Abstract
Osteoporosis is characterized by the loss of bone mass, deterioration of the bone microarchitecture, and an increased risk of fractures; these later complications are associated with significant morbidity and mortality. The asymptomatic and progressive nature of osteoporosis underscores the importance of identifying this entity in early stages. Despite the various treatments available, the prevention of the disease represents the most important aspect of management. An adequate intake of calcium and vitamin D as well as a healthy lifestyle is the basis for maintaining bone health. When osteoporosis is diagnosed, the choice of medications must be individualized considering characteristics of the patient and the risk of fractures. In this article, we review the main causes of osteoporosis, when and how to start treatment, and appropriate therapy and monitoring.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis affects 200 million people worldwide. Approximately 30% of postmenopausal women in the USA have osteoporosis, and 40% of them develop fragility fractures [1].
Three elements define osteoporosis: loss of bone mass (bone quantity), deterioration of bone microarchitecture (bone quality), and increasing fracture risk [2,3,4]. Fractures are morbid complications causing loss of independence, chronic pain, and need for rehabilitation. Hip fracture is the most devastating subtype, and 20–40% of individuals suffering hip fractures die within a year, and 10% of survivors fracture the contralateral hip [5].
The assessment of bone mass aids in diagnosis, risk prediction, and selection and monitoring of treatment. According to the International Society for Clinical Densitometry (ISCD), bone mineral density (BMD) measurement should be performed in women ≥ 65 and men ≥ 70 years old [6]. If several risk factors exist, BMD should be assessed in postmenopausal women younger than 65 and men younger than 70. The Fracture Risk Assessment Tool (FRAX ®) is an algorithm that predicts the 10-year incidence of hip and major osteoporotic fractures (clinical spine, forearm, hip, or shoulder fracture). FRAX is valuable in both assessing fracture risk and treatment. It combines individualized models integrated with clinical risk factors and BMD at the femoral neck [7]. The National Bone Health Alliance (NBHA) and the Clinical Diagnosis of Osteoporosis Working Group [8] recommend that postmenopausal women and men aged 50 years or older are diagnosed with osteoporosis if they have: T-score ≤ − 2.5 at the spine or hip; low-trauma hip fracture with or without BMD assessment, osteopenia by BMD with a low-trauma vertebral, proximal humerus, pelvis, or in some cases distal forearm fracture, and FRAX risk estimates above the country-specific threshold (US threshold: 10-year probability of major osteoporotic fracture ≥ 20% or 10-year probability of hip fracture ≥ 3%) [9].
When to initiate therapy
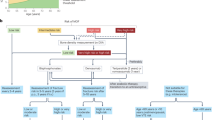
An antecedent fragility fracture is an indication for treatment regardless of T-score. Guidelines recommend a fracture risk based on BMD to define populations needing treatment, generally using a T-score ≤ − 2.5 alone or in combination with clinical risk calculations (i.e., FRAX®) [10,11,12] (see Table 1).
New diagnoses should prompt an evaluation of secondary causes of osteoporosis. A blood count, chemistry panel, intact parathyroid hormone, and a 24-h urine calcium should be assessed, and other testing should be considered according to clinical suspicion (see Table 2). Clinicians can stress modifying clinical risk factors and adjusting or suspending medications associated with loss of BMD or increased fall risk [4, 13, 14].
Choosing therapy depends on several factors: efficacy, tolerability, safety, adverse event profile, route of administration, frequency, non-skeletal benefits, and cost. Treatments should be approved by the Food and Drug Administration (FDA), the European Medicines Agency (EMA) and local regulatory authorities [13] (see Table 3).
Correcting vitamin D deficiency
Vitamin D deficiency is associated with secondary hyperparathyroidism, osteomalacia, altered bone turnover, osteoporosis, and increased risk of falls and fractures. Etiologies of vitamin D deficiency include inadequate sunlight exposure, altered absorption of vitamin D and medications [16, 17]. Vitamin D deficiency may result in poor responses to osteoporosis treatment [4, 16].
An average calcidiol [25(OH)D] level of 26.4 ng/ml (66 nmol/L) may reduce non-vertebral fracture risk in men and women over 65 years old, while an average level of 29.6 ng/ml (74 nmol/L) may reduce hip fracture risk [18]. Some experts suggest that calcidiol levels lower than 30–32 ng/ml (74.8–80 nmol/L) are suboptimal and the physiologic range begins above this value. The level estimated to suppress PTH is between 12.8–20 ng/ml (32–50 nmol/L) and 27.2–30 ng/ml (68–75 nmol/L) depending on the analytical approach [6, 18].
800–1000 IU/day of vitamin D is sufficient to achieve a calcidiol value of 30 ng/ml (75 nmol/L), and vitamin D2 (ergocalciferol) and D3 (cholecalciferol) are both viable supplements [13, 18]. However, when initial values are deficient, 800–1000 IU/day may not attain desired levels, so the practical alternative is administering high doses (50,000 IU/week) for 8 weeks, followed by the routine daily dose. Another option is administering daily doses of 6000 IU/day to reach levels above 30 ng/ml, followed by 1000–2000 IU/day to maintain that level [4, 16, 19].
Choosing an agent
Risks and benefits of each drug should be considered prior to prescription, and common pitfalls should be known. Parenteral drug adherence is optimal, which is useful when oral medication is contraindicated or poorly tolerated [20]. Zoledronic acid requires a single annual infusion; however, like all intravenous bisphosphonates, it may cause post-infusion arthralgias and flu-like symptoms. These symptoms are generally transient and only occur after the first infusion. Denosumab is administered subcutaneously every 6 months [21].
Alendronate and risedronate should be taken 30 min, and ibandronate 60 min, before any other medication or food or lying down [13]. Comorbidities inhibiting prolonged standing may relatively contraindicate oral bisphosphonates. Bedridden patients should avoid hormonal therapy due to risks of thromboembolism [13, 21]. Oral bisphosphonates are contraindicated when esophageal abnormalities exist, and renal insufficiency limits using any bisphosphonate. Hypocalcemia restricts bisphosphonates and denosumab and should be corrected prior to therapy. A history of bone neoplasms, metastases, or increased risk of osteosarcoma contraindicates PTH-analog therapy [21].
