Abstract
Bile acid prodrugs have served as a viable strategy for refining the pharmaceutical profile of parent drugs through utilizing bile acid transporters. A series of three ester prodrugs of the antiherpetic drug acyclovir (ACV) with the bile acids cholic, chenodeoxycholic and deoxycholic were synthesized and evaluated along with valacyclovir for their in vitro antiviral activity against herpes simplex viruses type 1 and type 2 (HSV-1, HSV-2). The in vitro antiviral activity of the three bile acid prodrugs was also evaluated against Epstein–Barr virus (EBV). Plasma stability assays, utilizing ultra-high performance liquid chromatography coupled with tandem mass spectrometry, in vitro cytotoxicity and inhibitory experiments were conducted in order to establish the biological profile of ACV prodrugs. The antiviral assays demonstrated that ACV-cholate had slightly better antiviral activity than ACV against HSV-1, while it presented an eight-fold higher activity with respect to ACV against HSV-2. ACV-chenodeoxycholate presented a six-fold higher antiviral activity against HSV-2 with respect to ACV. Concerning EBV, the highest antiviral effect was demonstrated by ACV-chenodeoxycholate. Human plasma stability assays revealed that ACV-deoxycholate was more stable than the other two prodrugs. These results suggest that decorating the core structure of ACV with bile acids could deliver prodrugs with amplified antiviral activity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acyclovir (ACV) is a potent agent used in the treatment of infections caused by herpes viruses such as Herpes simplex 1 and 2, Varicella-zoster virus, Herpesvirus type 6 and 7, EBV etc. (Kimberlin and Whitley 2007). Despite its potency, the very low aqueous solubility (< 1.3 mg/mL) and the poor oral bioavailability have hampered the exploitation of its full therapeutic window (de Miranda and Blum 1983; de Miranda et al. 1981, 1982; Bruni et al. 2013; Bras et al. 2001). ACV’s poor solubility, low gastrointestinal absorption and poor membrane permeability have been linked with its severe side effects after administering it at higher doses in humans, such as nausea, vomiting, diarrhea, loss of appetite, stomach pain etc. (de Miranda and Blum 1983; Klysik et al. 2018; Johnston et al. 2012). Furthermore, it has been reported that the oral bioavailability is highly variable among species ranging from 75.3 ± 1.3% in dogs to 3.7 ± 0.5% in rhesus monkey (de Miranda and Blum 1983; de Miranda et al. 1981, 1982). In humans ACV oral bioavailability is limited to only 20% due to its reduced intestinal permeability, thus for an effective treatment, 200 mg doses are required five times daily (de Miranda and Blum 1983; Kimberlin and Whitley 2007).
Therefore, several strategies including formulation and “prodrug” approaches have been developed as viable alternatives to enhance the delivery of ACV (Beauchamp et al. 1992; Park et al. 1992; Colla et al. 1983; Bando et al. 1994; Shao et al. 1994; Karaman et al. 2012; Santos et al. 2009). The active nutrient transport systems have become a promising target for prodrug design due to recent advances in the identification and cloning of cDNAs for individual carriers (Yang et al. 2001). Along these lines, it has been recently reported that oral administration of amino acid ester prodrugs of ACV increases its urinary excretion in rats without prodrug detection, indicating that these esters are subjected to extensive in vivo hydrolysis (Burnette and de Miranda 1994; Beauchamp et al. 1992; Beauchamp and Krenitsky 1993; Katragadda et al. 2008).
A successful prodrug approach has been described for valacyclovir, succeeding to enhance the oral bioavailability of the parent ACV to 54% (Soul-Lawton et al. 1995; Han et al. 1998; Bomgaars et al. 2008; Antman and Gudmundsson 2007). Valacyclovir is an l-valine ester prodrug of ACV targeting hPepT1 peptide transporters present in the intestine (Han et al. 1998; Guo et al. 1999). Also the oral administration of l-valine-ACV produced the greatest increase in urinary excretion of ACV (~ 63%) among the examined amino acid ester prodrugs of ACV (Soul-Lawton et al. 1995; Han et al. 1998).
Bile acids belong to another type of scaffolds that have been utilized in prodrug formulations by enhancing the absorption of the molecules that are conjugated to, via specific transporting routes. Specifically, bile acids, are steroid carboxylic acids derived from the catabolism of cholesterol, which facilitates the digestion and absorption of lipids and lipid-soluble vitamins and they are transferred by both active and passive transport processes during enterohepatic circulation (St-Pierre et al. 2001; Hofmann 1988; Carey and Cahalane 1988; Turley and Dietschy 1988; Petzinger 1994; Stedronsky 1994; Enhsen et al. 1998; Staels and Fonseca 2009; Chiang 2009). These transport processes occur through Na+-dependent transporters that mediate the compound uptake from the portal circulation into hepatocytes as also the compound reabsorption from the intestine and biliary epithelium and an ATP-dependent transporter that pumps bile acids into bile. In addition to these classes of transporters, Na+-independent organic anion carriers with broad multi-substrate specificities have been also identified (St-Pierre et al. 2001). Bile acids intestinal absorption involves the Na+/K+-ATPase and the human apical sodium-dependent bile acid transporter (hASBT) (Lack 1979; Love and Dawson 1998; Balakrishnan and Polli 2006; Duane et al. 2007). Several reports have appeared in an effort to take advantage of the organotropism of bile acids for pharmaceutical applications targeting primarily the bile acid transporters (Enhsen et al. 1998; Kramer and Wess 1996; Kramer and Glombik 2006; Stojančević et al. 2013; Elnaggar 2015). hASBT has been utilized as a prodrug target for enhancing the oral bioavailability and intestinal permeation of compounds with low intrinsic intestinal permeability. Bile acid transporters are a promising route for refining oral drug absorption through conjugating a drug to a bile acid, forming a prodrug. This prodrug approach has been successfully exploited with various compounds (Kagedahl et al. 1997; Kramer et al. 1992, 1997; Kim et al. 1993; Swaan et al. 1997a; Rais et al. 2011; Zhang et al. 2016; Sievanen 2007).
Formerly a successful example has been established demonstrating the potency of the bile acid prodrug strategy in improving the oral bioavailability of the intestinal permeability-limited compounds by the group of J. Polli (Tolle-Sander et al. 2004). In this work, ACV was conjugated to a bile acid via a valine linker and it was revealed that this ACV prodrug, upon utilizing the human apical sodium-dependent bile acid transporter, enhanced the oral bioavailability of ACV in rats by two-fold (Tolle-Sander et al. 2004).
