Abstract
Anthropogenic nitrogen (N) deposition has affected plant community composition and nutrient cycling in terrestrial ecosystems worldwide. This includes changes to the way plants use and recycle nutrients, including effects on nutrient resorption, which is a key process through which plants recover nutrients from tissue during senescence. Nutrient resorption has considerable adaptive and functional significance for plants and helps regulate core ecosystem processes such as decomposition. However, our understanding of how N deposition affects nutrient resorption and, in particular, of how N inputs alter ecosystem resorption via changes to existing species’ resorption compared with changes to community composition remains poor. To disentangle the role of species versus community composition controls driving variation in nutrient resorption responses to N inputs, we carried out an experiment with six different N addition rates in a temperate steppe. We found that species-scale nutrient resorption responses to N enrichment were variable; for example, only half of the measured species reduced both N and P resorption efficiency in response to increased N inputs. In contrast, community-scale responses consistently resulted in reduced N and P resorption. Still, N-induced changes in community composition were a weaker control on overall resorption responses than were the effects on individual species; however, it was the synergistic interaction between the two that resulted in the large total reductions of nutrient resorption in the face of increased N. Taken together, our results highlight that understanding and predicting nutrient resorption responses will be most accurately scaled by accounting not only for species’ reductions in resorption but also for changes in community composition.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Highlights
-
Increased nitrogen inputs reduced the nitrogen and phosphorus resorption of some species and dramatically changed plant community composition.
-
Interactions between changes to species-level resorption and plant community composition together determined overall changes in nutrient resorption.
-
Nitrogen enrichment reduced community-scale nitrogen and phosphorus resorption.
Introduction
One of the most globally widespread environmental changes is the increase in reactive nitrogen (N) deposition due to heightened human activities (Galloway and others 2004). Increased inputs of N can alter internal plant biogeochemistry; for example, affecting plant growth, carbon allocation, and foliar chemistry (Baer and others 2003; McNeil and others 2007; Farrer and others 2013). Nitrogen deposition also can drive dramatic changes to plant community composition, with implications for the maintenance of diversity, rare species, and community-level function (Wassen and others 2005; Bobbink and others 2010; Simkin and others 2016). For example, increased N availability has been shown to facilitate the growth of nitrophilic plants to the disadvantage of species with a conservative N use strategy (Suding and others 2005; Bobbink and others 2010). Yet, although changes to both individual plants and to community composition will determine the overall ecosystem effects of increased N deposition, our understanding of how these controls interact to govern ecosystem-level responses remains poor.
Nutrient resorption is a fundamental process through which plants withdraw nutrients from tissue during senescence and prior to abscission. Resorbed nutrients are available for subsequent plant use and thus reduce a plant’s dependence on nutrient uptake from the environment (Aerts and Chapin 2000; Killingbeck 2004; Brant and Chen 2015). Nutrient resorption is most commonly assessed in one of two ways. Nutrient resorption efficiency is the difference between the amount of a given nutrient in green versus fully senesced tissue relative to the amount in the green tissue, and nutrient resorption proficiency is the absolute level of nutrients found in senesced leaves (Killingbeck 1996). Nutrient efficiency and proficiency are different components of the same process, and both are ecologically important. Because they offer different insights into the evolution and functioning of resorption, these two measures of resorption are most powerfully used in concert (Killingbeck 1996).
Resorption plays a key role in regulating plant processes as diverse as competition, productivity, and nutrient uptake (Vitousek 1982; Killingbeck 1996). Because resorption also controls the chemistry, nutrient concentration, and stoichiometry of litterfall, which represents the largest source of nutrients to most terrestrial ecosystems (Moore and Braswell 1994; Cleveland and others 2013), resorption likely plays a role in regulating a wide range of ecosystem processes, including soil respiration, soil carbon storage, and N2 fixation, among others (Davidson and others 1993; Cleveland and Townsend 2006; Reed and others 2011). Indeed, it is critical to understand the consequences of intra-specific responses versus community-level responses for overall nutrient resorption, as community-level litter chemical quality is highly correlated with litter decomposition and nutrient cycling at the ecosystem scale (Garnier and others 2005; Garcia-Palacios and others 2016), even under N-enriched conditions (Pichon and others 2020). Despite the importance of this process, our understanding of the ways in which plant communities tune resorption to varying environmental conditions remains far from complete (Richardson and others 2005; Hayes and others 2014).
Although results from a global meta-analysis showed negative impacts of N enrichment on plant N resorption (Yuan and Chen 2015), empirical evidence at the species-level is highly variable, with negative, neutral, and positive effects reported (van Heerwaarden and others 2003; Lü and others 2013). Moreover, enhanced N availability can affect the resorption of other elements (Brant and Chen 2015; See and others 2015). For example, it could be expected that N enrichment would lead to increased phosphorus (P) resorption, as plants increase P conservation during a transition from N limitation to P limitation (Vitousek and others 2010; Reed and others 2012). However, N enrichment could also enhance soil P availability through stimulation of extracellular phosphatase enzyme activity (Marklein and Houlton 2012). There is evidence of N enrichment lowering P resorption, at least for some species (van Heerwaarden and others 2003; Lü and others 2013). Species-specific variability in nutrient resorption responses to N enrichment makes it difficult to give a clear projection of changes in nutrient resorption at the community-scale.
