Abstract
Background
Reports of post herpetic maxillofacial complications have been very rarely documented in the literature that includes periapical lesions, calcified and devitalized pulps, resorption of roots, osteonecrosis, and spontaneous exfoliation of teeth. The atypical feature of the case of concern to the dental surgeon is the rare complication of spontaneous tooth exfoliation following herpes zoster.
Case report
This case reports a male patient of age 47 years who reported to the Department of Periodontology with the chief complaint of mobility in the left upper central incisor. Patient history revealed herpes zoster infection that began 11 days earlier along with underlying diabetes mellitus condition. We hereby report a known diabetic patient with history of herpes zoster infection who presented with rare complication of spontaneous tooth exfoliation involving the maxillary division of the trigeminal nerve.
Discussion
Limited number of cases has been reported in the literature regarding spontaneous teeth exfoliation secondary to herpes zoster. The exact pathogenesis regarding the spontaneous exfoliation of teeth in herpes zoster patient is still controversial. Thus, an oral health care provider should be aware of this rare complication while managing a case of tooth mobility with the previous history of herpes zoster of trigeminal nerve.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The varicella-zoster virus is a ubiquitous human alpha herpes virus, which causes two clinical entities varicella (chicken pox) and herpes zoster (shingles). Varicella (or chickenpox) is a benign illness of childhood as a result of the primary infection of varicella-zoster virus. It is a highly contagious infection and is characterized by an exanthematous vesicular rash. When the varicella infection occurs, virus in the skin travels up the sensory nerves to become latent in the sensory ganglia [1, 2]. Once the varicella-zoster virus in its latent state is reactivated in the dorsal root ganglia, it results the inception of herpes zoster (HZ) infection that results in a localized cutaneous eruption [3] .Herpes virus (herpes, creeping) describes the nature of the pathologic lesion [4]. Little is known about the mechanisms that account for preservation of latency and subsequent reactivation of the virus. The spread of virus occurs through nerves to the skin, and a vesicular cutaneous eruption develops in the area innervated by the affected sensory nerve after the reactivation and active viral replication in the sensory ganglia. Among the cranial nerves affected by herpes zoster infection, trigeminal nerve is the most commonly affected (18.5 to 22 % of total cases), followed by glossopharyngeal nerve and hypoglossal nerve. It is conceivable that intraorally, symptoms appear when the second (maxillary) or third (mandibular) trigeminal divisions are affected [2, 5, 6].
Reports of post herpetic maxillofacial complications have been very rarely documented in the literature that includes periapical lesions, calcified and devitalized pulps, resorption of roots, osteonecrosis, and spontaneous exfoliation of teeth. Spontaneous exfoliation of teeth in the area innervated by the affected nerve has been reported rarely in the literature. It is considered to be an early [7, 8] episode by some researchers, while others consider it as delayed [5] complication of herpes zoster infection. We hereby report a case of spontaneous tooth exfoliation in a patient suffering from herpes zoster infection with underlying diabetic systemic condition.
Case description
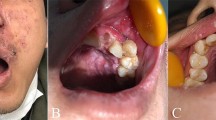
A 47-year-old male patient reported to the Department of Periodontology, Himachal Dental College, Sundernagar, with the chief complaint of mobility in the left upper central incisor. Patient also complained of numbness and tingling sensation in the upper left quadrant. Extraoral examination revealed residual crusty lesions of herpes infection over the left side of the face restricted to the distribution of the maxillary division of trigeminal nerve with no associated lymphadenopathy was noticed. There were also areas of healing with scar formation and hyperpigmentation (Fig. 1 and Fig. 2). On intraoral examination, slight redness of the alveolar mucosa was seen around the left upper central incisor along with grade III mobility with no alterations in the remaining teeth of the upper left quadrant (Fig. 3). Patient history revealed herpes zoster infection that began 11 days earlier with typical itching and paraesthetic sensation. After 3 days, vesicles appeared on an erythematous zone affecting the skin over the left side of face. The patient was being treated with oral (800 mg/daily for 10 days) acyclovir for the herpes zoster infection. Patient past medical history exhibits his underlying diabetes mellitus condition. He was on medication for the same from the past few years. He was asked to bring all the medical records, but the patient reported next day with exfoliated upper left incisor tooth. The intraoral examination revealed normal alveolar socket with no purulent exudates. The radiological examination revealed empty tooth socket on the left side of the maxilla with no sequestrum (Fig. 4).
Routine hematologic test investigations revealed marked lymphopenia with reactive lymphocytes present, suggestive of viral infection. Other laboratory investigations were within the normal limits (HIV, hepatitis B, and routine urine analysis). The values of fasting blood sugar were slightly raised (140 mg/dl). The patient was prescribed on antibiotic therapy (amoxicillin 500 mg, three times daily for 10 days) and was instructed to rinse with 0.12 % chlorhexidine digluconate.
