Abstract
Objective
This study aims to examine the ability of optical coherence tomography (OCT) to differentiate ex vivo epithelial structure of benign disorders, dysplastic, and oral squamous cell carcinoma (OSCC) in comparison with the structure of normal marginal mucosa of oral biopsies. As a secondary objective, we examined the inter- and intra-observer variations of OCT measurements of two calibrated assessors.
Materials and methods
Oral biopsies (n = 44) were scanned using the swept source OCT (SSOCT) and grouped by pathology diagnosis to benign, dysplasia or carcinoma. Two trained and calibrated assessors scored on the five OCT variables: thickness of keratin layer (KL), epithelial layer (EL), homogeneity of lamina propria (LP), basement membrane integrity (BMI), and the degree of reflection of the epithelial layer (Ep Re). Chi-square tests and Fischer’s exact method were used to compare the data.
Results
The OCT images showed breached BM status in all the OSCC samples (100%). Epithelial reflection was noted to be hyper-reflective in all the OSCC and oral dysplasia samples (100%). An increase in KL in 66.67% of the OSCC and 100% of the oral dysplasia samples was found. EL was increased in all the OSCC samples (100%) and 85.72% of the oral dysplasias. Kappa values showed that there was very good agreement (over 0.7) when scoring individual parameters between the two assessors.
Conclusion
The study showed that the BM status was a key parameter in the detection of SCC and for differentiating SCC from oral dysplasia or benign disorders.
Clinical relevance
OCT is a non-invasive and non-radioactive adjunct diagnostic tool that can provide immediate results on the structure of oral mucosa. The BM status measured ex vivo was a key parameter in the detection of SCC and for differentiating SCC from oral dysplasia or benign disorders.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Squamous cell carcinoma is the most common form of malignancy of the lip and oral cavity, and is often preceded by oral potentially malignant disorders (OPMD) [1]. Early diagnosis of oral cancer can improve survival rates, and the presence of an OPMD provides opportunities to identify patients who have an increased risk of oral cancer [2]. Conventional clinical oral examination (COE) is recommended to detect cancer and OPMDS in the oral cavity [3]. A Cochrane review analysing 10 studies confirmed that the COE showed very high specificity (98%) and was deemed to be better at correctly classifying the absence of OPMDs or oral cavity cancer in disease-free individuals than classifying the presence of disease [4]. Oral lesions detected by COE need a scalpel biopsy—the current diagnostic standard—the test is painful for patients and involves a delay in order to process the tissue and render a histologic diagnosis.
Optical diagnostic tests are available that are less invasive, and some can provide immediate results. The non-invasive optical diagnostic systems applied as adjuncts to COE include tissue auto-fluorescence imaging (VELscope) [5], tissue reflectance (ViziLite) [6], optical biopsy (narrow band imaging (NBI)) [7], micro-Raman spectroscopy [8], optical coherence microscopy (OCM) [9] and optical coherence tomography (OCT) [10].
Optical coherence tomography (OCT) was first developed by Fujimoto et al. in 1991 [11, 12]. It is a non-invasive and non-radioactive adjunct diagnostic tool based on interferometers [12]. OCT and ultrasound share the same concept [13], both being capable of providing real-time cross-sectional sub-surface tissue images. The difference is that OCT uses broadband light, while ultrasound uses sound waves [11, 14]. Besides, modern OCT systems outweigh ultrasound by having around 10 times higher axial resolution [15]. Moreover, OCT uses a low-coherence broadband near-infrared light source, so it gets an excellent spatial resolution (~ 20 μm) and real-time images [12, 16].
The use of OCT for diagnosing oral premalignancy and malignancy in human subjects is reported in the literature [17]. A research group in London has investigated ways of measuring the epithelial morphology of oral biopsies (in vivo) using OCT [18,19,20]. The objective of this study was to examine the ability of OCT to differentiate ex vivo epithelial structure of benign, dysplastic, and oral cancer biopsies. As a secondary objective, we examined the inter- and intra-observer variation of OCT measurements of the two calibrated assessors.