Alendronate, risedronate, and zoledronic acid are currently considered first-line agents due to their body of evidence showing fracture incidence reduction. In older individuals, when efficacy at multiple sites (spine, hip, and non-vertebral) is desired, alendronate, risedronate, and zoledronic acid all are good options [14, 21,22,23] (see Table 4). Some guidelines consider denosumab a first-line agent [4, 15, 23], but others prefer its use for high-risk individuals and therapeutic failures. Denosumab is favored in the setting of advanced renal insufficiency [24]. The UK clinical guideline for osteoporosis prevention and treatment considers alendronate and risedronate first-line and intravenous bisphosphonates or denosumab the most appropriate alternatives [12].
Teriparatide is a PTH-analog, an anabolic agent approved by FDA for patients with high fracture risk, specifically a very low T-score and/or failure or intolerance to previous treatments [4, 15, 22]. Teriparatide protects against vertebral and non-vertebral (non-hip) fractures but has not shown efficacy in reducing hip fracture risk [4, 12, 25]. Abaloparatide is a synthetic analog of PTH-related peptide (PTHrP) that retains anabolic activity with less bone resorption compared with PTHrP. In the ACTIVE trial, abaloparatide was more effective preventing vertebral fractures compared to placebo at 18 months, and in a secondary endpoint, significantly reduced the non-vertebral fracture risk over placebo [26]. Duration of PTH-analog therapy should be less than 24 months and followed by antiresorptive therapy to preserve gains in BMD [4, 25].
Estrogens and selective estrogen receptor modulators (SERMs) are no longer considered first-line therapy but may be a preventative option for women with significant risk who cannot receive non-estrogenic treatment, have climacteric symptoms, or have menopause-associated vaginal atrophy [13, 17, 21, 22, 28]. Raloxifene reduces the risk of vertebral fractures in women with osteopenia and osteoporosis but failed to consistently reduce non-vertebral fracture risk [21].
Novel therapies include romosozumab, a monoclonal antibody-producing anabolic effect by inhibiting sclerostin, an inhibitor of Wnt pathways that suppresses the proliferation of osteoblasts [29]. Romosozumab was more effective than alendronate at preventing vertebral fractures in postmenopausal women, but safety signals revealed increased risks of serious cardiovascular events, thus delaying approval pending further review [30]. Next, odanacatib selectively inhibits cathepsin K, an extracellular protease produced by osteoclasts to degrade bony matrix. This mechanism hypothetically spares anabolic effects of osteoblasts by preserving cell-to-cell signals from osteoclasts [31, 32]. The study evaluating odanacatib also had to be discontinued because of major adverse cardiovascular events [33].
Combination treatment
Osteoporosis therapies have been combined to attain greater yields in increasing bone density. In severe, advanced, or refractory disease, combining therapies may be considered, especially administering anabolic therapy (i.e., abaloparatide or teriparatide) followed by antiresorptive drug (i.e., bisphosphonates or denosumab). This combination strategy has shown superiority over monotherapy in increasing BMD [25, 34,35,36].
The DATA (Denosumab and Teriparatide Administration) study examined denosumab and teriparatide over 12 months and compared BMD in individuals receiving each alone or a combination of the two. The combination group had the greatest increases in BMD in the spine, femoral neck and total hip, as measured by dual-energy X-ray absorptiometry (DXA) [35]. In the DATA-HRpQTC study, the combination of denosumab and teriparatide after 12 months demonstrated improvement in bone quality compared with either alone via high-resolution quantitative tomography. At 24 months, this combination continued to demonstrate improvements in bone microarchitecture suggesting that efficacy may improve during the second year [37,38,39].
Special populations
Osteoporosis in men
Osteoporosis in men is a growing problem due to an aging population. An estimated 1–8% of men suffer from osteoporosis in industrialized countries [40] and about 1 in 8 men over 50 years of age will suffer an osteoporotic fracture [41]. Mortality after fracture was higher for men than women, and some authors report that men have twofold mortality rates after hip fractures [42, 43]. Male-specific determinants of bone mass like the peak bone mass occur later in life because of later onset of puberty. Males also have larger periosteal deposition of bone conferring a better resistance to mechanical forces [44].
Trabecular bone mass decreases with age in both sexes, but this loss is accelerated by menopause. Cortical bone mass remains stable until menopause for women and even later ages for men. Women mainly experience trabecular bone perforation and loss of connectivity, while men predominantly experience trabecular thinning. Perforation is structurally more detrimental and could partially explain the higher fracture risks of women [41].
Two hundred thirty-two patients were studied to describe the etiology and characteristics of osteoporosis in men, and results revealed 57% had idiopathic osteoporosis, which was more common under the age of 60. Forty-three percent of men developed secondary osteoporosis, which was more frequent in individuals older than 60 years. The risk factors of osteoporosis are similar despite sex; however, men with idiopathic osteoporosis have higher frequencies of hypercalciuria and family history [45].
Common secondary causes of osteoporosis in men are excessive consumption of alcohol, hypogonadism, tobacco use, and prolonged treatment with corticosteroids [45,46,47,48,49]. Kanis et al. found up to 68% of both sexes suffering a hip fracture and 38% suffering any fragility fracture excessively consumed alcohol (≥ 3 units/day) [45, 46]. A meta-analysis including 3,730,424 participants revealed that heavy alcohol consumption was associated with a trend toward increased hip fracture risk, but there was significant heterogeneity between studies (P < 0.001, I2 = 72.6%). Analyses based on amount of alcohol consumption revealed the RR of hip fractures was 0.88 (95% CI 0.83–0.89) with light alcohol consumption (0.01–12.5 g/day), 1.00 (95% CI 0.85–1.14) with moderate consumption (12.6–49.9 g/day), and 1.71 (95% CI: 1.41–2.01) with heavy consumption (≥ 50 g/day) [47].
A meta-analysis of 59,232 participants found an association between smoking and the risk of any fracture (RR = 1.25, 95% CI 1.15–1.36). The relative risk (RR) of suffering a hip fracture was 1.84 (95% CI 1.52–2.22). The male subgroup (26% of the study population) had elevated risks of osteoporotic fractures at any site [48].
Another risk common in older men is androgenic hormone suppression in individuals with prostate cancer. Hormonal therapy is associated with a fracture risk of 20% in the first 5 years [49] and a rapid loss of bone mass after the first year of treatment (approximately 2–4% in the lumbar spine and hip) [50]. Clinically, we assume that BMD scores (T-scores ≤ − 2.5) can be used similarly in both men age 50 and older and postmenopausal women; however, fractures in men have been associated with higher absolute BMD values in comparison to women [43, 49, 50].
