Abstract
Woody plant demographics provide important insight into ecosystem state-shifts in response to changing fire regimes. In Australian tropical savannas, the switch from patchy landscape burning by Aborigines to unmanaged wildfires within the past century has been implicated in biodiversity declines including the fire-sensitive conifer, Callitris intratropica. C. intratropica commonly forms small, closed-canopy groves that exclude fire and allow recruitment of conspecifics and other fire-sensitive woody plants. C. intratropica groves provide a useful indicator of heterogeneity and fire regime change, but the mechanisms driving the species’ persistence and decline remain poorly understood. We examined the hypothesis that C. intratropica population stability depends upon a regime of frequent, low-intensity fires maintained by Aboriginal management. We combined integral projection models of C. intratropica population behaviour with an environmental state change matrix to examine how vital rates, grove dynamics and the frequency of high- and low-intensity fires contribute to population stability. Closed-canopy C. intratropica groves contributed disproportionately to population growth by promoting recruitment, whereas singleton trees accounted for a larger proportion of adult mortality. Our patch-based population model predicted population declines under current fire frequencies and that the recruitment of new groves plays a critical role in the species’ persistence. Our results also indicated that reducing fire intensity, a key outcome of Aboriginal burning, leads to C. intratropica population persistence even at high fire frequencies. These findings provide insight into the relationship between ecosystem composition and human–fire interactions and the role of fire management in sustaining the mosaics that comprise ‘natural’ systems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The recruitment bottleneck imposed by fire is a major determinant of the demography of woody plants and, therefore, affects the composition and structural complexity of savannas and other grassland mosaic ecosystems worldwide (Bond 2008; Hoffmann and others 2009; Wood and others 2011; Murphy and Bowman 2012; Ellair and Platt 2013). For instance, fire suppression has been implicated in the encroachment of fire-sensitive shrubs into African and North American grasslands (Roques 2001; Heisler and others 2004), as well as continental-scale increases in the abundance of fire-sensitive trees in North America (Nowacki and Abrams 2008), whereas increases in fire intensity have been linked to the destruction and degradation of forest patches in South American and Australian savannas (Bilbao and others 2010; Russell-Smith and others 2012). Modelling the effects of fire on woody plant populations can therefore provide critical insight into how fire alters and interacts with the spatial heterogeneity of vegetation (Hoffmann 1999; Caswell and Kaye 2001; Keeley and others 2006).
Fire frequency alone remains the most common metric by which to describe fire regimes (Goldammer 1990; Gill and others 2000), frame human impacts on fire occurrence (Guyette and others 2002) and incorporate fire into demographic models (Hoffmann 1999; Caswell and Kaye 2001; Caswell 2006). Fire intensity, however, has critical effects on woody plant survival, growth and reproduction (Knapp and Keeley 2006; Hoffmann and others 2009) and is positively correlated with fire size (Bessie and Johnson 1995; Yates and others 2008). In regions with pronounced rainfall seasonality, the timing of fire occurrence can provide a useful proxy for fire intensity. Fire managers and indigenous landowners are well aware of this and often ‘jumpstart’ the process of landscape burning in the early dry season (EDS) before fuels completely cure to reduce the occurrence and extent of large, high-intensity fires that commonly occur in the late dry season (LDS; Gill and others 2000; Laris 2002; van Wilgen and others 2008; Bliege Bird and others 2012; Murphy and others 2015). The EDS/LDS dichotomy therefore provides a relative measure of fire intensity, which can be used to refine analyses of fire effects (Govender and others 2006; Knapp and Keeley 2006; Russell-Smith and Edwards 2006; Murphy and others 2015). Research in tropical savannas further indicates that intentional EDS burning alters fire intensity and patchiness, but has little effect on the percentage of area burned annually which is likely driven by climate variability (Gill and others 2000; Van Wilgen and others 2004; Archibald and others 2009). Thus, mean fire return interval or regional-scale fire frequency (sensu Li 2002) may be inadequate, on its own, to characterize human-mediated changes to savanna fire regimes.
Widespread mortality of Northern Cypress Pine Callitris intratropica R.T Baker & H.G. Smith (Cupressaceae) across northern Australian savannas provides strong evidence of region-wide increases in fire intensity in recent decades (Bowman and Panton 1993; Whitehead and others 2003; Edwards and Russell-Smith 2009; Trauernicht and others 2013). This has coincided with declines in other species assemblages in northern Australia, notably small mammals (Woinarski and others 2010) and granivorous birds (Franklin and others 2005). The persistence of a long-lived, fire-sensitive conifer in one of the most fire-prone biomes on Earth (Gill and others 2000; Yates and others 2008) has led to the assertion that C. intratropica establishment is dependent upon a regime of low-intensity, patchy fires maintained by Aboriginal burning (Price and Bowman 1994; Russell-Smith 2006; Trauernicht and others 2012). Proactive landscape burning to manipulate resources and mitigate destructive fires is prevalent among traditional management systems worldwide, often providing templates for contemporary land management (Trauernicht and others 2015).
C. intratropica adults can survive low-intensity EDS fires but are more vulnerable to damage from high-intensity LDS fires than the dominant Eucalyptus species with which they co-occur (for example, E. miniata and E. tetrodonta) due to low-growing crowns and a poor ability to resprout after burning (Prior and others 2007; Lawes and others 2011; Bowman and others 2014). The species is non-serotinous and forms no persistent seed banks in either the soil or retained in canopies. High-intensity fires severely reduce seed production (Hawkins 1966) and any burning results in high mortality among seedlings and saplings (Russell-Smith 2006; Lawes and others 2011). Unlike eucalypts, C. intratropica frequently occurs in very small (0.01–0.3 ha) clusters of adult trees (‘groves’) at a density of about 1 grove ha−1 within open savanna vegetation (Trauernicht and others 2015). Grove formation appears to be integral to C. intratropica persistence as closed-canopy groves alter fuel availability and can exclude low-intensity savanna fires (Trauernicht and others 2012). Through this vegetation–fire feedback, C. intratropica groves with intact, closed canopies promote both conspecific recruitment (Figure 1A, C) as well as the establishment of a distinct woody plant community with a greater abundance of other fire-sensitive species than that occur in the savanna matrix (Trauernicht and others 2013). High-intensity fires, however, cause C. intratropica mortality and canopy damage, and disrupt the fire exclusion feedback. This results in a state change in which grassy fuels, the probability of fire occurrence and plant community composition become similar to open savanna, even when adult C. intratropica individuals with damaged crowns persist (Figure 1B; Trauernicht and others 2012).
