Abstract
The etiology of systemic sclerosis (SSc) remains unknown; however, several occupational and environmental factors have been implicated. Our objective was to perform a meta-analysis of all studies published on SSc associated with occupational and environmental exposure. The review was undertaken by means of MEDLINE and SCOPUS from 1960 to 2014 and using the terms: “systemic,” “scleroderma,” or “systemic sclerosis/chemically induced” [MesH]. The Newcastle-Ottawa Scale was used for the qualifying assessment. The inverse variance-weighted method was performed. The meta-analysis of silica exposure included 15 case-control studies [overall OR 2.81 (95%CI 1.86–4.23; p < 0.001)] and 4 cohort studies [overall RR 17.52 (95%CI 5.98–51.37; p < 0.001)]; the meta-analysis of solvents exposure included 13 case-control studies (overall OR 2.00 [95%CI 1.32–3.02; p = 0.001); the meta-analysis of breast implants exposure included 4 case-control studies (overall OR 1.68 (95%CI 1.65–1.71; p < 0.001)) and 6 cohort studies (overall RR 2.13 (95%CI 0.86–5.27; p = 0.10)); the meta-analysis of epoxy resins exposure included 4 case-control studies (overall OR 2.97 (95%CI 2.31–3.83; p < 0.001)), the meta-analysis of pesticides exposure included 3 case-control studies (overall OR 1.02 (95%CI 0.78–1.32; p = 0.90)) and, finally, the meta-analysis of welding fumes exposure included 4 studies (overall OR 1.29 (95%CI 0.44–3.74; p = 0.64)). Not enough studies citing risks related to hair dyes have been published to perform an accurate meta-analysis. Silica and solvents were the two most likely substances related to the pathogenesis of SSc. While silica is involved in particular jobs, solvents are widespread and more people are at risk of having incidental contact with them.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Systemic sclerosis (SSc) is an autoimmune systemic disease of unknown origin. Although several different etiologic agents have been postulated, chemicals have been the most suggested etiologic agents. Among them, silica [1–27], solvents [5, 6, 17, 19, 21, 22, 24–26, 28–44], silicone breast implants [13, 16, 45–59], epoxy resins [19, 22, 25, 26, 60], welding fumes [21, 22, 25, 41], pesticides [17, 21, 25, 26], and hair dyes [21, 34, 61] have been suspected to be related to SSc development. Besides, scleroderma-like disorders related to other chemical agents such as vinyl chloride [62–64] and toxic oil syndrome [65] have also been reported, and isolated case reports suggesting a chemical-related origin have been published since the early twentieth century. Interestingly, more consistent studies have been published in the last three decades, including case-control studies, cohort studies, and meta-analyses.
Various occupational and environmental exposures are ubiquitous and it is almost impossible to avoid even a single contact. Several occupations are known to be at a higher risk, as workers have intense contact with these substances (Table 1). Accordingly, several reviews [66–81] and meta-analyses have been published so far [4, 30–32, 45–49], a few of which are systematic although incomplete and methodologically often highly debatable. The aim of our study was to perform a systematic review and meta-analysis of all these toxic substances related to scleroderma development.
Materials and methods
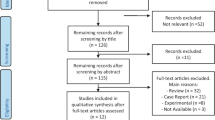
The search was performed by two independent investigators (R-R.M. and M.R.) through the MEDLINE and SCOPUS databases between January 1960 and November 2014, by using the terms: “systemic,” “scleroderma,” or “systemic sclerosis/ chemically induced” [MesH]. The search was completed by the bibliography review of every paper selected for full-text examination. No restriction related to language was performed initially but eventually a few papers from Eastern Europe and Asia were excluded from the analysis. The first search found 515 articles, of which 447 articles were non-selected assessing the title and/or abstract. Sixty-eight articles were finally selected for full-text review (Fig. 1).
In order to perform the meta-analysis, we selected those case-control studies (risk factor described as odds ratio or OR) and cohort studies (risk factor described as risk ratio or RR) separately. Those studies that belonged to other cohorts included in the same meta-analysis were rejected in order to avoid a bias due to data over expression. In such cases, the latest and larger study was selected. In the case of the studies by Bovenzi et al. [5, 6], data belonged to different periods of time, so there was no over expression of data. Eventually, 19 articles were included in the meta-analysis of silica (3 mortality studies were rejected) [5–26], 13 in the meta-analysis of solvents (1 mortality study was rejected) [5, 6, 19, 21, 22, 25, 26, 33, 34, 37–39, 41], 10 in the meta-analysis of breast implants [13, 16, 50–54, 56–59], 4 in the meta-analysis of epoxy resins [19, 22, 25, 26], 4 in the meta-analysis of pesticides [17, 21, 25, 26], and 4 in the meta-analysis of welding fumes [21, 22, 25, 41] (Fig. 1). Not enough studies citing risks related to hair dyes have been published during the study period to perform an accurate meta-analysis [21, 34].
Quality assessment was performed by means of the Newcastle-Ottawa Scale for observational studies [82].
