Abstract
Ileal pouch-anal anastomosis (IPAA) is the operation of choice following proctocolectomy for patients who suffer from ulcerative colitis and familial adenomatous polyposis. The aim of this study was to morphologically examine the neurons, endocrine cells and mast cells in the ileum of rats subjected to proctocolectomy followed by three different types of ileoanal anastomosis. Rats were subjected to either sham operation or proctocolectomy followed by ileoanal anastomosis end-to-end, side-to-end or IPAA (J-pouch). In comparison to sham-operated rats, the body weight was reduced in rats that underwent proctocolectomy with end-to-end or side-to-end, but not IPAA procedure. In all three models of ileoanal anastomosis, the ileum displayed crypt hyperplasia with a chronic inflammatory infiltrate located in the interstitium, hyperplasia of goblet cells, but reduced protein gene product 9.5 (PGP 9.5)-immunoreactive neurons in the mucosa as well as submucosa. Numbers of endocrine cells in the mucosa (chromogranin A immunostaining) and mast cells in the mucosa and submucosa (Astra blue staining) were unchanged after proctocolectomy. In conclusion, neurons, but neither endocrine cells nor mast cells, were reduced in the ileum of proctocolectomized rats followed by either of three different types of ileoanal anastomosis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Proctocolectomy is a definitive therapy for ulcerative colitis, reducing symptoms and removing the risk of colonic adenocarcinoma. Proctocolectomy with end ileostomy or continent ileostomy (Kock pouch) was the common procedure. As clinical experience accumulated, ileal pouch-anal anastomosis (IPAA, J-pouch) has become the most common operation up to date. Potential complications after the surgery are pelvic sepsis, intestinal obstruction, fistulas and pouchitis [1–3]. Pouchitis, which is a symptomatic inflammation in the reservoir, is the most frequent complication. In the long term, 40–50 % of the IPAA patients have one or more episodes of pouchitis [4–12]. Severe pouchitis may be accompanied by disabling symptoms and sometimes requiring proximal faecal diversion or reservoir removal [2, 13].
Dysplasia in the reservoir and carcinoma in the ileal pouch mucosa have been described [14–17]. Chronic inflammation displaying persistent severe atrophy in the pouch mucosa has been suggested to be at an increased risk for development of neoplastic transformations in the reservoir [15], although others reported that the risk of developing dysplasia and cancer might be low [17, 18].
The mechanisms behind the developments of pouchitis and of dysplasia have been studied mostly in man with respect to inflammatory mediators, adhesion molecules, matrix metalloproteinases, substance P, vasoactive intestinal polypeptide, neutrophils and mast cells, but not yet neurons and endocrine cells [19–25]. In the present study, we wanted to establish animal models of pouchitis in rats that underwent proctocolectomy followed by three types of ileoanal anastomosis, i.e., end-to-end (EEA), side-to-end (SEA) and IPAA, and then examine the ileum with focus on morphological changes in inflammation, neurons, endocrine cells and mast cells.
Materials and methods
Animals and experimental groups
Male Sprague–Dawley rats were purchased from Møllegaard (Skensved, Denmark), and housed in plastic cages (4 in each cage) under controlled conditions: 12 h/12 h light/dark, 20 °C and 40–60 % humidity. They were fed a standard rat food pellets (B&K universal, Norway) and had access ad libitum to filled tap water.
Forty rats were randomly divided into four groups: sham operation (controls), proctocolectomy with EEA, SEA, and IPAA (J-pouch) (Fig. 1a–c). Animals were operated and killed at 1 month postoperatively under anaesthesia with a subcutaneous injection of 0.02 ml/kg of a solution containing 2.5 mg/ml fluanison, 0.05 mg/ml fentanyl (Tamson Animal Health, Buckinghamshire, UK) and 1.25 mg/ml midazolam (Alpharma AS. Oslo, Norway). The animal experiment was performed after approval by the Norwegian National Animal Research Authority.