Characteristics of Callitris intratropica grove condition illustrating (A) a typical intact grove with closed-canopy and woody understory; (B) a degraded grove with damaged canopy and open, grassy understory; (C) kernel density plots of stem diameter distributions; and (D) density plots of basal area distribution (within a 10 m radius of each individual) for each C. intratropica grove condition.
Here we examine C. intratropica population behaviour in the context of grove dynamics and the frequency of low- and high-intensity fires to understand how Aboriginal burning may promote the species’ establishment and persistence. We hypothesize that population growth of C. intratropica depends on the ability of intact groves to exclude low-intensity fires and that the decline of C. intratropica is a sequential process from intact to degraded groves, then to singleton trees and eventually local extirpation. We first construct integral projection models (IPMs; Easterling and others 2000; Ellner and Rees 2006) to assess how ‘grove condition’ (singleton trees, intact groves and degraded groves) and fire disturbance (unburnt, low-intensity fire and high-intensity fire) affect the demography of C. intratropica. IPMs improve the accuracy of population projections for slow-growing species (Zuidema and others 2010) and provide the flexibility to model multiple predictors, which is providing novel insight into the effects of disturbance on population behaviour (Mandle and others 2015). We then combine IPMs of C. intratropica population behaviour with an environmental state change matrix that incorporates the frequency of low- and high-intensity fires observed in the field and the effect of these fires on transitions between grove conditions. Our approach builds on the ‘megamatrix’ approach pioneered with traditional population matrix models to incorporate patch dynamics and landscape-scale disturbance regimes (Horvitz and Schemske 1986; Valverde and Silvertown 1997; Pascarella and Horvitz 1998). Finally, by manipulating fire disturbance frequencies in the model, we examine how the cessation of Aboriginal burning may be implicated in C. intratropica decline, as well as the contemporary trend of homogenization of savanna vegetation in northern Australia (Russell-Smith and others 2012).
Materials and Methods
Study Location
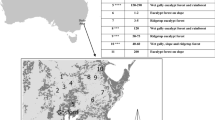
Censuses of C. intratropica growth and survival were conducted at Dukladjarranj (12°39′10.90″S, 134°18′1.40″E), an Aboriginal ‘estate’ on the north-eastern edge of the Arnhem Plateau owned and managed by a Gunei-speaking clan within the Djelk Indigenous Protected Area of Arnhem Land, Northern Territory, Australia (Figure 2A). The Arnhem Plateau is a low massif (<400 m elevation) of deeply dissected Middle Proterozoic quartzose sandstone and is a regional centre of species endemism (Freeland and others 1988; Woinarski and others 2006). The region is dominated by mesic savanna with annual rainfall (c. 1500 mm) driven by the Asian monsoon, with 90% of precipitation occurring from November to May. The region is characterized by very short fire return times of 1–3 years (Gill and others 2000). Detailed descriptions of the study area are provided elsewhere (Yibarbuk and others 2001; Murphy and Bowman 2007). Observations of cone and seed production used to estimate fecundity were made in a climatically and compositionally similar savanna in Litchfield National Park, about 200 km west of Dukladjarranj (Lawes and others 2013).
Maps of (A) the study site within the Djelk Indigenous Protected Area (I.P.A.) in Arnhem Land indicating the extent of the Arnhem Plateau ‘Stone Country’ (dark grey) and (B) the Callitris intratropica individuals surveyed in this study, illustrating the population extent (inset) and the spatial distribution of intact and degraded groves and singleton trees.
Demographic Surveys
We recorded the growth and survival of 722 C. intratropica individuals ranging from 1 to 650 mm in stem diameter (measured at 1 m from the ground) and 563 C. intratropica seedlings ranging from 35 to 1300 mm in height over three growing seasons between 2006 and 2009. The former, hereafter referred to as ‘adults’, were systematically sampled across a 2.5 km expanse of open savanna dominated by Eucalyptus tetrodonta and E. miniata with a grass understory (Figure 2B). Seedlings were tagged and monitored in thirteen 30 m2 circular plots in the same area. Sampling encompassed a range of grove conditions and fire disturbances. Adult trees were identified as occurring as lone, singleton trees (>5 m from any adult conspecific; N = 299) or within either an intact, closed-canopy grove (N = 215 among 12 groves) or a degraded, open-canopy grove (N = 208 among 24 groves). Degraded groves were distinguished from intact groves based on open, fire-damaged canopies and continuous grassy fuels in the understory, following Trauernicht and others (2012). LANDSAT satellite-derived fire scar maps (30 m resolution; Edwards and Russell-Smith 2009) were used to establish whether adult trees and seedling plots were exposed to low-intensity EDS fires (May–July), high-intensity LDS fires (August–October) or no fire in the year prior to measurements. These data were corroborated on the ground by recording fire damage for each individual. Fecundity was determined by measuring cone and seed production and seed germination rates from 31 trees sampled from closed-canopy groves, open-canopy groves and singleton trees (Lawes and others 2013).
Analyses of Vital Rates
We modelled C. intratropica adult survival and diameter growth, and seedling survival and height growth as continuous functions of initial tree diameter or initial seedling height using linear and generalized linear models, including random effects where applicable. We included (i) fire disturbance (unburnt, low-intensity fire, high-intensity fire) and (ii) grove condition (intact grove, degraded grove, singleton tree) as categorical covariates in our global models wherever appropriate (Table S1). Adult growth and survival models included both fire disturbance and grove condition as covariates. Seedling growth included fire disturbance and basal area of adult C. intratropica per seedling plot as covariates, whereas seedling survival included only low-intensify fire disturbance as a covariate (no high-intensity fires occurred during survival censuses; Table S1). We included individual and year as random effects to account for re-censusing individuals and annual climatic variation, respectively. Global models of adult growth and survival included two-way interactions between fire disturbance and grove condition and initial diameter and grove condition. Linear mixed models of both adult and seedling growth included exponential variance structures to account for heterogeneous variance (Zuur and others 2009).
