Abstract
Objectives
This study aimed to investigate the relationship between aging and tooth loss on masseter muscle quantity and quality.
Materials and methods
This cross-sectional study was conducted among 112 participants (in their 20s to 90s) who were independent in activities of daily living and were able to follow verbal commands. Exclusion criteria comprised participants with a lack of molar occlusal support, diseases that could affect muscle function, or temporomandibular disorder. Age, tooth loss, and weight were documented, and masseter muscle thickness (MMT) and masseter muscle echo intensity (MMEI) were measured using an ultrasonic diagnostic apparatus. A multiple regression analysis was used to determine the relationship between MMT, MMEI, aging, and tooth loss, among each sex. The significance level in the statistical analysis was p < 0.05.
Results
In males, aging was a significant explanatory variable for MMT (adjusted R2 = 0.27), while both aging and weight were significantly associated with MMEI (adjusted R2 = 0.54). In females, tooth loss and aging were significant explanatory variables for MMT (adjusted R2 = 0.36) and MMEI (adjusted R2 = 0.5), respectively. In both men and women, MMT and MMEI were highly correlated.
Conclusions
Masseter muscles in males were more likely to be attenuated by aging than in females. The main attenuation factors were observed to differ between MMT and MMEI in women.
Clinical relevance
In females, preservation of the natural dentition or prosthetic treatment may be effective for maintaining masseter muscle characteristics. In males, additional approaches, such as resistance exercise training, may be necessary.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Occlusal force and masticatory performance deteriorate mainly due to tooth loss [1], while reductions in tongue pressure are primarily associated with aging [2]. The impact of a decline in oral function extends beyond the oral cavity itself and can include serious health problems such as aspiration pneumonia, frailty, and malnutrition [3, 4]. A number of recent studies have reported on the relationship between oral function and systemic condition. Indeed, a previous study defined oral frailty as a condition with poor oral health, comprising tooth loss, and declines in occlusal force, tongue pressure, and masticatory ability [5]. Subjects with oral frailty were found to be more than twice as likely to have disability or mortality after 4 years, compared to subjects without oral frailty.
In addition to aging and tooth loss, the attenuation of perioral muscle characteristics is also considered to be a cause of oral function deterioration. Tongue thickness is positively associated with masticatory performance in healthy young dentate subjects [6], and with tongue pressure in healthy elders [7] and patients with neuromuscular disease [8]. The cross-sectional area of the geniohyoid muscle is also associated with masticatory performance [9] and jaw opening force [10] in community-dwelling subjects.
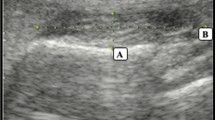
Ultrasonic apparatus can be also used to evaluate muscles, and has been shown to be a valid and reliable measure of both muscle quantity and quality [11]. Muscle quality is assessed by echo intensity; the whiter the muscle in the image, the more non-contractile tissue in the muscle and the lower the quality (Fig. 1) [12]. In limb muscles, the relationship between muscle characteristics (e.g., muscle quantity, muscle quality), muscle strength, and physical function has already been demonstrated [13]. A previous report has shown that sarcopenic patients have attenuated muscle quantity and quality [14]. Ultrasonic evaluation has also been demonstrated to be a practically convenient tool for perioral muscles (e.g., the masseter muscle), as there is no need for the subject to remove their clothes or change their posture during the assessment.
Perioral muscle characteristics vary with aging and tooth loss. In a study investigating the relationship between aging and quality of the suprahyoid muscle, as assessed by computed tomography, a deterioration due to increased intramuscular fat and atrophy was observed in an elderly group, compared to healthy adults [15]. Tooth loss has also been shown to affect MMT. Bhoyar et al. [16] documented a significantly thinner MMT among edentulous subjects aged 45–55 years, compared to healthy dentate adults. Nevertheless, no studies have compared masseter muscle characteristics (quantity and quality) among both adults and elders, and it remains unclear as to whether aging and tooth loss are independent factors associated with MMT and MMEI.