Therapeutic studies in men are scarce but bisphosphonates, denosumab, and teriparatide are FDA-approved options. Bisphosphonates, including alendronate, increase BMD and decrease bone turnover markers (BTMs) [12, 13, 51], and in a study of men with hypogonadism, alendronate reduced the incidence of vertebral fractures to 0.8% versus 7.1% of controls (P = 0.02) [52]. For men at high risk of vertebral fracture, especially with glucocorticoid-induced osteoporosis, bisphosphonates, denosumab, or teriparatide all seem reasonable [12].
Denosumab is efficacious in increasing BMD at the lumbar spine, total hip, femoral neck, trochanter, and radius. Denosumab was associated with a lower incidence of new vertebral fractures in men with prostate cancer receiving androgen deprivation therapy [53].
Glucocorticoid-induced osteoporosis
Glucocorticoid-induced osteoporosis (GIO) is a very common cause of secondary osteoporosis. Around 1–3% of the world population and 1% of the US adult population receives prolonged GC therapy [54, 55]. Fracture risk increases during the first 3–6 months of treatment, even with doses of 5 mg/day of prednisone, or its equivalent, due to high rates of trabecular bone loss [56].
GCs decrease function and promote apoptosis of osteoblasts via suppressing IGF-1 and TGF-β, both factors that promote bone formation. GCs also upregulate Wnt inhibitors, such as sclerostin and Dickkopf-1 (Dkk-1). Additionally, GCs prolong the lifespan of osteoclasts, increasing bone resorption [57, 58].
Doses of ≥ 7.5 mg/day of prednisolone (or equivalent) have an RR of 5.18 (95% CI 4.25–6.31) for vertebral fractures and 2.27 (2.16–3.10) for non-vertebral fractures [58]. An English cohort evaluated the RR of fracture of 244,235 patients on treatment with oral corticosteroids compared with 244,235 controls. Prednisolone ≥ 7.5 mg/day, or equivalent, produced a RR of 1.44 (95% CI 1.34–1.54) for non-vertebral fracture, 2.21 (95% CI 1.85–2.64) for hip fractures, and 2.83 (95% CI 2.35–2.40) for vertebral fractures, when compared to lower doses (< 2.5 mg/day) [59].
According to ACR guideline-based therapy, fracture risk should be stratified in each patient. For patients under 40 years of age, high-risk individuals are those with previous osteoporotic fractures, and moderate risk are those expected to continue GCs at > 7.5 mg/day for 6 months with either a hip or spine Z-score < −3 or a rapid decline in hip or spine BMD (≥ 10% loss in 1 year) during GC treatment. Low risk is assigned when no risk factor other than GC treatment is present [55]. In patients over 40 years of age, the risk outline is seen in Table 5.
All individuals receiving doses ≥ 2.5 mg/day of prednisone, or equivalent, for 3 months or more should optimize the intake of calcium (1000–1200 mg/day) and vitamin D (600–800 IU) along with enacting lifestyle modifications (maintain appropriate weight, smoking cessation, etc.). Those ≥ 40 years of age with a moderate risk of fracture may be treated with oral bisphosphonates (this recommendation is stronger in the high-risk population), but if oral therapy is contraindicated or poorly tolerated, intravenous bisphosphonates may be used. If bisphosphonates are contraindicated, teriparatide or denosumab are recommended, preferably in that order. If none of the above can be used, raloxifene may be considered in postmenopausal women [55].
Individuals < 40 years of age at moderate or high risk of fracture should be treated with oral bisphosphonates. If contraindications exist, the same alternative medications listed for adults ≥ 40 years of age are recommended; however, raloxifene should not be used in men or premenopausal women. The guidelines also include recommendations for special populations (women of childbearing potential, adults with organ transplantation, etc.) [55].
For individuals receiving ≥ 7.5 mg/day of prednisone (or equivalent), denosumab is superior to risedronate in increasing bone mineral density at the lumbar spine for patients on chronic GCs (4.4% [95% CI 3.8–5.0] vs. 2.3% [1.7–2.9], p < 0.0001) as well as those who had recently started GC therapy (3.8% [3.1–4.5] vs. 0.8% [0.2–1.5], p < 0.0001). Denosumab is also associated with improvements in cortical bone structure assessed by high resolution peripheral quantitative computed tomography (HR-pQCT) of the radius and tibia when compared to risedronate after 12 and 24 months of treatment [60].
Monitoring treatment
The National Osteoporosis Foundation (NOF) recommends monitoring with DXA every 2 years or less in certain clinical circumstances [13]. The ISCD recommends repeating a DXA in the following circumstances: once the expected change in BMD equals or exceeds the least significant change (LSC) of DXA measurement, 1 year after starting treatment, and/or when changing therapy. Subsequent intervals may be lengthened after therapeutic effect is established [6, 11]. AACE (American Association of Clinical Endocrinologists) recommends repeating bone densitometry 1 to 2 years after initiating treatment until BMD has stabilized [4].
Osteoporosis therapy induces rapid and large changes in BTMs; thus, they have the potential to predict treatment responses in individual cases, which could assist in treatment decisions. BTM assessment is recommended between 3 and 6 months after treatment initiation. Antiresorptive treatment significantly decreases markers of bone resorption within days or weeks followed by a decline in bone formation markers [62, 63]. Anabolic agents elevate markers of bone formation 1–3 months after treatment initiation [13]. βCTX is a preferred marker of bone resorption, and the PINP is a marker of bone formation. Changes in BTMs during treatment indicate adequate therapy and can be re-assessed earlier during treatment compared to BMD [61].
Defining therapeutic success and failure
Therapeutic success of treatment is poorly defined. Carey defines therapeutic success for bisphosphonates based on clinical, densitometric, and laboratory parameters (see Table 6) but notes that bisphosphonates reduce the risk of fracture even without elevation of BMD or significant changes in BTMs [64]. The Committee of Scientific Advisors of the International Osteoporosis Foundation (IOF) examined three parameters that may help define treatment failure: incident fractures, bone mineral density, and BTMs [65]. Previous fragility fractures confer high risk for incident fractures.
Gehlbach found that women with 1, 2, or ≥ 3 previous fractures had 1.8-, 3.0-, and 4.8-times greater risk of incident fracture, respectively, and women with ≥ 3 previous vertebral fractures have a 9.1-times greater risk of a new vertebral fracture [66]. Importantly, within the first 6 months of treatment, fractures may occur without implying a therapeutic failure [65, 67]. After an early fracture while on therapy, the incidence of second and third fractures is markedly reduced by 80–90%; therefore, the IOF considers a second fragility fracture, not the first, a marker of therapeutic failure [65].