Discussion
Herpes zoster affects all ages, but has been seen to be rare in children and adolescents [2]. It has no gender predilection but there is a slight male predominance [2, 5, 6], and the onset commonly occurs among individuals in the sixth through the eighth decades of life (five to 10 cases per 1000 persons) [5]. The incidence of HZ infection increases with age and in general population has been reported to be 5.4 % [9] while it has found to be up to 10 times higher in individuals suffering with HIV infection [10]. Moreover, studies suggest that prevalence of herpes zoster infection increases among immunocompromised patients such as patients infected with HIV infection, hematologic malignant disease, immune-mediated disorder, and organ transplant patients, and the risk of herpes zoster infection for such immune-suppressed patients also increases according to the age [6, 11, 12]. Other risk factors take account of external damage of the affected dermatomes, psychological stress, and race [12]. Among the affected dermatomes, it has been found that most commonly affected dermatomes are the thoracic, cervical, and trigeminal dermatome [11].
Intraorally, the lesions of herpes zoster appear when the mandibular or maxillary divisions of the trigeminal nerve are affected. The lesions of herpes zoster infection can appear on the face, in the mouth, in the eye, or on the tongue when the branches of the trigeminal nerve are involved [2]. The herpes zoster infections usually undergo three stages that includes the following: (1) Prodromal stage presenting as the sensations described as burning, tingling, itching, boring, prickly or knife like occurring in the skin over the affected nerve distribution which is usually preceded by the rash of the active stage by few hours or several days. (2) Active stage (or the acute stage) characterized by the emergence of the rash which progresses from erythematous papules and oedema to vesicles in 12 to 24 h and finally progresses to pustules within 1 to 7 days. The intraoral vesicles, scattered and surrounded by an erythematous zone, soon become ulcerated and covered by a white pseudo membrane. The vesicles dry and crust over after 1 to 2 weeks, but it may take several weeks for the skin to return to normal. (3) Chronic stage also termed as post herpetic neuralgia is the most significant complication of herpes zoster infection [2, 3, 8, 13, 14].
Besides herpes zoster infection, other predisposing conditions have included HIV/AIDS, malignancy, chemotherapy, radiation therapy, and diabetes mellitus [15]. Published reports have demonstrated increased incidence of herpes zoster in diabetic patients [16, 17]. In diabetic patients, occlusion of the vessels leading to tissue ischemia is present with associated predisposition to infection. Impairment of the neutrophil function particularly chemotaxis can be another contributing factor [15, 18]. It has been hypothesized that the increased risk for herpes zoster among patients with diabetes mellitus may also be related to decrease varicella zoster virus (VZV)-specific cell-mediated immunity. Yet more relevant studies are required to find the exact mechanism by which diabetes mellitus affects VZV-specific cell-mediated immunity [17]. The case reported in this paper had the medical history of diabetes which could be one of the predisposing factors to spontaneous tooth exfoliation.
The maxillofacial complications of herpes zoster infection including the spontaneous exfoliation of the teeth and the osteonecrosis have been described as rare phenomena [2, 6, 19]. These cases have rarely been reported in the literature, and a summary of the available data on patients with trigeminal herpes zoster infection associated with the exfoliation of teeth has been given in Table 1.
The first case concerning the relation of osseous alterations with the herpes zoster infection was reported in the literature by Rose in 1908 [2]. In 1922, it was Dechaume [20] et al. that accredited Gonnet as the pioneer to have reported on this rare complication. The enigma still remains regarding the time interval amid the outbreak of herpes zoster infection and the spontaneous exfoliation of teeth in the area innervated by the affected nerve. Published reports have demonstrated that spontaneous tooth exfoliation and bone necrosis occur as early as the first 2 weeks of initial infection while other authors consider this to be a delayed complication that will occur between the third and twelfth weeks after the onset [5, 7, 8, 15, 21, 22]. In the present case, tooth exfoliation occurred within 2 weeks of initial herpes zoster infection.
The underlying rationale for the spontaneous exfoliation of teeth can be attributed to the alveolar bone necrosis and/or to necrosis of the periodontal ligament [2, 23]. The uncertainty still exists regarding the process by which the HZ infection leads to the alveolar bone necrosis. Some researchers believe that it could be due to local vasculitis caused by direct extension of the neural inflammatory process to adjacent blood vessels and infarction of trigeminal vessels that accompany the trigeminal vessels supplying the jaws [24] or generalized infection of the trigeminal nerves supplying the periosteum and periodontium of the involved dermatome area [7]. Perhaps an ischemic problem with these attributes could result in more than one tooth affected [2], while others are of the opinion that pre-existing pulpal or periodontal inflammatory conditions or surgical procedures performed in the site of zoster infection [24, 25] or systemic viral infection of the odontoblasts causing degenerative tissue changes that could contribute to more destructive alveolar bone necrosis [5].