Materials and methods
Prior to this investigation, two assessors (an oral pathologist and an oral surgeon) were trained and calibrated using a training set of OCT images and light microscopic images of normal (obtained from the margins of SCC lesions) and different pathological conditions (n = 20) to enhance their knowledge in the field of OCT image interpretation. Both assessors were required to comment on five variables of the OCT images [18, 19], which included changes in the keratin layer (KL; increased or decreased or no change in thickness), changes in the epithelial layer (EP; increased or decreased or no change in thickness), changes in the lamina propria (LP; homogenous or non-homogenous), basement membrane identification (BMI; complete, partial or breached) and the degree of reflection of the epithelial layer (EP Re; normal reflection or hyper-reflective). Normal mucosa adjacent to the disease area was used as a standard reference.
Forty-four patients attending an oral surgery clinic with various oral mucosal disorders (Table 1) were included in this study. Based on the planned management, 18 needed excision, and 34 required incisional biopsies for pathology diagnoses. Following consenting, the size of each lesion was measured using a ruler; the biopsy site was marked out with a disposable surgical marker to include normal mucosa together with the lesion (Fig. 1a). Each biopsy specimen therefore had some amount of normal tissue at its edges. Intra-oral photographs of these oral lesions with their markings were documented, and the edges of the biopsy samples were sutured and taped to microscopic slides. These slides were photographed to record the orientation and to ensure an accurate co-registration (Fig. 1b) and were transferred in normal saline to the laboratory within an hour. In the laboratory, the specimens were blotted with tissue, and a line was drawn across the long axis of the specimen using a disposable surgical marker to ensure that the pathologist grossed the specimens in the same plane as used for OCT scanning (Fig. 1c and d). Immediately after scanning with OCT, the specimens were fixed in 10% buffered formalin and sent for histopathological diagnosis. The pathology diagnoses given by the reporting pathologist were used to group the biopsies to 3 categories—squamous cell carcinoma, epithelial dysplasia or benign disorders.
Photographs show a white plaque (with the surgical marking), with a mixed lichenoid appearance posteriorly. b Biopsy sample sutured and taped to the slide with orientation to ensure an accurate co-registration (P, posterior; S, superior; I, inferior; A, anterior). c OCT image of the biopsied tissue. d Photomicrograph of the biopsy sample (stain: H and E; original magnification × 15)
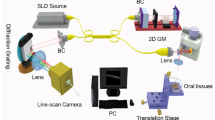
For this investigation, we used the swept source OCT (SSOCT) (Thorlabs OCS1300SS, USA) with an imaging wavelength of 1325 nm. An individual OCT scan was 10-mm wide, and the larger biopsy samples needed more than one scan along the long axis.
The two assessors were blinded to the clinical and histopathological diagnosis but were provided with the anatomical sites from where the biopsy samples were taken from. Both were asked to score on the five OCT variables referred to earlier based on the structural changes that they found during their OCT image assessments. The scoring of OCT images was carried out in two separate sessions to obtain repeat measurements. The cases were shuffled for the second session to blind them to the first recording. Wherever there were discrepancies in the scores, they were resolved by discussion between the two assessors. The research protocol was approved by the Medical Ethics Committee, Faculty of Dentistry, University of Malaya (DF OS1519/0077 P).
Statistical analysis
The percentage of architectural changes which were observed by the two observers were calculated and compared using Chi-square tests. The percentage of architectural changes among the three groups (benign lesions, epithelial dysplasia and OSCC) were calculated and tested by the Fischer’s exact method. The p value of less than 0.05 was considered statistically significant. The inter-observer and intra-observer agreements for the first and the second sessions were evaluated using kappa scores (poor = 0.00–0.40, good = 0.41–0.70, very good = 0.71–0.80, excellent = 0.81–1).
Results
Fifty-two tissue samples from 44 subjects were included in the study. Anatomical locations of biopsy sites and pathology diagnoses are listed in Table 1. The architectural characteristics that were scored by the two observers on the OCT images are shown in Table 2. The scorings were compatible, and no significant differences were observed between the two assessors for all 5 parameters.
Scanning different parts of normal oral mucosa revealed that the KL in most specimens appeared as a thin layer of hyper-reflection and looked brighter than the EP and LP (Fig. 2a), while in the pathological oral mucosa (dysplasia) with hyper-keratinization, the KL appeared thicker, brighter and more hyper-reflective (Fig. 3e).