Additionally, herpes simplex virus (HSV) strains are responsible for several health complications, with hepatitis being one of the most severe (Berger and Houff 2008; Pyles 2001; Arkin et al. 2009). Herpes infection of the liver can lead to liver failure (ALF), liver transplantation (LT) and even death in untreated cases (Norvell et al. 2007; Chaudhary et al. 2017). ACV has not demonstrated any promising results in the treatment of these diseases, due to its low cellular permeability and poor penetration in the liver cells (Longerich et al. 2005; Chaudhary et al. 2017). Furthermore, ACV’s side effects and nephrotoxicity, render the targeted delivery of acyclovir to the liver as a matter of great importance (Gupta et al. 2013). It has been shown that conjugates with bile acids have the ability to target the liver in a way that mimics the hepatic uptake of native bile acids (Vivian and Polli 2014). Moreover, the use of bile acid prodrugs could potentially substitute the established treatment with valacyclovir since it has been shown that hPEPT1 is primarily expressed in the small intestine and to a lesser degree in the liver cells (Bockman et al. 1997; Thamotharan et al. 1997; Liang et al. 1995; Wang et al. 2017).
Along these lines, the objective of the present study is to synthesize bile acid prodrugs of ACV and evaluate their potency in vitro against HSV-1, HSV-2 and EBV. We aimed to enhance the pharmaceutical profile of ACV following the bile acid prodrug strategy taking advantage of the established oral bioavailability of such prodrugs through using the human apical sodium-dependent bile acid transporter (Tolle-Sander et al. 2004). For this purpose, three bile acids (cholic, deoxycholic, chenodeoxycolic) were utilized for the synthesis of ACV prodrugs. The intrinsic amphiphilic character of bile acids, its rigid steroidal backbone along with the hydroxyl groups on the steroid ring which can be functionalized in various ways, enantiomeric purity and low cost make them ideal building blocks for conjugation to ACV (Davis 1993; Li and Dias 1997; Wallimann et al. 1997; Tamminen and Kolehmainen 2001; Virtanen and Kolehmainen 2004; Mukhopadhyay and Maitra 2004).
The three synthesized bile acid ACV prodrugs were evaluated for their biological efficiency against HSV-1, HSV-2 and EBV as also for their cytotoxic effect on normal cells. Also evaluation of the stability of the synthesized conjugates in human plasma was performed through establishing UHPLC–MS/MS assays.
Materials and methods
Chemicals and reagents
ACV (≥ 99% purity), valacyclovir hydrochloride (≥ 98% purity) and bile acids (cholic, deoxycholic, chenodeoxycholic) were purchased from Sigma-Aldrich. 4-Dimethylaminopyridine (DMAP) and N,N′-dicyclohexylcarbodiimide (DCC) were purchased from Merck. Formic acid (98%, LC–MS grade) and dimethylformamide (DMF) were obtained from Fluka. Acetonitrile, methanol, deionized water, LC–MS grade all, were purchased from Fisher Scientific. DMSO (LC–MS grade) was purchased from Thermo Scientific. Membrane filters with 0.2 μm pore size and 47 mm diameter were purchased from Phenomenex. High purity argon was used as collision gas. TLC analysis was performed on aluminum silica gel 60 F254 plates (Merck) and detection was performed using an UV lamp at 254 nm. Drug-free human plasma from healthy donors for the stability studies, was a kind offer from Blood Donation Center of the University Hospital of Ioannina.
General synthetic procedure for preparation of ACV prodrugs
A mixture of bile acids (0.480 g, 2 mmol) and DCC (0.191 g, 2 mmol) in DMF was stirred for 1 h at 0 °C under nitrogen atmosphere (Fig. 1). A solution of ACV (0.225 g, 1 mmol) and DMAP (0.244 g, 2 mmol) in DMF was added to the reaction mixture and stirred for 24 h. Then DMF was evaporated in vacuo, and the residue was purified on silica gel chromatography, using gradient of MeOH/CH2Cl2 (1:4). For the identification of the three analogues NMR and mass spectrometry were applied. The 1H, 13C NMR spectra were obtained on a Bruker Avance DRX-500 spectrometer, operating at 500.13 MHz. The ESI mass spectra were obtained on Bruker EVOQ Elite ER TQ. The IR spectra were obtained on a Thermo Scientific Nicolet iS10 FT-IR.
LC–MS/MS assays
Liquid chromatography conditions
Reversed phase liquid chromatography assay was performed using a Bruker Advance Ultra High Performance Liquid Chromatography (UHPLC) system (Bruker, Germany). Τhe separation of ACV esters and valacyclovir was performed on a Kinetex C18 column 100 mm × 2.1 mm, 2.6 um, with proguard column 2.1 mm (Phenomenex). Column temperature was maintained stable at 40 °C. The mobile phases were composed of deionized water with formic acid 0.1% (A) and acetonitrile with formic acid 0.1% (B). For gradient elution of the three bile acid esters the following profile, at constant flow rate of 200 uL/min, was used: initial phase (B) concentration 20%, increased to 100% within 2.0 min, then kept constant for 2 min and reduced to 20% till the end of the run at 5 min. For the separation of valacyclovir, the following gradient at constant flow rate of 200 uL/min was used: initial phase (B) concentration 5%, increased to 70% within 2.0 min, then kept constant for 2 min and reduced to 5% till the end of the run at 5 min. The injection volume was set at 5 uL while the temperature in auto-sampler was maintained at 25 °C.
Mass spectrometric conditions
For the ionization and detection of the four prodrugs, EVOQ Elite ER (Bruker) triple quadrupole mass spectrometer was operated in positive ionization electrospray mode (ESI+) in multiple reaction monitoring (MRM). Using MRM builder, a feature of MS workstation software of Bruker, the optimal MRM transitions selected for monitoring the four analogues were m/z 616 → 562.3 and 616 → 152.1 for ACV-cholate, 600 → 564.2 and 600 → 582.2 for ACV-deoxycholate, 600 → 582.3 and 600 → 564.3 for ACV-chenodeoxycholate and 325.1 → 152.2 and 325.1 → 146.3 for valacyclovir. Optimum ESI conditions were determined as follows: spray voltage 4500 V; heated probe gas flow, 50 units; heated probe temperature 350 °C; cone gas flow, 20 units; cone temperature, 350 °C; nebulizer gas flow, 50 units. Total control of LC and MS as also data acquisition were performed with MSWS software, version 8.2.1, provided by Bruker.