Statistical analysis
Categorical variables were described as absolute number and percentage. Continuous variables were described as mean and standard deviation. A few studies did not publish risk data, but they certainly published raw data enough to calculate the risk factor, so they were included. In those studies reflecting the OR or RR without confidence interval, it was estimated by the formula OR or RR ± 1.96 (OR or RR/√N). The study by Sverdrup et al. [43] showed confidence interval without RR, so it was estimated by the formula √ lower RR x upper RR [83, 84]. In the study by Thompson et al. [24], there was a typographical error in the case of silica exposure, and the RR was recalculated in accordance.
The inverse variance-weighted method was initially performed by using the fixed effects model (Fig. 2). Thereafter, between-study variability was measured by Tau2 parameter and, when confirmed (p ≤ 0.05), the analysis was completed by using the random effects model. In fact, random effects model assumes that there is an underlying effect for each study which varies randomly across studies, with the resulting overall effect an average of these [84].
Publication bias was ruled out by means of the Begg’s method (tau = −0.121 p = 0.547 for silica (case-control) and tau = 0.001 p = 1.000 for silica (cohorts); tau = 0.111 p = 0.677 for solvents; tau = −0.333 p = 0.497 for breast implants (case-control) and tau = 0.467 p = 0.188 for breast implants (cohorts); tau = −0.200 p = 0.624 for epoxy resins; tau = 0.333 p = 0.602 for pesticides; tau = 0.001 p = 1.000 for welding fumes) [85]. Statistical analysis was performed by SPSS 15.0.
Results
All studies included in the present meta-analysis were over 5 points in the Newcastle-Ottawa Scale, reflecting the quality of these papers.
Nineteen studies were included in the meta-analysis of silica (Table 2) [5–26], 4 cohort studies and 15 case-control studies. Three mortality studies were not included (Walsh et al. [14], Calvert et al. [15] and Gold et al. [17]). The 15 case-control studies reported data from 1336 patients (2 studies by Silman et al. [19] were based on the same cases but different controls), 835 (62.5%) were female and 501 (37.5%) were male. The mid-cohort years of every study ranged from 1960 to 2006 and the OR ranged from 0.9 (95%CI 0.2–3.2) to 21 (95%CI 4.7–101). The overall OR was 2.81 (95%CI 1.86–4.23; p < 0.001) by means of the random effects model. The overall OR for male gender was 3.06 (95%CI 1.90–4.91; p < 0.001) by the random effects model and the overall OR for female gender was 2.10 (95%CI 1.24–3.55; p = 0.005) by the fixed effects model. There were 4 cohort studies with risks described as RR. They showed data from 247,563 patients. The mid-cohort years ranged from 1977 to 1995 and RR ranged from 7.8 (95%CI 6.5–9.5) to 37 (95%CI 11.9–86.3). The overall RR was 17.52 (95%CI 5.98–51.37; p < 0.001) by means of the random effects model. There were not enough data to analyze by gender.
Thirteen studies were included in the meta-analysis of solvents (Table 3) [5, 6, 19, 21, 22, 25, 26, 33, 34, 37–39, 41]. All of them were case-control studies. The study by Laing et al. [35] belonged to the same cohort than the study by Lacey et al. [34], so we decided to include only the one with the largest number of patients. Czirjak et al. published two case-control studies [36, 39] which belonged to the same cohort, so the oldest was rejected. No study was excluded because of the quality assessment. These 13 case-control studies reported data from 2107 patients. The mid-cohort years ranged from 1983 to 2006 and the OR ranged from 0.56 to 9.28. The overall OR was 2.00 (95%CI 1.32–3.02; p = 0.001) by means of the random effects model. The OR for male gender was 2.40 (95%CI 1.44–4.01; p < 0.001) and for female gender 2.01 (95%CI 1.66–2.44; p < 0.001) with the fixed effects model. There were 2 more cohort studies describing RR of solvents as a risk factor for SSc, but they were not included in the present meta-analysis due to the limited representativeness of the sample [43, 44].
Ten studies were included in the meta-analysis of silicone breast implants (Table 4) [13, 16, 50–54, 56–59]. There were 7 cohort studies and 5 case-control studies. No study was excluded because of the quality assessment. Out of the 7 cohort studies the one by Fryzek et al. [55] was excluded since it showed data also reported in that reported by Kjoller et al. [53]. Therefore, the final 6 cohort studies reported data from 23,139 patients. All of them were women. The mid-cohort years ranged from 1978 to 1993 and RR ranged from 0.5 to 27.7. The overall RR was estimated in 2.13 (95%CI 0.86–5.27; p = 0.10) by means of the random effects model. Out of the 5 case-control studies, we excluded the one by Goldman et al. [54] since the calculated OR for SSc was zero because no cases of SSC were diagnosed among the 150 patients with breast implants of a representative sample of 721 women with a wide range of connective tissue diseases. Therefore, the final 4 case-control studies reported data from 1615 patients (1551 females and 64 males). The mid-cohort years ranged from 1988 to 1991 and the OR ranged from 1.01 to 1.68. The overall OR was 1.68 (95%CI 1.65–1.71; p < 0.001) by means of the fixed effects model.
