Abstract
Introduction
To investigate the differences in the incidence rates of suspected stage 0/1 osteonecrosis of the jaw (ONJ) and incidence risk of relevant clinical findings of suspected stage 0 ONJ between patients treated with sequential therapy comprising weekly teriparatide for 72 weeks followed by alendronate for 48 weeks vs. those who received monotherapy with alendronate for 120 weeks.
Materials and methods
Suspected stage 0/1 ONJ was defined by non-specific symptoms. Tooth mobility and periodontal symptoms (gingival bleeding, swelling, and/or pain) were selected as clinical findings of suspected stage 0 ONJ. Poisson regression models were applied to calculate the incidence rate ratios of suspected stage 0/1 between the teriparatide group (TG) and alendronate group (AG). Generalized linear models were used to calculate the risk ratios of clinical findings between groups.
Results
Two hundred and sixty-one participants in the TG and 344 in the AG answered a structured questionnaire on oral health and were included in this study. There were no significant differences between the groups in the incidence rate of suspected stage 0/1 ONJ at both 72 and 120 weeks. The risk ratio of the TG to AG for tooth mobility was 0.34 (95% confidence interval [CI] 0.13–0.88, p = 0.02) at 72 weeks and 0.90 (95% CI 0.40–2.03, p = 0.83) at 120 weeks. The incidence rate of tooth mobility related to periodontal symptoms decreased in the TG and increased in the AG during the study.
Conclusion
Tooth mobility accompanied by clinical periodontal symptoms may be a useful early sign of stage 0 ONJ.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In 2003, Marx first reported avascular necrosis of the jaw as a new adverse effect in patients with malignancy or osteoporosis who received high-dose intravenous bisphosphonate (BP), an antiresorptive agent [1]. In 2004, Ruggiero et al. reported a similar adverse effect of refractory osteomyelitis in patients with oncological disease or osteoporosis who had been treated with high-dose intravenous or low-dose oral BP therapy [2]. This adverse effect has subsequently been referred to as BP-related osteonecrosis of the jaw (BRONJ). Because osteonecrosis of the jaw (ONJ) is also found in patients who receive denosumab, which is another type of antiresorptive agent and a potent monoclonal antibody to RANK ligand [3], this disorder is called antiresorptive agent-related ONJ (ARONJ). ARONJ is now called medication-related ONJ (MRONJ) because other drugs, such as angiogenesis inhibitors and immunosuppressive agents, may play a part in the development of ONJ under antiresorptive agent therapy [4]. MRONJ is a rare adverse effect but obviously reduces the quality of life in patients receiving antiresorptive agents for malignancy or metabolic bone diseases such as osteoporosis, Paget’s disease, and osteogenesis imperfecta.
In 2007, the American Association of Oral and Maxillofacial Surgeons (AAOMS) first adopted the main definition for diagnosing BRONJ as persistent exposed necrotic bone in the oral cavity for more than 8 weeks, despite sufficient treatment, in a patient currently or previously receiving BP therapy [5]. Treatment-resistant persistent exposed necrotic bone in the oral cavity is a basic and important concept of BRONJ. However, in 2009, the AAOMS newly added stage 0 BRONJ, which shows no clinical evidence of exposed necrotic bone but has non-specific symptoms, clinical findings, or radiographic abnormalities that might be related to an antiresorptive agent [6]. The progression from stage 1 to stage 3 confirmed BRONJ has been reported in up to 50% of patients with stage 0 BRONJ [7]. As the definition of stage 0 BRONJ includes several non-specific symptoms (such as odontalgia related to a non-odontogenic cause) or non-specific clinical findings (such as tooth mobility), it may be difficult to identify patients with stage 0 BRONJ based on non-specific symptoms or clinical findings in clinical settings because the use of such non-specific diagnostic indicators may result in the overdiagnosis of ONJ [8].
Our previous study found no significant difference in the incidence rate of suspected stage 0/1 ONJ between postmenopausal Japanese women with osteoporosis treated with minodronic acid, a Japanese-made BP, or raloxifene, a selective estrogen receptor modulator [9]. However, as the total number of participants in this previous study was 3,229 (1,612 participants in the minodronic acid group and 1,617 in the raloxifene group) [9], the small sample size may have contributed to the absence of a significant difference between the groups in the incidence rate of suspected stage 0/1 ONJ. The results may also have been influenced by the relatively short observation period (2 years) and an unreliable and/or insensitive definition of ONJ, especially for suspected stage 0 ONJ [9]. The clinical symptoms of suspected stage 0 ONJ in this previous study were persistent dull pain in the jaws, persistent purulent discharge from an intraoral gingival fistula, and hypoesthesia of the lower lip and jaw [9]. These non-specific symptoms were selected in accordance with the 2014 AAOMS position paper and the findings reported by Fedele et al. [4, 7].
