Abstract
This study sought to investigate the prevalence of osteoporosis and the role of sex hormone levels in the determination of bone mineral density (BMD) and osteoporosis in a Vietnamese population of women and men. The cross-sectional study involved 269 women and 222 men aged 13–83 years, who were randomly selected from urban and rural areas in northern Vietnam. Serum concentrations of estradiol and testosterone were analyzed, and BMD was measured by dual X-ray absorptiometry. We found that the prevalence of osteoporosis in postmenopausal women was 18, 17, and 37 % for the femoral neck, total hip, and lumbar spine, respectively. For men aged 50 years or older, the corresponding values were 8, 7, and 12 %. In men, the most important predictors of BMD for the femoral neck and total hip were age, body mass index, and serum levels of estradiol. For the BMD of the lumbar spine, testosterone also had a significant influence. Determinants of osteoporosis in men for the total hip and lumbar spine were age, weight, and serum concentrations of estradiol and testosterone. In postmenopausal women, age, weight, and residence (urban vs rural) were the most important predictors of BMD and osteoporosis. For all women (including those of reproductive age), serum levels of estradiol were also significant. These data suggest that the prevalence of osteoporosis in the Vietnamese population is high also in men, and that estradiol levels are essential for bone mass in both men and women. The results should have clinical implications and increase awareness of an important health issue within Vietnamese society.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis and resulting fracture is a global public health problem, associated with increased mortality, concomitant morbidity, and reduced quality of life [1]. Most research on osteoporosis has focused on women, but this disorder is a serious problem also in men [2]. Bone mineral density (BMD) in men is decreased by age and is also a major determinant of fracture risk [3, 4]. We previously reported a prevalence of osteoporosis in Vietnamese women of similar magnitude as in Caucasian women [5]. In a recent cross-sectional study, we found a high prevalence of vitamin D deficiency not only in women but also in men from northern Vietnam [6]. Vitamin D has an important role in the regulation of calcium and bone metabolism [7], and low levels have been linked to low BMD in both women and men [6, 8].
Sex steroids, e.g., estrogen and testosterone, are other important factors for bone metabolism in both women and men [9]. In vitro and in vivo studies indicate estrogens and androgens act via different cellular mechanisms [10]. The bone-sparing effect of estrogen is antiresorption by inhibition of osteoclast activity. The skeletal effect of androgens may be partly mediated by local aromatization to estrogen. However, there are also data that support a direct androgen action on bone. The presence of specific androgen receptors in cultured osteoblasts has been reported [11]. Androgens were shown to stimulate proliferation and differentiation of osteoblasts and to inhibit apoptosis. In animal studies, treatment with dihydrotestosterone, which cannot be aromatized to estrogen, stimulated bone formation and thus supported a direct stimulatory effect of androgens on bone [12].
In women, reduced estrogen levels, e.g., following oophorectomy or natural menopause, are well known to increase bone loss, reduce bone mass, and increase the risk of osteoporosis and fracture [13]. In men, the relation between sex hormone levels, bone loss, and risk of osteoporosis is more uncertain. Also, the relative importance of testosterone versus estrogen for the maintenance of BMD in men is unclear. Men with gonadal insufficiency have lower BMD than healthy subjects of the same age, and testosterone replacement therapy has been suggested to reverse this condition [14]. However, the role of testosterone in BMD in men has also been questioned [15].
Risk factors for low BMD and for osteoporosis in Asian populations have not been well documented, particularly with respect to men. Genetic variations between Asian and Caucasian populations in sex steroid hormone metabolism regulating bone mass have been demonstrated [16]. Furthermore, there may be ethnic differences in gene polymorphisms involved in bone metabolism [17].
Vietnam is located in Southeast Asia, and owing to recent economic development the country has undergone rapid urbanization, in which there is a clear separation between urban and rural areas. The present study was designed to explore blood levels of estradiol and testosterone and the association of these hormones with BMD and osteoporosis in a representative sample of men and women of different ages from urban and rural areas in northern Vietnam.