Four studies were included in the meta-analysis of epoxy resins [19, 22, 25, 26], but the study by Silman et al. [19] included two case-control studies with the same cases but different controls (Table 5). No study was excluded because of the quality assessment. These 4 studies reported data from 264 patients, 147(55.7%) of them were women. The mid-cohort years ranged from 1984 to 2006 and the OR ranged from 0.5 to 4.24. The overall OR was 2.97 (95%CI 2.31–3.83; p < 0.001) by means of the fixed effects model. The OR for males was 2.92 (95%CI 2.26–3.78) and for females 1 (95%CI 0.02–12.72).
Three studies were included in the meta-analysis of pesticides (Table 5) [17, 21, 25, 26]. No study was excluded because of the quality assessment. These 3 studies reported data from 264 patients, 147(55.7%) of them were women. The mid-cohort years ranged from 1984 to 2006 and the OR ranged from 1 to 3.06. The overall OR was 1.02 (95%CI 0.78–1.32; p = 0.90) by means of the fixed effects model. The OR for males was 1.02 (95%CI 0.79–1.33; p = 0.86) and for females 3.06 (95%CI 0.22–43.34).
Four studies were included in the meta-analysis of welding fumes (Table 5) [21, 22, 25, 41]. All of them were case-control studies. No study was excluded because of the quality assessment. The study by Kutting et al. [41] reported OR for genders but did not report overall OR. However, it could be easily calculated. Inversely, the study by Maitre et al. [21] reported overall OR and we calculated those OR by genders. These 4 studies reported data from 448 patients, 371 (82.81%) of them were women. The mid-cohort years ranged from 1997 to 2006 and the OR ranged from 0.5 to 3.74. The overall OR was 1.29 (95%CI 0.44–3.74 p = 0.64) by means of the random effects model. The OR for males was 5.87 (95%CI 2.49–13.86) and for females 1.52 (95%CI 0.36–6.49).
Discussion
The present study constitutes the largest meta-analysis ever done before in occupational and environmental SSc. To date, only 8 meta-analyses had previously focused on this matter in medical literature: 1 on silica-related SSc, 3 on solvent-related SSc, 4 on silicone breast implant-related SSc, and none assessing epoxy resins nor pesticides nor welding fumes-related SSc. Differences between overall RR and OR were notorious, probably due to cohort studies just including a relatively small number of SSc cases and this fact can bias the results. Thus, in our opinion, case-control studies are a more appropriate method to study such low incidental disease.
Silica
Silica was the best and first well-known exposure related to SSc development [1, 2]. Silicon exists primarily in the form of silica dioxide (SiO2) and its three principal crystalline forms: quartz, tridymite, and cristobalite. Symptoms are undistinguishable from those recognized in the classical scleroderma, and autoimmune spectrum is similar as well. Antinuclear antibodies and anti-topoisomerase have been described in the majority of patients [3]. Only 1 meta-analysis has been reported by McCormic et al. [4], including 16 studies of different nature (case-control, cohort and mortality studies), initially evaluated all together and, thereafter, separately. Data were expressed as estimator of RR (CERR) but certainly the source risk was heterogenous. They estimated an overall CERR of 3.2 (95%CI 1.89–5.43) and of 2.24 (95%CI 1.65–3.31) for just the case-control studies. In the present meta-analysis, we showed data from 19 studies, 15 of which were case-control studies with an overall OR of 2.81 (95%CI 1.86–4.23; p < 0.001). The exposure levels differ from one patient to another and these differences were not taken into account in these studies.
Recently, Freire et al. reported clinical peculiarities in those patients with scleroderma exposed to silica [27]. Thus, these patients tended more often to be male and presenting with diffuse subset, topoisomerase antibodies, and interstitial lung disease.
Solvents
There is a very broad spectrum of different solvents potentially involved in the genesis of SSc related to chemical industry. Since its first description in 1957 [28], there have been many case reports and case series described [29]. Symptoms are also similar to those of classical SSc. Antinuclear antibodies with speckled pattern have been described and angiography reveals digital arterial narrowing.
There were 3 prior meta-analyses published in the last decades [30–32]. In 2002, Aryal et al. [30] reported the first systematic review focused on solvent exposure including 7 case-control studies and one cohort study, with an overall RR of 2.91 (95%CI 1.60–5.30). This review was done with only one database and data included were heterogenous, including both OR and RR in the same analysis. In 2007, Kettaneh et al. [31] reported a brilliant meta-analysis assessing 11 case-control studies with an overall OR of 2.41 (95%CI 1.73–3.37). Data were homogeneous and methodology was impeccable. Finally, in 2012, Barragán et al. [32] published the most recent meta-analysis from 4 different databases which included 8 case-control studies with an overall OR of 2.52 (95%CI 1.24–5.14). Among the revised studies, the one by Thompson et al. [24] showed data from different kinds of solvents but there was no category called “any solvent.” In the present meta-analysis, the representative sample of 13 case-control studies was larger and homogeneous, and data were expressed as OR in every study. Our overall OR was OR 2.00 (95%CI 1.32–3.02; p = 0.001), clearly reaffirming the results observed in previous studies. Again, the exposure levels differ from one patient to another and these differences were not taken into account in these studies.