Logically, increases of BMD during treatment should represent a good therapeutic response, but changes in BMD do not always correlate with changes in fracture risk [65, 68, 69]. Biological changes (gain or loss) in BMD are usually small, while the precision error of the measurement of BMD is much larger. But, BMD changes can be reliably detected when considering the least significant change (smallest change in BMD that is beyond the range of error of the machine). The IOF proposes that a decrease in BMD greater than the LSC is an indicator of treatment failure [65].
BTMs may predict fracture risk reduction over time. In a meta-analysis involving 18 clinical trials, antiresorptive therapy associated with a 70% decrease in BTMs conferred a 40% reduction in non-vertebral fracture risk [70].
The inter-laboratory variability and the precision error of BTMs may be significant, even when using the same method [71]. Taking into account variability of BTM measurement, the IOF proposes that a decrease in βCTX less than the least significant change (LSC) at 95% confidence is an indicator of antiresorptive treatment failure. Conversely, the increase in PINP less than the LSC at 95% confidence is an indicator of failure to respond to teriparatide [65].
Non-compliance can lead to treatment failure, and it is likely responsible for a significant number of “non-responders,” especially to oral bisphosphonates [64, 69]. Compliance should be addressed after therapy initiation and regularly at follow-up to detect poor tolerability [72,73,73,74, 76]. Despite the importance of adherence, only 25% or less of patients are adherent 1 year after initiating therapy [75]. Bisphosphonates are associated with an early decrease in the levels of PINP and CTX beyond the least significant change (LSC), so BTM assessment may predict adherence problems. The LSC is estimated as a decrease of more than 38% for PINP and 56% for CTX. If decreases do not exceed the LSC, adherence to treatment must be re-assessed and secondary causes of osteoporosis should be evaluated [63].
When to consider a change in treatment?
Prior to changing therapy, the IOF recommends ensuring good adherence and evaluating secondary causes of osteoporosis. After 1 year of therapy, a change from antiresorptive therapy should be considered in the following circumstances:
-
(1)
The occurrence of two or more incident fragility fractures (fractures of hand, digits, skull, feet, and ankle are not fragility fractures)
-
(2)
The occurrence of a single incident fracture along with either:
-
(a)
The lack of an appropriate reduction in serum βCTX or PINP
-
(b)
A significant decrease in BMD
-
(a)
-
(3)
The lack of a significant decrease in serum βCTX or PINP
-
(4)
A significant decrease in BMD.
A significant decrease in BMD is ≥ 5% at the lumbar spine and ≥ 4% at the proximal femur as measured by two DXA scans. Regarding BTM measurements (using the same assay), a significant decline is 25% from baseline for antiresorptive treatments. A 25% increase in BTMs is considered significant for anabolic agents after 6 months [65].
Long-term security aspects
The most common adverse events observed with oral bisphosphonates occur in the gastrointestinal tract. Acute influenza-like symptoms are commonly seen with intravenous bisphosphonates. All bisphosphonates have warnings or contraindications for use in patients with renal impairment [77]. Oral bisphosphonates are poorly absorbed (< 1%), so the risk of renal injury is higher with intravenous administration, it has been reported following zoledronic acid administration, and this may be related to rapid infusion rates or high dose. The recommended infusion time is at least 15 min with adequate hydration prior to administration [77, 78].
Long-term bisphosphonate or denosumab use is associated with a potential risk of medication-related osteonecrosis of the jaw, which may be related to cumulative high doses given for treatment of malignant disease [77, 79]. The incidence ranged from 1 to 15% in oncologic cohorts but only 0.001 to 0.01% in osteoporosis cohorts [80]. Risk factors include GC use, maxillary or mandibular bone surgery, poor oral hygiene, periodontal disease, diabetes mellitus, dental implants, suppuration, and dental extraction [79, 81]. Discussing this risk with patients and completing dental treatments 2 weeks before starting antiresorptive agents is advised. Good oral hygiene, the use of antibiotics post-procedure, mouth rinsing and appropriate wound closure following tooth extraction may reduce the risk [79, 81].
Atypical fractures of the femur (subtrochanteric or femoral diaphysis) are potentially related to the cumulative dose and have been reported with long-term bisphosphonate use and denosumab [4, 82].
The age-adjusted incidence rates for atypical femoral are 1.78/100,000 per year with a 2-year exposure or less; this increases to 113/100,000 per year with exposure of 8–9.9 years [83]. Receiving antiresorptive drugs in addition to bisphosphonates, GC use, and proton-pump inhibitors use increase the risk of atypical fracture. Some genetic susceptibilities have also been reported [83,84,85].
When to stop treatment
Bisphosphonates accumulate in bone resulting in a “residual effect” differing for each bisphosphonate. Effects on bone remodeling are preserved for months to years without therapy [13, 86]. A temporary suspension or “holiday” is common practice assuming the benefit of discontinuing treatment exceeds the risk of new fractures.
Treatment holidays reduce the risks of atypical fractures, but fragility fracture risk increases with drug holiday duration [87,88,89]. Drug holiday recommendations must consider the varied residual effects of each bisphosphonate. After 3–5 years of bisphosphonate therapy (3 years of zoledronic acid and 5 years of alendronate or risedronate), drug holidays are reasonable considerations [90,91,92]. Evidence-based guidance on the appropriate duration of holidays is lacking, but 2 to 3 years seems reasonable [90]. When offering a risedronate holiday, a shorter duration is preferred because 1 year or greater is associated with a significant loss of protection [93]. Holiday duration must be individualized weighing each overall fracture risk, the BMD and BTMs.
A theoretical holiday period applies only to bisphosphonates and no other therapies, like teriparatide and denosumab. Both act via different mechanisms and effects are rapidly reversed after discontinuation [94, 95]. Teriparatide should always be followed by antiresorptive treatment to prevent rapid loss of bone mass [4]. Several case reports and series suggest increased bone loss and rebound vertebral fractures after denosumab discontinuation [96, 97]. Denosumab discontinuation should be carefully planned, and bisphosphonate administration following denosumab cessation may inhibit rapid BMD loss [95, 97].
A working group from the European Calcified Tissue Society (ECTS) proposes re-evaluation after 5 years of denosumab treatment. In individuals with low fracture risk and increased BMD, cessation of denosumab followed by bisphosphonate therapy may be safe. Individuals still considered high-risk may continue denosumab for up to 10 years followed by a single infusion of zoledronic acid or one or more years of oral bisphosphonates. High-risk individuals wishing to stop denosumab after 5 years may be offered an additional 5 years of oral or 3 years of intravenous bisphosphonates [95].