Surgical drawing of rat models of proctocolectomy with end-to-end ileoanal anastomosis (EEA) (a), side-to-end ileoanal anastomosis (SEA) (b) and ileal J-pouch-anal anastomosis (IPAA) (c), and body weight at 1 month postsurgery (d). Mean ± SEM, *p < 0.05, **p = 0.01; NS not significant in comparison with sham-operated controls
Histology and immunohistochemistry
At euthanization, the tissue samples (ileum) were taken at 0.5–1 cm proximal to the ileocecal valve [26] and fixed in 4 % paraformaldehyde for 12 h at 4 °C. Formalin fixed samples were dehydrated and embedded in paraffin. Sections were cut at 4 μm thickness in a microtome and thawed onto Poly-l-Lysine-coated glass slides and allowed to dry over night. Goblet cells were stained by Periodic acid–Schiff (PAS) method. The sections were then incubated with periodic acid (Bancroft) for 2 min. After rinsed in distilled water the sections were incubated with Schiff’s reagent for 8 min and then rinsed in running tap water at 37 °C for 15 min. Sections were then incubated in hematoxylin for 2 min, rinsed in water for 5 min and then mounted in Assistent-Histokitt mounting media (Germany). Neurons and endocrine cells were examined by immunohistochemistry with antisera against PGP 9.5 and chromogranin A [27]. Immunohistochemistry was performed by the avidin–biotin complex (ABC) method using commercially available Vectastain® ABC kit (Vector Laboratories Inc., CA94010, USA). Slides were first dewaxed in xylen and rinsed in graded alcohol and then rehydrated in distilled water. Thereafter, antigen retrieval was performed using a commercial pressure cooker, boiling the sections for 1 min in 0.01 M citrate buffer (pH 6.0). Endogenous peroxidase activity was then blocked with 0.3 % hydrogen peroxide applied on the sections for 30 min and then rinsed in water for 5 min and washed in PBS buffer for 5 min. The sections were then incubated with normal serum from the ABC kit for 30 min and then incubated overnight with primary antisera: PGP 9.5 (code no. Z5116, DAKO, Denmark) in a final dilution of 1:30 and chromogranin A (code no.20086, DAKO, Denmark) in a final dilution of 1:500. The primary antibodies were diluted in PBS containing 0.25 % Triton X-100 and bovine serum albumin and incubated overnight at 4 °C. After washing in PBS for 10 min, the sections were incubated with a biotinylated antibody from the ABC-kit for 30 min, and then rinsed in PBS for 10 min and incubated with the ABC reagent for 30 min. After washing in PBS for 10 min the sections were incubated with 3-amino-9-ethylcarbazole (AEC, Vector Laboratories) as chromogen. The section was rinsed in water and counter-stained with hematoxylin for 1 min and rinsed in water for 5 min before mounting in mounting media (PBS:glycerol,1:4, DAKO) and examination under light microscope (Zeiss, Germany). The mast cells in mucosa and submucosa were stained with Astra Blue. Samples were incubated with Carnoy fixative for 2 h and then kept in 80 % alcohol overnight, dehydrated, paraffin embedded and then cut at 4 μm thickness in a microtome and thawed onto Poly-l-Lysine-coated glass slides and kept in a heating shelter over night at 37 °C to dry. Sections were incubated with 1 % Astra blue solution (pH 0.3) for 30 min, rinsed two times in 0.7 M HCl to decrease the background colour, and then counter-stained with 0.5 % Safranin O in 0.125 M HCl and mounted in Assistent-Histokitt mounting media (Germany).
All sections were examined under the light microscope (Zeiss) and minimum 5 randomly selected fields displaying full wall thickness were selected for morphometrical analysis. The number of immunoreactive cells at magnification 10× or 40× was counted and expressed as number of cells per visual field or volume density in percentage of the mucosa and submucosa.
Statistical analysis
The values are expressed as mean ± SEM. Statistically significant difference between sham-operated control and each of proctocolectomy procedures (EEA, SEA or IPAA) was determined by a one-way ANOVA followed by Dunnett’s test using SPSS version 15.0 (SPSS Inc., Chicago, IL, USA). A p value of <0.05 was considered statistically significant.
Results
In comparison with sham-operated control rats, rats that underwent proctocolectomy with EEA or SEA, but not IPAA, had less body weight (Fig. 1d). The rats with less body weight suffered from diarrhoea.
The proctocolectomized rats with EEA or SEA showed mild villous atrophy and numerous immune cells in the lamina propria in the area adjacent to the anastomosis. In IPAA rats, there was severe villous atrophy, mild crypt hyperplasia with a chronic inflammatory infiltrate located in the interstitium. Some neutrophils scattered around in the lamina propria were seen. The glandular structures were not infiltrated with immune cells. Muscularis propria was increased. The IPAA rats displayed macroscopic changes with great dilatation of the ileum proximal to the anastomosis (Fig. 2a–d). The ileal reservoir of IPAA contained large amounts of faeces.
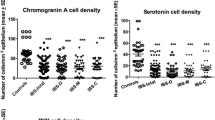
The goblet cells were increased in all proctocolectomized rats (Fig. 3). The volume density of PGP 9.5-positive neurons in the mucosa as well as submucosa was markedly reduced in all proctocolectomized rats (Fig. 4). The number of endocrine cells (chromogranin A-immunoreactive cells) in the mucosa was unchanged after either procedure of proctocolectomy (Fig. 5). The numbers of mast cells in the mucosa as well as submucosa were unchanged after either procedure of proctocolectomy, except for the number of mast cells in the mucosa which was statistically reduced after EEA (Fig. 6).