Human plasma stability of ACV esters and valacyclovir
The stock solutions of 1 mg/mL for each compound were prepared by dissolving the appropriate amounts in DMSO. The final working solutions of 60 uM where prepared by further dilution of the stock solutions with methanol. Plasma samples were prepared by incubating 10 uL of each analogue separately, with 90 uL human plasma (pH adjusted to 7.4) for 0, 1, 2, 3, 5, 7, 14, 18 h at 37 °C. In order to quench the reactions, 500 uL of cold-ice acetonitrile were added to each sample, giving a final concentration of 1 uM for each analyte. The samples were then vortex-mixed and centrifuged at 10,000g for 5 min. The supernatant was taken, filtered and transferred to vials for UHPLC-MS/MS analysis. Each sample was studied at triplicates and the plot of the percentage of the parent compound remaining in every time point in comparison to 0 time against time was designed for the four compounds.
Assays for the antiviral activity of the synthesized esters of ACV and valacyclovir against HSV-1 and HSV-2
Viruses and cells
The two laboratory strains, Da (HSV-1) and Bja (HSV-2), were kindly provided by Prof. S. Dundarov (National Center of Infectious and Parasitic Diseases, NCIPD, Bulgaria). Madin–Darby bovine kidney (MDBK) cells were cultured at 37 °C as monolayers in RPMI-1640 medium (Flow Laboratories, USA), supplemented with antibiotics (penicillin: 100 IU/mL, streptomycin: 100 ug/mL) and 10% bovine serum (NCIPD, Bulgaria). Serum concentration was reduced to 5% in order to examine the growth of the viruses and to identify the cytotoxicity of the synthesized esters and valacyclovir.
Cytotoxicity assay—determination of the maximum tolerable concentration (MTC)
In order to determine and compare the maximum tolerable (nontoxic) concentration (MTC) values of compounds to that of ACV, uninfected confluent cell monolayers were covered with media that contained different concentrations of the tested compounds (1.6–162.3 uM for ACV-cholate and 1.7–166.7 uM for ACV-deoxycholate and ACV-chenodeoxycholate) or ACV (4.4–88.8 uM) and cultured at 37 °C for 96 h. Samples of cells that grew in prodrug-free medium served as control. The maximum nontoxic concentration, which did not alter neither the morphology nor the viability of the cells, was established as MTC.
Antiviral assay
Experiments were performed under multicycle growth conditions. Confluent cell monolayers were washed and infected with 320 cell culture infectious doses (CCID50) per 0.1 mL of the appropriate virus strain (Da and Bja). After 1 h, cells were covered with maintenance media that included the four prodrugs (synthesized esters and valacyclovir) of ACV in the tested concentrations (1.6–162.3 uM for ACV-cholate, 1.7–166.7 uM for ACV-deoxycholate and ACV-chenodeoxycholate and 3.1–61.7 uM for valacyclovir). The effect on viral replication was determined after 48 h (for strains Da and Bja) culturing at 37 °C by reduction of infectious virus titres as compared to that of untreated viral control. The 50% inhibitory concentration (IC50) for virus-induced cytopathic effect (CPE) for each compound, including ACV, was determined by a dose–response curve charted out by the obtained data. To calculate the standard deviation of IC50 for every compound, each experiment was conducted in triplicate for HSV-1 (strain Da) and duplicate for HSV-2 (strain Bja).
Assay for the antiviral activity of the synthesized esters of ACV against EBV
Cells and compounds
For the evaluation of the three synthesized prodrugs of ACV against EBV, B95-8 cells (cotton-top tamarin monkey B-lymphoblastoid cell line, ATCC) were exposed to EBV infection. The resulting continuous cell line harbors the EBV genome in a latent state. However, infectious EBV is released in the culture supernatant, due to the fact that 5–8% of the cells spontaneously enter the lytic cycle of virus replication (Miller and Lipman 1973). Cells were maintained in RPMI 1640 medium (Sigma) supplemented with 10% fetal bovine serum (Sigma) and antibiotics (penicillin: 100 IU/mL, streptomycin: 100 ug/mL). All tested compounds were dissolved in DMF to a 3.3 mM concentration for ACV-deoxycholate and ACV-chenodeoxycholate and 3.2 mM for ACV-cholate while ACV was dissolved in DMSO to a 22.2 mM stock concentration. Stock solutions were further diluted in RPMI medium to reach the final concentrations required: 166.7, 66.7, 33.3, 16.7, 8.3, 1.7 uM for ACV-deoxycholate and ACV-chenodeoxycholate, 162.3, 64.9, 32.5, 16.2, 8.1, 1.6 uM for ACV-cholate and 44.4, 22.2, 8.9, 4.4 uM for ACV.
Cytotoxicity assay
Cells were seeded in 96-well plates (Nunc) at 2 × 103 cells per well. At 24 h post seeding, the conjugates were added to the medium to a final concentration ranging from 1.7 to 166.7 uM for ACV-deoxycholate and ACV-chenodeoxycholate, 1.6 to 162.3 uM for ACV-cholate and 4.4 to 44.4 uM for ACV. Mock-treated cells were used as controls. Cells cultured in the presence of DMF or DMSO were used to control solvent toxicity. The compound-related cellular toxicity was determined by using the CellTiter 96® AQueous One Solution Cell Proliferation Assay (Promega). The maximum (nontoxic) concentration that did not induce neither overt alteration on the cellular morphology nor cellular death, was established as MTC.
Antiviral activity assay
Cells were seeded in 10-cm dishes (Nunc) at 105 cells per dish. 24 h after seeding, cells were treated by adding the respective concentrations of conjugates (1.7–166.7 uM for ACV-deoxycholate and ACV-chenodeoxycholate and 1.6–162.3 uM for ACV-cholate) and ACV (4.4–44.4 uM) to the culture medium. Mock-treated cells were used as controls. At 72 h after the treatment, cells were harvested and stained against the EBV envelope glycoprotein gp350/220. For this purpose, the following antibodies were used: mouse monoclonal anti-gp350/220, 1:250 in PBS (ATCC, 7A21 hybridoma), and goat anti-mouse Cy3-coupled IgG, 1:300 in PBS (Dianova). Fluorescence was observed using a Leica fluorescent microscope (Leica, Germany) and the percentage of gp350/220-positive cells was determined for each experimental condition. The minimum inhibitory concentration (MIC) as well as the MTC/MIC ratio was determined for each compound.