Conclusions
Osteoporosis is a disabling disease and a serious health problem worldwide. Diagnosis in advanced stages is common, usually after fractures have occurred often reducing the quality of life indefinitely. Clinicians should focus on prevention and early recognition of risk factors leading to osteoporosis. A multidisciplinary approach is essential to prevent, treat, and rehabilitate fractures after occurrence [98].
The presence of a diminished bone mass measured by DXA is useful in predicting fracture risk, but adding clinical risk factors to DXA measurements, as used in the FRAX, improves overall risk predictions. Evaluation of secondary causes of osteoporosis helps ensure therapeutic responses. Vitamin D deficiency leads to poor treatment efficacy, and normal serum values are important for bone health. Values above 32 ng/ml (80 nmol/L) help reduce fracture risk in both sexes. Adequate calcium and vitamin D intake are vital in osteoporosis prevention.
Therapy should be individualized to promote adherence. Bisphosphonates are the most commonly prescribed agents, so awareness of the effectiveness and benefits of each drug is important. Choosing therapy to match patient comfort (route of administration, frequency, etc.) and associated comorbidities can mitigate risks of therapeutic failure. When confronting chronic kidney disease and/or gastrointestinal comorbidities, denosumab may offer therapeutic advantages. Combination therapy for severe and/or refractory osteoporosis consists of simultaneous or sequential administration of both anabolic and antiresorptive agents. Finally, clear guidance on stopping osteoporosis treatment is lacking, so providers must weigh the individualized risks of new fractures against treatment duration.
References
Kanis JA (2007) WHO Technical Report, University of Sheffield, UK, p 66
World Health Organization (1994) Assessment of fracture risk and its application to screening for postmenopausal osteoporosis: report of a WHO study group [meeting held in Rome from 22 to 25 June 1992]
NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy (2001) Osteoporosis prevention, diagnosis, and therapy. JAMA 285(6):785–795
Camacho PM, Petak SM, Brinkley N, Clarke BL, Harris ST, Hurley DL et al (2016) AACE/ACE Guidelines-American Association of Clinical Endocrinologists and American College of endocrinology clinical practice guidelines for diagnosis and treatment of postmenopausal osteoporosis. Endocr Pract 22(4):1–42
Ballane G, Cauley JA, Luckey MM, Fuleihan GEH (2014) Secular trends in hip fractures worldwide: opposing trends East versus West. J Bone Miner Res 29(8):1745–1755
International Society for Clinical Densitometry (ISCD). Official Positions of the ISCD as updated in 2015. http://www.iscd.org/official-positions/2013-iscd-official-positions-adult/. Updated: August 15, 2013 (accedido el 14 de septiembre del 2018)
Black DM, Rosen CJ (2016) Postmenopausal osteoporosis. N Engl J Med 374(3):254–262
Siris ES, Adler R, Bilezikian J, Bolognese M, Dawson-Hughes B, Favus MJ, Harris ST, Jan de Beur SM, Khosla S, Lane NE, Lindsay R, Nana AD, Orwoll ES, Saag K, Silverman S, Watts NB (2014) The clinical diagnosis of osteoporosis: a position statement from the National Bone Health Alliance Working Group. Osteoporos Int 25(5):1439–1443
Lewiecki EM (2018) Osteoporosis: clinical evaluation. [Updated 2018 Apr 23] In: De Groot LJ, Chrousos G, Dungan K et al (eds) Endotext. South Dartmouth: MDText.com, Inc. https://www.ncbi.nlm.nih.gov/books/NBK279049/
Sanfélix-Genovés J, Catalá-López F, Sanfélix-Gimeno G, Hurtado I, Baixauli C, Peiró S (2014) Variabilidad en las recomendaciones para el abordaje clínico de la osteoporosis. Med Clin (Barc) 142(1):15–22
Lewiecki EM (2012) To treat or not to treat: reducing fracture risk in postmenopausal women. Female Patient 37:11–16
Compston J, Cooper A, Cooper C, Gittoes N, Gregson C, Harvey N et al (2017) UK clinical guideline for the prevention and treatment of osteoporosis. Arch Osteoporos 12(1):43
Cosman F, De Beur SJ, LeBoff MS, Lewiecki EM, Tanner B, Randall S et al (2014) Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int 25(10):2359–2381
Compston J, Bowring C, Cooper A, Cooper C, Davies C, Francis R, Kanis JA, Marsh D, McCloskey E, Reid DM, Selby P, National Osteoporosis Guideline Group (2013) Diagnosis and management of osteoporosis in postmenopausal women and older men in the UK: National Osteoporosis Guideline Group (NOGG) update 2013. Maturitas 75(4):392–396
North American Menopause Society (2010) NAMS continuing medical education activity; management of osteoporosis in postmenopausal women: 2010 position statement. Menopause 17(1):23–56
Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, Murad MH, Weaver CM, Endocrine Society (2011) Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocr Metab 96(7):1911–1930
Moreira MLM, de Paula FJA (2017) What value is there in assessing postmenopausal women for vitamin D deficiency? Rev Bras Ginecol Obstet 39(11):585–586
Dawson-Hughes B, Mithal A, Bonjour JP, Boonen S, Burckhardt P, Fuleihan GH et al (2010) IOF position statement: vitamin D recommendations for older adults. Osteoporos Int 21(7):1151–1154
Del Valle HB, Yaktine AL, Taylor CL, Ross AC (eds) (2011) Dietary reference intakes for calcium and vitamin D. National Academies Press
Rizzoli R (2011) Bisphosphonates for post-menopausal osteoporosis: are they all the same? QJM-Int J Med 104(4):281–300
Kanis JA, McCloskey EV, Johansson H, Cooper C, Rizzoli R, Reginster JY (2013) European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int 24(1):23–57