Representative micrographs of PAS staining in rats subjected to sham operation (a), proctocolectomy with end-to-end ileoanal anastomosis (EEA) (b), side-to-end ileoanal anastomosis (SEA) (c) and ileal J-pouch-anal anastomosis (IPAA) (d). Note: example goblet cells indicated by arrows in a and d. Bars 50 μm. Numerical density (e) in rats subjected to sham operation, EEA, SEA and IPAA. Mean ± SEM, **p < 0.01, ***p = 0.001 in comparison with sham-operated controls
Representative micrographs of PGP 9.5 immunostaining in rats subjected to sham operation (a), proctocolectomy with end-to-end ileoanal anastomosis (EEA) (b), side-to-end ileoanal anastomosis (SEA) (c) and ileal J-pouch-anal anastomosis (IPAA) (d). Bars 50 μm. Note: example neurons indicated by arrows in a. Bars 50 μm. Volume density in the mucosa (e) and in the submucosa (f) in rats subjected to sham operation, EEA, SEA, and IPAA. Mean ± SEM, **p < 0.01, ***p = 0.001 in comparison with sham-operated controls
Representative micrographs of chromogranin A immunostaining in rats subjected to sham operation (a) and ileal J-pouch-anal anastomosis (IPAA) (b). Note: endocrine cell in the mucosa indicated by arrows in a and b. Bars 50 μm. Numerical density in the mucosa (c) in rats subjected to sham operation, in rats subjected to sham operation (Sham), proctocolectomy with end-to-end ileoanal anastomosis (EEA), side-to-end ileoanal anastomosis (SEA) and IPAA. Mean ± SEM, NS not significant in comparison with sham-operated controls
Representative micrographs of Astra blue staining in rats subjected to sham operation (a) and ileal J-pouch-anal anastomosis (IPAA) (b). Note: mast cells in the mucosa and submucosa indicated by arrows in a and b. Bars 50 μm. Numerical density in the mucosa (c) and the submucosa (d) in rats subjected to sham operation, proctocolectomy with end-to-end ileoanal anastomosis (EEA), side-to-end ileoanal anastomosis (SEA) and IPAA. Mean ± SEM, *p < 0.05; NS not significant in comparison with sham-operated controls
Discussion
This report describes rat models of three different fashions of ileal-anal anastomoses for studying ileal mucosa adaptation to proctocolectomy. The proctocolectomized rats with EEA or SEA, but not ileal J-pouch-anal anastomosis, suffered from diarrhoea and had less body weight, which could be explained by an adapted absorption in the J-pouch. The histological changes including villous atrophy and crypt hyperplasia with mild chronic inflammation were similar to the changes in the ileal mucosa in the reservoirs of patients who do not have symptoms of pouchitis.
Goblet cells are known to be important contributors to the mucosal defence barrier with their secretion of the protective sialomucin. In the present study, the IPAA postoperative morphological changes and the increase of goblet cells in the mucosa are in line with the observations in patients [28–30].
PGP 9.5-immunoreactive neurons (but not PGP 9.5-positive epithelial cells) were markedly reduced in the lamina propria of the ileal mucosa and the submucosa, including the submucosal nerve plexus and the myenteric nerve plexus. Mechanisms by which the neurons in the ileum were reduced after proctocolectomy are unknown, but this observation is of interest in further studying the risk of tumorigenesis in the reservoir. Very recently, we and others have demonstrated that innervation plays an important role in tumorigenesis in stomach [31] and prostate [32], and that denervation reduces the risk of tumorigenesis [31]. This may explain the observation that despite of chronic inflammation in the pouch mucosa, the risk of developing dysplasia and cancer seemed to be low probably due to the reduced neurons [17, 18]. The reduced neurons in the ileum after proctocolectomy may not be related to postsurgical diarrhoea as it did not occur after IPAA. However, the role of enteric nervous system in the functional disturbance that leads to diarrhoea should be better studied using the whole mount sample of tunica muscularis [33].
Endocrine cells in the intestine secrete a number of neuropeptides with a wide variance of functions. In the present study, there was no significant change in chromogranin A-immunoreactive endocrine cells in the mucosa after proctocolectomy. Possible functional link between the mast cells and neurons in the mucosa as well submucosa has been suggested [34, 35]. In the present study, the mast cells seemed to be unaffected after proctocolectomy, which was in line with clinical report of pouchitis [23]. The number of mast cells in the mucosa was statistically reduced after EEA, but it is unlikely to show any biological significance.