Silicone breast implants
Early after the introduction of this surgical procedure, concern grew about the risk of induction and development of associated connective tissue diseases. In early 60’s, paraffin, silicone, and petroleum jelly injections for augmentation mammoplasty (a technique widely used in Japan at that time) were related to SSc. Presence of antinuclear antibodies were described as well as improvement of the connective tissue disorder after implant removal.
Five meta-analyses [45–49] were reported in the medical literature prior to the present study. Perkins et al. [45] published the first meta-analysis in 1995, showing data from 7 studies (4 case-control and 3 cohort) from 4 databases. They found an overall RR of 0.98 (95%CI 0.57–1.64). However, these authors analyzed all these heterogenous studies together and did not reject any one of them with 0 cases reported. Hochberg et al. [46] published in 1995 a meta-analysis from 3 case-control studies by reviewing just one database (Medline), with an overall OR of 1.04 (95%CI 0.58–1.88). Publication bias was not assessed. Wong et al. [47] published in 1996 a meta-analysis including only 3 case-control studies from 2 databases, with an overall OR of 0.82 (95%CI 0.50–1.35). Whorton et al. [48] published their study in 1997. They did not specify the method for research and publication bias was not assessed. They showed data from 3 case-control studies with an overall OR of 0.82 (95%CI 0.50–1.35). Janowsky et al. [49] published in 2000 the most recent meta-analysis including 5 studies (4 case-control and 1 cohort) from 4 databases. They showed a summary RR of 1.30 (95%CI 0.86–1.96) but they certainly did not analyze homogenous studies. In our meta-analysis, the overall RR for cohort studies was 2.13 (95%CI 0.86–5.27; p = 0.10) and the overall OR for case-control studies was 1.68 (95%CI 1.65–1.71; p < 0.001). Although previous studies could not demonstrate a possible role of silicone breast implants inducing SSc, this possibility cannot be ruled out in our meta-analysis, in which results reached statistical significance when analyzing case-control studies. Another point of discussion would be the fact that the breast augmentation surgery has been changing over time and materials used today differ from those used in the past. They are expected to be safer, but unfortunately, the short number of studies performed and their heterogeneity does not allow identifying this progression over time.
Epoxy resins
These compounds are commonly used in construction. Their association with SSc was first described in 1980 by Yamakage et al. in 6 patients engaged in the polymerization process of epoxy resins, probably due to exposure to bis-(4-amino-3-methyl-cyclohexyl) methane [60]. These patients demonstrated skin sclerosis, telangiectasias, hyperpigmentation, muscle weakness, fatigue, pruritus, total loss of body hair, arthralgias, flexion contractures, diminished vital capacity in pulmonary function tests. Unlike scleroderma, neither Raynaud’s phenomenon nor antinuclear antibodies were detected. Angiography revealed narrowing and irregularity of the lumen in the left ulnar and interosseous arteries.
Since there were no prior systematic reviews on epoxy resins-induced SSc, the present study constitutes the first meta-analysis published to date. An overall OR of 2.97 (95%CI 2.31–3.83; p < 0.001) was found.
Welding fumes, pesticides and hair dyes
Few studies have focused on welding or metallic fumes as a causative agent of SSc. In particular, there were only 4 case-control studies previously published to our knowledge. Since the first detection of pesticides as a potential causative agent in 1996 [26], there have been only three case-control studies and one mortality study. Moreover, three case-control studies have focused on hair dyes as a causative agent to date. Thus, no previous meta-analyses have focused on welding fumes, pesticides or dyes exposure. Interestingly, and despite the low number of studies included, the present study constitutes the first meta-analysis evaluating pesticides and welding fumes-related SSc, with an overall OR of 1.02 (95%CI 0.78–1.32; p = 0.90) for the former and OR of 1.29 (95%CI 0.44–3.74; p = 0.64) for the latter. Conversely, it was considered that there were not enough published studies citing risks related to hair dyes to perform an accurate meta-analysis.
Drugs
A few drugs have been described as a case report or case series as potentially related to SSc or SSc-like development, namely some chemotherapy agents, appetite suppressants and L-5-hydroxytriptophan. Other drugs are reported as suspiciously related and in a few cases there is just an isolated case report so no conclusions can be made. In all these cases, symptoms are similar to those found in SSc, but autoimmunity is absent, so we should assume these cases are scleroderma-like disorders.
