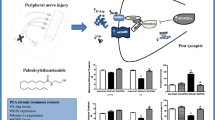
Abstract
d-Aspartate (d-Asp) is a free d-amino acid detected in multiple brain regions and putative precursor of endogenous N-methyl-d-aspartate (NMDA) acting as agonist at NMDA receptors. In this study, we investigated whether d-Asp (20 mM) in drinking solution for 1 month affects pain responses and pain-related emotional, and cognitive behaviour in a model of neuropathic pain induced by the spared nerve injury (SNI) of the sciatic nerve in mice. SNI mice developed mechanical allodynia and motor coordination impairment 30 days after SNI surgery. SNI mice showed cognitive impairment, anxiety and depression-like behaviour, reduced sociability in the three chamber sociability paradigm, increased expression of NR2B subunit of NMDA receptor and Homer 1a in the medial prefrontal cortex (mPFC). The expression of (post synaptic density) PSD-95 and Shank 1was instead unaffected in the mPFC of the SNI mice. Treatment with d-Asp drinking solution, started right after the SNI (day 0), alleviated mechanical allodynia, improved cognition and motor coordination and increased social interaction. d-Asp also restored the levels of extracellular d-Asp, Homer 1a and NR2B subunit of the NMDA receptor to physiological levels and reduced Shank1 and PSD-95 protein levels in the mPFC. Amitriptyline, a tricyclic antidepressant used also to alleviate neuropathic pain in humans, reverted mechanical allodynia and cognitive impairment, and unlike d-Asp, was effective in reducing depression and anxiety-like behaviour in the SNI mice and increased PSD protein level. Altogether these findings demonstrate that d-Asp improves sensorial, motor and cognitive-like symptoms related to chronic pain possibly through glutamate neurotransmission normalization in neuropathic mice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
l-Glutamate (Glu) is the major excitatory neurotransmitter in the mammalian brain and inappropriate regulation of glutamatergic neurotransmission drives to a spectrum of somatic, neurological and psychiatric disorders, including abnormal pain responses, motor impairment and negative emotional states such as anxiety, depression and psychosis (Coyle et al. 2003; Swanson et al. 2005). Glutamate transmission can be triggered by d-aspartate (d-Asp), which besides being the putative precursor of endogenous N-methyl-d-aspartate (NMDA) (D’Aniello et al. 2000b) activates itself NMDA receptors (NMDARs) on the orthosteric site with a relatively high affinity (K i 0.9 ± 0.4 μM) on each NR2A-D receptor subunits (Fagg and Matus 1984; Monahan and Michel 1987). d-Asp is synthesized in mammalian cells by the d-aspartate racemase (DR), the enzyme able to convert l-Asp in d-Asp (Kim et al. 2010; D’Aniello et al. 2011). d-Asp, in turn, is hypothesized to be converted in NMDA by d-aspartate methyl-transferase (NMDA synthetase) (D’Aniello et al. 2000a, b; Shibata et al. 2011). Free d-Asp has been found in the nervous system and endocrine tissues from mollusks to mammalians, including humans (D’Aniello 2007; Ota et al. 2012). In embryonic stage and in the first days of life d-Asp is found in high concentration which considerably decreases in adults (Dunlop et al. 1986; Neidle and Dunlop 1990; Hashimoto et al. 1993, 1995; Sakai et al. 1998; Lee et al. 1999; Wolosker et al. 2000; Huang et al. 2008). In mammals d-Asp can be detected in synaptic vesicles of nerve endings and has been demonstrated to act as a neurotransmitter (Spinelli et al. 2006; D’Aniello et al. 2011). d-Asp is selectively metabolized by d-aspartate oxidase (DDO), the only enzyme that degrades bicarboxylic d-amino acids, including N-methyl-d-aspartate (NMDA). DDO has been found in the central nervous system (CNS) at post-synaptic level and endocrine glands (Furuchi and Homma 2005; D’Aniello 2007). So far, apart from the effect of d-Asp in enhancing long-term potentiation (LTP) and rescuing the synaptic plasticity decay in the hippocampus of rodents (Errico et al. 2011a, b), the potential effect of exogenous d-Asp administration on brain pathophysiology is unknown. A putative role for endogenous d-Asp level in pain and pain-related affective/cognitive-like disorders has been just begin to be investigated in rodents (Boccella et al. 2015; Guida et al. 2015). Therefore we used the spare nerve injury (SNI) model of neuropathic pain to test the effects of 1 month d-Asp drinking solution on pain responses and pain-related affective and cognitive-like behaviour in mice. In some of the behavioural tests the effect of d-Asp was compared to that of amitriptyline, a tricyclic antidepressant which beside being effective towards depression and anxiety is widely used to treat neuropathic pain (Sindrup et al. 2005). The effect of d-Asp treatment on d-Asp and glutamate extracellular level and on glutamate postsynaptic density (PSD) proteins as well as on NR2A/NR2B subunits of NMDA receptor was evaluated in medial prefrontal cortex (mPFC), a forebrain area playing a crucial role in pain-related affective and cognitive disorders.