Materials and methods
Study design
The study was designed as a cross-sectional investigation with a multistage sampling scheme. Within the setting of northern Vietnam (latitude 21°N), two districts (Dong Da in Hanoi and Kim Bang in Hanam) were selected to represent urban and rural areas, respectively. From each of these districts, four communes were randomly selected, and a full list of all inhabitants was obtained from the local government authority, and this served as the sampling frame. The list of inhabitants was then sorted by age in 10-year groups. For each age group, a total of 140 individuals (70 women, 70 men) were randomly selected by computer-generated numbers and invited for a screening interview.
On the basis of published literature [18], where the prevalence of osteoporosis in men ranged between 10 and 20 %, we estimated that a sample size of 170 individuals would be adequate to calculate the prevalence within six percentage points of the true proportion with a 95 % confidence interval. In a subsample of at least 100 individuals (men aged 50 years or older) the corresponding figure was eight percentage points. To estimate the prevalence in both men and women, we aimed to recruit at least 400 individuals.
The research protocol and procedures were approved by the ethics councils of Hanoi Medical University and Karolinska Institutet, Stockholm, Sweden (no. 05/IRB and no. 97/HMU IRB). All participants were provided with adequate information about the objectives of the study and had given their oral informed consent to participate, according to the principles of medical ethics of the World Health Organization.
Data collection
A letter of invitation was sent to 980 individuals, among whom 823 came for the screening. Screening interviews were performed at the local health care center by health professionals from Hanoi Medical University, and participants were offered a free health checkup. Exclusion criteria were chronic diseases and disorders which affect bone metabolism, such as cancer, malabsorption syndrome, hepatic and renal disease, diabetes, ongoing pregnancy or lactation, use of medications influencing bone metabolism within the last 4 weeks, and a history of skeletal disease or immobility for more than 1 month. Women were excluded if they had undergone hysterectomy/oophorectomy.
After screening, 604 individuals fulfilled the inclusion criteria, and among them 559 ultimately participated in the study. A structured questionnaire was administered to each participant by health professionals from Hanoi Medical University. The questionnaire covered data on anthropometric variables, clinical history, lifestyle, and dietary habits. Height (in centimeters) without shoes was measured by a wall-mounted stadiometer. Weight (in kilograms), without shoes or clothing, was measured on an electronic scale. Body mass index (BMI) was then derived as the ratio of weight in kilograms divided by height in meters squared.
BMD was measured for the lumbar spine, left and right femoral neck, and total hip in all participants. The measurement was done with a dual-energy X-ray absorptiometry densitometer (Hologic Explorer 4500). The precision error (coefficient of variation as a percentage) in our laboratory was 1.75 % for lumbar spine and 1.50 % for hips. The densitometry was standardized by a phantom every time before measurement. In this analysis, BMD at the lumbar spine was estimated from L1 to L4. The femoral neck BMD and the total hip BMD used in the analysis were estimated from the right side. Estimates of peak BMD were based on bootstrap analysis of BMD in 118 women and 116 men in the age group 13–49 years [5]. From the peak BMD, the T score, which is the number of standard deviations from the peak BMD, was calculated for each individual in the study sample. The prevalence of osteoporosis was estimated for men aged 50 years or older and for postmenopausal women (more than 1 year since last menstruation). An individual was classified as having osteoporosis if the BMD T score was equal to or lower than −2.5 [19].
Serum analyses
Blood samples were drawn in the morning after fasting, and were centrifuged within 30 min after collection. Serum samples were then frozen at −80 °C until analysis. Serum concentrations of 17β-estradiol and testosterone were measured by electrochemiluminescence immunoassay (Roche Diagnostics). The intra-assay coefficient of variation was 5.2 % for estradiol and 5.7 % for testosterone. The interassay coefficients of variation were 9.2 and 9.9 %, respectively.