Vinyl chloride disease or occupational acro-osteolysis
Polymerized vinyl chloride is used by the plastic industry to produce manufactured goods and it was related to SSc from the beginning mainly to long-term reactor cleaning [62]. In the 1960s, first cases described presented with Raynaud’s phenomenon, acro-osteolysis on X-rays, clubbing, and scleroderma-like skin changes involving the hands, forearms and face, fatigue, artharlgias, myalgias, decreased grip, upper abdominal symptoms, dizziness and impotence and increased perspiration, carpal tunnel syndrome, hepatomegaly or splenomegaly, vasculitic purpura, abnormal liver function tests, thrombocytopenia, and leucopenia [63]. Non-cirrhotic portal hypertension and liver angiosarcoma appeared as late complications. All the patients were males. Lesions healed after withdrawal from vinyl chloride exposure [62]. Unlike scleroderma, calcinosis, telangiectasias, and esophageal, intestinal, cardiac or pulmonary involvement were not detected. Capillaroscopy revealed dilated capillary loops and avascular areas similar to those described in scleroderma. Antinuclear antibodies have been found in these patients [64]. Although there was evidence of similarities to SSc, there were differences in the presence of paresthesia, thrombocytopenia, splenomegaly, reticulocytosis, central nervous system involvement, leukopenia, angiosarcoma of the liver, absence of calcinosis and autoantibodies, so we should assume this entity as a scleroderma-like disorder.
Toxic oil syndrome
A worrisome circumscribed and extremely rare epidemic appeared in Spain in 1981 [65]. In its early stage, it appeared as an atypical pneumonia with fever, myalgia, rash, pruritus, arthralgia, headache, encephalopathy, thrombocytopenia, nausea, abdominal pain, purpura, lymphadenopathy, hepatomegaly, pancreatitis, pericardial effusion cough, shortness of breath, pleuritic chest pain with transudative pleural effusion, hemoptysis, and eosinophilia. The intermediate phase was characterized by myalgia, paraesthesias in lower extremities, pulmonary hypertension, severe thrombocytopenia, lymphocytosis, thromboembolic events, disseminated intravascular coagulation, dark yellow papoles on the skin, edema in the face and limbs. Finally, the late phase was characterized by scleroderma-like skin thickening with hyperpigmentation, Raynaud’s phenomenon, flexion contractures, dysphagia and sicca symptoms, acro-osteolysis, acro-osteosclerosis or osteopenia on X-rays, myalgia, dysesthesia, muscle atrophy, polyneuropathy, difficulty in swallowing, malabsorption, improving pulmonary hypertension. Most patients had positive antinuclear antibodies. Despite evident similarities to SSc, it differed on the presence of rash, pruritus, neuropathy, adenomegalies, eosinophilia, thrombocytopenia, lymphocytosis, so we should also assume this entity is a scleroderma-like disorder. Epidemiologic studies revealed cooking oil from toxic unlabelled plastic containers as the source of the disease. They contained a mixture of rapeseed oil (90%), liquefied aniline, and acetanilide. Initially, denaturalized with aniline and legally introduced in Spain for industrial use, the removal of aniline was attempted but unfortunately part of it was left behind, as well as acetanilide and oleoanilide.
In our opinion, there is enough data to step forward in our research. If we could measure these substances, we would be able to try to correlate these results with disease occurrence and even with clinical features according to the chemical involved. Safe levels of exposure could then be suggested by occupational hygienists and physicians.
As limitations of our study, there is between-study variability so random effects model was chosen for the assessment of risk in the case of silica, solvents, breast implants and welding fumes. All these studies are based on self-reported surveys and it is difficult to quantify—even by experts—the contact of any of these substances. Thus, since the exposure is difficult to quantify, this would be a good opportunity to perform a prospective cohort study based on particular jobs with a known exposure burden. Another limitation is the variable nature of the exposures over time as ALARA (as low as reasonably achievable) exposure levels that are increasingly brought into industry. Moreover, exposures in the non-occupational environment have not been taken into account in those studies.
Conclusion
In conclusion, the present study constitutes the largest meta-analysis ever done to date and concerning occupational and environmental SSc. We hereby reported a significant increased overall OR after exposure to silica, solvents, silicone breast implants, epoxy resins, pesticides, and welding fumes. These data support the suspicion that a chemical might be behind the diagnosis of scleroderma. Moreover, these chemicals can be present in many different occupations but widespread as well in our daily lives. In accordance, more efforts should be done to reveal these compounds in tissue samples of SSc patients and comparing with controls. It is hard work to make recommendations regarding occupations potentially related to SSc but, in our opinion, a change of job might be suggested after diagnosis. There are a few scleroderma-like disorders described so far, exhibiting common signs such as skin thickening, Raynaud’s phenomenon or even interstitial lung disease. Unlike SSc, they do not exhibit specific antibodies or other visceral involvements and, in contrast, they do exhibit atypical ancillary findings such as eosinophilia, rare cancers (angiosarcoma), or neuropathy.