Therefore, the purpose of this study was to compare masseter muscle characteristics by sex, among both adults and elders, and to investigate whether aging and tooth loss are independent predictors of these characteristics. The null hypothesis was that masseter muscle characteristics deteriorate with aging and tooth loss regardless of sex.
Methods
Participants
There were 112 participants, comprising 49 adults (24 males and 25 females in their 20s to 40s) and 63 elders (24 males and 39 females aged 65 years and over). Recruitment was conducted between November 2017 and May 2019. All adults were recruited from the Tokyo Medical and Dental University. Elderly participants were either recruited from patients of the Gerodontics clinic of the Dental Hospital at Tokyo Medical and Dental University who visited for the purpose of dental treatment or from participants of a health survey conducted in Koiwa city (Tokyo, Japan). Inclusion criteria comprised individuals who were independent in activities of daily living and were able to follow verbal commands. Exclusion criteria included participants with (1) diseases (e.g., cerebrovascular disorders with paralysis, neuromuscular diseases) that could affect muscle function; (2) a lack of molar occlusal support, whether from natural teeth or dentures; or (3) significant symptoms of temporomandibular disorder. A total of 124 participants were initially recruited, with two participants being excluded owing to lack of occlusal support in the molar region upon examination. An additional participant was excluded because she had temporomandibular symptoms (severe pain during jaw opening), and nine participants were excluded due to missing data. The final analyses were based on the data obtained from 112 participants. All the adults were classified as Eichner A. None of the participants in this study were undergoing orthodontic treatment and none had severe malocclusion. This study was approved by the Tokyo Medical and Dental University Ethics Committee (ref. D2014-047). The study protocol was explained to all participants, both verbally and in writing, and written informed consent was obtained.
Tooth loss evaluation
A dentist and two dental hygienists categorized tooth loss using the Eichner classification [17]. Both the dentist and the dental hygienists have more than 8 years of experience.
Ultrasound measurements of the masseter muscle
The masseter muscle was evaluated with an ultrasonic diagnostic apparatus (SonoSite M-turbo; Fujifilm, Tokyo, Japan), by two dentists (both were experienced in the assessment method). We have previously reported high intraclass correlation coefficients and high inter-rater reliability for ultrasonic measurements of the masseter muscle [6]. During the measurements, participants were instructed to relax while looking straight ahead in a sitting position. They were also instructed not to bite together.
A linear probe with a 6–13 MHz broadband frequency was used for masseter muscle measurement, with gain, depth, and frequency of echo being kept constant at appropriate levels. As based on a previous study, the probe was positioned approximately halfway between the zygomatic arch and the mandibular margin, parallel to the mandibular margin and perpendicular to the skin (Fig. 1) [18]. At the time of measurement, the probe was covered with a water-soluble transmission gel.
MMT and MMEI were measured using ImageJ (version 1.49, National Institutes of Health, Bethesda, MD, USA). The average of the two measurements was taken as the measurement value. MMT was measured from the surface layer of the masseter muscle to the mandibular ramus, with the thickest region (without fascia and bone) being recorded (Fig. 1) [18].
At the time of echo intensity measurement, the range of interest (ROI) included the maximum muscle without fascia and bone (Fig. 1) [19]. The intraclass correlation coefficient (ICC) was calculated to evaluate intra- and inter-examiner reliability for ultrasonic measurement of the masseter muscle in this study. The measurements were performed on eight volunteers. The intra-rater reliability was evaluated by calculating the ICC from two measurements performed by one examiner on different days. For the intra-rater reliability, the ICC of MMT was 0.97, and the ICC of MMEI was 0.83. The inter-rater reliability was evaluated by calculating the ICC from measurements by two examiners. For the inter-rater reliability, the ICC of MMT was 0.93 and that of MMEI was 0.84. All ICC values exceeded 0.8, indicating high reliability in ultrasonic measurements of the masseter muscle.
Statistical analysis
We calculated the sample size using G*Power3.1 software (Kiel University, Kiel, Germany). The required sample size was calculated to be 36 participants, based on an α of 0.05, power of 0.8, and an effect size of 0.35 reported in a previous study [19].