Photograph shows a OCT image of normal floor of mouth showing keratin layer (KL), epithelial layer (EP) and lamina propria (LP) which are clearly identified. EP looks translucent and less reflective than LP. The basement membrane (BM) is intact and identified clearly and completely. b Photomicrograph of normal floor of mouth (Stain: H and E; original magnification × 1.0). c OCT image of dorsal surface of the tongue showing papilla and epithelial ridges. d Photomicrograph of normal dorsal surface of tongue (stain: H and E; original magnification × 1.0)
Photograph shows a homogenous leukoplakia on right lateral border of tongue. b OCT image of biopsy of the tongue. Keratin layer (KL), epithelial layer (EP) and lamina propria (LP) are clearly identified. Basement membrane (BM) looks intact and identified clearly and completely. The epithelial layer (EP) increased in thickness (area defined by red arrows) when compared to its thickness on the right side of OCT image (area defined by white arrows). c Photomicrograph of lateral border of the tongue showing severe epithelial dysplasia (stain: H and E; original magnification × 1.5; inset, original magnification × 18). d Photograph of a white lesion (stained with toluidine blue) on the left buccal mucosa that was clinically diagnosed as oral lichen planus/lichenoid reaction. e OCT image of the lesion showing keratin layer (KL), epithelial layer (EP) and lamina propria (LP) all are clearly marked. The basement membrane (BM) is intact and identified clearly and completely. f Photomicrograph of left buccal mucosa showing OLP (stain: H and E; original magnification × 0.8, inset, stain: H and E; original magnification × 10)
The epithelial layer (EP) often looked translucent or less hyper-reflective than the upper keratin layer or lower lamina propria (Fig. 2a), while in most pathological oral tissues, it tended to be brighter and more hyper-reflective than the lower LP. In both carcinomas and dysplasias, the thickness of EP was mostly increased (Fig. 3b).
In normal mucosa, lamina propria (LP) appeared as a homogenous layer and was slightly brighter than the upper EP, with well-defined demarcation from the EP (Fig. 2a). In the SCC biopsies, it lacked the homogeneous appearance and could not be separately identified from the EP (Fig. 4b and e).
Photograph shows a a white patch on right lateral border of the tongue, the clinical diagnosis was leukoplakia. b OCT image of the biopsy. The marked area shows a breach in the basement membrane (BM) and an uneven surface epithelium. c Photomicrograph of right lateral border of the tongue. The area in black rectangle shows the invasion of the epithelial tumour cells into the underlying connective tissue (stain: H and E; original magnification × 0.8; inset, original magnification × 20). Pathology diagnosis SCC d photograph of non-healing ulcer on right ventral surface of the tongue, the clinical diagnosis was SCC. e OCT image of right ventral surface of the tongue. The areas in the white rectangles represent the focal invasion and breach of basement membrane (BM). The epithelial layer (EP) appears very bright, and its thickness is increased dramatically in the area of focal invasion. f Photomicrograph of right ventral surface of tongue showing moderately differentiated SCC (stain: H and E; original magnification × 0.8; inset, original magnification × 20)
The basement membrane (BM) was seen as an area between two different contrasts (EP and LP), and it could be identified clearly in most normal and benign oral mucosal tissues (Fig. 2a). Sometimes, BM showed some extensions down to the LP, which represented rete pegs such as in the dorsal surface of the tongue (Fig. 2c). The basement membrane was breached in all OSCC, in few dysplasia but only in 1 specimen of a benign disorder (1/30). Figure 4a to c illustrate a case of a white patch that was clinically diagnosed as oral leukoplakia which demonstrated a breach in BM in the OCT image that indicated the possibility of a malignancy and was confirmed by histopathology as SCC. Supplementary Table S1 gives the summary of OCT image assessments by clinical and histopathological diagnoses.
KL, EL, LP, BM and Ep Re scores by the two assessors were not significantly different (Table 2). Excluded in the analysis were few cases in which the change in the basement membrane was not recorded by some observers. Reference standard used was the normal mucosa found at the margin of each biopsy.