Statistical analysis
Characteristics of the participants are presented as the mean and standard deviation or median and range. For categorical data, frequency counts and percentages are presented. In comparisons between men and women, with respect to the characteristics of the participants, the t test for independent samples was used for continuous data and the chi square test was used for data measured on a nominal scale. All regression analyses were done separately for men, women of reproductive age, and postmenopausal women. Univariate and forward stepwise multiple linear regression analysis was used to evaluate to what extent the variation in different BMD measures could be explained by estradiol level, testosterone level, age, height, weight, BMI, residency, smoking, alcohol drinking, and coffee drinking. The results from the stepwise regression models are presented as the unstandardized regression coefficient, 95 % confidence interval, and R 2. To find the most important factors predicting the outcome of osteoporosis (T score less than or equal to −2.5), both univariate and stepwise multinomial logistic regression analyses were performed (osteoporosis vs normal). The predictors were age, height, weight, BMI, residency, smoking, alcohol drinking, coffee drinking, and the levels of estradiol and testosterone. The results from the logistic regression are presented as the odds ratio and the 95 % confidence interval. We considered p < 0.05 as statistically significant.
Results
The study involved 222 men and 269 women, aged between 13 and 83 years (Table 1). There were no differences between all women and men in terms of age and BMI. As expected, men had greater height and weight than women. The prevalence of smoking, alcohol consumption, and coffee consumption was several times higher in men than in women.
In men, serum levels of testosterone averaged 6.2 ng/mL, and no apparent change by age was recorded (Fig. 1). Average estradiol levels were similar to those of postmenopausal women, and there was a positive association with increasing age (r s = 0.26; p < 0.001) (Fig. 1). The BMD values for the femoral neck, total hip, and lumbar spine in men were similar to those quantified in women at reproductive age but were higher than in postmenopausal women (Table 1). The prevalence of osteoporosis among men aged 50 years or older was 7–12 % for the different sites (Table 1). There was no significant difference in osteoporosis frequency between those living in urban areas and those living in rural areas (data not shown).
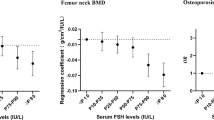
Among predictors of BMD in men, multiple regression analysis revealed age, BMI, and blood levels of estradiol to be the most important factors, and were estimated to explain 41 % of BMD at the femoral neck and 27 % for the total hip (Table 2). For BMD of the lumbar spine, age, BMI, and testosterone levels were the most important predictors. Similarly, age, weight, BMI, and the levels of estradiol and testosterone predicted osteoporosis at the different sites in men (Table 2).
As expected, sex hormone levels and BMD were lower in postmenopausal women than in women of reproductive age (Table 1). For the postmenopausal women, BMD values for the femoral neck and total hip were significantly higher in rural residents than in urban residents (p < 0.01) (Fig. 2). Furthermore, mean serum estradiol levels were higher (52 pg/mL vs 32 pg/mL, p < 0.05) in rural postmenopausal women than in urban postmenopausal women. A similar difference in BMD between rural and urban residence was not found in men. The prevalence of osteoporosis in postmenopausal women ranged between 17 and 37 % for the different sites measured.
When the hormonal status for women was controlled for by splitting women into reproductive and postmenopausal groups, different predictors of BMD were revealed (Table 3). For reproductive women, weight and residence were the most significant predictors of BMD at all three sites, and these two variables contributed to 12–14 % of the BMD variation. For the postmenopausal women, age, weight, and residence explained 40–46 % of the variation (Table 3). For the whole female population, multiple regressions revealed age, weight, residence, estradiol levels, and oral contraceptive use as the most important predictors of BMD and were estimated to explain around 50 % of the variation at the different sites (data not shown).