Given species-specific variation in nutrient resorption even in a single ecosystem, changes to plant community composition would also drive changes in community-scale nutrient resorption. For example, Richardson and others (2005) found a reduction in community-scale nutrient resorption along a soil chronosequence in a temperate rain forest that was caused by both community compositional changes and intra-specific plant responses. Similarly, changes to plant community composition contributed to variations in community-scale nutrient resorption along a 2-million-year dune chronosequence (Hayes and others 2014). Plant species resorption is related to a plant’s overall nutrient strategy, and plants with strategies aimed at acquisition are inherently less efficient in nutrient resorption than those with conservative strategies (Aerts and Chapin 2000). Thus, changes to plant community composition could occur in predictable ways (for example, nitrophiles overtaking conservative N users).
Most previous studies separately address intra-specific responses of nutrient resorption (van Heerwaarden and others 2003; Lü and others 2013; Gilliam and others 2018) and changes in plant community composition under N enrichment (Bobbink and others 2010; Zhang and others 2014; Gilliam and others 2016), regardless of the fact that both responses to increased N inputs have strong potential to affect overall resorption patterns. Opposing responses of inter- and intra-specific nutrient resorption to N enrichment, as indicated by a negative covariation, would result in a moderate or even neutral response at the community-scale (Figure 1). In contrast, consistent responses of inter- and intra-specific variability would reinforce one another and thus strengthen the community-level response. The role of inter- and intra-specific co-variation in driving community-scale responses to environmental change has been addressed for many traits (Auger and Shipley 2013; Volf and others 2016). For instance, Kinchenin and others (2013) found stable foliar N and P concentrations at the community-scale across an elevation gradient due to the strong but opposite changes at intra- and inter-specific responses. However, responses of chemical quality of living tissues do not fully reflect that of senesced tissue or plant litter, due to the sensitivity of nutrient resorption to the alteration of soil resources (Lü and others 2012a, b). Thus, there is a large knowledge gap regarding the relative roles of intra- and inter-specific variation in driving the variation of community-level nutrient resorption and litter quality.
Conceptual diagram of how intra- and inter-specific variations could drive community-level nutrient resorption responses (solid line) across a N addition gradient. Each dashed line represents the nutrient resorption responses of a different plant species. A If the intra-specific responses of nutrient resorption to N addition do not favor a community shift to species with lowered resorption, then the responses of community-level nutrient resorption could be neutral even if individual species reduce their resorption. B However, if N inputs facilitate an increased dominance of species with less efficient nutrient resorption, negative community-level resorption responses to increased N are strengthened.
Here, we examined the changes of species- and community-scale N and P resorption efficiency and proficiency from senescing shoots in response to N enrichment in a temperate steppe after 7 years of N addition. We focused on nutrient resorption from whole shoots instead of individual leaves because of the substantial amount of nutrients resorbed in culms and stems (Freschet and others 2010; Lü and others 2012a, b). We hypothesized that N enrichment would reduce nutrient (N and P) resorption at the community-level through both (1) intra-specific decreases of nutrient resorption for individual plant species and (2) changing species dominance, such that species more efficient in nutrient resorption would decline and the dominance of species less efficient in resorption would increase.
Materials and Methods
Study Site
The study site is a temperate semi-arid grassland in Xilinhot, Inner Mongolia, China (43° 38′ N, 116º 42′ E, 1255 m.a.s.l.). The mean annual temperature is 0.9°C, and mean annual precipitation is 337 mm (1980–2015). Vegetation is typical of temperate steppes, with dominance by Stipa grandis, Achnatherum sibiricum, Agropyron cristatum, Leymus chinensis, and Cleistogenes squarrosa. Traditional grassland management at the site has been livestock grazing in the growing season, lasting from April to September; however, the site has been fenced to exclude livestock grazing since 1999. The soil is classified as a Haplic Calcisol by the FAO system and as a Calcic-Orthic Aridisol in the US soil classification system. The rate of ambient total N deposition in this region was estimated to be about 1.5 g N m−2 y−1 for the last two decades (Zhao and others 2017).