The management of herpes zoster infection includes treatment with antiviral agents that have been shown to reduce the duration of the rash and severity of pain associated. Yet it has been reported that the benefits have only been seen in patients who were prescribed antiviral agents within 72 h after the onset of the rash [10]. The most significant complication of herpes zoster infection is post herpetic neuralgia which is generally a self-limited condition. The most debilitating complication of herpes zoster is osteonecrosis which is a rare phenomenon. Timely commencement of antiviral therapy and aggressive analgesic treatment can prevent this post herpetic complication of osteonecrosis. In cases of secondary infection with osteonecrosis, sequestrectomy along with the removal of inflammatory tissue and regular patient follow-up must be done [2, 5, 6]. When osteonecrosis of the jaw is accompanied with tooth exfoliation as rare complications, then active use of painkillers, regulation of topical factors, and proper extraction of dead bone and affected teeth can lead to better treatment outcomes [6].
In the present case scenario, the patient had no periapical lesion or periodontal involvement as depicted on radio graphical examination yet the authors are of the opinion that patients underlying diabetic condition may have contributed to the spontaneous tooth exfoliation along with herpes zoster infection. Thus, an oral health care provider should be aware of this rare complication while managing a case of tooth mobility with the previous history of herpes zoster of trigeminal nerve.
References
Tyler KL (1998) Aseptic meningitis, viral encephalitis and prion diseases. In: Fauci AS et al. (eds) Harrison’s principles of internal medicine, 17th edn. McGraw-Hill, New York, pp. 2439–2451
Mendieta C, Miranda J, Brunet LI, Gargallo J, Berini L (2005) Alveolar bone necrosis and tooth exfoliation following herpes zoster infection: a review of the literature and case report. J Periodontol 76:148–153
Gnann JW Jr, Whitley RJ (2002) Clinical practice. Herpes zoster. N Engl J Medn 34:340–346. doi:10.1056/NEJMcp1302674
Slots J (2005) Herpes viruses in periodontal diseases. Periodontol 2000(38):33–62
Jain MK, Manjunath KS, Jagadish SN (2010) Unusual oral complications of herpes zoster infection: report of a case and review of literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 110:e37–e41. doi:10.1016/j.tripleo.2010.04.026
Kim NK, Kim BC, Nam JW, Kim HJ (2012) Alveolar bone necrosis and spontaneous tooth exfoliation associated with trigeminal herpes zoster: a report of three cases. J Korean Assoc Oral Maxillofac Surg 38:177–183
Muto T, Tsuchiya H, Sato K, Kanazawa M (1990) Tooth exfoliation and necrosis of the mandible—a rare complication following trigeminal herpes zoster: report of a case. J Oral Maxillofac Surg 48:1000–1003
Schwartz O, Kvorning SA (1982) Tooth exfoliation, osteonecrosis of the jaw and neuralgia following herpes zoster of the trigeminal nerve. Int J Oral Surg 11:364–371
Hope-Simpson RE (1965) The nature of herpes zoster. A long term study and new hypothesis. Proc R Soc Med 58:9–20
Bandral MR, Chidambar YS, Telkar S, Japatti S, Choudary L, Dodamini A (2010) Oral complications of herpes zoster infection—report of 3 cases. Int J Dent Clin 2:70–73
Gershon AA, Gershon MD, Breuer J, Levin MJ, Oaklander AL, Griffiths PD (2010) Advances in the understanding of the pathogenesis and epidemiology of herpes zoster. J Clin Virol 48:S2–S7
Schmader KE, Dworkin RH (2008) Natural history and treatment of herpes zoster. J Pain 9:S3–S9
Hornstein OP, Gorlin RJ (1973) Oral infectious diseases. In: Gorlin RJ, Goldman HM (eds) Thoma’s oral pathology, edn. Salvat, Barcelona, pp. 822–824
Weaver BA (2009) Herpes zoster overview: natural history and incidence. J Am Osteopath Assoc 109:S2–S6
Meer S, Coleman H, Altini M, Alexander T (2006) Mandibular osteomyelitis and tooth exfoliation following zoster-CMV co-infection. Oral Surg Oral Med Oral Radiol Endod 101:70–75
Aldaz P, Díaz JA, Loayssa JR, Dronda MJ, Oscáriz M, Castilla J (2013) Herpes zoster incidence in diabetic patients. An Sist Sanit Navar 36:57–62