Results and discussion
Design and synthesis of ACV prodrugs
Formerly it was established that bile acid prodrugs of acyclovir conjugated by a valine linker presented a two-fold enhancement on acyclovir oral bioavailability by utilizing the human apical sodium-dependent bile acid transporter (hASBT) (Tolle-Sander et al. 2004). A hydrogen bond acceptor at C-24 as also a steroid nucleus have been identified as required structural determinants for hASBT recognition (Swaan et al. 1997b). To conform to these structural prerequisites and also to enable the formation of metabolically labile ester functioning as a hydrogen bond acceptor to retain recognition for hASBT we conjugated different bile acids, via their C24 carboxylic acid, to acyclovir alcohol through ester bonds. The structures of the new ACV prodrugs with the bile acids (cholic, deoxycholic, chenodeoxycholic) were confirmed by 1H NMR, 13C NMR, IR and ESI–MS spectrometry (Fig. 2). The three synthesized ACV prodrugs were identified through the NMR spectra which consisted of peaks derived both from the bile acid scaffold and the ACV part as listed below.
ACV-cholate (500 MHz NMR, DMSO-d6, 25 °C): δ 0.69 (3H, s, H-18), 0.86 (3H, s, H-19), 1.01 (3H, s, H-21), 2.53 (4H, d, J = 3.5 Hz, CH2), 3.23 (1H, m, 3α-OH), 3.42 (1H, s, 7α-OH), 4.01 (1H, s, 12α-OH); 4.21 (m, 2H, CH2OC(O), ACV), 5.01 (s, 2H, N–CH2–O, ACV), 6.43 (s, 2H, 2-NH2, ACV), 7.13 (s, 1H, H-8, ACV), 10.78 (s, 1H, ACV-NH); 13C-NMR (500 MHz, DMSO-d6, 25 °C) (35.25 –CH2), (31.0 –CH2), (71.4–CH), (39.9 –CH2), (41.9 –C), (35.7 –CH2), (70.8 –CH), (41.7 –CH), (26.6 –CH), (34.8 –C), (27.6 –CH2), (72 –CH), (46.2 –C), (39.85 –CH), (23.1 –CH2), (28.9 –CH2), (46.4 –CH), (12.7 –CH3), (22.9 –CH3), (35.25 –CH), (17.3 –CH3), (30.9 –CH2), (30.8 –CH2), (62.9 –C Acycl), (67.0 –C Acycl), (116.9 –C Acycl), (137.9 –C Acycl), (151.8 –C Acycl), (154.3 –C Acycl), (157.1 –C Acycl), (173.6 –C); IR, functional groups: OH (3334 cm−1), C–H (2931 cm−1), C–H (2866 cm−1), ester group (1688 cm−1), NH2 (1628 cm−1), C–H (1487 cm−1), C–H (1447 cm−1), Ether C–O (1308 cm−1), N–H (1221 cm−1); ESI MS: [M+H]+ was found 616, calculated 615.78; yield = 0.117 g (19%, white powder).
ACV-deoxycholate (500 MHz NMR, DMSO-d6, 25 °C): δ 0.61 (3H, s, H-18), 0.87 (3H, s, H-19), 1.01 (3H, s, H-21), 2.83–2.85 (4H, d, CH2), 3.57 (1H, m, 3α-OH), 4.02 (1H, s, 12α-OH), 4.02 [m, 2H, CH2OC(O), ACV], 5.24 (s, 2H, N–CH2–O, ACV), 6.34 (s, 2H, 2-NH2, ACV), 7.19 (s, 1H, H-8, ACV), 10.91 (s, 1H, ACV-NH); 13C-NMR (500 MHz, DMSO-d6, 25 °C) (36.5 –CH2), (31.2 –CH2), (72.6 –CH), (37.1 –CH2), (43.4 –C), (35.7 –CH2), (69.1 –CH), (40.1 –CH), (34.0 –CH), (36.1 –C), (21.8 –CH2), (41.0 –CH), (43.6 –C), (51.3 –CH), (24.5 –CH2), (29.1 –CH2), (57.1 –CH), (12.1 –CH3), (24.6 –CH3), (36.6 –CH), (18.7 –CH3), (32.4 –CH2), (32.3 –CH2), (62.5 –C Acycl), (66.5 –C Acycl), (118.0 –C Acycl), (138.0 –C Acycl), (151.2 –C Acycl), (153.9 –C Acycl), (157.1 –C Acycl), (173.0 –C); IR, functional groups: OH (3334 cm−1), C–H (2920 cm−1), C–H (2861 cm−1), ester group (1694 cm−1), NH2 (1628 cm−1), C–H (1487 cm−1), C–H (1447 cm−1), ether C–O (1307 cm−1), N–H (1219 cm−1); ESI MS: [M+H]+ was found 600, calculated 599.78; yield = 0.138 g (23%, white powder).
ACV-chenodeoxycholate (500 MHz NMR, DMSO-d6, 25 °C): δ 0.63 (3H, s, H-18), 0.90 (3H, s, H-19), 0.91 (3H, s, H-21), 2.83–2.85 (4H, d, CH2), 3.21 (1H, m, 3α-OH), 3.65 (1H, s, 7α-OH), 3.82 (t, 2H, CH2O, ACV), 4.28 [m, 2H, CH2OC(O), ACV], 5.16 (s, 2H, N–CH2–O, ACV), 6.73 (s, 2H, 2–123 NH2, ACV), 7.97 (s, 1H, H-8, ACV), 10.69 (s, 1H, ACV-NH); 13C-NMR (500 MHz, DMSO-d6, 25 °C) (36.3 –CH2), (30.8 –CH2), (72.4 –CH), (36.9 –CH2), (43.6 –C), (28.7 –CH2), (27.8 –CH), (37.1 –CH), (34.6 –CH), (35.1 –C), (29.8 –CH2), (74.0 –CH), (44.2 –C), (49.0 –CH), (24.7 –CH2), (28.1 –CH2), (48.1 –CH), (13.1 –CH3), (23.6 –CH3), (36.25 –CH), (17.5 –CH3), (32.9 –CH2), (32.0 –CH2), (61.9 –C Acycl), (67.0 –C Acycl), (117.7 –C Acycl), (139.1 –C Acycl), (150.8 –C Acycl), (154.4 –C Acycl), (156.9 –C Acycl), (173.2 –C); IR, functional groups: OH (3327 cm−1), C–H (2927 cm−1), C–H (2865 cm−1), ester group (1689 cm−1), NH2 (1628 cm−1), C–H (1487 cm−1), C–H (1447 cm−1), ether C–O (1308 cm−1), N–H (1221 cm−1); ESI MS: [M+H]+ was found 600, calculated 599.78; yield = 0.162 g (27%, white powder).