Fecundity was modelled as the count of cones produced as a function of adult tree diameter and grove condition, including an interaction term, using a generalized linear model with log-normal Poisson errors for overdispersed data (Elston and others 2001). We also incorporated cone presence/absence data from the adult population census to construct a size-based model of the probability of cone production. We then used mean counts of seeds per cone and percentage of seed germination for each grove type (Lawes and others 2013) to construct a size-based predictive model of viable seed production for adult C. intratropica trees. We determined the probability of seedling establishment as the proportion of newly established seedlings in our seedling plots to the total potential seed production of the C. intratropica adults in each plot based on this predictive model. C. intratropica is wind dispersed but the seeds are relatively large and Russell-Smith (2006) found that 94% of C. intratropica recruitment occurs within 10 m of adult trees. We therefore did not account for seed dispersal out of the plots. The best-supported models for analysis were determined via multi-model inference based on the Akaike Information Criterion (AICc) according to the Information-Theoretic approach (Anderson and Burnham 2002). All analyses were completed in R (vers. 3.1.1) using the lme4 (version 1.1) and nlme (version 3.1) packages. Model code and model selection tables are provided in the Supplementary Materials.
Modelling Population Behaviour
We used an IPM that combines model coefficients from the continuous, size-based functions of C. intratropica growth, survival and fecundity described above into a single function—the kernel—to project population behaviour (see Supplementary Methods; Easterling and others 2000; Ellner and Rees 2006). IPMs provide the same projections as traditional matrix models, such as long-term population stability, or lambda (λ), long-term population structure or stable stage distribution (SSD), in addition to sensitivity and elasticity analyses, but are considered more accurate for slow-growing plant species (Zuidema and others 2010). The use of fire disturbance and grove condition (see above) as categorical covariates in the kernel effectively resulted in nine separate IPMs, corresponding to all combinations of the three levels in each covariate. Seedling survival was parameterized using model results for unburnt conditions for intact groves across all fire conditions to represent the vegetation–fire feedback described above (Trauernicht and others 2012). The breakdown of this feedback due to high-intensity fires is ‘captured’ in the patch-based transitions among groves described below. Seedling survival for degraded groves and singleton trees was parametrized for no fires and low-intensity fires from census data and assumed to be zero under high-intensity fires based on field observations and prior work (Bowman and others 2014). We calculated the long-term population growth rate (λ) for each IPM with the popbio package in R (Stubben and Milligan 2007). Values of λ above 1 indicate a growing population whereas λ below 1 indicates a population expected to decline, although lambda values may provide a better measure of relative growth rate among populations than a prediction of actual future population growth (Menges 2000).
Environmental State Change Transition Matrix
To model C. intratropica patch dynamics relative to fire disturbance, we constructed matrix F, a 9 × 9 discrete-state Markov chain of transition probabilities from time t to time t + 1 for all grove condition categories across all fire disturbance categories (see Supplementary Methods; Horvitz and Schemske 1986; Valverde and Silvertown 1997; Pascarella and Horvitz 1998). Grove condition transitions were derived from the responses of intact groves, degraded groves and singleton C. intratropica trees to low- and high-intensity fires observed from surveys and experimental fires (Figure 3; Prior and others 2011; Trauernicht and others 2012, 2013). We calculated the probability of fire disturbance from LANDSAT satellite imagery (~30 m resolution) of EDS and LDS fire scars for each annual time-step between 1990 and 2011 across the entire 28,000 km2 region encompassing our field site and comprising the Western Arnhem Land Fire Abatement project (Edwards and Russell-Smith 2009). We used matrix F to project SSDs of each of the nine possible ‘fire-states’, from which we derived the predicted proportions of grove conditions across the landscape under known or hypothetical fire disturbance probabilities. We also used grove condition SSDs to examine the sensitivity of these outputs to changes in the grove condition transition parameters (see Supplementary Methods).
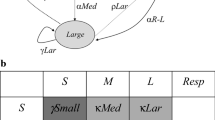
Flow chart of analyses employed in our patch-based model of Callitris intratropica population behaviour. Transitions among C. intratropica grove conditions (intact groves, degraded groves and singleton trees) based on fire disturbance were based on field observations. Fire disturbance Markov chain probabilities were derived from LANDSAT satellite imagery of early dry season (low-intensity) and late dry season (high-intensity) fires. Transitions from degraded groves and singletons to intact groves account for grove ‘recruitment’ and singleton mortality in the final Megamatrix accounts for grove ‘extinction’. Integral projection models (IPMs) of C. intratropica population behaviour under each condition of grove condition–fire disturbance were combined with the environmental state change matrix into a ‘megamatrix’ of transition probabilities of individual trees within and among size classes and environmental states. The megamatrix was used to project overall population stability (λ) of C. intratropica under actual and hypothetical probabilities of high- and low-intensity fire occurrence.
The Megamatrix
Finally, we combined our environmental state change matrix, F, with the IPMs derived for each of the nine combinations of fire disturbance and grove condition using a ‘megamatrix’ to project population demography across different environmental patches (Figure 3 and Supplementary Methods; Valverde and Silvertown 1997; Pascarella and Horvitz 1998). This approach produced the probabilities of a plant of a given size within a given environmental state (that is, grove condition × fire disturbance) transitioning to another combination of size and state instead of following populations within individual patches as they moved through successional states (Caswell 2006). The megamatrix enabled us to project long-term stability (λ) for the entire population across all environmental states, thereby incorporating demography, patch dynamics and fire disturbance into a single model. All code for the IPM, environmental state change matrix and megamatrix are provided in the Supplementary Materials.
Fire Regime Scenarios
To examine the effects of changing fire frequencies on our model projections, we first maintained the probabilities of no fire occurring at time t + 1 (established from the LANDSAT data in matrix D above) and altered the probabilities of fire occurring as a low-intensity or high-intensity fire at time t + 1. This allowed us to maintain the overall frequency of fire occurrence to better reflect the effects of actual savanna fire management which alters fire seasonality and intensity with little effect on total area burned (Gill and others 2000; Van Wilgen and others 2004). We also created hypothetical fire regime scenarios in which we varied both the absolute probability of fire occurring (overall fire frequency) and the probability of fire occurring as a low- versus high-intensity fire. This allowed us to examine whether and how the effects of moderating fire intensity on population stability decline when overall fire frequency increases.
Results
Vital Rates
Our analyses confirmed initial diameter, fire disturbance and grove condition as important predictors of both diameter growth and survival of C. intratropica adults (Size increment: Akaike weight (w i ) = 0.95, R 2 = 0.45, N = 2112, Figure 4A; Survival: w i = 0.65, R 2 = 0.11, N = 2342, Figure 4B). The effect of tree diameter on diameter growth differed among grove types, with a positive relationship in intact groves, a small negative relationship among singleton trees and almost no effect among degraded groves. Low-intensity fires increased diameter growth among trees in intact groves but had very little effect on singletons and trees in degraded groves. High-intensity fires decreased diameter growth with the greatest deleterious effect on intact groves (Figure 4C).