In conclusion, the establishment of these animal models of proctocolectomy helps us to better understand the postsurgical physiology and pathology. IPAA was better procedure than EEA or SEA as it did not result in diarrhoea and body weight loss. Neurons, but neither endocrine cells nor mast cells, were reduced in the ileal reservoir of proctocolectomized rats regardless of different anastomotic procedures.
References
Meagher AP, Farouk R, Dozois RR, Kelly KA, Pemberton JH (1998) J ileal pouch-anal anastomosis for chronic ulcerative colitis: complications and long-term outcome in 1310 patients. Br J Surg 85:800–803
Fazio VW, Ziv Y, Church JM, Oakley JR, Lavery IC, Milsom JW et al (1995) Ileal pouch-anal anastomoses complications and function in 1005 patients. Ann Surg 222:120–127
Farouk R, Pemberton JH, Wolff BG, Dozois RR, Browning S, Larson D (2000) Functional outcomes after ileal pouch-anal anastomosis for chronic ulcerative colitis. Ann Surg 231:919–926
Fleshman JW, Cohen Z, McLeod RS, Stern H, Blair J (1988) The ileal reservoir and ileoanal anastomosis procedure. Factors affecting technical and functional outcome. Dis Colon Rectum 31:10–16
Lohmuller JL, Pemberton JH, Dozois RR, Ilstrup D, van Heerden J (1990) Pouchitis and extraintestinal manifestations of inflammatory bowel disease after ileal pouch-anal anastomosis. Ann Surg 211:622–627 (discussion 7–9)
Shepherd NA, Hulten L, Tytgat GN, Nicholls RJ, Nasmyth DG, Hill MJ et al (1989) Pouchitis. Int J Colorectal Dis 4:205–229
Svaninger G, Nordgren S, Oresland T, Hulten L (1993) Incidence and characteristics of pouchitis in the Kock continent ileostomy and the pelvic pouch. Scand J Gastroenterol 28:695–700
Heuschen U, Schmidt J, Allemeyer E, Stern J, Heuschen G (2001) The ileo-anal pouch procedure: complications, quality of life, and long-term results. Zentralbl Chir 126(Suppl 1):36–42
Ferrante M, Declerck S, Coopmans T, De Hertogh G, Van Assche G, Penninckx F et al (2008) Development of pouchitis following ileal pouch-anal anastomosis (IPAA) for ulcerative colitis: a role for serological markers and microbial pattern recognition receptor genes. J Crohns Colitis 2:142–151
Hahnloser D, Pemberton JH, Wolff BG, Larson DR, Crownhart BS, Dozois RR (2007) Results at up to 20 years after ileal pouch-anal anastomosis for chronic ulcerative colitis. Br J Surg 94:333–340
Wasmuth HH, Trano G, Midtgard TM, Wibe A, Endreseth BH, Myrvold HE (2010) Long-term function after ileal pouch-anal anastomosis—function does not deteriorate with time. Colorectal Dis 12:e283–e290
Nilubol N, Scherl E, Bub DS, Gorfine SR, Marion J, Harris MT et al (2007) Mucosal dysplasia in ileal pelvic pouches after restorative proctocolectomy. Dis Colon Rectum 50:825–831
Keranen U, Luukkonen P, Jarvinen H (1997) Functional results after restorative proctocolectomy complicated by pouchitis. Dis Colon Rectum 40:764–769
Heuschen UA, Heuschen G, Autschbach F, Allemeyer EH, Herfarth C (2001) Adenocarcinoma in the ileal pouch: late risk of cancer after restorative proctocolectomy. Int J Colorectal Dis 16:126–130
Gullberg K, Stahlberg D, Liljeqvist L, Tribukait B, Reinholt FP, Veress B et al (1997) Neoplastic transformation of the pelvic pouch mucosa in patients with ulcerative colitis. Gastroenterology 112:1487–1492
Kariv R, Remzi FH, Lian L, Bennett AE, Kiran RP, Kariv Y et al (2010) Preoperative colorectal neoplasia increases risk for pouch neoplasia in patients with restorative proctocolectomy. Gastroenterology 139:806–812
Branco BC, Sachar DB, Heimann TM, Sarpel U, Harpaz N, Greenstein AJ (2009) Adenocarcinoma following ileal pouch-anal anastomosis for ulcerative colitis: review of 26 cases. Inflamm Bowel Dis 15:295–299
Borjesson L, Willen R, Haboubi N, Duff SE, Hulten L (2004) The risk of dysplasia and cancer in the ileal pouch mucosa after restorative proctocolectomy for ulcerative proctocolitis is low: a long-term term follow-up study. Colorectal Dis 6:494–498