The participants were classified as either adults or elders. The elderly group was further divided into three groups (Eichner A, B, or C) based on the degree of tooth loss. Thus, there were a total of four groups for both males and females (adults, elders with Eichner A, elders with Eichner B, and elders with Eichner C). The normality of each outcome measure was confirmed by the Shapiro–Wilk test. Comparisons for each outcome measure were made with the one-way analysis of variance and Kruskal–Wallis test. To determine which groups had significant differences in MMT and MMEI, a t test with Bonferroni correction was performed. A multiple regression analysis was carried out for each sex to examine the relationship between masseter muscle characteristics (MMT, MMEI) and potential explanatory variables (aging, weight, tooth loss). Both aging (adult group or elder group) and tooth loss (Eichner A, B, or C) were entered into the regression model as categorical variables. To avoid multicollinearity, the correlation between each dependent variable and explanatory variable was confirmed by Pearson’s correlation coefficient and Spearman’s rank correlation coefficient. The level of statistical significance was set at p < 0.05. For all statistical analyses, the Japanese version of SPSS for Windows (version 25J, IBM Inc., Tokyo, Japan) was used.
Results
Table 1 shows the background characteristics of the participants. Among males, there were 24 participants in the adult group, 6 participants in the elder group with Eichner A, 13 participants in the elder group with Eichner B, and 5 participants in the elder group with Eichner C. Among females, there were 25 participants in the adult group, 11 participants in the elder group with Eichner A, 18 participants in the elder group with Eichner B, and 10 participants in the elder group with Eichner C. All participants in the adult group were categorized as having an Eichner A classification. Age, weight, MMT, and MMEI were significantly different between the four groups among males. Similar results were observed among females, with the exception that the weight was not significantly different.
Figure 2 shows the results of comparisons of MMT and MMEI for each group among males and females. Among males, the MMT was significantly different between the adult group and the elder group with Eichner B, and the MMEI was significantly different between the adult group and the elder groups with Eichner A and Eichner B. Among females, the MMT was significantly different between the adult group and the elder group with Eichner C, and the MMEI was significantly different between the adult group and all elder groups. Table 2 shows the correlation between aging (adult group or elder group), weight, tooth loss, MMT, and MMEI for each sex. The highest correlation was for aging and tooth loss (r = 0.76, p < 0.01) in males. No correlations exhibited a r > 0.8. MMT and MMEI were highly correlated in both males (r = − 0.7, p < 0.01) and females (r = − 0.58, p < 0.01).
Masseter muscle thickness and masseter muscle echo intensity in each group. a Masseter muscle thickness in each group of males. b Masseter muscle echo intensity in each group of males. c Masseter muscle thickness in each group of females. d Masseter muscle echo intensity in each group of females. *p < 0.05, **p < 0.01. MMT, masseter muscle thickness; MMEI, masseter muscle echo intensity
Table 3 shows the results of multiple regression analyses by sex. In males, aging was the only significant explanatory variable for MMT. When MMEI was the dependent variable, aging and weight were significant explanatory variables. In females, tooth loss and weight were significant explanatory variables for MMT. When MMEI was the dependent variable, aging and weight were significant explanatory variables. All explanatory variables had a VIF < 2.5. In an analysis of MMEI among both males and females, multiple correlation coefficients (R) exceeded 0.7, indicating a high degree of association.
Discussion
In this study, MMT and MMEI in males were related to aging, while in females, MMT was related to tooth loss, and MMEI was related to aging (Table 3). Thus, these results suggested that the attenuation of masseter muscle quantity and quality differs based on sex. These results may be specific to the perioral muscles (e.g., the masseter muscle), as it has been previously demonstrated that both quantity and quality of limb muscles decrease with aging.