The severe overlapping of the signals from the bile acids leads to complicated patterns in the COSY spectrum. Much more useful was the one-bond hetero-correlation HSQC experiment, allowing easy assignment of proton chemical shifts, along with proton–carbon connectivity. Thus, for example, the signals at 72.2, 67.0 and 62.9 were assigned to the methylene fragments from the acyclovir and the three CH signal between 66 and 71 ppm were assigned to the OH-bearing fragments from the cholic acid. Although the full assignment of the compounds is far from trivial, the edited HSQC experiments is sufficient to confirm the structures of the compounds, when combined with DEPT135 experiment as complementary technique for 13C multiplicity determination.
Antiviral activity against HSV-1 and HSV-2
Cytotoxicity in vitro in MDBK cells
The synthesized prodrugs and valacyclovir were applied in concentration ranging from 1.6 to 32.5 uM for ACV-cholate, 1.7 to 33.3 uM for ACV-deoxycholate and ACV-chenodeoxycholate and 3.1 to 61.7 uM for valacyclovir. No morphological changes in the cell monolayer were observed within these concentration ranges. Due to lack of toxicity for the tested compounds at the defined concentration limits, MTC was not determined. This lack of toxicity could be attributed to the natural components (bile acids, valine) of the conjugates.
Inhibitory effect of the prodrugs on the replications of HSV-1 and HSV-2
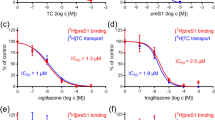
The ACV prodrugs were added in concentrations 1.6, 4, 8.1, 16.2, 32.5 uM for ACV-cholate, 1.7, 4.1, 8.3, 16.6, 33.3 uM for ACV-deoxycholate and ACV-chenodeoxycholate and 3.1, 7.7, 15.4, 30.8, 61.7 uM for valacyclovir in the MDBK cell cultures and exhibited similar or even enhanced inhibitory activity with respect to ACV against HSV-1 and HSV-2 strains. The experimental data suggested dose-depend effects. Valacyclovir presented enhanced antiviral properties with respect to the parent compound. The synthesized prodrugs significantly inhibited viral replication in dose-dependent manner without apparent cytotoxicity, presenting markedly similar effect with this of ACV. The results from treatment of the MDBK cells, infected with HSV-1 and HSV-2 strains, with the prodrugs are illustrated in Fig. 3. ACV-deoxycholate did not suppress the replication of HSV-1 and HSV-2. Against both viruses, most potent inhibition between the three synthesized prodrugs was monitored for the ACV-cholate with IC50 4.7 uM towards HSV-1 and 6.2 uM towards HSV-2. The results also pinpointed that ACV-cholate was approximately eight-fold more potent than ACV in the case of HSV-2 and slightly more effective in the case of HSV-1. Along the same lines, ACV-chenodeoxycholate exhibited approximately six-fold higher potency against HSV-2 than ACV presenting an IC50 8 uM with respect to 46.6 uM of ACV. The viral replication of both HSV-1 and HSV-2 after addition of 33.3 uM ACV-chenodeoxycholate and 32.5 uM ACV-cholate was completely suppressed.
Antiviral activity against EBV
Maximum tolerable concentration
ACV-deoxycholate and ACV-chenodeoxycholate were not toxic for B95-8 cells in the whole concentration range tested (1.7–166.7 uM). ACV-cholate demonstrated strong toxicity when it was applied at 64.9 and 162.3 uM. On the other hand, ACV suppressed B95-8 cell proliferation at the concentration of 44.4 uM. The MTC values were therefore determined as follows in Table 1.
Suppression of EBV lytic replication in B95-8 cells
The ability of the synthesized esters of ACV to suppress the spontaneous infectious EBV release from the infected B95-8 cells was determined through fluorescence microscopy. The percentage of envelope glycoprotein gp350/220-positive cells was compared between treated and non-treated cultures. As shown in Table 2, the three tested compounds inhibited the lytic EBV replication when applied in the concentration range of 33.3–166.7 uM for ACV-chenodeoxycholate, 66.7–166.7 uM for ACV-deoxycholate and 32.5 uM for ACV-cholate. On the other hand, ACV was active for a concentration rage of 8.9–44.4 uM. The calculated MIC values indicated that ACV-cholate and ACV-chenodeoxycholate had the best MIC values among the analogues.
The MTC/MIC ratio was also determined, with high values indicating low toxicity and strong antiviral activity. Taking into consideration the above results and under the experimental conditions described, ACV-chenodeoxycholate was found to be the most promising anti-EBV candidate since it demonstrated the best toxicity versus activity index among the prodrugs evaluated and is as effective in suppressing lytic EBV replication as ACV (Fig. 4).
Evaluation of human plasma stability of ACV prodrugs
Having determined the efficacy of the different prodrugs we then evaluated their human plasma stability. For this, we established novel UHPLC-MS/MS protocols to monitor the stability of the ACV prodrugs, as also to quantify their concentration in human plasma in a time dependent manner.
Several chromatography tests were conducted to maximize resolution for sharper peak shapes and shorter run times. These tests included various ratios of mobile phases, addition of salts (ammonium formate, ammonium acetate) in the aqueous phase and/or acids (formic, acetic). The presence of 0.1% formic acid in phase A and B improved peak shapes and increased the signal for all analogues. The total run time was 5 min and the elution time for ACV-cholate was 2.5 min for ACV-deoxycholate and ACV-chenodeoxycholate 2.8 min while for valacyclovir 2.3 min.
Tandem mass spectrometry was used for the detection and quantification of the targeted conjugates in positive electrospray ionization since the precursor > product transition for all analogues was optimum in this mode. Utilizing MRM builder, a feature of MSWS software of mass spectrometer, during direct infusion in mass spectrometer, the most abundant transitions in terms of sensitivity were found to be: m/z 616 → 562.3 and 616 → 152.1 for ACV-cholate, 600 → 564.2 and 600 → 582.2 for ACV-deoxycholate, 600 → 582.3 and 600 → 564.3 for ACV-chenodeoxycholate and 325.1 → 152.2 and 325.1 → 146.3 for valacyclovir. Along with chromatography tests, several trials with different ESI parameters like nebulizer, spray voltage and heated probe temperature were optimized in order to obtain a consistent and reliable response for all analytes.
