/or the snowstorm sign and/or the double contour sign were considered positive for the presence of gouty arthritis.
DECT examination
DECT examination of the symptomatic joints or areas was performed on the same day after crystals analysis and US scanning. A 128-row dual-source CT scanner (Somatom Definition Flash; Siemens Healthcare, Forchheim, Germany) equipped with two X-ray tubes and two corresponding detectors was used. Post-processing was performed using a commercial software program (“Gout,” Syngo CT Workplace, Siemens Medical Systems) to create material-selective images, where MSU deposits were color-coded as green. The results were analyzed by one DECT reader with more than 5 years of experience in musculoskeletal radiology who was blind to the results of both synovial fluid analysis and US examination.
Statistical analyses
Variables are described as mean and standard deviation or proportion. The difference of positive rate between DECT and US was analyzed using chi-squared analysis. A p value of < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS 17 (IBM, Cary NC, USA).
Results
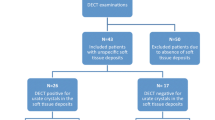
A total of 41 consecutive patients with a clinical suspicion of acute gouty arthritis were enrolled in this study. Synovial fluid analysis found MSU crystals in 37 patients who were diagnosed with gouty arthritis. Two patients with calcium pyrophosphate deposition arthritis and two patients with septic arthritis were excluded from the study. Among the 37 patients (36 males, aged 22 to 81 years) with acute gouty arthritis, MSU crystals were not found at the initial aspiration in 2 patients but were found at the second aspiration.
The clinical characteristics of the 37 patients are shown in Table 1. The disease duration in the 37 patients ranged from 1 day to 21 years, and in 4 patients, this was the first attack of gout. Patients were divided into three groups: early stage (n = 15), middle stage (n = 12), and late stage (n = 10). These 37 patients with 55 affected joints or areas underwent US and DECT examinations. The most symptomatic joint or area was knee (60%), followed by the foot (21.8%) and the hand (14.5%). The average scores by ACR/EULAR gout classification system of 2015 were 10.8 ± 2.1. According to this classification system, gout is diagnosed in patients with a score ≥ 8 or MSU positivity [7].
As shown in Table 2, US findings were positive for gouty arthritis in 27 of 37 patients (72.9%), including 10 patients (66.7%) in the early stage, 9 patients (75%) in the middle stage, and 8 patients (80%) in the late stage. In 21 of 37 patients (56.6%), DECT examinations were positive, including 4 patients (26.6%) in the early stage, 8 patients (66.6%) in the middle stage, and 9 patients (90%) in the late stage. In the early-stage group, the sensitivity of US was significantly higher than that of DECT in identifying MSU deposition (66.7% vs 26.6%, p < 0.05), while in the middle and late-stage groups, the sensitivity of US and DECT was similar.
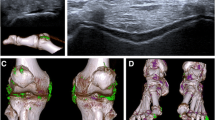
In the early-stage group, the US results were positive in 9 joints (4 with double contour sign, 4 with snowstorm sign, and 1 with both double contour sign and snowstorm sign), while DECT did not show any urate crystal deposits. In the middle-stage group, the US results were positive in 3 joints, while DECT results were negative, and in 1 joint, the DECT findings were positive, while the US examination was negative. In the late-stage group, the US findings in 3 joints were positive, while DECT examination was negative, and in 2 joints, the DECT results were positive, while the US examination was negative. Figs. 1 and 2 showed the typical findings of DECT and US.
Discussion
In this study, microscopy of synovial fluid demonstrated MSU crystals in 35 of 37 patients (94.6%) at the initial aspiration and in the remaining 2 patients at the second aspiration. Repeated aspiration from different involved joints or the same joint at different times can improve the detection rate of MSU crystals [11]. Consistent with our results, MSU crystals were observed in more than 95% of patients with acute gouty arthritis in an earlier study by Lawry and Bluestone [12]. Unfortunately, the high dependence on microscopic analyses of synovial fluid is not without problems: joint aspiration is invasive, obtaining adequate specimens from small joints may not be feasible, and sometime it may be hard to find the MSU crystals in patients with a low synovial fluid leukocyte count after an acute attack [13, 14].
Previous studies assessing the test characteristics of US and DECT in patients with gout have shown different results concerning the diagnostic accuracy of each imaging modality [15]. We thought this discrepancy might be partly due to the inclusion of patients with different durations of gout, so we divided patients in our study into three groups according to the duration of gout. In addition, subjects with tophi, a clinical situation where detection of MSU deposition by US or DECT scanning is usually not required for diagnostic confirmation, were excluded from our study.
As we know, MSU crystal deposition can be identified on US by one of the three signs: the presence of tophi and/or the snowstorm sign and/or the double contour sign, while MSU deposits are color-coded as green on DECT [16, 17]. In our study, US had a significantly higher sensitivity than DECT for the presence of MSU deposition in the early-stage group, in whom US demonstrated the snowstorm sign in four joints and the double contour sign in five joints, while DECT did not show any urate crystal deposits. However, in the other two groups, the DECT finding was positive in three joints, while the US examination was negative. These results suggest that the snowstorm sign in US represented free MSU crystals in the synovial fluid, which were hard to see on DECT. The presence of tophi, which indicate a long-standing duration of gout, can easily be seen with both US and DECT. The double contour sign in US can be seen more than half of the time by DECT. DECT can also find some deep anatomical MSU crystals which are undetectable by US.
Our results were similar to those of several previous studies. Bongartz et al. showed that DECT appears to have limited sensitivity in patients with acute gout and no prior episodes of gouty arthritis, and 20% of patients with early disease did not demonstrate evidence of MSU deposition on DECT images [18]. Wang et al. found that DECT may miss crystal deposition on the surface of the cartilage, which was the most frequent US manifestation in patients [19]. Ogdie et al. found that US features of MSU crystal deposition had high specificity and high positive predictive value but more limited sensitivity for early gout [20].