MMT was found to be associated with aging in males and tooth loss in females (Table 3). In terms of the relationship between MMT and aging, a previous study compared MMT among the following five age groups: group I (7–12 years old), group II (13–20 years old), group III (21–40 years old), group IV (41–60 years old), and group V (61–80 years old) [20]. Among males, MMT gradually increased from group I to group IV, but it was thinner in group V compared to group IV. In females, the average value of MMT in group III was lower than that in group IV, and the differences between these two age groups was not as profound as that observed in males. Notably, this study was limited to fully dentate participants, and no adjustments were made for systemic variables such as weight. Nevertheless, these results provide support for the present study, which found an association of MMT with aging for males, but not for females.
This study showed an association between MMT and tooth loss in women (Table 3). A previous study reported that the MMT of edentulous subjects was significantly thinner when compared to dentate subjects [16]. In addition, when edentulous subjects wore complete dentures, a significant increase in MMT was observed 3 months after denture insertion. While recovery of MMT may be achieved with prosthodontic treatments such as dentures, the maintenance of occlusal support with natural teeth is the most effective way of maintaining MMT.
Gender-based differences in MMT deterioration may possibly be due to changes in the endocrine system with aging. Muscle atrophy may be attributed to various factors such as aging and disuse, as well as hormones. Testosterone is an essential hormone for muscle repair and strengthening, but it decreases with aging [21]. Indeed, masseter muscle mass has been shown to be very sensitive to testosterone. In a study using rats, it was reported that masseter muscle mass was reduced by castration and increased by 38% with testosterone injection [22]. Notably, this increase was greater than that observed in limb muscles. Testosterone secretion is overwhelmingly higher in men than women [23, 24]. Thus, based on the above, the decrease in testosterone with aging may have a potentially significant effect on masseter muscle mass, especially in men.
MMEI was strongly related to aging, regardless of tooth loss in both males and females (Table 1, Table 3, Fig. 2). Muscle echo intensity represents non-contractible tissue in muscle such as intramuscular fat and fibrous tissue [12]. The interaction between muscle satellite cells and mesenchymal progenitor cells has been demonstrated as the mechanism of fat formation in muscles. Skeletal muscle has a very high regenerative ability, due to muscle satellite cells [25]. Muscle satellite cells create muscle fibers that prevent mesenchymal progenitor cells from differentiating into fat [26]. Mesenchymal progenitor cells promote muscle differentiation of muscle satellite cells [27], and this interaction is important in maintaining homeostasis of muscle tissue. Therefore, if muscle satellite cells decrease or muscle atrophy progresses, the inhibitory signal for adipose differentiation of mesenchymal progenitor cells is weakened and fat differentiation occurs. There are two types of muscle fibers (type 1 and type 2), with type 2 atrophy and a decrease in the number of type 2 muscle satellite cells being particularly associated with aging [28]. As type 2 fibers make up 40% of all muscle fibers in the masseter muscle [29], it was not unexpected that an increase in echo intensity was observed with aging, regardless of sex.
Observation with an ultrasound diagnostic apparatus is simple and non-invasive and can be used by paramedical stuff, such as nurses and therapists. Understanding masseter muscle characteristics using an ultrasonic diagnostic apparatus is useful because masseter muscle characteristics can be a predictor of systemic condition as well as oral condition. MMT has been previously related not only to occlusal force [30], but also to skeletal muscle mass [31], and grip strength [32]. Further, a 2-year observational study conducted in trauma patients over 65 years of age reported that masseter muscle mass could be an excellent indicator of sarcopenia [33]. A cross-sectional study of healthy elders found that masseter muscle echo intensity (MMEI) was independently associated with MMT, grip strength, and walking speed and suggested that MMEI may be a predictor of dynapenia [19]. Performing similar examinations not only for the masseter muscle but also for other peroral muscles such as the tongue could lead to an interdisciplinary approach to oral medicine.
Some limitations are acknowledged in this study. Firstly, since this was a cross-sectional study, longitudinal studies are required to determine cause-and-effect relationships between aging and changes in masseter muscle characteristics. Secondly, this study was limited in terms of the sample size employed, especially for males among Eichner groups A and C. Additional studies with larger samples sizes are required to confirm the results of this study.