Ohbayashi et al. reported that 6 months of treatment with teriparatide results in a significant improvement in patients with osteoporosis with stage 2/3 MRONJ [10]. The effectiveness of teriparatide therapy in stage 0/1 MRONJ compared with stage 2/3 MRONJ was also reported in a recent systematic review and meta-analysis [11]. These results suggest that there would be a difference in the non-specific symptoms or clinical findings of suspected stage 0/1 ONJ between patients with osteoporosis who have received teriparatide vs. BP therapies if the non-specific symptoms or clinical findings of stage 0/1 ONJ are valid and/or sensitive signs. Therefore, if we can identify the sensitive signs of stage 0 ONJ, we may be able to prevent stage 0 ONJ from progressing to exposed ONJ.
The present study investigated the differences in the incidence rate ratios of suspected stage 0/1 ONJ and incidence risk ratios of clinical findings shown in suspected stage 0 ONJ between patients with osteoporosis treated with sequential therapy (from teriparatide to alendronate: teriparatide group) or monotherapy with alendronate (alendronate group) in the Japanese Osteoporosis Intervention Trial-05 (JOINT-05). In addition, we evaluated whether the non-specific symptoms or clinical findings were sensitive indicators of stage 0 ONJ.
Materials and methods
Study design and ethical considerations
The design and rationale for the JOINT-05, a Japanese multicenter, open-label, randomized controlled, head-to-head trial, have been reported previously [12]. The primary endpoint was the incidence of morphometric vertebral fractures. In addition, oral health was followed as one of the secondary endpoints. Data obtained from the JOINT-05, which enrolled 1011 patients from 113 institutes nationwide in Japan between October 2014 and December 2017, were used in the present study. The JOINT-05 was conducted in accordance with the Declaration of Helsinki and the Clinical Trials Act of the Japanese Ministry of Health, Labour, and Welfare. The protocol of the JOINT-05 was approved by the certified review board of Toranomon Hospital and the central ethics committee of the Adequate Treatment of Osteoporosis research group. Written informed consent was obtained from all individual participants included in the study. The JOINT-05 was registered with the Japan Registry of Clinical Trials (registration number jRCTs031180235) and the University Hospital Medical Information Network-Clinical Trials Registry (registration number UMIN000015573).
Study participants
Japanese women aged at least 75 years were eligible for the JOINT-05 if they had primary osteoporosis and were at high risk of fracture. Primary osteoporosis was diagnosed in accordance with the revised 2012 Diagnostic Criteria for Primary Osteoporosis of the Japanese Society for Bone and Mineral Research. Patients at high risk of fracture were defined as those who had one of the following: (i) bone mineral density < 60% of the young adult mean or less than − 3.3 standard deviations; (ii) at least two vertebral fractures in the area from the fourth thoracic vertebra to the fourth lumbar vertebra; (iii) grade 3 prevalent fracture; or (iv) past hip fracture. The exclusion criteria were listed in our previous study [12].
Patients were randomly assigned in a 1:1 ratio to receive sequential therapy (once weekly subcutaneous injection of teriparatide 56.5 µg for 72 weeks followed by alendronate for 48 weeks) or monotherapy with alendronate for 120 weeks. Alendronate was administered as the following formulations: 5 mg tablet (administered orally once daily), 35 mg tablet or jelly (administered orally once weekly), or 900 µg infusion bag (administered intravenously once every 4 weeks). Nature Made Vitamin D400 supplements were also provided to patients in both arms throughout the entire treatment period.
A structured questionnaire on oral health status that was used in the JOINT-04 was applied to identify non-specific symptoms or clinical findings related to suspected stage 0/1 ONJ [9]. In the present study, suspected stage 0 ONJ was determined on the basis of three non-specific symptoms identified in our previous study [9]: persistent dull pain in the jaws, persistent purulent discharge from an intraoral gingival fistula, or hypoesthesia of the lower lip and jaw. Suspected stage 1 ONJ was defined as the presence of exposed bone of the jaws.
In the present study, tooth mobility was selected as a non-specific clinical finding that may be related to suspected stage 0 ONJ in accordance with the 2022 AAOMS position paper [13]. As the Japanese position paper revised in 2016 reported that patients with stage 0 ARONJ may have deep periodontal pockets, oral mucosal ulceration, swelling, abscess formation, and non-odontogenic pain [14], we also selected persistent gingival bleeding, swelling, and/or pain as a potential clinical finding related to suspected stage 0 ONJ.
Based on the oral health questionnaire administered in the JOINT-05 (version 10), participants who had periodontal symptoms, peri-implantitis, incompatible dentures, non-specific symptoms related to the jaws, and/or bone exposure of the jaws at 72 weeks and 120 weeks were referred to general dental practitioners to confirm and treat these conditions. When the presence of ONJ was doubted, the dentists evaluated dental radiographs to confirm the presence of ONJ.