The scores from OCT images showed an increase in KL in 66.67% of the OSCC and 100% of the oral dysplasia samples. EL was increased in all the OSCC samples (100%) and 85.72% of the oral dysplasia samples. There was no change in EL in 63.34% of the benign samples. With regard to the BM, 57.14% of the oral dysplasia and 83.34% of the benign samples showed complete BM status. Breached BM status was seen in all the OSCC samples (100%). Epithelial reflection was noted to be hyper-reflective in all the OSCC and oral dysplasia samples (100%). The variations in the thickness of the keratin layers were not significantly different in the 3 groups of pathologies. The EL, LP, BM status and EP Re showed significant differences among the three groups (Table 3).
Kappa values showed that there was very good agreement when scoring different parameters between the two assessors as well as in the scores from the repeat session (Table S2). Scores reached over 0.7 in all comparisons. Better inter-observer agreement was noted in the pathologist’s scoring.
Discussion
During COE, small cancers can sometimes be missed, overlooked, or diagnosed as benign disease resulting in late detection of oral malignancy [21]. Moreover, COE alone cannot differentiate between benign and potentially malignant oral lesions (OPMDs) or oral cancer, because many OPMDs and SCCs are capable of mimicking the clinical features of benign oral lesions or infections of the oral mucosa [22]. In addition, clinicians depend on COE to visually select the most suspicious site for biopsy, and wrong sampling could lead to under diagnosis. For these reasons, there is a continuous need for new studies regarding the combination of plural techniques for the early clinical detection of oral cancer and diagnosis of OPMDs [23].
Several scanning techniques have been developed that can detect the structural changes in the pathological tissues [13]. Currently available scanning methods with varying depths of imaging include ultrasound (100–500 μm), CT (500–1000 μm) and MRI (500–1000 μm), but these techniques are still insufficient for an accurate identification of the structural changes that occur during the pathological process in squamous lining epithelia [10]. Compared to these above scanning techniques, OCT shows the highest resolution [24] and is considered the only optical method that is available with a resolution of up to 10 μm (10). In addition, OCT provides cross-sectional images of the healthy and pathological tissues without the necessity of obtaining biopsy or subjecting the patient to any ionizing radiation [25]. An earlier study of OCT on hamster cheek pouches showed a good resolution up to 3-mm depth and in concordance with histologic images [25,26,27].
In our OCT scans, we noted a degree of differential tissue contrast information. Different tissue components of the oral mucosa gave different OCT reactivity signals that allowed differentiation of the surface epithelium from the lamina propria that could be resolved without any staining. An earlier study that evaluated the OCT scans in the assessment of oral mucosal tissue stated that the epithelium thickness was increased dramatically in malignant lesions, while its thickness was variable in oral dysplasia. Drawing some definitive criteria based on epithelial thickness alone for the differentiation between benign, potentially malignant disorders and cancer was difficult [28]. The oral mucosa has a complex arrangement of different structural layers, i.e. surface keratin layer, the rest of the epithelium, the basement membrane and the lamina propria [29]. Gross structural differences in these different layers may help to differentiate various oral mucosal pathologies [30].
The scalpel and punch are the most widely used methods of obtaining diagnostic biopsies for assessing oral and maxillofacial pathologies. In this study, we utilised incision and excision biopsies using the scalpel and did not opt for punch biopsies. A recent systematic review of four publications has shown evidence that punch biopsies are less likely to produce artefacts, such as crush, fragmentation, splits and haemorrhage, than biopsies obtained with a scalpel [31].
The KL thickness and reflectivity was variable in different pathological conditions, but its status alone was not discriminative in identifying the pathological changes. These findings are confirmative of a previous OCT study conducted to assess the structural changes of oral mucosa which stated that KL layer was hyper-reflective in mild and moderate oral dysplasias, while it was hypo-reflective in sever dysplasias and carcinoma in situ [28].
The epithelial layer (EP) was hyper-reflective in all dysplasia and SCC and some benign cases, while it was less hyper-reflective than the LP in all the normal tissue biopsies (Fig. 4a). Therefore, the degree of EP reflectivity was a good parameter in differentiating normal tissue from the other pathological conditions.
Our study also showed that EP was increased in thickness and more hyper-reflective than the LP in all the OCT images of oral dysplasia and SCC. However, the EP thickness and the EP Re still could not be definitive diagnostic parameters for the differentiation between oral dysplasia and other benign lesions; the EP thickness and its degree of reflection were also increased in some benign conditions.