References
Bramwell B (1914) Diffuse sclerodermia: its frequency; its occurrence in stone-masons; its treatment by fibrolysin-elevations of temperature due to fibrolysin injections. Edinburgh Med J 12:387–340
Erasmus LD (1957) Scleroderma in gold-miners on the Witwatersrand with particular reference to pulmonary manifestations. S Afr J Lab Clin Med 3:209–231
McHugh NJ, Whyte J, Harvey G, Haustein UF (1994) Anti-topoisomerase I antibodies in silica associated systemic sclerosis. A model for autoimmunity. Arthritis Rheum 37:1198–1205
McCormic ZD, Khuder SS, Aryal BK, Ames AL, Khuder SA (2010) Occupational silica exposure as a risk factor for scleroderma: a meta-analysis. Int Arch Occup Environ Health 83:763–769
Bovenzi M, Barbone F, Betta A, Tommasini M, Versini W (1995) Scleroderma and occupational exposure. Scand J Work Environ Health 21:289–292
Bovenzi M, Barbone F, Pisa FE, Betta A, Romeo L, Tonello A (2004) A case-control study of occupational exposures and systemic sclerosis. Int Arch Occup Environ Health 77:10–16
Rodnan GP, Benedek TG, Medsger TA Jr, Cammarata RJ (1967) The association of progressive systemic sclerosis (scleroderma) with coal miner’s pneumoconiosis and other forms of silicosis. Ann Intern Med 66:323–334
Sluis-Cremer GK, Hessel PA, Nizdo EH, Churchill AR, Zeiss EA (1985) Silica, silicosis and progressive systemic sclerosis. BJ Ind Med 42:838–843
Ziegler V, Enderlein G, Mehlhorn J (1997) Retrospective epidemiological analysis of the relation between progressive systemic scleroderma and the exposure to quartz dust out of uranium ore mining in East Germany. Zentralbl Arbeitsmed 47:7–13
Brown LM, Gridley J, Olsen JH, Mellemkjaer L, Linet MS, Fraumeni JF Jr (1997) Cancer risk and mortality patterns among silicotic men in Sweden and Denmark. J Occup Environ Med 39:633–638
Mehlhorn J, Enderlein G, Conrad K (1999) Analysis for the association between progressive systemic scleroderma exposure to quartz dust and silicosis in east German uranium mining. Zbl Arbeitsmed 49:134–147
Englert H, Small-Mcmahon J, Davis K et al (2000) Male systemic sclerosis and occupational silica exposure—a population-based study. Aust NZ J Med 30:215–220
Burns CJ, Laing TJ, Gillespie BW et al (1996) The epidemiology of scleroderma among women: assessment of risk from exposure to silicone and silica. J Rheumatol 23:1904–1911
Walsh SJ (1999) Effects of non-mining occupational silica exposure on proportional mortality from silicosis and systemic sclerosis. J Rheumatol 26:2179–2185
Calvert GM, Rice FL, Boiano JM, Sheehy JW, Sanderson WT (2003) Occupational silica exposure and risk of various diseases: an analysis using death certificates from 27 states of the United States. Occup Environ Med 60:122–129
Lacey JV, Laing TJ, Gillespie BW (1997) Epidemiology of scleroderma among women: assessment of risk from exposure to silicone and silica. J Rheumatol 24:1854–1855
Gold LS, Ward MH, Dosemeci M, De Roos AJ (2007) Systemic autoimmune disease mortality and occupational exposures. Arthritis Rheum 56:3189–3201
Rosenman KD, Moore-Fuller M, Reilly MJ (1999) Connective tissue disease and silicosis. Am J Ind Med 35:375–381
Silman AJ, Jones S (1992) What is the contribution of occupational environmental factors to the occurrence of scleroderma in men? Ann Rheum Dis 51:1322–1324
Makol A, Reilly MJ, Rosenman KD (2011) Prevalence of connective tissue disease in silicosis (1985-2006)-a report from the state of Michigan surveillance system for silicosis. Am J Ind Med 54:255–262
Maitre A, Hours M, Bonneterre V et al (2004) Systemic sclerosis and occupational risk factors: role of solvents and cleaning products. J Rheumatol 31:2395–2401
Diot E, Lesire V, Guilmot JL et al (2002) Systemic sclerosis and occupational risk factors: a case control study. Occup Environ Med 59:545–549
Mora G. 2003 Esclerodermia inducida por sílice. Tesis Doctoral. Facultad de Medicina, Universidad de Buenos Aires, Argentina.