Callitris intratropica (A) diameter growth and (B) survival as a function of initial diameter across fire conditions (unburnt, low-intensity fires and high-intensity fires) and stand types (Intact stands, damaged stands and singleton trees). Solid lines illustrate model predictions and dotted lines represent upper and lower 95% confidence intervals for model predictions. Akaike weights (w i ) are presented relative to all candidate models.
Fire disturbance increased C. intratropica mortality with high-intensity fires having the strongest effect on tree mortality (Figure 4). Trees in degraded groves experienced slightly higher mortality than in intact groves, whereas singleton trees experienced much higher rates of mortality, being most pronounced for high-intensity fires (Figure 4F). Although we did not explicitly include density effects in our models, intact and degraded C. intratropica had higher tree densities (Figure 2B) and basal area (for example, mean of 12.65 m2/ha for intact groves, 12.18 m2/ha for degraded groves and 4.99 m2/ha for singletons; Figure 1D) and therefore including grove condition as an explanatory variable in our analyses incorporated potential effects of density on vital rates.
Our analysis of C. intratropica seedling growth only supported initial height and basal area of adult C. intratropica trees as important factors, with no effect of fire (w i = 0.82, R 2 = 0.91, N = 367). The effect of adult tree basal area was incorporated into the IPM using mean values for each grove type. Analyses of seedling survival identified both initial height and fire disturbance (low-intensity fires only) as important correlates (w i = 0.70, R 2 = 0.08, N = 972, Figure 5A), with low-intensity fires negatively affecting survival. In our models of fecundity, only tree diameter was supported as a predictor of both the count of cones produced (w i = 0.55, R 2 = 0.024, N = 31, Figure 5B) and the probability of cone production (w i > 0.99, R 2 = 0.42, N = 1227).
Population Dynamics
The individual IPMs enabled us to examine population behaviour for each grove condition under each fire disturbance. These models alone are inadequate for assessing the overall population stability of C. intratropica because projections assume constant fire disturbance (that is, no fires vs. annual high- and low-intensity fires) as opposed to variable patterns of disturbance among patches across the landscape. The λ values from these individual IPMs (Figure 6A), however, do indicate how different grove conditions contribute to overall population stability as well as susceptibility of each grove condition to fire disturbance. Our models indicated positive population growth (λ > 1) among all grove conditions in the absence of fire and for intact groves under low-intensity fires. IPMs projected negative population growth (λ < 1) for all other combinations of grove condition and fire disturbance, with high-intensity fires having a strong negative effect on the population stability of degraded groves and singleton trees.
Results from integral projection models (IPM), the environmental state change matrix (EM) and the patch-based ‘megamatrix’ model of Callitris intratropica population behaviour illustrating: (A) population growth (λ) derived from IPMs for each combination of fire disturbance type (unburnt, low-intensity and high-intensity fires) and grove condition (intact groves, degraded groves and singleton trees) ignoring patch dynamic effects; (B) observed proportions and stable stage (projected) proportions of grove conditions derived from the EM under different probabilities that fire occurs at low- vs. high-intensity, keeping overall fire frequency constant (c. 40% burned annually); (C) the response of λ to changing the probability that fire occurs at low-intensity, keeping overall fire frequency constant (c. 40% land area burned); and (D) a surface plot of the response of lambda to hypothetical changes in both total fire frequency (y-axis) and the probability that fire occurs at low-intensity (x-axis). Values of λ above 1 indicates positive population growth and λ below 1 indicates populations are declining. The dashed vertical lines in (B) and (C) indicates actual observed probabilities that fire occurs as low-intensity (early dry season) fires versus high-intensity (late dry season) fires, derived from satellite imagery.
Patch Dynamics and the Megamatrix
The grove condition SSDs (projected proportions) from the environmental state change matrix, F, were robust to changes in the transition probabilities of intact groves under low-intensity fires, and for both intact and degraded groves under high-intensity fires (Supplementary Methods; Figure S1). Grove condition SSDs were sensitive to changes in the recruitment of intact groves (that is, increasing contributions from degraded groves and singleton trees to intact groves; Figure S1d). However, the probabilities of intact grove recruitment used in matrix F were kept at low levels (1% probability) and were assumed to occur only under conditions of no fire.
Grove condition SSD under current fire probabilities projected higher proportions of singleton trees and lower proportions of intact and degraded groves (Intact: 5%, Degraded: 11%, Singletons: 84%) than the actual proportions of the population among grove conditions observed in the field (Intact: 30%, Degraded: 29%, Singletons: 41%). It must be noted, however, that the environmental state change matrix does not include singleton mortality—this is accounted for in the megamatrix by including singleton tree mortality from the IPM. Therefore, some proportion of singletons projected by the SSD (that is, 84%) actually represent patches that are unoccupied by C. intratropica. Increasing the proportion of fires that occur as low-intensity fires resulted in a greater projected proportion of intact C. intratropica groves in the landscape (Figure 6B).
The megamatrix projected negative population growth (λ = 0.98) for the overall C. intratropica population across all grove types under observed probabilities of fire disturbance. The λ values for overall C. intratropica population stability increased as the prevalence of low-intensity increased relative to high-intensity fires (Figure 6C). Altering both the probability of fire occurrence and low- versus high-intensity fires, indicated that decreasing total fire frequency in the model also increased C. intratropica population stability. However, these results also suggested that increasing the prevalence of low-intensity fires may increase C. intratropica population growth even under very high total fire frequencies (Figure 6D)
Discussion
The demographic patterns of C. intratropica support the hypothesis that the patchy burning that characterizes Aboriginal management enabled this fire-sensitive conifer to establish and persist in highly fire-prone vegetation (Bowman and Panton 1993; Trauernicht and others 2012). The differences in C. intratropica population stability among grove conditions (intact, degraded and singleton trees) indicate that overall population growth depends on the ability of closed-canopy, intact groves to exclude fire (Figure 6). Although this feedback may enable population growth under the high fire frequencies that characterize northern Australian savannas (that is, 1–3 years between fires; Gill and others 2000), it is most effective under conditions of low-intensity fire (Trauernicht and others 2012). Areas still managed by Aboriginal communities, including our study site, have some of the lowest reported fuel loads among Australian savannas (600–1200 kg ha−1; Bowman and others 2007; Trauernicht and others 2012); increases in fire intensity due to the cessation of Aboriginal patch burning is a widely cited cause of declines in C. intratropica and other fire-sensitive vegetation, as well as declines in the structural complexity and plant diversity of Australian savannas more generally (Bowman and Panton 1993; Edwards and Russell-Smith 2009; Russell-Smith and others 2012; Trauernicht and others 2013; Murphy and others 2015). Projected declines of C. intratropica under contemporary frequencies of low- and high-intensity fires in our model further corroborate these observations. Our results indicate that both vegetation–fire feedbacks and human-imposed limitations on fire intensity contribute to the persistence of fire-sensitive vegetation in Australian savannas.