Body JJ, Bergmann P, Boonen S, Boutsen Y, Devogelaer JP, Goemaere S, Kaufman JM, Rozenberg S, Reginster JY (2010) Evidence-based guidelines for the pharmacological treatment of postmenopausal osteoporosis: a consensus document by the Belgian Bone Club. Osteoporos Int 21(10):1657–1680
Papaioannou A, Morin S, Cheung AM, Atkinson S, Brown JP, Feldman S (2010) Collab; Scientific Advisory Council of Osteoporosis Canada. 2010 clinical practice guidelines for the diagnosis and management of osteoporosis in Canada: summary. CMAJ 182(17):1864–1873
Silverman S, Christiansen C (2012) Individualizing osteoporosis therapy. Osteoporos Int 23(3):797–809
Cosman F, Miller PD, Williams GC, Hattersley G, Hu MY, Valter I, Fitzpatrick LA, Riis BJ, Christiansen C, Bilezikian JP, Black D (2017) Eighteen months of treatment with subcutaneous abaloparatide followed by 6 months of treatment with alendronate in postmenopausal women with osteoporosis: results of the ACTIVExtend trial. Mayo Clin Proc 92(2):200–210
Miller PD, Hattersley G, Riis BJ, Williams GC, Lau E, Russo LA, Alexandersen P, Zerbini CAF, Hu MY, Harris AG, Fitzpatrick LA, Cosman F, Christiansen C, for the ACTIVE Study Investigators (2016) Effect of abaloparatide vs placebo on new vertebral fractures in postmenopausal women with osteoporosis: a randomized clinical trial. JAMA 316(7):722–733
Saag KG, Wagman RB, Geusens P, Adachi JD, Messina OD, Emkey R, Chapurlat R, Wang A, Pannacciulli N, Lems WF (2018) Denosumab versus risedronate in glucocorticoid-induced osteoporosis: a multicentre, randomised, double-blind, active-controlled, double-dummy, non-inferiority study. Lancet Diabetes Endocrinol 6(6):445–454. https://doi.org/10.1016/S2213-8587(18)30075-5
Mendoza N, Sánchez-Borrego R, Villero J, Baró F, Calaf J, Cancelo MJ, Coronado P, Estévez A, Fernández-Moya JM, González S, Llaneza P, Neyro JL, del Pino J, Rodríguez E, Ruiz E, Cano A, Spanish Menopause Society (2013) 2013 up-date of the consensus statement of the Spanish menopause society on postmenopausal osteoporosis. Maturitas 76(1):99–107
McClung MR, Grauer A, Boonen S, Bolognese MA, Brown JP, Diez-Perez A et al (2014) Romosozumab in postmenopausal women with low bone mineral density. N Engl J Med 370(5):412–420
Saag KG, Petersen J, Brandi ML, Karaplis AC, Lorentzon M, Thomas T, Maddox J, Fan M, Meisner PD, Grauer A (2017) Romosozumab or alendronate for fracture prevention in women with osteoporosis. N Engl J Med 377(15):1417–1427
Zerbini CAF, McClung MR (2013) Odanacatib in postmenopausal women with low bone mineral density: a review of current clinical evidence. Ther Adv Musculoskelet Dis 5(4):199–209. https://doi.org/10.1177/1759720X13490860
Bone HG, McClung MR, Roux C, Recker RR, Eisman JA, Verbruggen N, Hustad CM, DaSilva C, Santora AC, Ince BA (2010) Odanacatib, a cathepsin-K inhibitor for osteoporosis: a two-year study in postmenopausal women with low bone density. J Bone Miner Res 25(5):937–947
Eisman JA, Bone HG, Hosking DJ, McClung MR, Reid IR, Rizzoli R et al (2011) Odanacatib in the treatment of postmenopausal women with low bone mineral density: three-year continued therapy and resolution of effect. J Bone Miner Res 26(2):242–251
Cosman F, Nieves JW, Dempster DW (2017) Treatment sequence matters: anabolic and antiresorptive therapy for osteoporosis. J Bone Miner Res 32(2):198–202
Tsai JN, Uihlein AV, Lee H, Kumbhani R, Siwila-Sackman E, McKay EA et al (2013) Teriparatide and denosumab, alone or combined, in women with postmenopausal osteoporosis: the DATA study randomised trial. Lancet 382(9886):50–56
Nakamura Y, Suzuki T, Kamimura M, Ikegami S, Murakami K, Uchiyama S, Taguchi A, Kato H (2017) Two-year clinical outcome of denosumab treatment alone and in combination with teriparatide in Japanese treatment-naive postmenopausal osteoporotic women. Bone Res 5:16055
Tsai JN, Uihlein AV, Burnett-Bowie SAM, Neer RM, Zhu Y, Derrico N, Lee H, Bouxsein ML, Leder BZ (2015) Comparative effects of teriparatide, denosumab, and combination therapy on peripheral compartmental bone density, microarchitecture, and estimated strength: the DATA-HRpQCT study. J Bone Miner Res 30(1):39–45
Leder BZ, Tsai JN, Uihlein AV, Wallace PM, Lee H, Neer RM, Burnett-Bowie SAM (2015) Denosumab and teriparatide transitions in postmenopausal osteoporosis (the DATA-Switch study): extension of a randomised controlled trial. Lancet 386(9999):1147–1155
Tsai JN, Uihlein AV, Burnett-Bowie SM, Neer RM, Derrico NP, Lee H, Bouxsein ML, Leder BZ (2016) Effects of two years of teriparatide, denosumab, or both on bone microarchitecture and strength (DATA-HRpQCT study). J Clin Endocr Metab 101(5):2023–2030
Wade SW, Strader C, Fitzpatrick LA, Anthony MS, O’Malley CD (2014) Estimating prevalence of osteoporosis: examples from industrialized countries. Arch Osteoporos 9(1):182
Giusti A, Bianchi G (2015) Treatment of primary osteoporosis in men. Clin Interv Aging 10:105
Bliuc D, Nguyen ND, Milch VE, Nguyen TV, Eisman JA, Center JR (2009) Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA 301:513–521
Cawthon PM (2011) Gender differences in osteoporosis and fractures. Clin Orthop Relat Res 469(7):1900–1905
Bonjour JP, Chevalley T, Ferrari S, Rizzoli R (2009) The importance and relevance of peak bone mass in the prevalence of osteoporosis. Salud Publica Mex 51(Suppl 1):S5–S17
Peris P, Martinez-Ferrer A, Monegal A, Martinez de Osaba MJ, Alvarez L, Ros I et al (2008) Aetiology and clinical characteristics of male osteoporosis. Have they changed in the last few years? Clin Exp Rheumatol 26(4):582–588
Kanis JA, Johansson H, Johnell O, Oden A, De Laet C, Eisman JA et al (2005) Alcohol intake as a risk factor for fracture. Osteoporos Int 16:737–742