Statistical analyses
In descriptive analyses, binary variables were summarized as frequencies and proportions, while continuous variables were summarized as means and standard deviations. The independent t test or Fisher’s exact test was used to investigate the differences in the background and oral health characteristics between the teriparatide and alendronate groups. In addition, the differences between the groups in the number of teeth extracted in the previous year and clinical symptoms of peri-implantitis at 72 and 120 weeks were investigated to evaluate the influence of tooth extraction and peri-implantitis on ONJ.
The incidence rates of ONJ were estimated by the person-year method. In the comparison of the incidence rates of ONJ between treatment arms, Poisson regression models were fit to calculate the rate ratios of teriparatide to alendronate, 95% confidence intervals (CIs), and p values. For calculation of the p values, Fisher’s exact test was used if there were zero cells; otherwise, the Wald test of Poisson regression models was used. To compare binary outcomes, generalized linear models with the log link were fit to calculate the risk ratios of teriparatide to alendronate, 95% CIs, and p values based on Fisher’s exact test. Missing data were handled by complete-case analysis without imputation.
As periodontal disease, especially periodontal abscess, may cause tooth mobility, we used Fisher’s exact test to evaluate the association of tooth mobility with clinical symptoms of periodontal disease (persistent gingival bleeding, swelling, and/or pain) in both groups at baseline, 72 weeks, and 120 weeks. All comparisons were two sided with the level of significance set as p = 0.05. Statistical analysis was performed using SAS version 9.4 (SAS Institute, Cary, NC, USA).
Results
Participant characteristics at baseline
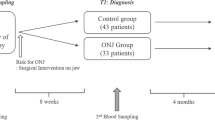
Of 1,011 participants (505 in the teriparatide group and 506 in the alendronate group) in the JOINT-05, 605 participants (261 participants in the teriparatide group and 344 in the alendronate group) answered a structured questionnaire on oral health and were included in the present study (Fig. 1). The background characteristics of the study participants at baseline are shown in Table 1. Characteristics related to the oral health of the study participants at baseline are shown in Table 2. The background characteristics were similar between the groups; the rate of diabetes mellitus tended to be lower in the teriparatide group than the alendronate group (6.9% vs. 9.3%), although this difference did not reach statistical significance (p = 0.30).
There were no non-specific symptoms related to suspected stage 0/1 ONJ in either the teriparatide group or the alendronate group at baseline (Table 2). The rate of tooth mobility at baseline tended to be higher in the teriparatide group (10.2%) than the alendronate group (6.5%), although this difference did not reach statistical significance (p = 0.13). The rate of persistent gingival bleeding, swelling, and/or pain at baseline was similar in both groups (5.0% in the teriparatide group vs. 4.4% in the alendronate group, p = 0.70). The rate of persistent pain caused by dentures at baseline tended to be higher in the teriparatide group than in the alendronate group (5.0% vs. 2.8%), although this difference did not reach statistical significance (p = 0.24). Three participants in the teriparatide group had peri-implantitis at baseline, although there were many missing data on dental implants in both groups.
There were no significant differences between the groups in the number of teeth extracted in the previous year at 72 weeks (p = 0.84) and 120 weeks (p = 0.29) (Table 3). No participant in either group had clinical symptoms of peri-implantitis at 72 weeks. There was no significant difference between the groups in the rate of clinical symptoms of peri-implantitis at 120 weeks (p = 0.47).
Difference between the groups in the incidence rate of suspected stage 0/1 ONJ
At 72 weeks, the incidence rates of suspected stage 0 ONJ in the teriparatide and alendronate groups were 1.39 and 0.84, respectively. There was no significant difference between the groups in the incidence rate of suspected stage 0 ONJ (incidence rate ratio of the teriparatide to alendronate groups = 1.66, 95% CI 0.45–6.18, p = 0.45) (Table 4). At 120 weeks, the incidence rates of suspected stage 0 ONJ in the teriparatide and alendronate groups were 1.33 and 0.63, respectively. There was no significant difference between the groups in the incidence rate of suspected stage 0 ONJ (incidence rate ratio of the teriparatide to alendronate groups = 2.09, 95% CI 0.68–6.40, p = 0.20).
There were two participants with suspected stage 1 ONJ in the alendronate group during 72 weeks of follow-up (incidence rate = 0.42) (Table 4). After that, there were no patients with bone exposure in the alendronate group (incidence ratio = 0.25 at 120 weeks). There were no patients in the alendronate group with confirmed stage 1 ONJ. The teriparatide group had no patients with bone exposure during 120 weeks of follow-up. There were no significant differences between the groups in the incidence rates of suspected stage 1 ONJ at 72 weeks (p = 0.51) and 120 weeks (p = 0.51).