Patch Dynamics and C. intratropica Vital Rates
The strongest impacts of fire on C. intratropica demography is via seedling and sapling mortality (Bowman and Panton 1993; Price and Bowman 1994; Russell-Smith 2006; Prior and others 2007, 2011). Adult trees are highly fecund (Figure 5B) and the species recruits abundantly in fire-protected sites (Bowman and others 1988; Lawes and others 2013). Yet even low-intensity fires significantly reduce seedling survival (Figure 5A; Russell-Smith 2006) and frequent savanna fires impose a strong recruitment bottleneck on C. intratropica (Price and Bowman 1994; Prior and others 2007). By maintaining positive population growth under low-intensity fires, our analyses indicate that intact groves are a key driver of population stability via their ability to exclude fires (Figure 6A). These dynamics highlight the importance of applying a patch-based approach to modelling C. intratropica demography that incorporates the fire exclusion feedback imposed by intact groves (Trauernicht and others 2012).
Several processes may be affecting C. intratropica growth. Higher growth rates of smaller C. intratropica singletons than of individuals of the same size occurring in intact groves (Figure 5) suggest that grove condition may be correlated with a density-dependent reduction in growth rate. Arrested growth in fire-protected, high-density C. intratropica stands supports this interpretation (Bowman and others 1988; Lawes and others 2013). In addition, reduced growth of large trees occurring as singletons versus those among intact groves may be due to accumulated canopy damage due to exposure to repeated fires (Bowman and others 2014). The limited effect of tree size on growth among degraded groves suggests that these trees may experience the worst of both worlds, with competition and canopy damage reducing growth for trees across all size classes. The legacy effects of canopy damage from previous fires, where living foliage is restricted to the upper most branches (Bowman and others 2014), probably also explain the negligible effects of high-intensity fires on growth rates of singletons and trees in degraded groves.
Whereas intact groves contribute disproportionately to C. intratropica seedling and sapling recruitment (Figure 1C), singleton trees conversely accounted for a larger proportion of the mortality events than adult trees occurring in groves, especially under high-intensity fires (Figure 4B). These findings support our hypothesis that degraded groves and singleton trees represent sequential stages in the process of C. intratropica decline. In the absence of the fire exclusion feedback, degraded groves and singleton trees do little to promote seedling/sapling survival (Figure 1C) and therefore do not represent intermediate stages of grove formation. Moreover, the dependence of seedling/sapling recruitment on fire exclusion suggests that most extant C. intratropica adults in open savanna vegetation—whether currently part of a grove or singletons—originated as part of a grove.
New groves of seedling/sapling cohorts are occasionally observed in the field (C. Trauernicht, pers. obs.). Any extant adult tree may contribute to grove formation as a seed source—our patch dynamic model allows for this by including the probability of transitioning from degraded groves and singletons to intact groves (Figure 3). However, new grove formation ultimately depends on the availability of habitat patches that remain unburnt long enough for seedling cohorts to establish and suppress fuels, and thereby alter the local disturbance regime. Habitat modelling suggests increasing the spatial heterogeneity of fire disturbance—which is also associated with regimes of low-intensity fires—can increase patch age diversity and thus the availability of older, unburnt habitat for C. intratropica recruitment (Trauernicht and others 2015). Yet the inevitability of high-intensity fires, even when low-intensity fires predominate, suggests that C. intratropica groves will not persist indefinitely and, therefore, the rate of grove establishment is a critical driver of grove dynamics (Figure S1d; see below) and long-term population stability.
Population Dynamics and Fire Feedbacks
Individual IPMs for each combination of fire disturbance and grove condition illustrate how under conditions of no fire, all grove types are capable of maintaining similar positive population growth (Figure 6A). The IPMs also revealed that despite little change in tree growth rates among degraded groves and singletons exposed to low-intensity fires (Figure 4B), the reduction in seedling survival (Figure 5A) is sufficient to project negative population growth (λ < 1; Figure 6A). Further increases in tree mortality under high-intensity (LDS) fires drove IPM projections well below levels of stable population behaviour across all grove conditions.
Combining fire-dependent transition probabilities among C. intratropica groves with fire frequencies observed in the field (environmental state matrix F) provided a landscape context from which to consider the individual IPMs. First, the difference between projected (SSDs) and observed proportions of grove conditions indicates that, under observed fire frequencies, the distribution of C. intratropica patches in the field is not in a stable state and is ‘drifting’ towards lower proportions of intact and degraded groves and higher proportions of singleton trees (Figure 6B). Given the high rates of singleton mortality, these projections alone suggest that the current fire regime will lead to C. intratropica extirpation. However, the patch dynamic model also suggested that increasing the prevalence of low-intensity fires may increase the abundance of intact C. intratropica groves (Figure 6B), which has been linked to greater structural heterogeneity and diversity of savanna vegetation (Trauernicht and others 2013).
Grove condition SSDs derived from environmental state matrix F were robust to altering transition probabilities (Figure S1), suggesting that our parameterizations were conservative with respect to the process of transitioning from intact groves to degraded groves to singleton trees with fire occurrence. Increasing ‘recruitment’ rates of intact groves (that is, transitions from degraded groves and singleton trees) dramatically altered projected grove proportions (Figure S1d). We based grove recruitment rates in our model on the frequency of patches of seedling/sapling cohorts (that is, new groves) relative to mature groves observed across a landscape of variable fire conditions (Trauernicht and others 2015). We therefore suggest that these estimates were similarly conservative because grove establishment under conditions of no fire was likely underestimated.