Zhang X, Yu Z, Yu M, Qu X (2015) Alcohol consumption and hip fracture risk. Osteoporos Int 26(2):531–542
Kanis JA, Johnell O, Odén A, Johansson H, De Laet C, Eisman JA et al (2005) Smoking and fracture risk: a meta-analysis. Osteoporos Int 16(2):155–162
Adler RA (2014) Osteoporosis in men: a review. Bone Res 2:14001
Ebeling PR (2014) Osteoporosis in men: why change needs to happen. International Osteoporosis Foundation
Orwoll ES, Miller PD, Adachi JD, Brown J, Adler RA, Kendler D, Bucci-Rechtweg C, Readie A, Mesenbrink P, Weinstein RS (2010) Efficacy and safety of a once-yearly i.v. infusion of zoledronic acid 5 mg versus a once-weekly 70-mg oral alendronate in the treatment of male osteoporosis: a randomized, multicenter, double-blind, active-controlled study. Journal of bone and mineral research. J Bone Miner Res 25:2239–2250
Orwoll E, Ettinger M, Weiss S, Miller P, Kendler D, Graham J, Adami S, Weber K, Lorenc R, Pietschmann P, Vandormael K, Lombardi A (2000) Alendronate for the treatment of osteoporosis in men. New Engl J Med 343:604–610
Willson T, Nelson SD, Newbold J, Nelson RE, LaFleur J (2015) The clinical epidemiology of male osteoporosis: a review of the recent literature. J Clin Epidemiol 7:65
McDonough AK, Curtis JR, Saag KG (2008) The epidemiology of glucocorticoid-associated adverse events. Curr Opin Rheumatol 20(2):131–137
Buckley L, Guyatt G, Fink HA, Cannon M, Grossman J, Hansen KE, Humphrey MB, Lane NE, Magrey M, Miller M, Morrison L, Rao M, Byun Robinson A, Saha S, Wolver S, Bannuru RR, Vaysbrot E, Osani M, Turgunbaev M, Miller AS, McAlindon T (2017) 2017 American College of Rheumatology Guideline for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res 69(8):1095–1110
Rossini M, Viapiana O, Vitiello M, Malavolta N, La Montagna G, Bongi SM et al (2017) Prevalence and incidence of osteoporotic fractures in patients on long-term glucocorticoid treatment for rheumatic diseases: the Glucocorticoid Induced OsTeoporosis TOol (GIOTTO) study. Reumatismo 69(1):30–39
Mitra R (2011) Adverse effects of corticosteroids on bone metabolism: a review. PM R 3(5):466–471
Lekamwasam S, Adachi JD, Agnusdei D, Bilezikian J, Boonen S, Borgström F et al (2012) A framework for the development of guidelines for the management of glucocorticoid-induced osteoporosis. Osteoporos Int 23(9):2257–2276
Van Staa TP, Leufkens HGM, Abenhaim L, Zhang B, Cooper C (2000) Oral corticosteroids and fracture risk: relationship to daily and cumulative doses. Rheumatology 39(12):1383–1389
Geusens P, Goemaere S, Pannacciulli N, Lane NE, Lespessailles E, Messina OD, Chapurlat R, Yin X, Wagman R, van den Bergh JPW (2018) Effect of denosumab versus risedronate on cortical and trabecular bone microarchitecture by high resolution peripheral quantitative computed tomography (HR-pQCT) in glucocorticoid-treated individuals. FRI-0906 Friday, September 28, 2018-ASBMR 2018 Annual Meeting, Montreal
Vasikaran S, Eastell R, Bruyère O, Foldes AJ, Garnero P, Griesmacher A et al (2011) Markers of bone turnover for the prediction of fracture risk and monitoring of osteoporosis treatment: a need for international reference standards. Osteoporos Int 22(2):391–420
Romero Barco CM, Manrique Arija S, Rodríguez Pérez M (2012) Marcadores bioquímicos en osteoporosis. Utilidad en la práctica clínica. Reumatol Clin 8(3):149–152
Diez-Perez A, Naylor KE, Abrahamsen B, Agnusdei D, Brandi ML, Cooper C et al (2017) International osteoporosis foundation and european calcified tissue society working group. Recommendations for the screening of adherence to oral bisphosphonates. Osteoporos Int 28(3):767–774
Carey JJ (2005) What is a ‘failure’ of bisphosphonate therapy for osteoporosis? Cleve Clin J Med 72(11):1033–1039
Diez-Perez A, Adachi JD, Agnusdei D, Bilezikian JP, Compston JE, Cummings SR et al (2012) Treatment failure in osteoporosis. Osteoporos Int 23(12):2769–2774
Gehlbach S, Saag KG, Adachi JD, Hooven FH, Flahive J, Boonen S, Chapurlat RD, Compston JE, Cooper C, Díez-Perez A, Greenspan SL, LaCroix AZ, Netelenbos JC, Pfeilschifter J, Rossini M, Roux C, Sambrook PN, Silverman S, Siris ES, Watts NB, Lindsay R (2012) Previous fractures at multiple sites increase the risk for subsequent fractures: the global longitudinal study of osteoporosis in women. J Bone Miner Res 27(3):645–653
Díez-Pérez A, Olmos JM, Nogués X, Sosa M, Díaz-Curiel M, Pérez-Castrillón JL, Pérez-Cano R, Muñoz-Torres M, Torrijos A, Jodar E, del Rio L, Caeiro-Rey JR, Farrerons J, Vila J, Arnaud C, González-Macías J (2012) Risk factors for prediction of inadequate response to antiresorptives. J Bone Moner Res 27(4):817–824
Pasco JA, Seeman E, Henry MJ, Merriman EN, Nicholson GC, Kotowicz MA (2006) The population burden of fractures originates in women with osteopenia, not osteoporosis. Osteoporos Int 17(9):1404–1409
Vondracek SF, Minne P, McDermott MT (2008) Clinical challenges in the management of osteoporosis. Clin Interv Aging 3(2):315–329
Hochberg MC, Greenspan S, Wasnich RD, Miller P, Thompson DE, Ross PD (2002) Changes in bone density and turnover explain the reductions in incidence of nonvertebral fractures that occur during treatment with antiresorptive agents. J Clin Endocr Metab 87(4):1586–1592
Hlaing TT, Compston JE (2014) Biochemical markers of bone turnover–uses and limitations. Ann Clin Biochem 51(2):189–202
Chapurlat RD, Palermo L, Ramsay P, Cummings SR (2005) Risk of fracture among women who lose bone density during treatment with alendronate. The fracture intervention trial. Osteoporos Int 16(7):842–848
Caro JJ, Ishak KJ, Huybrechts KF, Raggio G, Naujoks C (2004) The impact of compliance with osteoporosis therapy on fracture rates in actual practice. Osteoporos Int 15(12):1003–1008