Materials and methods
Animals
Male 5 week-old CD1 mice (25–30 g) were housed under controlled illumination (12/12 h light/dark cycle; light on 06:00 hours) and environmental conditions (room temperature 20–22 °C, humidity 55–60 %) for at least 1 week before the commencement of experiments. Mouse chow and tap water were available ad libitum. The experimental procedures were conducted in conformity with protocols approved by the Animal Ethics Committee of the Second University of Naples. Animal care was in compliance with the IASP and European Community (E.C. L358/1 18/12/86) guidelines on the use and protection of animals in experimental research. All efforts were made to minimise animal suffering and to reduce the number of animals used.
Spared nerve injury
Mono-neuropathy was induced according to the method of Decosterd and Woolf (2000). Mice were anaesthetised with sodium pentobarbital (50 mg/kg, i.p.), the sciatic nerve was exposed at the level of its trifurcation and the tibial and common peroneal nerves were tightly ligated with 5.0 silk thread and then transected just distal to the ligation, leaving the sural nerve intact. Sham mice were anaesthetised, the sciatic nerve was exposed at the same level, but not ligated. The quantification of d-Asp content in the brain, the protein levels and all behavioural tests were performed 30 days after the SNI or sham surgery by an experimenter blind to the treatment.
Total brain d-Asp quantification
Mice were killed and the brain immediately stored at −80 °C. The determination of d-Asp was performed by HPLC technique, based on the diastereomeric separation of d-Asp from the l-form and other l-amino acids, as previously described by D’Aniello et al. (2000b). Data were analysed using ANOVA or Student’s t test where appropriate and data expressed as mean ± standard error of the mean (SEM).
Mechanical allodynia
Dynamic plantar aesthesiometer
Mechanical allodynia was measured by the dynamic plantar aesthesiometer device (Ugo Basile, Varese, Italy). Mice were allowed to move freely in one of the two compartments of the enclosure, positioned on the metal mesh surface and allowed to adapt to the testing environment before the experimental procedure. After 30 min habituation period, the mechanical stimulus, a steel rod (2 mm) pushed with ascending force (0–30 g in 10 s), was delivered to the plantar surface of the hind paw of the mouse from below the floor of the test chamber by an automated testing device. The cut-off was set to 30 g to avoid excessive ascending force. When the mouse withdrew its hind paw, the mechanical stimulus was automatically stopped and the force was recorded to the nearest 0.1 g. Data were expressed as mechanical withdrawal threshold (MWT) in grams and measured for 2 h every 15 min.
Von Frey filaments
Mechanical allodynia was also measured with a series of calibrated nylon von Frey filaments (Stoelting, Wood Dale, IL, USA), ranging from 0.6 to 10 g. The animals were placed in plastic cages with a wire-mesh floor approximately 5–10 min before testing to allow behavioral accommodation. The von Frey filaments were applied in ascending order to the mid-plantar surface of the injured hind paw through the mesh floor. If the use of the filament three times did not induce a reaction, the next filament with higher pressure was used. The time interval before the application of the next filament was at least 5 s. Data were expressed as mechanical withdrawal threshold (MWT) in grams.