Experimental Design and Plant Sampling
The N addition experiment was started in 2008, and detailed information about the experimental design can be found in Zhang and others (2018). Briefly, there were nine rates of N enrichment ranging from 0 to 50 g N m−2 y−1, with each treatment replicated ten times across ten blocks under a completely randomized block design. Such a broad and multi-level gradient of N addition rates was used to find the thresholds for the responses of biodiversity and ecosystem functioning to N enrichment. The area of each plot was 8 m × 8 m. Blocks were separated by 2 m buffers, and plots within each block were separated by 1 m buffers. Nitrogen (NH4NO3) was added twice each year: once in June (growing season) in a dissolved form and once in November (non-growing season) in a dry form. In the beginning of June, half of the NH4NO3 was mixed with distilled water (9 l for each plot) and sprinkled with a sprayer to simulate wet N deposition. In the beginning of November, another half of the NH4NO3 was mixed with sand (0.5 kg sand for each plot) and broadcasted evenly by hand. We added N in both wet and dry forms to better mimic expected N deposition at the site. To maintain a study design feasible for the resorption work described here, we chose six different N addition rates and used six randomly selected blocks from the ten N addition rates and ten blocks of the larger experiment. We used six blocks each of 0, 2, 5, 10, 20, and 50 g N m−2 y−1, for a total of 36 plots.
This study was carried out in 2015, 7 years after the establishment of this long-term experiment. In mid-August, community aboveground biomass and species composition were assessed in each of the 36 plots using a 1 m × 1 m quadrat. For each plot, the quadrat was randomly placed inside the plot avoiding spatial overlap with sampling quadrats from previous years. Moreover, the quadrats were placed no less than 50 cm inside of the borders of each plot. Aboveground biomass was sampled by clipping all vascular plants at the soil surface. The timing of plant clipping was concurrent with peak biomass, a point after which plant nutrient uptake would be limited. Thus, clipping was unlikely to significantly affect the nutrient status of plants living at the border of clipped quadrats. All plants were sorted by species and oven-dried at 65°C for 48 h. The dry mass of each species was then weighed. Shoot samples from those quadrats were used as green tissue for nutrient analyses.
The senesced shoots were sampled after the growing season in October 2015 when they were fully senesced for all species in this area. All species represented in the August biomass quadrat were sampled. For each species, 10–30 senesced shoots were clipped near the August quadrat in each plot. All samples were oven-dried at 65°C for 48 h. Shoot samples from those quadrats were used as senesced tissue for nutrient analyses.
Dry samples of both green (August samples) and senesced shoots (October samples) were ground using a ball mill (Retsch MM 400; Retsch, Haan, Germany). Total N concentrations of plant samples were analyzed by an Alpkem autoanalyzer (Kjektec System 1026 Distilling Unit, Sweden) (Gallaher and others 1976). For assessing total P concentrations, the ground samples were digested with H2SO4–H2O2 and measured colorimetrically at 880 nm after reaction with molybdenum blue (Kuo 1996).
Data Analysis
The species-level nutrient resorption efficiency (RE) was quantified by the proportional changes of nutrient concentrations between green and senesced shoots following the equation:
In which Nutrientsenesced and Nutrientgreen is the N or P concentration in the October (senesced) and August (green) shoots, respectively. Here, we did not account for the mass loss of shoots during senescence due to the difficulty of accurately measuring shoot-level mass loss in the field, which would underestimate the actual values of nutrient resorption. Nutrient resorption proficiency was quantified as Nutrientsenesced (Killingbeck 1996), and we used the reciprocal of this value so that, as with nutrient resorption efficiency, a higher value would signify higher efficiency. The community biomass-weighed mean values (CWM) of nutrient resorption efficiency in each plot were calculated based on the relative biomass and resorption efficiency of each species present. The relative biomass for each species in each plot was calculated based on the aboveground biomass data of the August sampling.
Data were tested for normality using the Shapiro–Wilk test and for equality of variance using Levene’s test. The relationships between N addition rates and changes in species- and community-level nutrient concentrations and resorption efficiency were tested by linear and nonlinear regression; results of the best-fitting curve are given. The relative contributions of inter- and intra-specific trait variation effects on community-level nutrient concentration and resorption efficiency along the N addition experimental gradient were assessed following Lepš and others (2011), which is based on the division of total sum of squares (SSspecific) of the community-level trait variance related to an environmental factor (the N addition gradient here) into three different effects: ‘fixed’ (SSfixed), ‘intra-specific’ (SSintra), and ‘covariation’ (SScov). The ‘specific’ community-average trait values in each community were calculated using species trait values as measured in the particular community, which includes both inter- and intra-specific effects. The ‘fixed’ community-average trait values were calculated with a fixed trait value (averaged across all plots along the N addition gradient), which thus only included the inter-specific variation effect. The ‘intra-specific’ community-average trait values were calculated as the difference between ‘specific’ and ‘fixed’ community-average trait values. For each of the traits examined here (N and P concentrations in green and senesced shoots, N resorption efficiency, and P resorption efficiency), we ran three generalized linear model regressions for the ‘specific,’ ‘fixed,’ and ‘intra-specific’ community averages with N addition rate as an explanatory variable. From such regressions, we extracted the sums of squares for each of the three community-average measures (SSspecific, SSfixed, and SSintra) explained by N addition. Then, we calculated the SScov by subtracting SSfixed and SSintra from SSspecific. All analyses were conducted with SPSS 13.0 (SPSS Inc., Chicago, IL, USA).