Chernev I, Gomez E (2014) Herpes zoster and diabetes mellitus. Korean J Pain 27:92. doi:10.3344/kjp.2014.27.1.92
Murrah VA (1985) Diabetes mellitus and associated oral manifestations: a review. J Oral Pathol 14:271–281. doi:10.1111/j.1600-0714.1985.tb00494.x
Okumara K, Inui M, Inoue J, Wakabayashi H, Tagawa T (2015) A case of herpes zoster associated with meningoencephalitis and tooth exfoliation by alveolar bone resorption. J Oral Maxfac Surg Med Pathol 27:61–64. doi:10.1016/j.ajoms.2013.07.009
Dechaume M, Descrozailles C, Garlopeau F, Robert J (1955) Localized mandibular necrosis during herpes zoster infection (in French). Rev Stomatol 56:516–521
Mintz SM, Anavi Y (1992) Maxillary osteomyelitis and spontaneous tooth exfoliation after herpes zoster. Oral Surg Oral Med Oral Pathol 73:664–666
Siwamogstham P, Kuansuwan C, Reichart PA (2006) Herpes zoster in HIV infection with osteonecrosis of the jaw and tooth exfoliation. Oral Dis 12:500–505
Toshitaka M, Tsuchiya H, Sato K, Kanazawa M (1990) Tooth exfoliation and necrosis of the mandible—a rare complication following trigeminal herpes zoster: report of a case. J Oral Maxillofac Surg 48:1000–1003
Wright WE, Davis ML, Geffen DB, Martin SE, Nelson MJ, Straus SE (1983) Alveolar bone necrosis and tooth loss. A rare complication associated with herpes zoster infection of the fifth cranial nerve. Oral Surg Oral Med Oral Pathol 56:39–46
Cooper JC (1977) Tooth exfoliation and osteonecrosis of the jaw following herpes zoster. Br Dent J 143:297–300
Delaire J, Billet J (1959) New observations of alveolar necrosis during a zona of the mandibular nerve (in French). Rev Stomatol 60:550–553
Hall HD, Jacobs JS, O’Malley JP (1974) Necrosis of maxilla in a patient with herpes zoster. Oral Surg Oral Med Oral Pathol 37:657–662. doi:10.1016/0030-4220(74)90128-5
Chemitz JE (1976) Herpes zoster in Hodgkin’s disease: unusual oral sequelae. J Dent Child 43:181–186
Vickery IM, Midda M (1976) Dental complications of cytotoxic therapy in Hodgkin’s disease. Br J Oral Surg 13:282–288
Delbrouck-Poot F, Reginster JP (1979) Trigeminal herpes zoster and maxillary necrosis. Dermatologica 158:210–213
Manz HJ, Canter HG, Melton J (1986) Trigeminal herpes zoster causing mandibular osteonecrosis and spontaneous tooth exfoliation. South Med J 79:1026–1028
Mostofi R, Marchmont-Robinson H, Freije S (1987) Spontaneous tooth exfoliation and osteonecrosis following a herpes zoster infection of the fifth cranial nerve. J Oral Maxillofac Surg 45:264–266. doi:10.4103/0019-5154.110878
Penarrocha M, Bagan JV, Sanchis JM (1992) Herpes zoster and maxillary osteonecrosis (in Spanish). Rev Actual Odontoestomatol Esp 418:49–55
Owotade FJ, Ugboko VI, Kolude B (1999) Herpes zoster infection of the maxilla: case report. J Oral Maxillofac Surg 57:1249–1251. doi:10.1016/S0278-2391(99)90497-4
Lambade P, Lambade D, Saha TK, Dolas RS, Pandilwar PK (2012) Maxillary osteonecrosis and spontaneous teeth exfoliation following herpes zoster. Oral Maxillofac Surg 16:369–372. doi:10.1007/s10006-011-0303-8
Mahajan VK, Ranjan N, Sharma S, Sharma NL (2013) Spontaneous tooth exfoliation after trigeminal herpes zoster: a case series of an uncommon complication. Indian J Dermatol 58:244. doi:10.4103/0019-5154.110878
Cloarec N, Zaegel-Faucher O, Bregigeon S et al (2014) Mandibular osteonecrosis and dental exfoliation after trigeminal zoster in an HIV-infected patient: case report and review of literature. AIDS 28:448–449. doi:10.1097/QAD.0000000000000122
Patil S, Al Zarea BK (2015) Spontaneous exfoliation and osteonecrosis following herpes zoster infection in an HIV-seropositive subject: a case report. Austin J Dent 2:1015
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaur, R., Rani, P., Malhotra, D. et al. A rare case report and appraisal of the literature on spontaneous tooth exfoliation associated with trigeminal herpes zoster. Oral Maxillofac Surg 20, 331–336 (2016). https://doi.org/10.1007/s10006-016-0561-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10006-016-0561-6