Overall, as two participants with bone exposure also showed clinical symptoms of suspected stage 0 ONJ, the incidence rate ratios of the teriparatide to alendronate groups for suspected 0/1 ONJ were the same as those for suspected stage 0 ONJ at both 72 and 120 weeks (Table 4).
Risk ratios between the groups for clinical findings related to suspected stage 0 ONJ
At 72 weeks, the number (%) of participants with persistent gingival bleeding, swelling, and/or pain were eight (3.40%) in the teriparatide group and 22 (6.88%) in the alendronate group. The risk ratio of the teriparatide to alendronate groups was 0.50 (95% CI 0.22–1.09, p = 0.09) (Table 5). At 120 weeks, the number (%) of participants with persistent gingival bleeding, swelling, and/or pain were six (2.91%) in the teriparatide group and eight (2.91%) in the alendronate group. The risk ratio of the teriparatide to alendronate groups was 1.00 (95% CI 0.35–2.84, p = 1.00).
At 72 weeks, the number (%) of participants with new-onset tooth mobility were five (2.30%) in the teriparatide group and 21 (6.84%) in the alendronate group. The risk ratio of the teriparatide to alendronate groups was 0.34 (95% CI 0.13–0.88, p = 0.02) (Table 5). At 120 weeks, the number (%) of participants with new-onset tooth mobility were nine (4.71%) in the teriparatide group and 14 (5.24%) in the alendronate group. The risk ratio of the teriparatide to alendronate groups was 0.90 (95% CI 0.40–2.03, p = 0.83).
Association between tooth mobility and clinical periodontal symptoms
There were no significant associations between tooth mobility and clinical periodontal symptoms (persistent gingival bleeding, swelling, and/or pain) in the teriparatide group at baseline (p = 0.63), 72 weeks (p = 1.00), and 120 weeks (p = 1.00) (Table 6). No participant in the teriparatide group had tooth mobility related to clinical periodontal symptoms at 72 and 120 weeks, although two (7.69%) participants had tooth mobility at baseline.
There were significant associations between tooth mobility and clinical periodontal symptoms in the alendronate group at 72 weeks (p = 0.04) and 120 weeks (p = 0.01), but not at baseline (p = 1.00). The incidence rate of tooth mobility related to clinical periodontal symptoms in the alendronate group increased from 4.55% at baseline to 19.05% at 72 weeks and 21.43% at 120 weeks.
Discussion
In the present study, there were no significant differences between the teriparatide and alendronate groups in the incidence rates of suspected stage 0 and stage 1 ONJ for up to 120 weeks. However, the incidence rate of tooth mobility was significantly smaller in the teriparatide group than the alendronate group at 72 weeks but was similar in both groups at 120 weeks. The significant effectiveness of teriparatide therapy for stage 0 and stage 1 MRONJ has recently been reported [11]. However, the present study had a small sample size and short study period (only 72 weeks) for the group receiving teriparatide therapy alone. In the present study, there was no significant difference between the teriparatide and alendronate groups in the incidence rate of tooth mobility at 120 weeks. Our results may suggest that new-onset tooth mobility may be prevented by teriparatide therapy and may appear early in patients with stage 0 ONJ.
The presence of periodontal disease is associated with tooth mobility. In particular, periodontal abscess is an acute destructive process in the periodontium that largely contributes to tooth mobility. However, in the present study, there was no significant association between tooth mobility and clinical symptoms of periodontal disease at baseline. It is likely that participants had no awareness of the clinical symptoms of mild periodontal disease at baseline, although there may be accumulated alveolar bone resorption associated with tooth mobility. However, the rate of tooth mobility accompanied by clinical symptoms of periodontal disease increased in the alendronate group during the study, suggesting that alendronate may increase tooth mobility related to clinical symptoms of periodontal disease. Conversely, no participants in the teriparatide group had tooth mobility accompanied by clinical symptoms of periodontal disease, suggesting that teriparatide may reduce tooth mobility related to clinical symptoms of periodontal disease. These findings suggest that tooth mobility caused by severe periodontal disease (such as periodontal abscess) may be a useful clinical sign of stage 0 ONJ, although Patel et al. reported the clinical finding of stage 0 BRONJ as tooth mobility not caused by periodontal disease [15]. Fedele et al. emphasized that use of the traditional definition of ONJ may result in one-quarter of patients remaining undiagnosed [16].