Results
Changes of Species Dominance in Response to N Addition
The dominance of plant species varied greatly in treatments with different N addition rates (Table 1). Among the seven dominant plant species, which together contributed more than 98% of the total biomass averaged across the gradient, S. grandis was the most dominant in the control plots and in plots with N addition rates less than 10 g N m−2 y−1, contributing about 40% of the total community biomass in those plots. In contrast, S. grandis contributed less than 10% of total biomass in plots with addition rates of 20 and 50 g N m−2 y−1. Nitrogen addition increased the contribution of a perennial rhizome grass, L. chinensis, and an annual forb, C. glaucum (Table 1). L. chinensis was the third most dominant species in the control plots, but was the most dominant plant in plots with the highest N addition rate (contributing 70% of total community biomass). C. glaucum was the least dominant species in the control plots, but was the second most dominant species in plots with N addition rates higher than 10 g N m−2 y−1.
Species-Scale Nutrient Resorption
Nitrogen resorption efficiency (NRE) was negatively correlated with N addition rates for three out of the seven species (Table 2). The correlation between N addition rate and NRE was marginally significant for A. sibiricum and not significant for C. glaucum, C. korshinskyi, and C. squarrosa. Nitrogen addition rates were negatively correlated with P resorption efficiency (PRE) for five species (marginally significant for A. sibiricum) and were not significantly correlated with PRE for C. glaucum and S. grandis (Table 2). Averaged across all the treatments, L. chinensis and C. glaucum had the lowest NRE values, whereas A. sibiricum, C. squarrosa, and S. grandis had the highest NRE values (Figure S1a). For P resorption, S. grandis was the most efficient, C. squarrosa was the least efficient, and L. chinensis had an intermediate value (Figure S1b). For all species, resorption proficiencies of both N and P were negatively correlated with N addition rates, except P resorption proficiency in A. sibiricum (Table 2; Figure S2).
Community-Scale Nutrient Concentrations and Nutrient Resorption
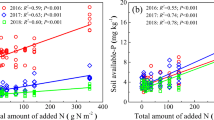
Nitrogen addition significantly increased community-scale biomass-weighted N and P concentrations in both green and senesced shoots (Figure 2A, B) but decreased both NRE and PRE (Figure 2C). N/P resorption efficiency ratios significantly decreased with increased N addition rates (Figure 2D).
Changes in biomass-weighted community-level means for N (A) and P concentrations in green and senesced shoots (B), as well as nutrient resorption efficiency for N and P (C) and the N resorption efficiency/P resorption efficiency ratio (D) across the N addition gradient (0–50 g N m−2 y−1) shown on a log10 scale. The P- and R2 values for the best fit regressions of the relationships between N addition rate and each measured factor are shown.
The contributions of species- and community-level variability were each statistically significant in explaining the response of nutrient concentrations and resorption efficiencies to N addition rates (Figure 3). Further, there were positive covariations between the effects of species- and community-level variability on changes to the concentration and resorption efficiency of both N and P (Figure 3C). The relative contribution of intra-specific variability in explaining total variation was the highest for both nutrient concentration and resorption. However, the relative contribution of covariation in explaining total variation was much higher than that of inter-specific variability alone for both NRE and PRE (NRE: 36% vs. 7%; PRE: 39% vs. 11%, respectively).
Separation of the total variation in community N and P concentrations in green shoots (A) and senesced shoots (B) and the resorption efficiency (C) explained by N addition rates, with explanatory values parsed into inter-specific, intra-specific and covariation effects. Co-variation strength is indexed by the interval between the ‘total variability’ and the sum of inter- and intra-specific variability effects. The statistical significance (***P < 0.001; **P < 0.01; *P < 0.05) of inter-specific, intra-specific, and total variability effects is indicated on the graph when significant.
Discussion
Our results showed negative effects of N enrichment on community-level resorption efficiency for both N and P in the temperate steppe. Such changes were the result of reductions of species-level nutrient resorption efficiency (intra-specific variability), changes in community composition (inter-specific variability), and the positive covariation between intra- and inter-specific variability (Figure 3). Although previous studies have found that both inter- and intra-specific variability contributed to changes in community-scale nutrient resorption across natural soil nutrient availability gradients (Richardson and others 2005; Hayes and others 2014), this study disentangled their relative contributions to the responses of community-scale N and P resorption with N enrichment. More nutrients (both N and P) remained in plant litter with reduced resorption efficiency and proficiency following increasing additions of N, suggesting important changes to plant-driven nutrient inputs into soil in conjunction with the changes to inputs via anthropogenic N deposition. Given the well-established positive relationship between substrate nutrient concentrations and litter decomposition (Melillo and others 1982) and the importance of internal nutrient recycling in driving primary production (Cleveland and others 2013), the reduction in community-level nutrient resorption could have important consequences for ecosystem nutrient cycling.