In conclusion, our study demonstrated that US had higher sensitivity in the presence of MSU crystals for early gout than DECT. Based on this, in early gout, DECT is not as helpful for diagnosis and will not easily give you a 4-point score based on the 2015 criteria [7]. However, using the 2015 criteria, US may be sufficient. We suggest that US should be the first choice for the diagnosis of acute gouty arthritis, especially in patients with early-stage disease. However, data integration on a larger cohort is necessary in the future, given the small sample size of the patients analyzed in the present.
References
Brook RA, Forsythe A, Smeeding JE, Lawrence Edwards N (2010) Chronic gout: epidemiology, disease progression, treatment and disease burden. Curr Med Res Opin 26:2813–2821
Zhang W, Doherty M, Pascual E, Bardin T, Barskova V, Conaghan P, Gerster J, Jacobs J, Leeb B, Lioté F, McCarthy G, Netter P, Nuki G, Perez-Ruiz F, Pignone A, Pimentão J, Punzi L, Roddy E, Uhlig T, Zimmermann-Gòrska I, EULAR Standing Committee for International Clinical Studies Including Therapeutics (2006) EULAR evidence based recommendations for gout. part I: diagnosis. Report of a task force of the Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT). Ann Rheum Dis 65:1301–1311
Gamala M, Jacobs JWG, Linn-Rasker SF et al (2019) The performance of dual-energy CT in the classification criteria of gout: a prospective study in subjects with unclassified arthritis. Rheumatology (Oxford):2019
Schueller-Weidekamm C, Schueller G, Aringer M, Weber M, Kainberger F (2007) Impact of sonography in gouty arthritis: comparison with conventional radiography, clinical examination, and laboratory findings. Eur J Radiol 62:437–443
Choi HK, Burns LC, Shojania K, Koenig N, Reid G, Abufayyah M, Law G, Kydd AS, Ouellette H, Nicolaou S (2012) Dual energy CT in gout: a prospective validation study. Ann Rheum Dis 71:1466–1471
Dalbeth N, Choi HK (2013) Dual-energy computed tomography for gout diagnosis and management. Curr Rheumatol Rep 15:301
Neogi T, Jansen TL, Dalbeth N, Fransen J, Schumacher HR, Berendsen D, Brown M, Choi H, Edwards NL, Janssens HJ, Lioté F, Naden RP, Nuki G, Ogdie A, Perez-Ruiz F, Saag K, Singh JA, Sundy JS, Tausche AK, Vaquez-Mellado J, Yarows SA, Taylor WJ (2015) 2015 gout classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann Rheum Dis 74:1789–1798
Gruber M, Bodner G, Rath E, Supp G, Weber M, Schueller-Weidekamm C (2014) Dual-energy computed tomography compared with ultrasound in the diagnosis of gout. Rheumatology (Oxford) 53:173–179
Huppertz A, Hermann KG, Diekhoff T, Wagner M, Hamm B, Schmidt WA (2014) Systemic staging for urate crystal deposits with dual-energy CT and ultrasound in patients with suspected gout. Rheumatol Int 34:763–771
Zhu L, Wu H, Wu X, Sun W, Zhang T, Ye L, Wang W, Wang J (2015) Comparison between dual-energy computed tomography and ultrasound in the diagnosis of gout of various joints. Acad Radiol 22:1497–1502
Mader R (1998) Repeated synovial fluid analysis may be needed to find crystals in gout. Clin Rheumatol 17:549–550
Lawry GV 2nd, Fan TP, Bluestone R (1988) Polyarticular versus monoarticular gout: a prospective analysis of clinical features. Medicine (Baltimore) 67:335–342
Pascual E, Sivera F, Andrés M (2011) Synovial fluid analysis for crystals. Curr Opin Rheumatol 23:161–169
Swan AAH, Dieppe P (2002) The value of synovial fluid assays in the diagnosis of joint disease: a literature survey. Ann Rheum Dis:493–498
Ogdie A, Taylor WJ, Weatherall M et al (2015) Imaging modalities for the classification of gout: systematic literature review and meta-analysis. Ann Rheum Dis 74:1868–1874
Grassi W, Okano T, Filippucci E (2015) Use of ultrasound for diagnosis and monitoring of outcomes in crystal arthropathies. Curr Opin Rheumatol 27:147–155
Glazebrook KN, Guimaraes LS, Murthy NS et al (2011) Identification of intraarticular and periarticular uric acid crystals with dual-energy CT: initial evaluation. Radiology 261:516–524
Bongartz T, Glazebrook KN, Kavros SJ et al (2015) Dual-energy CT for the diagnosis of gout: an accuracy and diagnostic yield study. Ann Rheum Dis 74:1072–1077
Wang Y, Deng X, Xu Y et al (2018) Detection of uric acid crystal deposition by ultrasonography and dual-energy computed tomography: a cross-sectional study in patients with clinically diagnosed gout. Medicine (Baltimore) 97:e12834
Ogdie A, Taylor WJ, Neogi T et al (2017) Performance of ultrasound in the diagnosis of gout in a multicenter study: comparison with monosodium urate monohydrate crystal analysis as the gold standard. Arthritis Rheumatol 69:429–438
Funding
This study was partly supported by the grants from the National Science Foundation of Zhejiang (LY19H100002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, B., Yang, M. & Wang, H. Diagnostic value of ultrasound versus dual-energy computed tomography in patients with different stages of acute gouty arthritis. Clin Rheumatol 39, 1649–1653 (2020). https://doi.org/10.1007/s10067-020-05014-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05014-6