In our previous study comparing suspected stage 0 ONJ between participants treated with minodronic acid and raloxifene, we defined suspected stage 0 ONJ as persistent dull pain in the jaws, persistent purulent discharge from an intraoral gingival fistula, or hypoesthesia of the lower lip and jaw [9]. Fedele et al. reported that the most common clinical symptom of non-exposed BRONJ (i.e., stage 0 BRONJ) is jawbone pain (91.6%) followed by sinus tract formation in the jaw (51%) [7]. A magnetic resonance imaging study reported that non-exposed BRONJ is characterized by hypointensity on T1-weighted images and hyperintensity on T2-weighted and short tau inversion recovery images, which suggest the pattern of osteomyelitis [17]. Hypoesthesia of the lower lip and jaw sometimes occurs in patients with osteomyelitis. The 2014 AAOMS position paper reports altered neurosensory function as a symptom of stage 0 MRONJ [4]. However, our current study findings suggest that persistent dull pain in the jaws, persistent purulent discharge from an intraoral gingival fistula, or hypoesthesia of the lower lip and lower jaw are unreliable or insensitive indicators of suspected stage 0 ONJ in comparison to the finding of tooth mobility related to clinical periodontal symptoms. This unreliable definition may be associated with the lack of difference in the incidence rate of suspected stage 0 ONJ between participants treated with minodronic acid and raloxifene in our previous study [9].
Two participants in the alendronate group had bone exposure in the jaw at 72 weeks that disappeared between 72 and 120 weeks, and had symptoms defined as suspected stage 0 ONJ. As stage 1, ONJ is defined as exposed and necrotic bone in patients who are asymptomatic and have no evidence of infection, these two suspected cases in our study were not defined as stage 1 ONJ. In our previous study comparing suspected stage 1 ONJ between participants treated with minodronic acid vs. raloxifene, bone exposure of the jaw was present at baseline in 11 patients (0.7%) in the minodronic acid group and six (0.4%) in the raloxifene group [9]. This suggests that bone exposure of the jaw may appear in patients with osteoporosis without exposure to antiresorptive agents like BP and denosumab. Kunihara et al. recently reported that the annual incidence rate of ONJ without antiresorptive agent use is 5.1 per 100,000, which is larger than expected [18]. They considered that the major cause of ONJ in these cases is continuous bacterial infection of the jaw caused by inadequate oral hygiene management [18]. Patients with osteoporosis have a higher risk of periapical and/or periodontal diseases that might influence exposure of the jawbone [19, 20]. Additionally, we previously demonstrated that Japanese older adults with fragility fractures without antiresorptive agents may have a higher risk of problematic delayed wound healing after tooth extraction than those without fractures [21, 22]. Thus, delayed wound healing after tooth extraction may result in the development of ONJ in patients with osteoporosis without exposure to BP and/or denosumab.
There were no patients with jawbone exposure in the teriparatide group. A recent study reported the significant effectiveness of teriparatide therapy for stage 1 MRONJ [11]. Thus, it is possible that jawbone exposure may have been prevented by the teriparatide therapy in the present study. However, as the sample size of our current study was too small, further study of more patients receiving teriparatide therapy is needed to clarify whether teriparatide reduces the risk of bone exposure of the jaw.
Ideally, a regular oral hygiene management should be performed for all osteoporosis patients who receive antiresorptive therapy to reduce the incidence of ONJ. Beth-Tasdogan et al. concluded that dental examinations at 3-month intervals and preventive treatments including tooth extraction may be more effective than standard care for reducing the incidence of MRONJ in cancer patients taking intravenous BPs [23]. Our recent study conducted in Japan Osteoporosis Society (JOS) revealed that the rate of physicians who requested oral health care by a dentist has increased from 32.9% in the 2015 survey to 72.7% in the 2022 survey because the physicians who belong to JOS wanted to reduce the incidence of ONJ as possible [24]. However, it may be difficult for physicians to refer all their patients with osteoporosis to dentists in the real world. If self-reported tooth mobility associated with clinical symptoms of periodontal disease is one kind of clue, physicians may be able to select the patients at risk of ONJ and refer them to dentists before or during the treatment.
The present study had some limitations. Of the 1,011 participants (505 in the teriparatide group and 506 in the alendronate group), only 261 (51.7%) participants in the teriparatide group and 344 (68.0%) in the alendronate group participated in the oral health study. The small number of participants as well as the disproportionate ratio of participants in the two groups may have influenced the present results. Compared with the alendronate group, the teriparatide group had a smaller mean number of teeth present at baseline (10.4 vs. 12.0), higher rate of tooth mobility at baseline (10.2% vs. 6.5%), and higher rate of persistent pain caused by dentures at baseline (5.0% vs. 2.8%). These findings suggest that the teriparatide group may have had worse oral health than the alendronate group. There was no information on oral health awareness and practices such as routine dental checkups, although there was no significant difference between the teriparatide and alendronate groups in the number of times the teeth were brushed per day (daily toothbrushing sessions in Table 2). Nonetheless, the teriparatide group had a significantly lower risk of tooth mobility than the alendronate group in the short term (within 72 weeks).