We observed marked community shifts with different N addition rates, with higher dominance of L. chinensis and lower dominance of S. grandis in treatments with higher rates of N addition (Table 1). For example, S. grandis made up more than 40% of the biomass in plots receiving 5 g N m−2 y−1 or less, but the species represented less than 1% of the biomass in plots receiving 50 g N m−2 y−1. Given the substantial inter-specific variation in nutrient resorption (Figure S1), such shifts in species assemblage have important consequences for community-level nutrient resorption. Indeed, it was shifts in community composition and species-specific responses to increased N together that determined the community-level changes observed (Figure 3). Although some work has highlighted the role of community composition in controlling responses of ecosystem function to environmental change (Wedin and Tilman 1996; Austin and Zanne 2015), our findings present an additional pathway through which species composition can alter ecosystem biogeochemical cycles in the face of anthropogenic change: nutrient resorption. Assessing the changes of community-level functional traits across environmental gradients facilitates our understanding of the subsequent changes to ecosystem processes, as indicated by the stronger relationship between community-level functional traits and fundamental ecosystem properties relative to the relationships with species-level traits alone (Garnier and others 2005; Castro-Díez 2012).
Another important aspect of species composition changes driving community-scale nutrient resorption was reflected by the positive covariation between species- and community-level effects, the relative contribution of which was much greater than the direct contribution of species composition alone (Figure 3). This positive covariation resulted from a consistency in the direction of inter- and intra-specific selection by environmental factors (Figure 1B; Cornwell and Ackerly 2009; Lepš and others 2011; Siefert and others 2014). At the community-scale, N enrichment facilitated the growth of L. chinensis, the species with the lowest NRE. Meanwhile, N enrichment reduced the dominance of S. grandis, the species with highest nutrient resorption (Figure S1). Thus, in addition to its direct effects on how different species resorb nutrients, overall, N deposition shifted the community from a species that had traits associated with high N resorption to one with low N resorption. Our findings show that changes in community composition had a strong effect via a strengthening of the negative effects of N enrichment on individual plant species’ nutrient resorption.
Our hypothesis that N addition would lead to reduced nutrient resorption at the species-scale was supported; however, there was significant variability among species and among resorption methods. Resorption proficiency of both N and P (NRP and PRP) in all the examined species was negatively correlated with N addition rates, and thus this measure suggested strong, consistent reductions in N and P resorption proficiency following 7 years of N addition. Previous studies have suggested resorption proficiency may be more sensitive to nutrient addition relative to nutrient resorption efficiency (for example, Ratnam and others 2008; Rejmánková 2005) and less variable across time (Killingbeck 1996). In comparison with resorption proficiency, three out of seven species showed significant reduction in NRE and six of seven species showed reduction in PRE. Our simultaneous assessment of resorption efficiency and proficiency at the community-level highlights the importance of intra- and inter-specific variation in modulating community-level changes of nutrient conservation during plant senescence, as well as that of litter quality following plant senescence.
In particular, the NRE of four perennial grasses with tall stature were highly sensitive to N enrichment. In contrast, three species with low stature showed neutral responses of NRE to N enrichment. These results are in line with findings that tall perennial grasses are generally facilitated by N enrichment in diverse herbaceous ecosystems due to their competitive advantage for light (Gough and others 2012). There are at least two factors that could account for the neutral response of NRE in the three species with low stature. First, competition for soil available N is intense due to the rapid increases of biomass of perennial tall grasses (Zhang and others 2015) and thus the added N could be less available for less competitive species. Second, the reduction in light availability in the sub-canopy following N enrichment could mediate the effects of N enrichment on nutrient resorption in understory species through changing the composition and degradation of leaf proteins (Yasumura and others 2007).
Our results further underscore the capacity for plant nutrient resorption to vary among species even within a single site (Figure S1), which adds emphasis to the role of community composition in dictating overall ecosystem responses. Negative responses of PRE and PRP to N enrichment seem to conflict with predictions that increased N inputs could result in increased likelihood of P limitation and thus greater resorption of P. Greater soil P availability following N enrichment due to increased phosphatase enzyme activity (Marklein and Houlton 2012; Lü and others 2013) could account for the negative effects of N enrichment on PRE and PRP. Alternatively, P availability may not be limiting enough in this ecosystem to overcome N’s control over the species- and community-level responses of N addition, or nutrients other than P could be playing an interactive role (Crowley and others 2012).
It should be noted that, while the plants were experiencing treatments for 7 years, we measured nutrient resorption in a single year and thus nutrient resorption and the role of species versus community effects could vary among years (Drenovsky and others 2019). Although we would expect these interannual fluctuations to be low relative to the overall effects of the N deposition treatments, further work should examine to what extent the relative contributions of inter-specific variability, intra-specific variability, and their co-variation to community-scale nutrient resorption change across years. Moreover, N deposition is declining or is predicted to decline for some developed countries (Stevens 2016; Gilliam and others 2019); thus, it is important to understand the recovery process for N-enriched ecosystems. The importance of plant community composition in driving the responses of ecosystem-scale nutrient resorption to N enrichment, as found here, implies that there may be a long way to go for restoring N-enriched ecosystems because plant community composition recovers more slowly than soil nutrient status after the cessation of N inputs (Stevens 2016).