Thompson AE, Pope JE (2002) Increased prevalence of scleroderma in southwestern Ontario: a cluster analysis. J Rheumatol 29:1867–1873
Marie I, Gehanno JF, Bubenheim M et al (2014) Prospective study to evaluate the association between systemic sclerosis and occupational exposure and review of the literature. Autoimmun Rev 13:151–156
Zachariae H, Bjerring P, Søndergaard KH, Halkier-Sørensen L (1997) Occupational systemic sclerosis in men. Ugeskr Laeger 159:2687–2689
Freire M, Alonso M, Rivera A, Sousa A, Soto A, Gómez-Sousa JM, Baroja A, Vázquez-Triñanes C, Sopeña B (2015) Clinical peculiarities of patients with scleroderma exposed to silica: a systematic review of the literature. Semin Arthritis Rheum 45:294–300
Reinl W (1957) Scleroderma caused by trichloroethylene effects. Zentralbl Arbeitsmed 7:58–60
Walder G (1965) Solvents and scleroderma. Lancet 28:436–437
Aryal BK, Khuder SA, Schaub EA (2001) Meta-analysis of systemic sclerosis and exposure to solvents. Am J Ind Med 40:271–274
Kettaneh A, Al Moufti O, Tiev KP, Chayet C, Tolédano C, Fabre B et al (2007) Occupational exposure to solvents and gender-related risk of systemic sclerosis: a meta-analysis of case-control studies. J Rheumatol 34:97–103
Barragán-Martínez C, Speck-Hernández CA, Montoya-Ortiz G, Mantilla RD, Anaya JM, Rojas-Villarraga A (2012) Organic solvents as risk factor for autoimmune diseases: a systematic review and meta-analysis. PLoS One 7:1–18
Garabrant DH, Lacey JV, Laing TJ et al (2003) Scleroderma and solvent exposure among women. Am J Epidemiol 157:493–500
Lacey JV, Garabrant DH, Gillespie BW (1997) Self-reported exposure to solvents in women with systemic sclerosis. Arthritis Rheum 40:1021
Laing TJ, Gillespie B, Burns C (1995) Risk factors for scleroderma among Michigan women. Arthritis Rheum 38(suppl 9):S341
Czirjak L, Bokk A, Csontos G, Lörincz G, Szegedi G (1989) Clinical findings in 61 patients with progressive systemic sclerosis. Acta Derm Venereol 69:533–536
Goldman JA (1996) Connective tissue disease in people exposed to organic chemical solvents. Systemic sclerosis (scleroderma) in dry cleaning plant and aircraft industry workers. J Clin Rheumatol 2:185–190
Nietert PJ, Sutherland SE, Silver RM et al (1998) Is occupational organic solvent exposure a risk factor for scleroderma? Arthritis Rheum 41:1111–1118
Czirják L, Kumánovics G (2002) Exposure to solvents in female patients with scleroderma. Clin Rheumatol 21:114–118
Arnaud J, De Gaudemaris R, Carpentier P (2002) Sclérodermie et facteurs professionneles de l’environnment : étude cas-témoins en population générale. Arch Mal Prof 63:144
Kütting B, Uter W, Drexler H (2006) Is occupational exposure to solvents associated with an increased risk for developing systemic scleroderma? J Occup Med Toxicol 1:1–6
Garabrant DH, Dumas C (2000) Epidemiology of organic solvents and connective tissue disease. Arthritis Res 2:5–15
Sverdrup B (1984) Do workers in the manufacturing industry run an increased risk of getting scleroderma? Int J Dermatol 23:629
Lundberg I, Alfredsson L, Plato N, Sverdrup B, Klareskog L, Kleinau S (1994) Occupation, occupational exposure to chemicals and rheumatological disease. Scand J Rheumatol 23:305–310
Perkins LL, Clark BD, Klein PJ, Cook RR (1995) A meta-analysis of breast implants and connective tissue disease. Ann Plast Surg 35:561–570
Hochberg MC, Perlmutter DL (1996) The association of augmentation mammoplasty with connective tissue disease, including systematic sclerosis (scleroderma): a meta-analysis. Curr Top Microbiol Immunol 210:411–417
Wong O (1996) A critical assessment of the relationship between silicone breast implants and connective tissue diseases. Regul Toxicol Pharmacol 23:74–85
Whorton D, Wong O (1997) Scleroderma and silicone breast implants. West J Med 167:159–165
Janowsky EC, Kupper LL, Hulka BS (2000) Meta-analysis of the relation between silicone breast implants and the risk of connective-tissue diseases. N Engl J Med 342:781–790
Gabriel SE, O’Fallon M, Kurland LT, Beard CM, Woods JE, Melton LJ 3rd (1994) Risk of connective-tissue diseases and other disorders after breast implantation. N Engl J Med 24:1697–1702
Brinton L, Buckley LM, Dvorkina O, Lubin JH, Colton T, Murray MC (2004) Risk of connective tissue disorders among breast implant patients. Am J Epidemiol 160:619–627
McLaughlin JK, Fraumeni JF, Olsen J, Mellemkjaer L (1994) Breast implants, cancer and systemic sclerosis. J Natl Cancer Inst 86:424
Kjoller K, Friis S, Mellemkjaer L et al (2001) Connective tissue disease and other rheumatic conditions following cosmetic breast implantation in Denmark. Arch Intern Med 161:973–979
Goldman JA, Greenblatt J, Joines R, White L, Aylward B, Lamm SH (1995) Breast implants, rheumatoid arthritis, and connective tissue diseases in a clinical practice. J Clin Epidemiol 48:571–582
Fryzek JP, Holmich L, Mclaughlin JK et al (2007) A nationwide study of connective tissue disease and other rheumatic conditions among Danish women with long-term cosmetic breast implantation. Ann Epidemiol 17:374–379