Discussion
This cross-sectional study was implemented to investigate the possible associations between BMD, osteoporosis, and the serum levels of testosterone and estradiol in Vietnamese men and women of different age and residence. We found that the prevalence of osteoporosis in postmenopausal women and in men aged 50 years or older was similar to that of Caucasian populations but was substantially higher than that in Chinese and Japanese populations [5, 20, 21]. In men, significant predictors of BMD for the femoral neck and total hip were age, BMI, and serum levels of estradiol, whereas determinants of osteoporosis at these sites were age, weight, and serum levels of estradiol and testosterone. For the BMD at the lumbar spine, age, BMI, and testosterone had a significant influence, whereas for osteoporosis BMI and estradiol were important factors. In postmenopausal women, age, weight, and residence (urban vs rural) were the most important predictors of BMD and osteoporosis. For all women (including those of reproductive age), blood levels of estradiol and testosterone were also significant determinants.
Our study supports the notion that estradiol levels have a stronger association and are more important as a predictor of BMD and osteoporosis than testosterone levels in both men and women. The critical role of estrogen in bone metabolism of both sexes is consistent with several previous reports [8, 22]. Whereas BMD and osteoporosis in men were previously considered to correlate mainly with testosterone [23], recent findings rather indicate a direct correlation between BMD and estradiol rather than between BMD and testosterone in healthy men regardless of their age [24]. In men, estrogens are produced via conversion from androgens by aromatase activity in peripheral tissues, including bone [25]. Apart from age and BMI, we found only estradiol levels to be significantly predictive of BMD of the femoral neck and total hip in men. This is in agreement with a case report of an adult man with estrogen resistance caused by a mutation in the estrogen receptor gene [26]. Although blood levels of testosterone were within the normal range, this man had increased bone resorption and osteopenia [26].
Our observation that testosterone remained a significant predictor of BMD at the lumbar spine but not at the femoral neck is in agreement with previous data. Men older than 50 years gained 8–14 % of BMD at the lumbar spine but not at the femoral neck after receiving intramuscular testosterone therapy for moderate hypogonadism [27]. Testosterone, like estrogen, appears to stimulate bone turnover, acting directly or indirectly via conversion into estradiol in human osteoblasts to increase androgen receptor expression and stimulate bone cell proliferation and mineralization [28, 29].
We previously reported a high prevalence of osteoporosis in Vietnamese women, and this finding was confirmed in the present study of another sample [5]. The proportions of postmenopausal women and men aged 50 years or older who were classified as having osteoporosis were similar to what has been reported for many Western Caucasian populations [5, 30]. We also found the prevalence of vitamin D deficiency to be higher among women living in urban areas [6]. Here in postmenopausal women, BMD of the femoral neck and BMD of the total hip were significantly lower among urban residents. Working and living indoors and also air pollution in cities and as a consequence reduced sun exposure of the skin could contribute to a lower average BMD in urban postmenopausal women. However, in the present study we also found urban postmenopausal women to have lower levels of circulating estradiol than rural postmenopausal women. The explanation for this difference is not clear, but it could possibly reflect differences in physical activity and dietary habits [31]. None of the postmenopausal women used hormone replacement therapy.
The present results should be interpreted within the context of strengths and potential limitations. To the best of our knowledge, this is the first study to investigate the correlation between sex steroid hormones and osteoporosis in Vietnamese men and women. The clinical subjects were randomly recruited from both rural and urban areas according to a rigorous selection scheme. Furthermore, the present study population had a wide age range, from 13 to 83 years, which allowed the comparison of the relation between sex hormones and BMD in different age groups. BMD was measured by dual-energy X-ray absorptiometry, which should ensure reliable results. Nevertheless, the study had potential limitations. Serum levels of sex-hormone-binding globulin were not quantified, and thus the concentrations of free sex steroid hormones which are bioactive in target tissues could not be evaluated [32]. In addition, as the study design was cross-sectional, conclusions on the long-term influence of sex hormones on BMD and osteoporosis could not be drawn. Ideally, this relationship should be determined in a representative cohort where a large number of men and women are followed.