The rate of persistent gingival bleeding, swelling, and/or pain decreased after 72 weeks in both groups. In particular, the rate was 6.9% at 72 weeks and 2.9% at 120 weeks in the alendronate group. However, it is unknown whether some of the participants with persistent gingival bleeding, swelling, and/or pain at 72 weeks discontinued the present study. Previous studies have suggested that BP therapy has protective effects against periodontal disease in postmenopausal women [25, 26]. This protective effect may appear between 72 and 120 weeks in both groups.
There was a significant difference in the incidence rate of tooth mobility between the teriparatide and alendronate groups at 72 weeks in the present study, and this difference between the groups disappeared at 120 weeks. As teriparatide basically contributes to the progression of bone formation, this may simply improve the deterioration of alveolar bone [27], resulting in a decreased risk of tooth mobility. The finding of tooth mobility may not directly be an early sign of stage 0 ONJ. However, this finding was detected in the small number of participants with osteoporosis during short-term follow-up. Future research is needed to investigate this clinical finding in patients receiving antiresorptive therapy for osteoporosis or malignancy.
The patients cannot precisely answer the number and extent of symptoms/clinical findings in the questionnaire by themselves. Although the details like the number and the extent might be the important sign associated with MRONJ, the questionnaire-based oral health examination in JOINT-05 did not seek the detail information of symptoms/clinical findings in this study. Further study which includes the number and extent of symptoms/clinical findings would be necessary to clarify the results of our study.
In conclusion, there was no significant difference between the teriparatide and alendronate groups in the incidence rate of suspected stage 0/1 ONJ defined by non-specific symptoms. These non-specific symptoms may be too unreliable or insensitive to define stage 0 ONJ. However, the teriparatide group had a significantly lower risk of tooth mobility related to clinical periodontal symptoms than the alendronate group. The clinical finding of tooth mobility accompanied by periodontal disease may be a useful early sign for identifying stage 0 ONJ.
References
Marx RE (2003) Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: a growing epidemic. J Oral Maxillofac Surg 61:1115–1117
Ruggiero SL, Mehrotra B, Rosenberg TJ, Engroff SL (2004) Osteonecrosis of the jaws associated with the use of bisphosphonates: a review of 63 cases. J Oral Maxillofac Surg 62:527–534
Stopeck AT, Lipton A, Body JJ, Steger GG, Tonkin K, de Boer RH, Lichinitser M, Fujiwara Y, Yardley DA, Viniegra M, Fan M, Jiang Q, Dansey R, Jun S, Braun A (2010) Denosumab compared with zoledronic acid for the treatment of bone metastases in patients with advanced breast cancer: a randomized, double-blind study. J Clin Oncol 28:5132–5139
Ruggiero SL, Dodson TB, Fantasia J, Goodday R, Aghaloo T, Mehrotra B, O’Ryan F, American Association of Oral and Maxillofacial Surgeons (2014) American Association of Oral and Maxillofacial Surgeons position paper on medication-related osteonecrosis of the jaw–2014 update. J Oral Maxillofac Surg 72:1938–1956
Advisory Task Force on Bisphosphonate-Related Osteonecrosis of the Jaws, American Association of Oral and Maxillofacial Surgeons (2007) American Association of Oral and Maxillofacial Surgeons position paper on bisphosphonate-related osteonecrosis of the jaws. J Oral Maxillofac Surg 65:369–376
Ruggiero SL, Dodson TB, Assael LA, Landesberg R, Marx RE, Mehrotra B, American Association of Oral and Maxillofacial Surgeons (2009) American Association of Oral and Maxillofacial Surgeons position paper on bisphosphonate-related osteonecrosis of the jaws–2009 update. J Oral Maxillofac Surg 67:2–12
Fedele S, Porter SR, D’Aiuto F, Aljohani S, Vescovi P, Manfredi M, Arduino PG, Broccoletti R, Musciotto A, Di Fede O, Lazarovici TS, Campisi G, Yarom N (2010) Nonexposed variant of bisphosphonate-associated osteonecrosis of the jaw: a case series. Am J Med 123:1060–1064
Khan AA, Morrison A, Hanley DA, Felsenberg D, McCauley LK, O’Ryan F, Reid IR, Ruggiero SL, Taguchi A, Tetradis S, Watts NB (2015) Diagnosis and management of osteonecrosis of the jaw: a systematic review and international consensus. J Bone Miner Res 30:3–23
Taguchi A, Uemura Y, Imai T, Tanaka S, Ohta H, Nakamura T, Orimo H, Sugimoto T, Soen S, Shiraki M, Adequate Treatment of Osteoporosis (A-TOP) research group (2019) Incidence of osteonecrosis of the jaw in Japanese osteoporosis patients taking minodronic acid. J Bone Miner Metab 37:886–892