In conclusion, results from this study have important implications for understanding the effects of species-specific and community composition controls over ecosystem function. Changes to nutrient resorption have myriad consequences for carbon and nutrient cycling and these findings suggest community composition and species responses together dictate nutrient resorption patterns with increasing anthropogenic N inputs. Further, the community-level changes to nutrient resorption in response to N enrichment could not have been deduced from the arithmetic means of the community’s composing species. In turn, in the absence of plant community information, species-specific responses of nutrient resorption to N enrichment would not be enough to find the overall, quantitative effect. Given the co-existence of diverse species in natural ecosystems, results from this community perspective facilitate the construction of conceptual and numerical models of how species and communities function and respond to change. Further, while the direct role of species composition in driving ecosystem functioning is well recognized (Wedin and Tilman 1996), our results join others in highlighting their indirect role through the positive covariation between inter- and intra-specific effects. The functional identity of species and their sensitivity to environmental change together determine the impacts of N deposition on ecosystem functioning.
References
Aerts R, Chapin FS. 2000. The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. Adv Ecol Res 30:1–67.
Auger S, Shipley B. 2013. Inter-specific and intra-specific trait variation along short environmental gradients in an old-growth temperate forest. J Veg Sci 24:419–28.
Austin AT, Zanne AE. 2015. Whether in life or in death: fresh perspectives on how plants affect biogeochemical cycling. J Ecol 103:1367–71.
Baer SG, Blair JM, Collins SL, Knapp AK. 2003. Soil resources regulate productivity and diversity in newly established tallgrass prairie. Ecology 84:724–35.
Bobbink R, Hicks K, Galloway J, Spranger T, Alkemade R, Ashmore M, Bustamante M, Cinderby S, Davidson E, Dentener F, Emmett B, Fenn M, Gilliam F, Pardo L, De Vries W. 2010. Global assessment of nitrogen deposition effects on terrestrial plant diversity: a synthesis. Ecol Appl 20:30–59.
Brant AN, Chen HYH. 2015. Patterns and mechanisms of nutrient resorption in plants. Crit Rev Plant Sci 34:471–86.
Castro-Díez P. 2012. Functional traits analyses: scaling-up from species to community level. Plant Soil 357:9–12.
Cleveland CC, Houlton BZ, Smith WK, Marklein AR, Reed SC, Parton W, Del Grosso S, Running SW. 2013. Patterns of new versus recycled primary production in the terrestrial biosphere. Proc Natl Acad Sci U S A 110:12733–7.
Cleveland CC, Townsend AR. 2006. Nutrient additions to a tropical rain forest drive substantial carbon dioxide losses to the atmosphere. Proc Natl Acad Sci U S A 104:10316–21.
Cornwell WK, Ackerly DD. 2009. Community assembly and shifts in plant trait distributions across an environmental gradient in coastal California. Ecol Monogr 79:109–26.
Crowley KF, McNeil BE, Lovett GM, Canham CD, Driscoll CT, Rustad LE, Denny E, Hallett RA, Arthur MA, Boggs JL, Goodale CL, Kahl JS, McNulty SG, Ollinger SV, Pardo LH, Schaberg PG, Stoddard JL, Weand MP, Weathers KC. 2012. Do nutrient limitation patterns shift from nitrogen toward phosphorus with increasing nitrogen deposition across the Northeastern United States? Ecosystems 15:940–57.
Davidson EA, Matson PA, Vitousek PM, Riley R, Dunkin K. 1993. Processes regulating soil emissions of NO and N2O in a seasonally dry tropical forest. Ecology 74:130–9.
Drenovsky RE, Pietrasiak N, Short TH. 2019. Global temporal patterns in plant nutrient resorption plasticity. Glob Ecol Biogeogr 28:728–43.
Farrer EC, Herman DJ, Franzova E, Pham T, Suding KN. 2013. Nitrogen deposition, plant carbon allocation, and soil microbes: changing interactions due to enrichment. Am J Bot 100:1458–70.
Freschet GT, Cornelissen JHC, van Logtestijn RSP, Aerts R. 2010. Substantial nutrient resorption from leaves, stems and roots in a subarctic flora: what is the link with other resource economics traits? New Phytol 186:879–89.
Gallaher RN, Weldon CO, Boswell FC. 1976. A semi-automated procedure for total nitrogen in plant and soil samples. Soil Sci Soc Am J 40:887–9.
Galloway JN, Dentener FJ, Capone DG, Boyer EW, Howarth RW, Seitzinger SP, Asner GP, Cleveland CC, Green PA, Holland EA, Karl DM, Michaels AF, Porter JH, Townsend AR, Vorosmarty CJ. 2004. Nitrogen cycles: past, present, and future. Biogeochemistry 70:153–226.
Garcia-Palacios P, McKie BG, Handa IT, Frainer A, Hattenschwiler S. 2016. The importance of litter traits and decomposers for litter decomposition: a comparison of aquatic and terrestrial ecosystems within and across biomes. Funct Ecol 30:819–29.