In summary, the present data suggest that the prevalence of osteoporosis in the Vietnamese population is high not only in women but also in men, and that estrogen levels are essential for bone mass in both men and women. The results should have clinical implications and increase awareness of an important health issue within Vietnamese society.
References
Center JR, Nguyen TV, Schneider D, Sambrook PN, Eisman JA (1999) Mortality after all major types of osteoporotic fracture in men and women: an observational study. Lancet 353:878–882
Chang KP, Center JR, Nguyen TV, Eisman JA (2004) Incidence of hip and other osteoporotic fractures in elderly men and women: dubbo osteoporosis epidemiology study. J Bone Miner Res 19:532–536
Kanis JA, Oden A, Johnell O, Johansson H, De Laet C et al (2007) The use of clinical risk factors enhances the performance of BMD in the prediction of hip and osteoporotic fractures in men and women. Osteoporos Int 18:1033–1046
Nguyen TV, Eisman JA, Kelly PJ, Sambrook PN (1996) Risk factors for osteoporotic fractures in elderly men. Am J Epidemiol 144:255–263
Nguyen HTT, von Schoultz B, Pham DMT, Nguyen DB, Le QH et al (2009) Peak bone mineral density in Vietnamese women. Arch Osteoporos 2:9–15
Nguyen HT, von Schoultz B, Nguyen TV, Dzung DN, Duc PT et al (2012) Vitamin D deficiency in northern Vietnam: prevalence, risk factors and associations with bone mineral density. Bone 51:1029–1034
Lips P (2006) Vitamin D physiology. Prog Biophys Mol Biol 92:4–8
Clarke BL, Khosla S (2010) Physiology of bone loss. Radiol Clin North Am 48:483–495
Kuchuk NO, van Schoor NM, Pluijm SM, Smit JH, de Ronde W et al (2007) The association of sex hormone levels with quantitative ultrasound, bone mineral density, bone turnover and osteoporotic fractures in older men and women. Clin Endocrinol (Oxf) 67:295–303
Clarke BL, Khosla S (2009) Androgens and bone. Steroids 74:296–305
Huber DM, Bendixen AC, Pathrose P, Srivastava S, Dienger KM et al (2001) Androgens suppress osteoclast formation induced by RANKL and macrophage-colony stimulating factor. Endocrinology 142:3800–3808
Vanderschueren D, Vandenput L, Boonen S, Lindberg MK, Bouillon R et al (2004) Androgens and bone. Endocr Rev 25:389–425
Riggs BL, Khosla S, Melton LJ 3rd (2002) Sex steroids and the construction and conservation of the adult skeleton. Endocr Rev 23:279–302
Khosla S (2006) Editorial: sex hormone binding globulin: inhibitor or facilitator (or both) of sex steroid action? J Clin Endocrinol Metab 91:4764–4766
Khosla S, Melton LJ 3rd, Atkinson EJ, O’Fallon WM (2001) Relationship of serum sex steroid levels to longitudinal changes in bone density in young versus elderly men. J Clin Endocrinol Metab 86:3555–3561
Jakobsson J, Ekstrom L, Inotsume N, Garle M, Lorentzon M et al (2006) Large differences in testosterone excretion in Korean and Swedish men are strongly associated with a UDP-glucuronosyl transferase 2B17 polymorphism. J Clin Endocrinol Metab 91:687–693
Gorai I, Inada M, Morinaga H, Uchiyama Y, Yamauchi H et al (2007) CYP17 and COMT gene polymorphisms can influence bone directly, or indirectly through their effects on endogenous sex steroids, in postmenopausal Japanese women. Bone 40:28–36
Melton LJ 3rd, Chrischilles EA, Cooper C, Lane AW, Riggs BL (1992) Perspective. How many women have osteoporosis? J Bone Miner Res 7:1005–1010
Kanis JA, Gluer CC (2000) An update on the diagnosis and assessment of osteoporosis with densitometry. Committee of Scientific Advisors, International Osteoporosis Foundation. Osteoporos Int 11:192–202