OCT was uniquely able to identify all the SCC samples that showed breach of BM and discriminate them from dysplasia or benign samples in which the BM was identified partially or as complete without any signs of breach. Therefore, BM status was a key parameter in detecting the early invasion of oral cancer (Fig. 4b and e). This finding is confirmative and consistent with another study conducted earlier using the OCT to measure the epithelial thicknesses within oral cavity which stated that the BM was a basic critical parameter in the differentiation between the malignant and non-malignant tissue [32]. This observation allows us to interpret that OCT imaging of oral mucosa could allow investigation of the basement membrane of a lesion to study whether it is intact or breached without the need to remove tissue. The study supports the potential application of surface OCT probe in clinical experimental use [33]. Kappa agreement between the pathologist and the clinician on BM assessment reached 0.675 and was closely similar to an earlier study which reported a kappa of 0.656 [19].
In our study, we evaluated KL, EL, LP, BM and Ep Re scores. A recent in vivo study has undertaken evaluation of seven criteria that included examining the integrity and the profile of the epithelial surface as well as the grade of vascular supply in the lamina propria. The mean vascular density was found to be highest in the sublingual mucosa in comparison with other anatomical sites [34]. A denser red pattern of vascularisation when compared to normal mucosa, indicative of a higher blood inflow typical of a chronically inflamed mucosa, was shown in a study of atrophic and erosive oral lichen planus [35]. Advances in OCT technology exploring the use of three-dimensional (3D) OCT combined with intelligent machine learning systems to evaluate mucosal changes have also been described recently [36].
While COE alone cannot alone accurately differentiate between OPMDs or oral cancer (22), a recent critical review commented on an overall high sensitivity (80–90%) and specificity (85–98%) reported for OCT to intercept oral potentially malignant disorders and oral squamous cell carcinoma [37]. Several OCT systems have received the Food and Drug Administration (FDA) approval for clinical use. Based on the current evidence, OCT technique may potentially benefit in the early detection of oral malignancies (based on breach of BM), selection of biopsy sites and in identification of margins. However, some challenges that limit the wide scale application of this technique beyond research studies need considerations: (1) the need to calibrate clinicians to read and interpret the images, (2) discomfort during the examination process and (3) initial high cost of the scanning tool compared with other commercially available adjunctive tools or low-cost vital staining techniques.
There were some limitations in our study that were encountered which requires caution. (i) The correlation between histopathological and OCT images was not always possible due to the amount of shrinkage found in the biopsy samples caused by its preservation in formalin. (ii) The resolution of OCT images was affected by the ex vivo nature of the study particularly in the clear identification of epithelial and sub-epithelial structures of oral mucosa. (iii) The sample size was small, which limits the generalisation of our findings.
Conclusions
The findings of this study confirm previous observations reported in ex vivo studies on oral mucosa using OCT. We found good calibration between two assessors to record OCT findings with little inter-observer variation, and repeat measurements had good kappa values showing good inter-observer agreement in all parameters. The main outcome of this study was that the BM status observed in OCT images was a key parameter in the detection of SCC and for differentiating SCC from oral dysplasia or benign disorders.