The degradation rate of the four analogues, after incubation in human plasma for 0, 1, 2, 3, 5, 7, 14 and 18 h is presented in Fig. 5. It is notable that after 18 h of plasma incubation, all analogues were completely eliminated from plasma esterases activity. Valacyclovir presented the highest stability profile among the four prodrugs. ACV-chenodeoxycholate demonstrated the fastest degradation rate while ACV-deoxycholate presented the slowest rate in comparison to the three synthesized prodrugs of ACV. Despite the limited monitored stability of ACV-cholate, this prodrug along with ACV-chenodeoxycholate, demonstrated similar antiviral activity with ACV against HSV-1 and HSV-2. Same applies for ACV-chenodeoxycholate since this analogue exhibited the most promising profile as antivirus agent against EBV. This could be attributed to the fact that all analogues are prodrugs and their beneficial effects are based on the optimum release of the drug, in this case of ACV. On the contrary, ACV-deoxycholate demonstrated the most stable stability profile, nonetheless, the slow release of ACV, potentially rationalize its diminished biological activity against HSV-1, HSV-2 and EBV.
Conclusions
Three ACV prodrugs with bile acids (cholic, deoxycholic, chenodeoxycolic) were synthesized and their antiviral activity against three human herpesviruses was evaluated. Even if, it was revealed that the ACV-cholate prodrug adopted similar antiviral activity with ACV against HSV-1, it demonstrated an approximately eight-fold higher activity against HSV-2. Furthermore, an approximately six-fold higher antiviral activity for ACV-chenodeoxycholate against HSV-2 with respect to ACV was observed. The bile acids prodrugs were also evaluated against EBV, and ACV-chenodeoxycholate presented the highest antiviral effect. To explore the stability of the different prodrugs in human plasma UHPLC-MS/MS assays were established and revealed ACV-deoxycholate to be more stable than the other two bile acid prodrugs, potentially corroborating to the monitored diminished antiviral activity against HSV-1, HSV-2 and EBV. Thus, although the bile acid prodrug strategy has been established as a tool to improve the oral bioavailability of intestinal permeability-limited compounds, we hereby suggest that it can also amplify the antiviral activity of the parent antiviral compounds.
Abbreviations
- ACV:
-
Acyclovir
- HSV-1:
-
Herpes simplex virus type 1
- HSV-2:
-
Herpes simplex virus type 2
- EBV:
-
Epstein–Barr virus
- LC–MS/MS:
-
Liquid chromatography–tandem mass spectrometry
- RT:
-
Retention time
- ΜRΜ:
-
Multiple reaction monitoring
- MTC:
-
Maximum tolerable concentration
- MIC:
-
Minimum inhibitory concentration
References
Antman MD, Gudmundsson OS (2007) Case study: valacyclovir: a prodrug of acyclovir. In: Stella VJ, Borchardt RT, Hageman MJ, Oliyai R, Maag H, Tilley JW (eds) Prodrugs: challenges and rewards part 1. Springer, New York, pp 1369–1376. https://doi.org/10.1007/978-0-387-49785-3_54
Arkin LM, Castelo-Soccio L, Kovarik C (2009) Disseminated herpes simplex virus (HSV) hepatitis diagnosed by dermatology evaluation. Int J Dermatol 48(9):1020–1021. https://doi.org/10.1111/j.1365-4632.2009.04084.x
Balakrishnan A, Polli JE (2006) Apical sodium dependent bile acid transporter (ASBT, SLC10A2): a potential prodrug target. Mol Pharm 3(3):223–230. https://doi.org/10.1021/mp060022d
Bando H, Yamashita F, Takakura Y, Hashida M (1994) Skin penetration enhancement of acyclovir by prodrug-enhancer combination. Biol Pharm Bull 17(8):1141–1143
Beauchamp LM, Krenitsky TA (1993) Acyclovir prodrugs: the road to valaciclovir. Drugs Future 18(7):619–628
Beauchamp LM, Orr GF, de Miranda P, Bumette T, Krenitsky TA (1992) Amino acid ester prodrugs of acyclovir. Antiviral Chem Chemother 3(3):157–164. https://doi.org/10.1177/095632029200300305
Berger JR, Houff S (2008) Neurological complications of herpes simplex virus type 2 infection. Arch Neurol 65(5):596–600. https://doi.org/10.1001/archneur.65.5.596
Bockman DE, Ganapathy V, Oblak TG, Leibach FH (1997) Localization of peptide transporter in nuclei and lysosomes of the pancreas. Int J Pancreatol 22(3):221–225. https://doi.org/10.1007/BF02788388
Bomgaars L, Thompson P, Berg S, Serabe B, Aleksic A, Blaney S (2008) Valacyclovir and acyclovir pharmacokinetics in immunocompromised children. Pediatr Blood Cancer 51(4):504–508. https://doi.org/10.1002/pbc.21638
Bras AP, Sitar DS, Aoki FY (2001) Comparative bioavailability of acyclovir from oral valacyclovir and acyclovir in patients treated for recurrent genital herpes simplex virus infection. Can J Clin Pharmacol 8(4):207–211
Bruni G, Maietta M, Maggi L, Mustarelli P, Ferrara C, Berbenni V, Milanese C, Girella A, Marini A (2013) Preparation and physicochemical characterization of acyclovir cocrystals with improved dissolution properties. J Pharm Sci 102(11):4079–4086. https://doi.org/10.1002/jps.23721
Burnette TC, de Miranda P (1994) Metabolic disposition of the acyclovir prodrug valaciclovir in the rat. Drug Metab Dispos 22(1):60–64
Carey MC, Cahalane MJ (1988) In: Arias IM, Jakoby WB, Popper H, Schachter D, Shafritz DS (eds) In the liver: biology and pathobiology. Raven Press, New York, pp 573–616
Chaudhary D, Ahmed S, Liu N, Marsano-Obando L (2017) Acute liver failure from herpes simplex virus in an immunocompetent patient due to direct inoculation of the peritoneum. ACG Case Rep J 4:e23. https://doi.org/10.14309/crj.2017.23
Chiang JY (2009) Bile acids: regulation of synthesis. J Lipid Res 50(10):1955–1966. https://doi.org/10.1194/jlr.R900010-JLR200