Nevertheless, this was the first study to examine the relationships between MMT, MMEI, aging, and tooth loss among both males and females. In males, aging was associated with both MMT and MMEI, while tooth loss was not. This suggested that masseter muscle characteristics in males were more susceptible to the effects of aging than in females. Maintenance of masseter muscle characteristics in males may require not only the preservation of the natural dentition, but also resistance exercises to increase the amount of available testosterone [34]. However, in females, MMT was related to tooth loss, and MMEI was related to aging. Thus, good muscle quantity may equate to good muscle quality in the masseter muscle. Preservation of the natural molars or replacement of missing molars with prostheses is important for maintenance of good masseter muscle characteristics.
References
Ikebe K, Matsuda K, Kagawa R, Enoki K, Okada T, Yoshida M, Maeda Y (2012) Masticatory performance in older subjects with varying degrees of tooth loss. J Dent 40:71–76. https://doi.org/10.1016/j.jdent.2011.10.007
Utanohara Y, Hayashi R, Yoshikawa M, Yoshida M, Tsuga K, Akagawa Y (2008) Standard values of maximum tongue pressure taken using newly developed disposable tongue pressure measurement device. Dysphagia 23:286–290. https://doi.org/10.1007/s00455-007-9142-z
Castrejón-Pérez RC, Jiménez-Corona A, Bernabé E, Villa-Romero AR, Arrivé E, Dartigues JF, Gutiérrez-Robledo LM, Borges-Yáñez SA (2017) Oral disease and 3-year incidence of frailty in Mexican older adults. J Gerontol A Biol Sci Med Sci 72:951–957. https://doi.org/10.1093/gerona/glw201
Sheiham A, Steele JG, Marcenes W, Lowe C, Finch S, Bates CJ, Prentice A, Walls AW (2001) The relationship among dental status, nutrient intake, and nutritional status in older people. J Dent Res 80:408–413. https://doi.org/10.1177/00220345010800020201
Tanaka T, Takahashi K, Hirano H, Kikutani T, Watanabe Y, Ohara Y, Furuya H, Tetsuo T, Akishita M, Iijima K (2018) Oral frailty as a risk factor for physical frailty and mortality in community-dwelling elderly. J Gerontol A Biol Sci Med Sci 73:1661–1667. https://doi.org/10.1093/gerona/glx225
Yamaguchi K, Tohara H, Hara K, Chantaramanee A, Nakagawa K, Yoshimi K, Nakane A, Minakuchi S (2019) Tongue thickness is associated with masticatory performance of perioral muscles: ultrasonographic study of perioral muscle characteristics in healthy young subjects. J Oral Rehabil. https://doi.org/10.1111/joor.12909
Yoshimi K, Hara K, Tohara H, Nakane A, Nakagawa K, Yamaguchi K, Kurosawa Y, Yoshida S, Ariya C, Minakuchi S (2018) Relationship between swallowing muscles and trunk muscle mass in healthy elderly individuals: a cross-sectional study. Arch Gerontol Geriatr 79:21–26. https://doi.org/10.1016/j.archger.2018.07.018
Umemoto G, Furuya H, Arahata H, Sugahara M, Sakai M, Tsuboi Y (2016) Relationship between tongue thickness and tongue pressure in neuromuscular disorders. Neurol Clin Neurosci 4:142–145. https://doi.org/10.1111/ncn3.12058
Baba T, Goto T, Fujimoto K, Suito H, Yagi K, Nagao K, Ichikawa T (2018) The geniohyoid muscle directly affects masticatory function: evaluation with decision-tree analysis. Int J Prosthodont 31:569–572. https://doi.org/10.11607/ijp.5862
Kajisa E, Tohara H, Nakane A, Wakasugi Y, Hara K, Yamaguchi K, Yoshimi K, Minakuchi S (2018) The relationship between jaw-opening force and the cross-sectional area of the suprahyoid muscles in healthy elderly. J Oral Rehabil 45:222–227. https://doi.org/10.1111/joor.12596