The megamatrix approach allowed us to effectively ‘insert’ the demographic models of C. intratropica (the IPMs) into the environmental state change matrix F (Horvitz and Schemske 1986; Valverde and Silvertown 1997; Pascarella and Horvitz 1998). This approach does not account for the full complexity of savanna dynamics. However, the megamatrix M provided a model of population behaviour that incorporates fire exclusion by intact groves and is ultimately driven by real world probabilities of low- and high-intensity fire occurrence. Given the evidence that management has little influence on annual area burned (that is, mean fire return interval) but has major effects on the intensity and heterogeneity of fire events (Gill and others 2000; Knapp and Keeley 2006; Russell-Smith and Edwards 2006; Archibald and others 2009), our model helps disentangle the effects of how a landscape burns (that is, fire severity sensu Keeley 2009) from simply how much of it burns (that is, area-based fire frequency sensu Li 2002).
The negative population growth (λ = 0.98) projected by the megamatrix under current frequencies of low- and high-intensity fires corroborates the documented declines in C. intratropica across northern Australia (Bowman and Panton 1993; Edwards and Russell-Smith 2009; Trauernicht and others 2013). Our findings also provide strong and novel evidence that increasing the frequency of low-intensity fires relative to high-intensity fires increases the population stability of C. intratropica (Figure 6C). Increasing the predominance of low-intensity fires contributed to C. intratropica population stability as the proportion of the total landscape burnt increases as well (Figure 6D). This supports the application of fire management to mitigate increases in overall fire occurrence, which appear to be driven more by climate than management practices (for example, Bliege Bird and others 2012; Murphy and others 2015).
Patch Burning
Our research indicates that understanding the influence of Aboriginal burning on Australian savannas requires exploring the effects of multiple fire regime metrics. In Australian savannas, there is a clear relationship between the abundance of intact C. intratropica groves and the severity and heterogeneity of fire disturbance, availability of refugia for other fire-sensitive species and overall structural complexity and plant diversity (Trauernicht and others 2012, 2013). The relationship between fire intensity and C. intratropica persistence evident in our model suggests a potential mechanism by which limiting high-intensity fires through patch burning promotes fire feedbacks that drive mosaics of fire-sensitive and fire-tolerant vegetation in fire-prone ecosystems. Indeed, recent analyses of the effects of prescribed burning programmes on the persistence of fire-sensitive vegetation in northern Australian savannas confirm these findings (Murphy and others 2015).
Our results illustrate the potential of human management to influence fundamental aspects of ecosystem composition through the intentional use of fire, a practice reported across a wide diversity of indigenous land management systems (Trauernicht and others 2015). Therefore, understanding the relationship between disturbance and species composition may require reshaping our perspective of what constitutes the ecological baseline in these systems. The persistence of species such as C. intratropica through more than 40,000 years of Aboriginal management strongly implicates the cessation of patch burning as a contributing factor to contemporary declines of birds, mammals and fire-sensitive plants in northern Australia (Franklin and others 2005; Edwards and Russell-Smith 2009; Woinarski and others 2010). Our findings provide critical insight into a millennial-scale relationship between human–fire interactions and ecosystem composition, and highlight the role of fire management in sustaining ‘natural’ systems.
References
Anderson DR, Burnham KP. 2002. Avoiding pitfalls when using information-theoretic methods. J Wildl Manag 66:912–18.
Archibald S, Roy DP, van Wilgen BW, Scholes RJ. 2009. What limits fire? An examination of drivers of burnt area in Southern Africa. Glob Change Biol 15:613–30.
Bessie W, Johnson E. 1995. The relative importance of fuels and weather on fire behavior in subalpine forests. Ecology 76:747–62. http://www.esajournals.org/doi/abs/10.2307/1939341. Last accessed 02/11/2013.
Bilbao BA, Leal AV, Mendez CL. 2010. Indigenous use of fire and forest loss in Canaima National Park, Venezuela. Assessment of and tools for alternative strategies of fire management in Pemón indigenous lands. Human Ecol 38:663–73.
Bliege Bird R, Codding BF, Kauhanen PG, Bird DW. 2012. Aboriginal hunting buffers climate-driven fire-size variability in Australia’s spinifex grasslands. Proc Natl Acad Sci USA 109:10287–92. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3387077&tool=pmcentrez&rendertype=abstract. Last accessed 13 Feb 2013.
Bond WJ. 2008. What limits trees in C 4 grasslands and savannas? Ann Rev Ecol Evol Syst 39:641–59. http://www.annualreviews.org/doi/abs/10.1146/annurev.ecolsys.39.110707.173411. Last accessed 11 Feb 2013.
Bowman D, McDermott HJ, Nichols SC, Murphy BP. 2014. A grass–fire cycle eliminates an obligate-seeding tree in a tropical savanna. Ecol Evol 4:4185–94. http://onlinelibrary.wiley.com/doi/10.1002/ece3.1285/full. Last accessed 02 Mar 2015.
Bowman DMJS, Franklin DC, Price OF, Brook BW. 2007. Land management affects grass biomass in the Eucalyptus tetrodonta savannas of monsoonal Australia. Aust Ecol 32:446–52.
Bowman DMJS, Panton WJ. 1993. Decline of Callitris intratropica R. T. Baker & H. G. Smith in the Northern Territory: implications for pre- and post-European colonization fire regimes. J Biogeogr 20:373–81.
Bowman DMJS, Wilson BA, Davis GW. 1988. Response of Callitris intratropica R.T. Baker & H.G. Smith to fire protection, Murgenella, northern Australia. Aust J Ecol 13:147–59.
Caswell H, Kaye T. 2001. Stochastic demography and conservation of an endangered perennial plant (Lomatium bradshawii) in a dynamic fire regime. Adv Ecol Res 32:1–51. http://www.sciencedirect.com/science/article/pii/S006525040132010X. Last accessed 10 Feb 2013.
Caswell H. 2006. Matrix population models. Sunderland, MA: Sinauer Associates, Inc. http://onlinelibrary.wiley.com/doi/10.1002/9780470057339.vam006m/full. Last accessed 17 Feb 2013.
Easterling MR, Ellner SP, Dixon PM. 2000. Size-specific sensitivity: applying a new structured population model. Ecology 81:694–708.
Edwards AC, Russell-Smith J. 2009. Ecological thresholds and the status of fire-sensitive vegetation in western Arnhem Land, northern Australia: implications for management. Int J Wildland Fire 18:127–46.