Stallmach A, Chan CC, Ecker KW, Feifel G, Herbst H, Schuppan D et al (2000) Comparable expression of matrix metalloproteinases 1 and 2 in pouchitis and ulcerative colitis. Gut 47:415–422
Boerr LA, Sambuelli AM, Filinger E, Peredo H, Graziano A, Valero J et al (1996) Increased mucosal levels of leukotriene B4 in pouchitis: evidence for a persistent inflammatory state. Eur J Gastroenterol Hepatol 8:57–61
Stallmach A, Schafer F, Hoffmann S, Weber S, Muller-Molaian I, Schneider T et al (1998) Increased state of activation of CD4 positive T cells and elevated interferon gamma production in pouchitis. Gut 43:499–505
Okabe T, Hide M, Koro O, Nimi N, Yamamoto S (2001) The release of leukotriene B4 from human skin in response to substance P: evidence for the functional heterogeneity of human skin mast cells among individuals. Clin Exp Immunol 124:150–156
Keranen U, Jarvinen H, Karkkainen P, Kiviluoto T, Kivilaakso E, Soinila S (1996) Substance P—an underlying factor for pouchitis? Prospective study of substance P- and vasoactive intestinal polypeptide-immunoreactive innervation and mast cells. Dig Dis Sci 41:1665–1671
Keranen U, Jarvinen H, Kiviluoto T, Kivilaakso E, Soinila S (1996) Substance P- and vasoactive intestinal polypeptide-immunoreactive innervation in normal and inflamed pouches after restorative proctocolectomy for ulcerative colitis. Dig Dis Sci 41:1658–1664
Singh S, Sharma PK, Loftus EV Jr, Pardi DS (2013) Meta-analysis: serological markers and the risk of acute and chronic pouchitis. Aliment Pharmacol Ther 37:867–875
Johannessen H, Kodama Y, Zhao CM, Sousa MM, Slupphaug G, Kulseng B et al (2013) Eating behavior and glucagon-like peptide-1-producing cells in interposed ileum and pancreatic islets in rats subjected to ileal interposition associated with sleeve gastrectomy. Obes Surg 23:39–49
Day IN, Hinks LJ, Thompson RJ (1990) The structure of the human gene encoding protein gene product 9.5 (PGP9.5), a neuron-specific ubiquitin C-terminal hydrolase. Biochem J 268:521–524
Corfield AP, Warren BF, Bartolo DC, Wagner SA, Clamp JR (1992) Mucin changes in ileoanal pouches monitored by metabolic labelling and histochemistry. Br J Surg 79:1209–1212
Shepherd NA, Healey CJ, Warren BF, Richman PI, Thomson WH, Wilkinson SP (1993) Distribution of mucosal pathology and an assessment of colonic phenotypic change in the pelvic ileal reservoir. Gut 34:101–105
Corfield AP, Myerscough N, Longman R, Sylvester P, Arul S, Pignatelli M (2000) Mucins and mucosal protection in the gastrointestinal tract: new prospects for mucins in the pathology of gastrointestinal disease. Gut 47:589–594
Zhao CM, Hayakawa Y, Kodama Y, Muthupalani S, Westphalen CB, Andersen GT et al (2014) Denervation suppresses gastric tumorigenesis. Sci Transl Med 6:250ra115
Magnon C, Hall SJ, Lin J, Xue X, Gerber L, Freedland SJ et al (2013) Autonomic nerve development contributes to prostate cancer progression. Science 341:1236361
Collins J, Borojevic R, Verdu EF, Huizinga JD, Ratcliffe EM (2014) Intestinal microbiota influence the early postnatal development of the enteric nervous system. Neurogastroenterol Motil 26:98–107
Perdue MH, Marshall J, Masson S (1990) Ion transport abnormalities in inflamed rat jejunum. Involvement of mast cells and nerves. Gastroenterology 98:561–567
McCafferty DM, Sharkey KA, Wallace JL (1994) Beneficial effects of local or systemic lidocaine in experimental colitis. Am J Physiol 266:G560–G567
Acknowledgments
The present study was supported by funding from the Liaison Committee between the Central Norway Regional Health Authority (RHA) and the Norwegian University of Science and Technology (NTNU). The authors thank Dr. Rune Søberg for his assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, CM., Myrvold, H.E. & Chen, D. Reduced neurons in the ileum of proctocolectomized rat models. Med Mol Morphol 48, 155–163 (2015). https://doi.org/10.1007/s00795-014-0093-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00795-014-0093-9