Ohbayashi Y, Iwasaki A, Nakai F, Mashiba T, Miyake M (2020) A comparative effectiveness pilot study of teriparatide for medication-related osteonecrosis of the jaw: daily versus weekly administration. Osteoporos Int 31:577–585
Dos Santos FL, Abreu LG, Calderipe CB, Martins MD, Schuch LF, Vasconcelos ACU (2021) Is teriparatide therapy effective for medication-related osteonecrosis of the jaw? A systematic review and meta-analysis. Osteoporos Int 32:2449–2459
Tanaka S, Mori S, Hagino H, Sugimoto T (2020) Design of a randomized trial of teriparatide followed by alendronate: Japanese Osteoporosis Intervention Trial-05 (JOINT-05). J Bone Miner Metab 38:412–417
Ruggiero SL, Dodson TB, Aghaloo T, Carlson ER, Ward BB, Kademani D (2022) American Association of Oral and Maxillofacial Surgeons’ position paper on medication-related osteonecrosis of the jaws–2022 update. J Oral Maxillofac Surg 80:920–943
Yoneda T, Hagino H, Sugimoto T, Ohta H, Takahashi S, Soen S, Taguchi A, Nagata T, Urade M, Shibahara T, Toyosawa S, Japanese Allied Committee on Osteonecrosis of the Jaw (2017) Antiresorptive agent-related osteonecrosis of the jaw: Position Paper 2017 of the Japanese Allied Committee on Osteonecrosis of the Jaw. J Bone Miner Metab 35:6–19
Patel S, Choyee S, Uyanne J, Nguyen AL, Lee P, Sedghizadeh PP, Kumar SK, Lytle J, Shi S, Le AD (2012) Non-exposed bisphosphonate-related osteonecrosis of the jaw: a critical assessment of current definition, staging, and treatment guidelines. Oral Dis 18:625–632
Fedele S, Bedogni G, Scoletta M, Favia G, Colella G, Agrillo A, Bettini G, Di Fede O, Oteri G, Fusco V, Gabriele M, Ottolenghi L, Valsecchi S, Porter S, Fung PP, Saia G, Campisi G, Bedogni A (2015) Up to a quarter of patients with osteonecrosis of the jaw associated with antiresorptive agents remain undiagnosed. Br J Oral Maxillofac Surg 53:13–17
Bedogni A, Blandamura S, Lokmic Z, Palumbo C, Ragazzo M, Ferrari F, Tregnaghi A, Pietrogrande F, Procopio O, Saia G, Ferretti M, Bedogni G, Chiarini L, Ferronato G, Ninfo V, Lo Russo L, Lo Muzio L, Nocini PF (2008) Bisphosphonate-associated jawbone osteonecrosis: a correlation between imaging techniques and histopathology. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 105:358–364
Kunihara T, Tohmori H, Tsukamoto M, Kobayashi M, Okumura T, Teramoto H, Hamasaki T, Yamasaki T, Nakagawa T, Okimoto N, Fujiwara S (2023) Incidence and trend of antiresorptive agent-related osteonecrosis of the jaw from 2016 to 2020 in Kure, Japan. Osteoporos Int. https://doi.org/10.1007/s00198-023-06732-8
Xu S, Zhang G, Guo JF, Tan YH (2021) Associations between osteoporosis and risk of periodontitis: a pooled analysis of observational studies. Oral Dis 27:357–369
Katz J, Rotstein I (2021) Prevalence of periapical lesions in patients with osteoporosis. J Endod 47:234–238
Taguchi A, Kamimura M, Nakamura Y, Sugino N, Ichinose A, Maezumi H, Fukuzawa T, Ashizawa R, Takahara K, Gushiken S, Mukaiyama K, Ikegami S, Uchiyama S, Kato H (2016) Delayed wound healing after tooth extraction and self-reported kyphosis in Japanese men and women. Sci Rep 6:36309
Taguchi A, Ikegami S, Tokida R, Kamimura M, Sakai N, Horiuchi H, Takahashi J, Kato H (2020) Fragility fractures and delayed wound healing after tooth extraction in Japanese older adults. J Bone Miner Metab 38:357–362
Beth-Tasdogan NH, Mayer B, Hussein H, Zolk O, Peter JU (2022) Interventions for managing medication-related osteonecrosis of the jaw. Cochrane Database Syst Rev 7:CD012432
Taguchi A, Hagino H, Inoue D, Endo N, Society JO (2023) Cooperation between physicians and dentists for osteonecrosis of the jaw: a 2022 Japanese survey. J Bone Miner Metab. https://doi.org/10.1007/s00774-023-01458-3
Penoni DC, Leão ATT, Torres SR, Farias MLF, Fernandes TM, Crivelli M, Vettore MV (2018) Effects of bone fragility and antiresorptive drugs on periodontal disease and tooth loss: a longitudinal study. JDR Clin Trans Res 3:378–387
Taguchi A, Shiraki M, Tanaka S, Ohshige H, Nakamura T (2019) Improved periodontal disease and prevention of tooth loss in osteoporosis patients receiving once-yearly zoledronic acid: a randomized clinical trial. Menopause 26:1277–1283
Bashutski JD, Eber RM, Kinney JS, Benavides E, Maitra S, Braun TM, Giannobile WV, McCauley LK (2010) Teriparatide and osseous regeneration in the oral cavity. N Engl J Med 363:2396–2405
Acknowledgements
The authors thank the clinical investigators in the JOINT-05, members of the central ethics committee, and members of the data monitoring committee. The authors would also like to express their sincere thanks to the chairman, Dr. Itsuo Gorai, and the members of the central ethics committee for the JOINT trials. Finally, the authors thank Mr. Teruhiko Miyazaki and Ms. Yuko Iwata for their secretarial help, and Kelly Zammit, BVSc, from Edanz (https://jp.edanz.com/ac), for editing a draft of this manuscript.