Ellair D, Platt W. 2013. Fuel composition influences fire characteristics and understorey hardwoods in pine savanna. J Ecol 101:192–201. http://onlinelibrary.wiley.com/doi/10.1111/1365-2745.12008/full. Last accessed 26 Feb 2013.
Ellner SP, Rees M. 2006. Integral projection models for species with complex demography. Am Nat 167:410–28.
Elston D, Moss R, Boulinier T. 2001. Analysis of aggregation, a worked example: numbers of ticks on red grouse chicks. Parisitology 122:563–9. http://journals.cambridge.org/production/action/cjoGetFulltext?fulltextid=82702. Last accessed 21 Feb 2013.
Franklin DC, Whitehead PJ, Pardon G, Matthews J, McMahon P, McIntyre D. 2005. Geographic patterns and correlates of the decline of granivorous birds in northern Australia. In: Fire and savanna management, Darwin, Australia, July 2002. pp 399–408.
Freeland WJ, Winter JW, Raskin S. 1988. Australian rock-mammals: a phenomenon of the seasonally dry tropics. Biotropica 20:70–9.
Gill AM, Ryan PG, Moore PHR, Gibson M. 2000. Fire regimes of world heritage Kakadu National Park, Australia. Aust Ecol 25:616–25.
Goldammer J. 1990. Fire in the tropical biota. Berlin: Springer http://scholar.google.com/scholar?hl=en&q=coutinho+fire+1990&btnG=&as_sdt=1,5&as_sdtp=#1. Last accessed 11 Feb 2013.
Govender N, Trollope WSW, Van Wilgen BW. 2006. The effect of fire season, fire frequency, rainfall and management on fire intensity in savanna vegetation in South Africa. J Appl Ecol 43:748–58.
Guyette RP, Muzika RM, Dey DC. 2002. Dynamics of an anthropogenic fire regime. Ecosystems 5:472–86.
Hawkins P. 1966. Seed production and litter fall studies of Callitris columellaris. Aust For Res 2:3–16. http://scholar.google.com/scholar?hl=en&q=hawkins+callitris&btnG=&as_sdt=1,5&as_sdtp=#0. Last accessed 17 Feb 2013.
Heisler J, Briggs J, Knapp A, Blair J, Seery A. 2004. Direct and indirect effects of fire on shrub density and aboveground productivity in a mesic grassland. Ecology 85:2245–57. http://www.esajournals.org/doi/pdf/10.1890/03-0574. Last accessed 19 Feb 2013.
Hoffmann WA, Adasme R, Haridasan M, de Carvalho MT, Geiger EL, Pereira M a B, Gotsch SG, Franco AC. 2009. Tree topkill, not mortality, governs the dynamics of savanna-forest boundaries under frequent fire in central Brazil. Ecology 90:1326–37. http://www.ncbi.nlm.nih.gov/pubmed/19537552.
Hoffmann WA. 1999. Fire and population dynamics of woody plants in a neotropical savanna: matrix model projections. Ecology 80:1354–69.
Horvitz C, Schemske D. 1986. Seed dispersal and environmental heterogeneity in a neotropical herb: a model of population and patch dynamics. In: Estrada A, Fleming T, editors. Frugivores and seed dispersal. Rotterdam: Springer Netherlands. pp 169–86. http://www.bio.miami.edu/horvitz/bil235/brasil08/HORVITZCC_SCHEMSKEDW1986_OP.PDF. Last accessed 16 Feb 2013.
Keeley JE, Fotheringham CJ, Baer-Keeley M. 2006. Demographic patterns of postfire regeneration in Mediterranean-climate shrublands of California. Ecol Monogr 76:235–55.
Keeley JE. 2009. Fire intensity, fire severity and burn severity: a brief review and suggested usage. Int J Wildland Fire 18:116–26.
Knapp EE, Keeley JE. 2006. Heterogeneity in fire severity within early season and late season prescribed burns in a mixed-conifer forest. Int J Wildland Fire 15:37–45.
Laris P. 2002. Burning the seasonal mosaic: preventative burning strategies in the wooded savanna of southern Mali. Human Ecol 30:155–86.
Lawes M, Taplin P, Bellairs S, Franklin D. 2013. A trade-off in stand size effects in the reproductive biology of a declining tropical conifer Callitris intratropica. Plant Ecol 214:169–74. http://cat.inist.fr/?aModele=afficheN&cpsidt=26697698. Last accessed 17 Feb 2013.
Lawes MJ, Richards A, Dathe J, Midgley JJ. 2011. Bark thickness determines fire resistance of selected tree species from fire-prone tropical savanna in north Australia. Plant Ecol 212:2057–69. http://www.springerlink.com/index/10.1007/s11258-011-9954-7. Last accessed 12 Feb 2013.
Li C. 2002. Estimation of fire frequency and fire cycle: a computational perspective. Ecol Model 154:103–20.
Mandle L, Ticktin T, Zuidema P. 2015. Resilience of palm populations to disturbance is determined by interactive effects of fire, herbivory and harvest. J Ecol 103:1032–43.
Menges E. 2000. Population viability analyses in plants: challenges and opportunities. Trends Ecol Evol 15:51–6. http://www.sciencedirect.com/science/article/pii/S0169534799017632. Last accessed 17 Feb 2013.
Murphy B, Cochrane MA, Russell-Smith J. 2015. Prescribed burning protects endangered tropical heathlands of the Arnhem Plateau, northern Australia. J Appl Ecol . doi:10.1111/1365-2664.12455.
Murphy BP, Bowman DMJS. 2007. The interdependence of fire, grass, kangaroos and Australian Aborigines: a case study from central Arnhem Land, northern Australia. J Biogeogr 34:237–50.
Murphy BP, Bowman DMJS. 2012. What controls the distribution of tropical forest and savanna? Ecol Lett 15:748–58.
Nowacki GJ, Abrams MD. 2008. The demise of fire and “mesophication” of forests in the eastern United States. Bioscience 58:123–38.
Pascarella J, Horvitz C. 1998. Hurricane disturbance and the population dynamics of a tropical understory shrub: megamatrix elasticity analysis. Ecology 79:547–63. http://www.esajournals.org/doi/abs/10.1890/0012-9658(1998)079[0547:HDATPD]2.0.CO;2. Last accessed 13 Feb 2013.
Price O, Bowman DMJS. 1994. Fire-stick forestry: a matrix model in support of skilful fire management of Callitris intratropica R.T. Baker by north Australian Aborigines. J Biogeogr 21:573–80.