Garnier E, Cortez J, Billes G, Navas ML, Roumet C, Debussche M, Laurent G, Blanchard A, Aubry D, Bellmann A, Neill C, Toussaint JP. 2005. Plant functional markers capture ecosystem properties during secondary succession. Ecology 85:2630–7.
Gilliam FS, Welch NT, Phillips AH, Billmyer JH, Peterjohn WT, Fowler ZK, Walter CA, Burnham MB, May JD, Adams MB. 2016. Twenty-five-year response of the herbaceous layer of a temperate hardwood forest to elevated nitrogen deposition. Ecosphere 7:e01250.
Gilliam FS, May JD, Adams MB. 2018. Response of foliar nutrients of Rubus allegheniensis to nutrient amendments in a central Appalachian hardwood forest. For Ecol Manag 411:101–7.
Gilliam FS, Burns DA, Driscoll CT, Frey SD, Lovett GM, Watmough SA. 2019. Decreased atmospheric nitrogen deposition in eastern North America: predicted responses of forest ecosystems. For Ecol Manag 244:560–84.
Gough L, Gross KL, Cleland EE, Clark CM, Collins SL, Fargione JE, Pennings SC, Suding KN. 2012. Incorporating clonal growth form clarifies the role of plant height in response to nitrogen addition. Oecologia 169:1053–62.
Hayes P, Turner BL, Lambers H, Laliberte E. 2014. Foliar nutrient concentrations and resorption efficiency in plants of contrasting nutrient-acquisition strategies along a 2-million-year dune chronosequence. J Ecol 102:396–410.
Kinchenin E, Wardle DA, Peltzer DA, Morse CW, Freschet GT. 2013. Contrasting effects of plant inter- and intraspecific variation on community-level trait measures along an environmental gradient. Funct Ecol 27:1254–61.
Killingbeck KT. 1996. Nutrients in senesced leaves: keys to the search for potential resorption and resorption proficiency. Ecology 77:1716–27.
Killingbeck KT. 2004. Nutrient resorption. In: Nooden LD, Ed. Plant Cell Death Processes. San Diego, California, USA: Academic Press.
Kuo S. 1996. Phosphorus. In: Bridgham JM, Ed. Methods of soil analysis, part 3. Madison: ASA and SSSA. p 869–919.
Lepš J, de Bello F, Smilauer P, Dolezal J. 2011. Community trait response to environment: disentangling species turnover versus intraspecific trait variability effects. Ecography 34:856–63.
Lü XT, Freschet GT, Flynn DFB, Han XG. 2012a. Plasticity in leaf and stem nutrient resorption proficiency potentially reinforces plant-soil feedbacks and microscale heterogeneity in a semi-arid grassland. J Ecol 100:144–50.
Lü XT, Kong DL, Pan QM, Simmons ME, Han XG. 2012b. Nitrogen and water availability interact to affect leaf stoichiometry in a semi-arid grassland. Oecologia 168:301–10.
Lü XT, Reed S, Yu Q, He NP, Wang ZW, Han XG. 2013. Convergent responses of nitrogen and phosphorus resorption to nitrogen inputs in a semiarid grassland. Glob Change Biol 19:2775–84.
Marklein AR, Houlton BZ. 2012. Nitrogen inputs accelerate phosphorus cycling rates across a wide variety of terrestrial ecosystems. New Phytol 193:696–704.
McNeil BE, Read JM, Driscoll CT. 2007. Foliar nitrogen responses to elevated atmospheric nitrogen deposition in nine temperate forest canopy species. Environ Sci Technol 41:5191–7.
Melillo JM, Aber JD, Muratore JF. 1982. Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology 63:621–6.
Moore B, Braswell BH. 1994. Planetary metabolism: understanding the carbon-cycle. Ambio 23:4–12.
Pichon NA, Cappelli SL, Soliveres S, Holzel N, Klaus VH, Kleinebecker T, Allan E. 2020. Decomposition disentangled: a test of the multiple mechanisms by which nitrogen enrichment alters litter decomposition. Funct Ecol 34:1485–96.
Ratnam J, Sankaran M, Hanan NP, Grant RC, Zambatis N. 2008. Nutrient resorption patterns of plant functional groups in a tropical savanna: variation and functional significance. Oecologia 157:141–51.
Reed SC, Cleveland CC, Townsend AR. 2011. Functional ecology of free-living nitrogen fixation: a contemporary perspective. Annu Rev Ecol Evol Syst 42:489–512.
Reed SC, Townsend AR, Davidson EA, Cleveland CC. 2012. Stoichiometric patterns in foliar nutrient resorption across multiple scales. New Phytol 196:173–80.
Rejmánková E. 2005. Nutrient resorption in wetland macrophytes: comparison across several regions of different nutrient status. New Phytol 167:471–82.
Richardson SJ, Peltzer DA, Allen RB, McGlone MS. 2005. Resorption proficiency along a chronosequence: responses among communities and within species. Ecology 86:20–5.