Funding
This Japanese Osteoporosis Intervention Trial-05 (JOINT-05) study was sponsored by the Public Health Research Foundation. This study was also supported in part by funding through the Project Promoting Clinical Trials for Development of New Drugs (19lk0201061t0004) from the Japan Agency for Medical Research and Development (AMED) to Shiro Tanaka and partially funded by Japan Society for the Promotion of Science (JSPS) KAKENHI grants provided to A.T. (grant nos. 21K09885 and 22K10208).
Author information
Authors and Affiliations
Consortia
Contributions
AT: conceptualization, methodology, validation, formal analysis, investigation, resources, writing—original draft, writing—review and editing, and funding acquisition. ST: methodology, software, formal analysis, writing—original draft. TO: methodology, software, formal analysis. KA: methodology, software, formal analysis. SM: conceptualization, methodology, validation, project administration, writing—review and editing. HO: investigation, resources, data curation, visualization, project administration, writing—review and editing. HH: conceptualization, methodology, validation, project administration, writing—review and editing. ST: conceptualization, methodology, validation, project administration, writing—review and editing. MS: investigation, visualization, project administration, writing—review and editing. TN: conceptualization, methodology, investigation, writing—review and editing. SS: conceptualization, methodology, validation, project administration, writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
A. Taguchi has received lecture fees from Asahi Kasei Pharma Corp., Daiichi Sankyo Co. Ltd., Chugai Pharmaceutical Co. Ltd., and Teijin Pharma Ltd. He has received a consultation fee from Media Corp. Ltd. Shiro Tanaka has received lecture fees from Bayer Yakuhin, Amgen Astellas BioPharma K.K., and the Research Institute of Healthcare Data Science. He has received consultation fees and outsourcing fees from Daiichi Sankyo Company, Limited, Boehringer Ingelheim, Satt, and the Public Health Research Foundation. He has received research grants from the Japan Agency for Medical Research and Development, the Japanese Ministry of Health Labor and Welfare, the Japanese Ministry of Education, Science, and Technology, and Novo Nordisk. He engaged in a research project of the Japan Agency for Medical Research and Development. H. Hagino has received lecture fees or grants outside the submitted work from Amgen Inc., Asahi Kasei Pharma Corp., Astellas Pharma Inc., Chugai Pharmaceutical Co., Ltd., Daiichi Sankyo Co., Ltd., Eisai Co., Ltd., Mochida Pharma Corp., Taisho Pharmaceutical Co., Ltd., Teijin Pharma Co., Ltd., and UCB Japan. S. Soen has received consulting fees, speaking fees, and/or honoraria from Amgen, Asahi Kasei Pharma, Astellas Pharma, Chugai, Daiichi Sankyo, Eisai, Ono, Teijin Pharma, and UCB Japan. T. Ozaki, K. Arai, S. Mori, H. Ohta, Sakae Tanaka, M. Shiraki, and T. Nakamura have no conflicts of interests.
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The protocol of the Japanese Osteoporosis Intervention Trial-05 (JOINT-05) was approved by the certified review board of Toranomon Hospital and the central ethics committee of the Adequate Treatment of Osteoporosis (A-TOP) research group.
Consent to participate
All patients provided written informed consent.
Consent for publication
All patients provided written informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Taguchi, A., Tanaka, S., Ozaki, T. et al. Reliability of early stage symptoms/clinical findings of osteonecrosis of the jaw: Japanese Osteoporosis Intervention Trial-05 (JOINT-05). J Bone Miner Metab 41, 854–864 (2023). https://doi.org/10.1007/s00774-023-01466-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-023-01466-3