Colla L, De Clercq E, Busson R, Vanderhaeghe H (1983) Synthesis and antiviral activity of water-soluble esters of acyclovir [9-[(2-hydroxyethoxy)methyl]guanine). J Med Chem 26(4):602–604. https://doi.org/10.1021/jm00358a029
Davis AP (1993) Cholaphanes et al.; steroids as structural components in molecular engineering. Chem Soc Rev 22(4):243–253. https://doi.org/10.1039/cs9932200243
de Miranda P, Blum MR (1983) Pharmacokinetics of acyclovir after intravenous and oral administration. J Antimicrob Chemother 12 Suppl B:29–37
de Miranda P, Krasny HC, Page DA, Elion GB (1981) The disposition of acyclovir in different species. J Pharmacol Exp Ther 219(2):309–315
de Miranda P, Krasny HC, Page DA, Elion GB (1982) Species differences in the disposition of acyclovir. Am J Med 73(1A):31–35
Duane WC, Xiong W, Wolvers J (2007) Effects of bile acids on expression of the human apical sodium dependent bile acid transporter gene. Biochim Biophys Acta 1771(11):1380–1388. https://doi.org/10.1016/j.bbalip.2007.09.003
Elnaggar YS (2015) Multifaceted applications of bile salts in pharmacy: an emphasis on nanomedicine. Int J Nanomed 10:3955–3971. https://doi.org/10.2147/IJN.S82558
Enhsen A, Kramer W, Wess G (1998) Bile acids in drug discovery. Drug Discov Today 3(9):409–418
Guo A, Hu P, Balimane PV, Leibach FH, Sinko PJ (1999) Interactions of a nonpeptidic drug, valacyclovir, with the human intestinal peptide transporter (hPEPT1) expressed in a mammalian cell line. J Pharmacol Exp Ther 289(1):448–454
Gupta S, Agarwal A, Gupta NK, Saraogi G, Agrawal H, Agrawal GP (2013) Galactose decorated PLGA nanoparticles for hepatic delivery of acyclovir. Drug Dev Ind Pharm 39(12):1866–1873. https://doi.org/10.3109/03639045.2012.662510
Han H, de Vrueh RL, Rhie JK, Covitz KM, Smith PL, Lee CP, Oh DM, Sadee W, Amidon GL (1998) 5′-Amino acid esters of antiviral nucleosides, acyclovir, and AZT are absorbed by the intestinal PEPT1 peptide transporter. Pharm Res 15(8):1154–1159
Hofmann A (1988) In: Arias IM, Jakoby WB, Popper H, Schachter D, Shafritz DA (eds) The liver: biology and pathobiology. Raven Press, New York, pp 553–572
Johnston C, Saracino M, Kuntz S, Magaret A, Selke S, Huang ML, Schiffer JT, Koelle DM, Corey L, Wald A (2012) Standard-dose and high-dose daily antiviral therapy for short episodes of genital HSV-2 reactivation: three randomised, open-label, cross-over trials. Lancet 379(9816):641–647. https://doi.org/10.1016/S0140-6736(11)61750-9
Kagedahl M, Swaan PW, Redemann CT, Tang M, Craik CS, Szoka FC Jr, Oie S (1997) Use of the intestinal bile acid transporter for the uptake of cholic acid conjugates with HIV-1 protease inhibitory activity. Pharm Res 14(2):176–180
Karaman R, Dajani KK, Qtait A, Khamis M (2012) Prodrugs of acyclovir—a computational approach. Chem Biol Drug Des 79(5):819–834. https://doi.org/10.1111/j.1747-0285.2012.01335.x
Katragadda S, Jain R, Kwatra D, Hariharan S, Mitra AK (2008) Pharmacokinetics of amino acid ester prodrugs of acyclovir after oral administration: interaction with the transporters on Caco-2 cells. Int J Pharm 362(1–2):93–101. https://doi.org/10.1016/j.ijpharm.2008.06.018
Kim DC, Harrison AW, Ruwart MJ, Wilkinson KF, Fisher JF, Hidalgo IJ, Borchardt RT (1993) Evaluation of the bile acid transporter in enhancing intestinal permeability to renin-inhibitory peptides. J Drug Target 1(4):347–359. https://doi.org/10.3109/10611869308996094
Kimberlin DW, Whitley RJ (2007) Antiviral therapy of HSV-1 and -2. In: Arvin A, Campadelli-Fiume G, Mocarski E et al (eds) Human herpesviruses: biology, therapy, and immunoprophylaxis. Cambridge University Press, Cambridge
Klysik K, Pietraszek A, Karewicz A, Nowakowska M (2018) Acyclovir in the treatment of herpes viruses—a review. Curr Med Chem. https://doi.org/10.2174/0929867325666180309105519
Kramer W, Glombik H (2006) Bile acid reabsorption inhibitors (BARI): novel hypolipidemic drugs. Curr Med Chem 13(9):997–1016
Kramer W, Wess G (1996) Bile acid transport systems as pharmaceutical targets. Eur J Clin Invest 26(9):715–732
Kramer W, Wess G, Schubert G, Bickel M, Girbig F, Gutjahr U, Kowalewski S, Baringhaus KH, Enhsen A, Glombik H et al (1992) Liver-specific drug targeting by coupling to bile acids. J Biol Chem 267(26):18598–18604
Kramer W, Wess G, Enhsen A, Falk E, Hoffmann A, Neckermann G, Schubert G, Urmann M (1997) Modified bile acids as carriers for peptides and drugs. J Control Release 46(1):17–30. https://doi.org/10.1016/S0168-3659(96)01599-4
Lack L (1979) Properties and biological significance of the ileal bile salt transport system. Environ Health Perspect 33:79–89
Li Y, Dias JR (1997) Dimeric and oligomeric steroids. Chem Rev 97(1):283–304
Liang R, Fei YJ, Prasad PD, Ramamoorthy S, Han H, Yang-Feng TL, Hediger MA, Ganapathy V, Leibach FH (1995) Human intestinal H+/peptide cotransporter. Cloning, functional expression, and chromosomal localization. J Biol Chem 270(12):6456–6463
Longerich T, Eisenbach C, Penzel R, Kremer T, Flechtenmacher C, Helmke B, Encke J, Kraus T, Schirmacher P (2005) Recurrent herpes simplex virus hepatitis after liver retransplantation despite acyclovir therapy. Liver Transpl 11(10):1289–1294. https://doi.org/10.1002/lt.20567
Love MW, Dawson PA (1998) New insights into bile acid transport. Curr Opin Lipidol 9(3):225–229
Miller G, Lipman M (1973) Release of infectious Epstein–Barr virus by transformed marmoset leukocytes. Proc Natl Acad Sci USA 70(1):190–194
Mukhopadhyay S, Maitra U (2004) Chemistry and biology of bile acids. Curr Sci 87(12):1666–1683
Norvell JP, Blei AT, Jovanovic BD, Levitsky J (2007) Herpes simplex virus hepatitis: an analysis of the published literature and institutional cases. Liver Transpl 13(10):1428–1434. https://doi.org/10.1002/lt.21250