Young HJ, Jenkins NT, Zhao Q, Mccully KK (2015) Measurement of intramuscular fat by muscle echo intensity. Muscle Nerve 52:963–971. https://doi.org/10.1002/mus.24656
Pillen S, Tak RO, Zwarts MJ, Lammens MM, Verrijp KN, Arts IM, van der Laak JA, Hoogerbrugge PM, van Engelen BG, Verrips A (2009) Skeletal muscle ultrasound: correlation between fibrous tissue and echo intensity. Ultrasound Med Biol 35:443–446. https://doi.org/10.1016/j.ultrasmedbio.2008.09.016
Rech A, Radaelli R, Goltz FR, da Rosa LH, Schneider CD, Pinto RS (2014) Echo intensity is negatively associated with functional capacity in older women. Age (Dordr) 36:9708. https://doi.org/10.1007/s11357-014-9708-2
Yamada M, Kimura Y, Ishiyama D, Nishio N, Abe Y, Kakehi T, Fujimoto J, Tanaka T, Ohji S, Otobe Y, Koyama S, Okajima Y, Arai H (2017) Differential characteristics of skeletal muscle in community-dwelling older adults. J Am Med Dir Assoc 18:807.e9. https://doi.org/10.1016/j.jamda.2017.05.011
Feng X, Todd T, Lintzenich CR, Ding J, Carr JJ, Ge Y, Browne JD, Kritchevsky SB, Butler SG (2013) Aging-related geniohyoid muscle atrophy is related to aspiration status in healthy older adults. J Gerontol A Biol Sci Med Sci 68:853–860. https://doi.org/10.1093/gerona/gls225
Bhoyar PS, Godbole SR, Thombare RU, Pakhan AJ (2012) Effect of complete edentulism on masseter muscle thickness and changes after complete denture rehabilitation: an ultrasonographic study. J Investig Clin Dent 3:45–50. https://doi.org/10.1111/j.2041-1626.2011.0088.x
Eichner K (1955) Über eine Gruppeneintelung des lückengebisses für die prothetik. Dtsch Zahnarztl Z 10:1831–1834
Serra MD, Duarte Gavião MB, dos Santos Uchôa MN (2008) The use of ultrasound in the investigation of the muscles of mastication. Ultrasound Med Biol 34:1875–1884. https://doi.org/10.1016/j.ultrasmedbio.2008.05.009
Yamaguchi K, Tohara H, Hara K, Nakane A, Yoshimi K, Nakagawa K, Minakuchi S (2019) Factors associated with masseter muscle quality assessed from ultrasonography in community dwelling elderly individuals:a cross-sectional study. Arch Gerontol Geriatr 82:128–132. https://doi.org/10.1016/j.archger.2019.02.003
Palinkas M, Nassar MS, Cecílio FA, Siéssere S, Semprini M, Machado-de-Sousa JP, Hallak JE, Regalo SC (2010) Age and gender influence on maximal bite force and masticatory muscles thickness. Arch Oral Biol 55:797–802. https://doi.org/10.1016/j.archoralbio.2010.06.016
Albani D, Batelli S, Polito L, Vittori A, Pesaresi M, Gajo GB, De Angeli S, Zanardo A, Gallucci M, Forloni G (2009) A polymorphic variant of the insulin-like growth factor 1 (IGF-1) receptor correlates with male longevity in the Italian population: a genetic study and evaluation of circulating IGF-1 from the "Treviso Longeva (TRELONG)" study. BMC Geriatr 9:19. https://doi.org/10.1186/1471-2318-9-19
Widmer CG, Morris-Wiman J (2010) Limb, respiratory, and masticatory muscle compartmentalization: developmental and hormonal considerations. Prog Brain Res 187:63–80. https://doi.org/10.1016/B978-0-444-53613-6.00005-8
Sato Y, Tanda H, Kato S, Onishi S, Nakajima H, Nanbu A, Nitta T, Koroku M, Akagashi K, Hanzawa T, Shinozaki T, Terao N, Fujisaki N, Kuwabara M, Niimura K (2006) Serum testosterone levels using the radioimmunoassay method in healthy Japanese male volunteers. Reprod Med Biol 5:37–41. https://doi.org/10.1111/j.1447-0578.2006.00121.x