References
Warnakulasuriya S, Kujan O, Aguirre-Urizar JM, Bagan JV, González-Moles MÁ, Kerr AR, Lodi G, Mello FW, Monteiro L, Ogden GR, Sloan P, Johnson NW (2020) Oral potentially malignant disorders: a consensus report from an international seminar on nomenclature and classification, convened by the WHO Collaborating Centre for Oral Cancer. Oral Dis. https://doi.org/10.1111/odi.13704
Barasch A, Epstein JB (2020) Early diagnosis of oral cancer. In: Warnakulasuriya S, Greenspan J (eds) Textbook of Oral Cancer. Springer, Switzerland, pp 195–200
Speight PM, Epstein J, Kujan O, Lingen MW, Nagao T, Ranganathan K, Vargas P (2016) Screening for oral cancer – a perspective from the global oral cancer forum. Oral Surg Oral Med Oral Pathol Oral Radiol 123:680–687
Walsh T, Liu JL, Brocklehurst P, Glenny AM, Lingen M, Kerr AR, Ogden G, Warnakulasuriya S, Scully C (2013) Clinical assessment to screen for the detection of oral cavity cancer and potentially malignant disorders in apparently healthy adults. Cochrane Database Syst Rev 11:CD010173
Awan K, Morgan P, Warnakulasuriya S (2011) Evaluation of an autofluorescence based imaging system (VELscope™) in the detection of oral potentially malignant disorders and benign keratoses. Oral Oncol 47:274–277
Oh ES, Laskin DM (2007) Efficacy of the ViziLite system in the identification of oral lesions. J. Oral Maxillofac Surg 65:424–426
Watanabe A, Taniguchi M, Tsujie H, Hosokawa M, Fujita M, Sasaki S (2008) The value of narrow band imaging endoscope for early head and neck cancers. Otolaryngol. Head Neck Surg 138:446–451
Krishna CM, Sockalingum G, Kurien J, Rao L, Venteo L, Pluot M, Manfait M, Kartha V (2004) Micro-Raman spectroscopy for optical pathology of oral squamous cell carcinoma. Appl Spectrosc 58:1128–1135
Clark AL, Gillenwater A, Alizadeh-Naderi R, El-Naggar AK, Richards-Kortum R (2004) Detection and diagnosis of oral neoplasia with an optical coherence microscope. J Biomed Opt 9(6):1271–1280
Ridgway JM, Armstrong WB, Guo S, Mahmood U, Su J, Jackson RP, Shibuya T, Crumley RL, Gu M, Chen Z (2006) In vivo optical coherence tomography of the human oral cavity and oropharynx. Arch Otolaryngol Head Neck Surg 132:1074–1081
Huang D, Swanson EA, Lin CP, Schuman JS, Stinson WG, Chang W, Hee MR, Flotte T, Gregory K, Puliafito CA (1991) Optical coherence tomography. Science 254:1178–1181
Fujimoto JG (2003) Optical coherence tomography for ultrahigh resolution in vivo imaging. Nat Biotechnol 21:361–1367
Tadrous PJ (2000) Methods for imaging the structure and function of living tissues and cells: 1. Optical coherence tomography. J Pathol 191:115–119
Ding Z, Ren H, Zhao Y, Nelson JS, Chen Z (2002) High-resolution optical coherence tomography over a large depth range with an axicon lens. Optics Letters 27:243–245
Schmitt JM (1999) Optical coherence tomography (OCT): a review. IEEE Journal of Selected Topics in Quantum Electronics 5:1205–1215
Wojtkowski M, Srinivasan V, Fujimoto JG, Ko T, Schuman JS, Kowalczyk A, Duker JS (2005) Three-dimensional retinal imaging with high-speed ultrahigh-resolution optical coherence tomography. Ophthalmology 112:1734–1746
Wilder-Smith P, Lee K, Guo S, Zhang J, Osann K, Chen Z, Messadi D (2009) In vivo diagnosis of oral dysplasia and malignancy using optical coherence tomography: preliminary studies in 50 patients. Lasers Surg Med 41:353–357
Jerjes W, Upile T, Conn B, Hamdoon Z, Betz CS, McKenzie G, Radhi H, Vourvachis M, El Maaytah M, Sandison A (2010) In vitro examination of suspicious oral lesions using optical coherence tomography. Br J Oral Maxillofac Surg 48:18–25
Hamdoon Z, Jerjes W, Upile T, McKenzie G, Jay A, Hopper C (2013) Optical coherence tomography in the assessment of suspicious oral lesions: an immediate ex vivo study. Photodiagnosis Photodyn Ther 10:17–27
Jerjes W, Hamdoon Z, Al Rawi N, Hopper C (2019) OCT in the diagnosis of head and neck pre-cancerous and cancerous cutaneous lesions: an immediate ex vivo study. Photodiagnosis Photodyn Ther 27:481–486
Bagan JV, Bagan-Debon L (2020) Clinical presentation and differential diagnosis of oral cancer. In: Warnakulasuriya S, Greenspan J (eds) Textbook of Oral Cancer. Springer, Switzerland, pp 47–54