Englert HJ, Brooks P (1994) Scleroderma and augmentation mammoplasty- a causal relationship? Aust NZ J Med 24:74–79
Hochberg MC, Perlmutter DL, Medsger T et al (1996) Lack of association between augmentation mammoplasty and systemic sclerosis (scleroderma). Arthritis Rheum 39:1125–1131
Wigley FM, Miller R, Hochberg MC (1992) Augmentation mammoplasty in patients with systemic sclerosis: data from the Baltimore scleroderma research center and Pittsburgh scleroderma data bank. Arthritis Rheum 35(Suppl 9):S46
Hennekens CH, Lee IM, Cook NR et al (1996) Self-reported breast implants and connective-tissue diseases in female health professionals. JAMA 275:616–621
Yamakage A, Ishikawa H, Saito Y, Hattori A (1980) Occupational scleroderma like disorder occurring in men engaged in the polymerization of epoxy resins. Dermatologica 161:33–44
Freni-Titulaer LWJ, Kelley DB, Grow AG, McKinley TW, Arnett FC, Hochberg MC (1989) Connective tissue disease in southeastern Georgia: a case-control study of etiologic factors. Am J Epidemiol 130:404–409
Wilson RH, McCormick WE, Tatum CF, Creech JL (1967) Occupational acroosteolysis. Report of 31 cases. JAMA 201:577–581
Veltman G, Lange CE, Jühe S, Stein G, Bachner U (1975) Clinical manifestations and course of vinyl chloride disease. Ann N Y Acad Sci 246:6–17
Ward AM, Udnoon S, Watkins J, Walker AE, Darke CS (1976) Immunological mechanisms in the pathogenesis of vinyl chloride disease. Br Med J 1:936–938
Posada de la Paz M, Philen RM, Abaitua I (2001) Toxic oil syndrome: the perspective after 20 years. Epidemiol Rev 23:231–247
Mayes MD (1999) Epidemiologic studies of environmental agents and systemic autoimmune diseases. Environ Health Perspect 107:743–748
Hess EV (2002) Environmental chemicals and autoimmune disease: cause and effect. Toxicology 181-2:65–70
Magnant J, Diot E (2006) Sclérodermie systémique : épidémiologie et facteurs environnementaux. Presse Med 35:1894–1901
Haustein UF, Ziegler V (1985) Environmentally induced systemic sclerosis-like disorders. Int J Dermatol 24:147–151
Fishman SJ, Russo GG (1991) The toxic pseudosclerodermas. Int J Dermatol 30:837–842
Cooper GS, Miller FW, Germolec DR (2002) Occupational exposures and autoimmune diseases. Int Immunopharmacol 2:303–313
Silman AJ, Hochberg MC (1996) Occupational and environmental influences on scleroderma. Rheum Dis Clin N Am 4:737–747
Steen VD (1999) Occupational scleroderma. Curr Opin Rheumatol 11:490–494
Hallé O, Schaeverbeke T, Bannwarth B, Dehais J (1997) Les facteurs d’environment et les éléments iatrogènes dans la sclérodermie systémique et les syndromes apparentés. Revue de la littérature. Rev Méd Interne 18:219–229
Granel B, Zemour F, Lehucher-Michel MP, Moulin P, Disdier P, Durand JM et al (2008) Évaluation de l’exposition toxique professionnelle de patients atteints de sclérodermie systémique. Revue de la lilttérature et résultat d’un auto-questionnaire. Rev Méd Interne 29:891–900
Straniero NR, Furst DE (1989) Environmentally-induced systemic sclerosis-like illness. Baillieres Clin Rheumatol 3:63–79
Nietert PJ, Silver RM (2000) Systemic sclerosis: environmental and occupational risk factors. Curr Opin Rheumatol 12:520–526
Miller FW, Alfredsson L, Costenbader KH, Kamen DL, Nelson LM, Norris JM et al (2012) Epidemiology of environmental exposures and human autoimmune diseases: findings from a National Institute of Environmental Health Sciences expert panel workshop. J Autoimmun 39:259–271
D’Cruz D (2000) Autoimmune diseases associated with drugs, chemicals and environmental factors. Toxicol Lett 112-113:421–432
Dospinescu P, Jones GT, Basu N (2013) Environmental risk factors in systemic sclerosis. Curr Opin Rheumatol 25:179–183
Schaeverbeke T, Hallé O, Moride Y (1995) Systemic sclerosis and prolonged exposure to toxics. Arthritis Rheum 38(Suppl 9):S332
Wells G, Shea B, O’Connell D. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomized studies in meta-analysis. http://www.ohri.ca/programs/clinical_epidemiology /oxford.htm.
Kahn HA, Sempos CT.1989. Statistical Q14 methods in epidemiology. Oxford University Press.
Higgins JPT, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560
Begg CB, Berlin JA (1989) Publication bias and dissemination of clinical research. J Natl Cancer Inst 81:107–115
Acknowledgements
Special acknowledgment is given to Sandra Stroo from Dallas, Texas (USA), for the revision of the present study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is neither financial support nor conflict of interest.
The authors declare no conflict of interest, no source of funding or sponsors or any relationship to organizations that could potentially influence the present work.
Financial support
None
Rights and permissions
About this article
Cite this article
Rubio-Rivas, M., Moreno, R. & Corbella, X. Occupational and environmental scleroderma. Systematic review and meta-analysis. Clin Rheumatol 36, 569–582 (2017). https://doi.org/10.1007/s10067-016-3533-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-016-3533-1