Siris E, Rosen CJ, Harris ST, Abbott TA, Barr CE, Silverman S (2005, April) Adherence to bisphosphonate therapy: relationship to bone fractures at 24 months in women with postmenopausal osteoporosis. In National Osteoporosis Foundation 6th International Symposium on Osteoporosis, pp 6–9
McCombs JS, Thiebaud P, McLaughlin-Miley C, Shi J (2004) Compliance with drug therapies for the treatment and prevention of osteoporosis. Maturitas 48:271–287
Becker CB (2016) Osteoporosis: managing patients who fracture on osteoporosis treatment. Meet-the-professor: endocrine case management. https://doi.org/10.1210/MTP5.9781943550043.ch14
Boonen S, Ferrari S, Miller PD, Eriksen EF, Sambrook PN, Compston J, Reid IR, Vanderschueren D, Cosman F (2012) Postmenopausal osteoporosis treatment with antiresorptives: effects of discontinuation or long-term continuation on bone turnover and fracture risk—a perspective. J Bone Miner Res 27(5):963–974
Eriksen EF, Díez-Pérez A, Boonen S (2014) Update on long-term treatment with bisphosphonates for postmenopausal osteoporosis: a systematic review. Bone 58:126–135
Yoneda T, Hagino H, Sugimoto T, Ohta H, Takahashi S, Soen S et al (2017) Anti-resorptive agent-related osteonecrosis of the jaw: Position Paper 2017 of the Japanese Allied Committee on Osteonecrosis of the Jaw. J Bone Miner Metab 35(1):6–19. https://doi.org/10.1007/s00774-016-0810-7
Svejda B, Muschitz C, Gruber R, Brandtner C, Svejda C, Gasser RW, Santler G, Dimai HP (2016) Position paper on medication-related osteonecrosis of the jaw (MRONJ). Wien Med Wochenschr 166(1–2):68–74
Khan AA, Morrison A, Hanley DA, Felsenberg D, McCauley LK (2015) Diagnosis and management of osteonecrosis of the jaw: a systematic review and international consensus. J Bone Min Res 30:3–23
Adler RA, Fuleihan G, Bauer DC, Camacho PM, Clarke BL et al (2016) Managing osteoporosis in patients on long-term bisphosphonate treatment: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 31:16–35
Dell RM, Adams AL, Greene DF, Funahashi TD, Silverman SL, Eisemon EO et al (2012) Incidence of atypical nontraumatic diaphyseal fractures of the femur. J Bone Min Res 27:2544–2550
Shane E, Burr D, Abrahamsen B, Adler RA, Brown TD, Cheung AM, Cosman F, Curtis JR, Dell R, Dempster DW, Ebeling PR, Einhorn TA, Genant HK, Geusens P, Klaushofer K, Lane JM, McKiernan F, McKinney R, Ng A, Nieves J, O'Keefe R, Papapoulos S, Howe TS, van der Meulen MCH, Weinstein RS, Whyte MP (2014) Atypical subtrochanteric and diaphyseal femoral fractures: second report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 29(1):1–23
Nguyen HH, van de Laarschot DM, Verkerk A, Milat F, Zillikens MC, Ebeling PR (2018) Genetic risk factors for atypical femoral fractures (AFFs): a systematic review. JBMR Plus 2:1–11
Xu XL, Gou WL, Wang AY, Wang Y, Guo QY, Lu Q, Lu SB, Peng J (2013) Basic research and clinical applications of bisphosphonates in bone disease: what have we learned over the last 40 years? J Transl Med 11(1):303
Compston JE, Bilezikian JP (2012) Bisphosphonate therapy for osteoporosis: the long and short of it. J Bone Miner Res 27(2):240–242
Curtis JR, Chen R, Li Z, Arora T, Saag KG, Wright NC et al (2018) The impact of the duration of bisphosphonate drug holidays on hip fracture rates. Ann Rheum Dis 77:58
Anagnostis P, Paschou S, Mintziori G, Lambrinoudaki I, Goulis D (2017) Efficacy and safety of bisphosphonate discontinuation in postmenopausal osteoporosis: a systematic review. Endocr Abstr. https://doi.org/10.1530/endoabs.49.EP214
Anagnostis P, Paschou SA, Mintziori G, Ceausu I, Depypere H et al (2017) Drug holidays from bisphosphonates and denosumab in postmenopausal osteoporosis: EMAS position statement. Maturitas 10:23–30
Ott SM (2011) What is the optimal duration of bisphosphonate therapy? Cleve Clin J Med 78(9):619–630
Briot K, Trémollières F, Thomas T, Roux C (2007) How long should patients take medications for postmenopausal osteoporosis? Joint Bone Spine 74(1):24–31
Black DM, Bauer DC, Schwartz AV, Cummings SR, Rosen CJ (2012) Continuing bisphosphonate treatment for osteoporosis—for whom and for how long? N Engl J Med 366(22):2051–2053
Leder BZ, Tsai JN, Jiang LA, Lee H (2017) Importance of prompt antiresorptive therapy in postmenopausal women discontinuing teriparatide or denosumab: the Denosumab and Teriparatide Follow-up study (DATA-Follow-up). Bone 98:54–58
Tsourdi E, Langdahl B, Cohen-Solal M, Aubry-Rozier B, Eriksen EF (2017) Discontinuation of denosumab therapy for osteoporosis: a systematic review and position statement by ECTS. Bone 105:11–17
Anastasilakis AD, Polyzos SA, Makras P, Aubry-Rozier B, Kaouri S, Lamy O (2017) Clinical features of 24 patients with rebound-associated vertebral fractures after denosumab discontinuation: systematic review and additional cases. J Bone Miner Res 32:1291–1296
Gonzalez Rodriguez E, Delphine St, Aubry-Rozier B, Olivier L (2018) Thirty-one men and women with 145 spontaneous vertebral fractures after denosumab discontinuation: a single center observational study. In 20th European Congress of Endocrinology. Endocrine Abstracts. https://doi.org/10.1530/endoabs.56.GP59
Albergaria BH, Chalem M, Clark P, Messina OD, Pereira RMR, Vidal LF (2018) Consensus statement: osteoporosis prevention and treatment in Latin America—current structure and future directions. Arch Osteoporos 13(1):90
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Vidal, M., Thibodaux, R.J., Neira, L.F.V. et al. Osteoporosis: a clinical and pharmacological update. Clin Rheumatol 38, 385–395 (2019). https://doi.org/10.1007/s10067-018-4370-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-018-4370-1