Epstein JB, Güneri P, Boyacioglu H, Abt E (2012) The limitations of the clinical oral examination in detecting dysplastic oral lesions and oral squamous cell carcinoma. J Am Dent Assoc 143:1332–1342
Liu D, Zhao X, Zeng X, Dan H, Chen Q (2016) Non-invasive techniques for detection and diagnosis of oral potentially malignant disorders. TOHOKU J Exp Med 238:165–177
Sinescu C, Negrutiu M, Todea C, Hughes M, Tudorache F, Podoleanu AG (2008) Fixed partial dentures investigated by optical coherent tomography. Biomedical Optics (BiOS), International Society for Optics and Photonics, pp. 684707-684707-10
Wilder-Smith P, Jung WG, Brenner M, Osann K, Beydoun H, Messadi D, Chen Z (2004) In vivo optical coherence tomography for the diagnosis of oral malignancy. Lasers Surg Med 35:269–275
Matheny ES, Hanna NM, Jung W, Chen Z, Wilder-Smith P, Mina-Araghi R, Brenner M (2004) Optical coherence tomography of malignancy in hamster cheek pouches. J Biomed Opt 9:978–981
Wilder-Smith P, Krasieva T, Jung W-G, Zhang J, Chen Z, Osann K, Tromberg B (2005) Noninvasive imaging of oral premalignancy and malignancy. J Biomed Opt 10(5), 051601-051601-8
Hamdoon Z, Jerjes W, Al-Delayme R, McKenzie G, Jay A, Hopper C (2012) Structural validation of oral mucosal tissue using optical coherence tomography. Head Neck Oncol 6(4):29
Squier CA, Johnson NW, Hopps RM (1976) Human oral mucosa; development, structure and function. Blackwell Scientific, Oxford, pp 7–15
Cruchley AT, Bergmeier LA (2018) In: Bergmeier LA (ed) Structure and functions of the oral mucosa, Oral Mucosa in Health and Disease. Springer, pp 1–18
Rezende Oliveira S, Fernandes Araújo Almeida T, Aparecida da Silva T, Alves Mesquita R, Guimarães Abreu L (2020) Comparison of tissue artifacts in punch and scalpel biopsies of oral and maxillofacial lesions: a systematic review and meta-analysis. J Stomatol Oral Maxillofac Surg 121(6):704–712
Prestin S, Rothschild SI, Betz CS, Kraft M (2012) Measurement of epithelial thickness within the oral cavity using optical coherence tomography. Head Neck 34:1777–1781
DeCoro M, Wilder-Smith P (2010) Potential of optical coherence tomography for early diagnosis of oral malignancies. Expert Rev Anticancer Ther 10(3):321–329
Albrecht M, Schnabel C, Mueller J, Golde J, Koch E, Walther J (2020) In vivo endoscopic optical coherence tomography of the healthy human oral mucosa: qualitative and quantitative image analysis. Diagnostics (Basel) 10(10):827
Gambino A, Cabras M, Cafaro A, Broccoletti R, Carossa S, Hopper C, Chiusa L, El Haddad G, Porter SR, Arduino PG (2020) In-vivo usefulness of optical coherence tomography in atrophic-erosive oral lichen planus: comparison between histopathological and ultrastructural findings. J Photochem Photobiol B 211:112009
Heidari AE, Pham TT, Ifegwu I, Burwell R, Armstrong WB, Tjoson T, Whyte S, Giorgioni C, Wang B, Wong BJF, Chen Z (2020) The use of optical coherence tomography and convolutional neural networks to distinguish normal and abnormal oral mucosa. J Biophotonics 13(3):e201900221
Ilhan B, Lin K, Guneri P, Wilder-Smith P (2020) Improving oral cancer outcomes with imaging and artificial intelligence. J Dent Res 3:241–248
Acknowledgement
The authors gratefully acknowledge University Malaya through Post Graduate Research Grant (PPP) (PG095-2015A) for providing an opportunity to conduct this research.
Funding
The work was supported by the funding obtained from the Faculty of Dentistry, University of Malaya (PPP) (PG095-2015A), Kuala Lumpur, Malaysia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The research protocol was approved by the Medical Ethics Committee, Faculty of Dentistry, University of Malaya (DF OS1519/0077 P).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Obade, A.Y., Pandarathodiyil, A.K., Oo, A.L. et al. Application of optical coherence tomography to study the structural features of oral mucosa in biopsy tissues of oral dysplasia and carcinomas. Clin Oral Invest 25, 5411–5419 (2021). https://doi.org/10.1007/s00784-021-03849-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-021-03849-0