Prior LD, Bowman DMJS, Brook BW. 2007. Growth and survival of two north Australian relictual tree species, Allosyncarpia ternata (Myrtaceae) and Callitris intratropica (Cupressaceae). Ecol Res 22:228–36.
Prior LD, McCaw WL, Grierson PF, Murphy BP, Bowman DMJS. 2011. Population structures of the widespread Australian conifer Callitris columellaris are a bio-indicator of continental environmental change. For Ecol Manag 262:252–62.
Roques K. 2001. Dynamics of shrub encroachment in an African savanna: relative influences of fire, herbivory, rainfall and density dependence. J Appl Ecol 38:268–80. http://onlinelibrary.wiley.com/doi/10.1046/j.1365-2664.2001.00567.x/full. Last accessed 19 Feb 2013.
Russell-Smith J, Edwards A. 2006. Seasonality and fire severity in savanna landscapes of monsoonal northern Australia. Int J Wildland Fire 15:541–50. http://www.publish.csiro.au/?paper=WF05111. Last accessed 08 Mar 2013.
Russell-Smith J, Edwards AC, Price OF. 2012. Simplifying the savanna: the trajectory of fire-sensitive vegetation mosaics in northern Australia. J Biogeogr 39:1303–7.
Russell-Smith J. 2006. Recruitment dynamics of the long-lived obligate seeders Callitris intratropica (Cupressaceae) and Petraeomyrtus punicea (Myrtaceae). Aust Bot 54:479–85.
Stubben C, Milligan B. 2007. Estimating and analyzing demographic models using the popbio package in R. J Stat Softw 22:1–23. http://www.jstatsoft.org/v22/i11/paper. Last accessed 19 Feb 2013.
Trauernicht C, Brook BW, Murphy BP, Williamson GJ, Bowman DMJS. 2015. Local and global pyrogeographic evidence that indigenous fire management creates pyrodiversity. Ecol Evol 5:1908–18.
Trauernicht C, Murphy BP, Portner TE, Bowman DMJS. 2012. Tree cover–fire interactions promote the persistence of a fire-sensitive conifer in a highly flammable savanna. J Ecol 100:958–68.
Trauernicht C, Murphy BP, Tangalin N, Bowman DMJS. 2013. Cultural legacies, fire ecology, and environmental change in the Stone Country of Arnhem Land and Kakadu National Park, Australia. Ecol Evol 3:286–97.
Valverde T, Silvertown J. 1997. An integrated model of demography, patch dynamics and seed dispersal in a woodland herb, Primula vulgaris. Oikos 80:67–77.
Whitehead PJ, Bowman DMJS, Preece N, Fraser F, Cooke P. 2003. Customary use of fire by indigenous peoples in northern Australia: its contemporary role in savanna management. Int J Wildland Fire 12:415–25.
Van Wilgen BW, Govender N, Biggs HC, Ntsala D, Funda XN. 2004. Response of savanna fire regimes to changing fire management policies in a large African national park. Conserv Biol 18:1533–40.
Van Wilgen BW, Govender N, MacFadyen S. 2008. An assessment of the implementation and outcomes of recent changes to fire management in the Kruger National Park. Koedoe 50:22–31.
Woinarski JCZ, Armstrong M, Brennan K, Fisher A, Griffiths AD, Hill B, Milne DJ, Palmer C, Ward S, Watson M, Winderlich S, Young S. 2010. Monitoring indicates rapid and severe decline of native small mammals in Kakadu National Park, northern Australia. Wildl Res 37:116–26.
Woinarski JCZ, Hempel C, Cowie I, Brennan K, Kerrigan R, Leach G, Russell-Smith J. 2006. Distributional pattern of plant species endemic to the Northern Territory, Australia. Aust J Bot 54:627–40.
Wood SW, Murphy BP, Bowman D. 2011. Firescape ecology: how topography determines the contrasting distribution of fire and rain forest in the south-west of the Tasmanian Wilderness World Heritage Area. J Biogeogr 38:1807–20.
Yates C, Edwards A, Russell-Smith J. 2008. Big fires and their ecological impacts in Australian savannas: size and frequency matters. Int J Wildland Fire 17:768–81.
Yibarbuk D, Whitehead PJ, Russell-Smith J, Jackson D, Godjuwa C, Fisher A, Cooke P, Choquenot D, Bowman DMJS. 2001. Fire ecology and Aboriginal land management in central Arnhem Land, northern Australia: a tradition of ecosystem management. J Biogeogr 28:325–43.
Zuidema PA, Jongejans E, Chien PD, During HJ, Schieving F. 2010. Integral Projection Models for trees: a new parameterization method and a validation of model output. J Ecol 98:345–55.
Zuur A, Ieno E, Walker N, Saveliev A, Smith G. 2009. Mixed effects models and extensions in ecology with R. New York: Springer http://books.google.com/books?hl=en&lr=&id=vQUNprFZKHsC&oi=fnd&pg=PA1&dq=zuur+mixed+models&ots=kawEBQ2D4o&sig=7TqA_N3YpS1Ha-GIA7yNhaykERo. Last accessed 13 Feb 2013.
Acknowledgments
We are grateful to the Rostron family, Wesley Campion and the Djelk Aboriginal Ranger group for support and permission to work in Arnhem Land. These analyses would not have been possible without the initial guidance of Orou Gaoue and Carol Horvitz, made possible by a grant to CT from the National Climate Change Adaptation Research Facility of Australia, as well as the patient feedback of Lisa Mandle. Thanks to Talia Portner, Fay Johnston and Clancy Bowman for help in the field. Grants from the Commonwealth Environmental Research Facilities (CERF) Program of Australia, ANZ Holsworth Endowment, Australian Geographic, the Ecological Society of Australia and the Australian Research Council (LP110200006 and DE130100434) funded this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
Clay Trauernicht performed research, analysed data, contributed new models and wrote the paper. Brett Murphy performed research and analysed data. Mike Lawes performed research and analysed data. Lynda Prior designed study and performed research. David Bowman conceived of study and performed research.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Trauernicht, C., Murphy, B.P., Prior, L.D. et al. Human-Imposed, Fine-Grained Patch Burning Explains the Population Stability of a Fire-Sensitive Conifer in a Frequently Burnt Northern Australia Savanna. Ecosystems 19, 896–909 (2016). https://doi.org/10.1007/s10021-016-9973-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-016-9973-2