Haring R, Hannemann A, John U, Radke D, Nauck M, Wallaschofski H, Owen L, Adaway J, Keevil BG, Brabant G (2012) Age-specific reference ranges for serum testosterone and androstenedione concentrations in women measured by liquid chromatography-tandem mass spectrometry. J Clin Endocrinol Metab 97:408–415. https://doi.org/10.1210/jc.2011-2134
Hawke TJ, Garry DJ (2001) Myogenic satellite cells: physiology to molecular biology. J Appl Physiol 91:534–551. https://doi.org/10.1152/jappl.2001.91.2.534
Uezumi A, Fukada S, Yamamoto N, Takeda S, Tsuchida K (2010) Mesenchymal progenitors distinct from satellite cells contribute to ectopic fat cell formation in skeletal muscle. Nat Cell Biol 12:143–152. https://doi.org/10.1038/ncb2014
Joe AW, Yi L, Natarajan A, Le Grand F, So L, Wang J, Rudnicki MA, Rossi FM (2010) Muscle injury activates resident fibro/adipogenic progenitors that facilitate myogenesis. Nat Cell Biol 12:153–163. https://doi.org/10.1038/ncb2015
Verdijk LB, Koopman R, Schaart G, Meijer K, Savelberg HH, van Loon LJ (2007) Satellite cell content is specifically reduced in type II skeletal muscle fibers in the elderly. Am J Physiol Endocrinol Metab 292:E151–E157. https://doi.org/10.1152/ajpendo.00278.2006
Osterlund C, Thornell LE, Eriksson PO (2011) Differences in fibre type composition between human masseter and biceps muscles in young and adults reveal unique masseter fibre type growth pattern. Anat Rec (Hoboken) 294:1158–1169. https://doi.org/10.1002/ar.21272
Raadsheer MC, van Eijden TM, van Ginkel FC, Prahl-Andersen B (1999) Contribution of jaw muscle size and craniofacial morphology to human bite force magnitude. J Dent Res 78:31–42
Umeki K, Watanabe Y, Hirano H, Edahiro A, Ohara Y, Yoshida H, Obuchi S, Kawai H, Murakami M, Takagi D, Ihara K, Igarashi K, Ito M, Kawai Y (2018) The relationship between masseter muscle thickness and appendicular skeletal muscle mass in Japanese community-dwelling elders: a cross-sectional study. Arch Gerontol Geriatr 78:18–22. https://doi.org/10.1016/j.archger.2018.05.014
Yamaguchi K, Tohara H, Hara K, Nakane A, Kajisa E, Yoshimi K, Minakuchi S (2018) Relationship of aging, skeletal muscle mass, and tooth loss with masseter muscle thickness. BMC Geriatr 18:67. https://doi.org/10.1186/s12877-018-0753-z
Wallace JD, Calvo RY, Lewis PR, Brill JB, Shackford SR, Sise MJ, Sise CB, Bansal V (2017) Sarcopenia as a predictor of mortality in elderly blunt trauma patients: comparing the masseter to the psoas using computed tomography. J Trauma Acute Care Surg 82:65–72. https://doi.org/10.1097/TA.0000000000001297
Kraemer WJ, Ratamess NA, Nindl BC (2017) Recovery responses of testosterone, growth hormone, and IGF-1 after resistance exercise. J Appl Physiol 122:549–558. https://doi.org/10.1152/japplphysiol.00599.2016
Acknowledgments
The authors would like to thank Editage (http://www.editage.com) for editing and proofreading this manuscript for English language and all staff members and participants of the study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Kohei Yamaguchi, Chizuru Namiki, and Chantaramanee Ariya. The first draft of the manuscript was written by Kohei Yamaguchi and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (Tokyo Medical and Dental University Ethics Committee [ref: D2014-047]) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yamaguchi, K., Hara, K., Nakagawa, K. et al. Association of aging and tooth loss with masseter muscle characteristics: an ultrasonographic study. Clin Oral Invest 24, 3881–3888 (2020). https://doi.org/10.1007/s00784-020-03255-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-020-03255-y