Park G-B, Shao Z, Mitra AK (1992) Acyclovir permeation enhancement across intestinal and nasal mucosae by bile salt-acylcarnitine mixed micelles. Pharm Res 9(10):1262–1267. https://doi.org/10.1023/a:1015845031488
Petzinger E (1994) Transport of organic anions in the liver. An update on bile acid, fatty acid, monocarboxylate, anionic amino acid, cholephilic organic anion, and anionic drug transport. Rev Physiol Biochem Pharmacol 123:47–211
Pyles RB (2001) The association of herpes simplex virus and Alzheimer’s disease: a potential synthesis of genetic and environmental factors. Herpes J IHMF 8(3):64–68
Rais R, Fletcher S, Polli JE (2011) Synthesis and in vitro evaluation of gabapentin prodrugs that target the human apical sodium-dependent bile acid transporter (hASBT). J Pharm Sci 100(3):1184–1195. https://doi.org/10.1002/jps.22332
Santos CR, Capela R, Pereira CSGP, Valente E, Gouveia L, Pannecouque C, De Clercq E, Moreira R, Gomes P (2009) Structure–activity relationships for dipeptide prodrugs of acyclovir: implications for prodrug design. Eur J Med Chem 44(6):2339–2346. https://doi.org/10.1016/j.ejmech.2008.08.009
Shao Z, Hoffman AJ, Mitra AK (1994) Biodegradation characteristics of acyclovir 2′-esters by respiratory carboxylesterases: implications in prodrug design for intranasal and pulmonary drug delivery. Int J Pharm 112(2):181
Sievanen E (2007) Exploitation of bile acid transport systems in prodrug design. Molecules 12(8):1859–1889
Soul-Lawton J, Seaber E, On N, Wootton R, Rolan P, Posner J (1995) Absolute bioavailability and metabolic disposition of valaciclovir, the L-valyl ester of acyclovir, following oral administration to humans. Antimicrob Agents Chemother 39(12):2759–2764
Staels B, Fonseca VA (2009) Bile acids and metabolic regulation: mechanisms and clinical responses to bile acid sequestration. Diabetes Care 32(Suppl 2):S237–S245. https://doi.org/10.2337/dc09-S355
Stedronsky ER (1994) Interaction of bile acids and cholesterol with non-systemic agents having hypocholesterolemic properties. Biochim Biophys Acta 1210(3):255–287
Stojančević M, Pavlović N, Goločorbin-Kon S, Mikov M (2013) Application of bile acids in drug formulation and delivery. Front Life Sci 7(3–4):112–122. https://doi.org/10.1080/21553769.2013.879925
St-Pierre MV, Kullak-Ublick GA, Hagenbuch B, Meier PJ (2001) Transport of bile acids in hepatic and non-hepatic tissues. J Exp Biol 204(Pt 10):1673–1686
Swaan PW, Hillgren KM, Szoka FC, Øie S (1997a) Enhanced transepithelial transport of peptides by conjugation to cholic acid. Bioconj Chem 8(4):520–525. https://doi.org/10.1021/bc970076t
Swaan PW, Szoka FC Jr, Oie S (1997b) Molecular modeling of the intestinal bile acid carrier: a comparative molecular field analysis study. J Comput Aided Mol Des 11(6):581–588
Tamminen J, Kolehmainen E (2001) Bile acids as building blocks of supramolecular hosts. Molecules 6(1):21
Thamotharan M, Lombardo YB, Bawani SZ, Adibi SA (1997) An active mechanism for completion of the final stage of protein degradation in the liver, lysosomal transport of dipeptides. J Biol Chem 272(18):11786–11790
Tolle-Sander S, Lentz KA, Maeda DY, Coop A, Polli JE (2004) Increased acyclovir oral bioavailability via a bile acid conjugate. Mol Pharm 1(1):40–48
Turley DS, Dietschy JM (1988) In: Arias IM, Jakoby WB, Popper H, Schachter D, Shafritz DS (eds) In the liver: biology and pathobiology. Raven Press, New York, pp 617–641
Virtanen E, Kolehmainen E (2004) Use of bile acids in pharmacological and supramolecular applications. Eur J Org Chem 16:3385–3399. https://doi.org/10.1002/ejoc.200300699
Vivian D, Polli JE (2014) Synthesis and in vitro evaluation of bile acid prodrugs of floxuridine to target the liver. Int J Pharm 475(1–2):597–604. https://doi.org/10.1016/j.ijpharm.2014.09.014
Wallimann P, Marti T, Fürer A, Diederich F (1997) Steroids in molecular recognition. Chem Rev 97(5):1567–1608. https://doi.org/10.1021/cr960373b
Wang J, Yan X, Lu R, Meng X, Nie G (2017) Peptide transporter 1 (PepT1) in fish: a review. Aquac Fish 2(5):193–206. https://doi.org/10.1016/j.aaf.2017.06.007
Yang C, Tirucherai GS, Mitra AK (2001) Prodrug based optimal drug delivery via membrane transporter/receptor. Expert Opin Biol Ther 1(2):159–175. https://doi.org/10.1517/14712598.1.2.159
Zhang D, Li D, Shang L, He Z, Sun J (2016) Transporter-targeted cholic acid-cytarabine conjugates for improved oral absorption. Int J Pharm 511(1):161–169. https://doi.org/10.1016/j.ijpharm.2016.06.139
Acknowledgements
We thank Dr. Regina Feederle from the German Cancer Research Center, Heidelberg for assistance with immunofluorescence staining, and Dr. Yavor Mitrev from Laboratory “Bulgarian NMR Centre” Institute of Organic Chemistry with Centre of Phytochemistry, Sofia for helpful discussions.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
All authors listed have contributed to conception, design, gathering, analysis, or interpretation of data, and have contributed to the writing and intellectual content of the article. All authors gave informed consent to the submission of this manuscript.
Additional information
Handling Editor: G. J. Peters.
Rights and permissions
About this article
Cite this article
Chayrov, R.L., Stylos, E.K., Chatziathanasiadou, M.V. et al. Tailoring acyclovir prodrugs with enhanced antiviral activity: rational design, synthesis, human plasma stability and in vitro evaluation. Amino Acids 50, 1131–1143 (2018). https://doi.org/10.1007/s00726-018-2590-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-018-2590-y