See CR, Yanai RD, Fisk MC, Vadeboncoeur MA, Quintero BA, Fahey TJ. 2015. Soil nitrogen affects phosphorus recycling: foliar resorption and plant–soil feedbacks in a northern hardwood forest. Ecology 96:2488–98.
Siefert A, Fridley JD, Ritchie ME. 2014. Community functional responses to soil and climate at multiple spatial scales: when does intraspecific variation matter? PLoS ONE 9:e111189.
Simkin SM, Allen EB, Bowman WD, Clark CM, Belnap J, Brooks ML, Cade BS, Collins SL, Geiser LH, Gilliam FS, Jovan SE, Pardo LH, Schulz BK, Stevens CJ, Suding KN, Throop HL, Waller DM. 2016. Conditional vulnerability of plant diversity to atmospheric nitrogen deposition across the United States. Proc Natl Acad Sci U S A 113:4086–91.
Stevens CJ. 2016. How long do ecosystems take to recover from atmospheric nitrogen deposition? Biol Conserv 200:160–7.
Suding KN, Collins SL, Gough L, Clark C, Cleland EE, Gross KL, Michunas DG, Pennings S. 2005. Functional- and abundance-based mechanisms explain diversity loss due to N fertilization. Proc Natl Acad Sci U S A 102:4387–92.
van Heerwaarden LM, Toet S, Aerts R. 2003. Nitrogen and phosphorus resorption efficiency and proficiency in six sub-arctic bog species after 4 years of nitrogen fertilization. J Ecol 91:1060–70.
Vitousek P. 1982. Nutrient cycling and nutrient use efficiency. Am Nat 119:553–72.
Vitousek PM, Porder S, Houlton BZ, Chadwick OA. 2010. Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen–phosphorus interactions. Ecol Appl 20:5–15.
Volf M, Redmond C, Albert AJ, Le Bagousse-Pinguet Y, Biella P, Gotzenberger L, Hrazsky Z, Janecek S, Klimesova J, Leps J, Sebelikova L, Vlasata T, de Bello F. 2016. Effects of long- and short-term management on the functional structure of meadows through species turnover and intraspecific trait variability. Oecologia 180:941–50.
Wassen MJ, Venterink HO, Lapshina ED, Tanneberger F. 2005. Endangered plants persist under phosphorus limitation. Nature 437:547–50.
Wedin DA, Tilman D. 1996. Influence of nitrogen loading and species composition on the carbon balance of grasslands. Science 274:1720–3.
Yasumura Y, Hikosaka K, Hirose T. 2007. Nitrogen resorption and protein degradation during leaf senescence in Chenopodium album grown in different light and nitrogen conditions. Funct Plant Biol 34:409–17.
Yuan ZY, Chen HYH. 2015. Negative effects of fertilization on plant nutrient resorption. Ecology 96:373–80.
Zhang YH, Feng JC, Isbell F, Lü XT, Han XG. 2015. Productivity depends more on the rate than the frequency of N addition in a temperate grassland. Sci Rep 5:12558.
Zhang YH, Lu XT, Isbell F, Stevens CJ, Han X, He NP, Zhang GM, Yu Q, Huang JH, Han XG. 2014. Rapid plant species loss at high rates and at low frequency of N addition in temperate steppe. Glob Change Biol 20:3520–9.
Zhang YH, Wang J, Stevens CJ, Lü XT, He NP, Wang CH, Han XG. 2018. Effects of the frequency and the rate of N enrichment on community structure in a temperate grassland. J Plant Ecol 11:685–95.
Zhao YH, Zhang L, Chen YF, Liu XJ, Xu W, Pan YP, Duan L. 2017. Atmospheric nitrogen deposition to China: a model analysis on nitrogen budget and critical load exceedance. Atmos Environ 153:32–40.
Acknowledgements
We acknowledge the staff of the Inner Mongolia Grassland Ecosystem Research Station for their support. Comments from two anonymous reviewers contributed substantially to improving the manuscript. This work was supported by the National Natural Science Foundation of China (31822006 and 31770503), K.C.Wong Education Foundation (GJTD-2019-10), Strategic Priority Research Program of the Chinese Academy of Sciences (XDA23070103), and the Key Research Program from CAS (QYZDB-SSW-DQC006). S.R. was supported by the U.S. Geological Survey Ecosystems Mission Area. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interests.
Additional information
Authors Contributions
XTL, SLH and XGH designed the study. SLH, YYH, HWW, ZWZ, ZYL and GJY collected the data. XTL, SCR, JXY and SLH analyzed the data. XTL, JXY and SCR wrote the first draft of the manuscript. All authors discussed the results and substantially contributed to manuscript revisions.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lü, XT., Hou, SL., Reed, S. et al. Nitrogen Enrichment Reduces Nitrogen and Phosphorus Resorption Through Changes to Species Resorption and Plant Community Composition. Ecosystems 24, 602–612 (2021). https://doi.org/10.1007/s10021-020-00537-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-020-00537-0