Wu XP, Liao EY, Huang G, Dai RC, Zhang H (2003) A comparison study of the reference curves of bone mineral density at different skeletal sites in native Chinese, Japanese, and American Caucasian women. Calcif Tissue Int 73:122–132
Kaneki M, Hedges SJ, Hosoi T, Fujiwara S, Lyons A et al (2001) Japanese fermented soybean food as the major determinant of the large geographic difference in circulating levels of vitamin K2: possible implications for hip-fracture risk. Nutrition 17:315–321
Khosla S, Melton LJ 3rd, Atkinson EJ, O’Fallon WM, Klee GG et al (1998) Relationship of serum sex steroid levels and bone turnover markers with bone mineral density in men and women: a key role for bioavailable estrogen. J Clin Endocrinol Metab 83:2266–2274
Faustini-Fustini M, Rochira V, Carani C (1999) Oestrogen deficiency in men: where are we today? Eur J Endocrinol 140:111–129
Hoppe E, Morel G, Biver E, Borg S, Chopin F et al (2011) Male osteoporosis: do sex steroids really benefit bone health in men? Joint Bone Spine 78(Suppl 2):S191–S196
Ongphiphadhanakul B, Rajatanavin R, Chanprasertyothin S, Piaseu N, Chailurkit L (1998) Serum oestradiol and oestrogen-receptor gene polymorphism are associated with bone mineral density independently of serum testosterone in normal males. Clin Endocrinol (Oxf) 49:803–809
Smith EP, Boyd J, Frank GR, Takahashi H, Cohen RM et al (1994) Estrogen resistance caused by a mutation in the estrogen-receptor gene in a man. N Engl J Med 331:1056–1061
Tracz MJ, Sideras K, Bolona ER, Haddad RM, Kennedy CC et al (2006) Testosterone use in men and its effects on bone health. A systematic review and meta-analysis of randomized placebo-controlled trials. J Clin Endocrinol Metab 91:2011–2016
Takeuchi M, Kakushi H, Tohkin M (1994) Androgens directly stimulate mineralization and increase androgen receptors in human osteoblast-like osteosarcoma cells. Biochem Biophys Res Commun 204:905–911
Kasperk CH, Wergedal JE, Farley JR, Linkhart TA, Turner RT et al (1989) Androgens directly stimulate proliferation of bone cells in vitro. Endocrinology 124:1576–1578
Ho-Pham LT, Nguyen UD, Pham HN, Nguyen ND, Nguyen TV (2011) Reference ranges for bone mineral density and prevalence of osteoporosis in Vietnamese men and women. BMC Musculoskelet Disord 12:182
Devlin MJ (2011) Estrogen, exercise, and the skeleton. Evol Anthropol 20:54–61
Elmlinger MW, Kuhnel W, Wormstall H, Doller PC (2005) Reference intervals for testosterone, androstenedione and SHBG levels in healthy females and males from birth until old age. Clin Lab 51:625–632
Acknowledgments
We thank Sida for research funds, the Swedish Research Council (20324 to A.L.H.), Hanoi Medical University, Dinh Tien Hoang Institute of Medicine, Bach Mai Hospital, Karolinska Institutet, and the Garvan Institute. We are grateful to all members of the Friendship and Science for Health group for assistance in data collection. We especially thank Nguyen Thu Hoai for technical assistance in the analysis of estradiol and testosterone, and Dang Ngoc Dzung, Hoang Hoa Son, Le Tuan Thanh, Pham Hong Duong, and Vu Thanh Thuy for logistical arrangements in some parts of the study. We also thank Elisabeth Berg for statistical support.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Nguyen, H.T.T., von Schoultz, B., Nguyen, T.V. et al. Sex hormone levels as determinants of bone mineral density and osteoporosis in Vietnamese women and men. J Bone Miner Metab 33, 658–665 (2015). https://doi.org/10.1007/s00774-014-0629-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-014-0629-z