Motor coordination
Motor coordination was evaluated by Rota-rod (Ugo Basile, Varese, Italy). Mice were placed on a rotary cylinder and the latency time of equilibrium before falling was measured in seconds. The five sections for cylinder allowing to screen five animals per each test. The platform below the cylinder was connected to a magnet thus recording the time of permanence on the cylinder. After a period of adaptation of 30 s, the spin speed gradually increased from 5 to 40 rpm for the maximum time of 5 min. The time of permanence of the mouse on the cylinder was expressed as duration in seconds.
Cognitive performance
Novel object recognition
Novel object recognition consisted in a period of habituation, an acquisition trial, and a test trial. During habituation mice were allowed to freely explore for 1 h the apparatus which consists of a rectangular open box (40 × 30 × 30 cm; width × length × height) made of grey polyvinyl chloride (PVC) illuminated by a dim light. During the acquisition trial each mouse was allowed to explore two identical objects positioned in the back left and right corners for 5 min. A camera recorded the time spent on exploration of each object. In the test trial, which was carried out for 2 h after the acquisition, one of the two objects was replaced with a new different object. The time spent exploring the object was considered as the time the mouse spent with its nose directed to, and within 1 cm from, the object. The recognition index (R.I.) expressed as the percentage of the time the mouse spent exploring the novel object/(the time the mouse spent exploring the novel object + the time the mouse spent exploring the familiar object) was recorded.
Y maze
The apparatus consisted of three enclosed arms (30 × 5 × 15 cm; length x width x height) converging on an equilateral triangular center (5 × 5 × 5 cm). At the beginning of each experimental session, each mouse was placed in the center platform and the number of spontaneous alternations (defined as number of successive triplet entry into each of the three arms without any repeated entries) was monitored in a 5 min test session. The percentage of alternation was calculated as the percentage of the ratio of the number of alternations/(total number of arm entries − 2).
Depression-like behavior
Tail suspension
Mice were individually suspended by the tail on a horizontal bar (50 cm from floor) using adhesive tape placed approximately 4 cm from the tip of the tail. The duration of immobility was recorded in seconds over a period of 6 min by a time recorder. Immobility time was defined as the absence of escape-oriented behavior.
Forced swimming test
Mice were placed in a large cylinder (30 cm × 45 cm) filled with water at a temperature of 27 °C, for a 6-min period. The duration of immobility was monitored during the last 4-min of the 6-min test and defined as the absence of escape-oriented behavior (mice were floating in the water without struggling and making only those movements necessary to keep head above the water). Immediately after the trial mice were placed under a heating lamp to dry.
Anxiety-like behaviour
Marble burying
Mice were individually placed in a plastic cage (21 × 38 × 14 cm; length × width × height) containing 5 cm layer of sawdust bedding and 15 glass marbles (1.5 cm in diameter) arranged in three rows. Mice were left undisturbed for 15 min under dim light. An observer blind to the treatment counted the time spent in digging behavior, the number of marbles buried (at least two-third buried in the sawdust) and the number of digging events. At the end of the test the mouse was removed to its own cage.
Light–dark box
The light–dark box (600 × 300 × 300 mm; length × width × height) apparatus consisted into two equally sized compartments: the dark compartment (black perspex) was covered, whereas the light compartment (white perspex) was open and brightly lit from above (~150 lux). Access between compartments was allowed through a partition door (70 × 70 mm). At the beginning of the session, mice were placed in the dark compartment and were free to explore for 10 min. The time spent in each compartment, transitions into each compartment, latency of first entry into the light compartment were recorded.
Three chamber sociability
This test allows to evaluate two distinct aspects of the social behaviour: the social affiliation (sociability) and the social recognition memory. A plexiglass three-chambered box was custom-built as follows: doorways in the two dividing walls had sliding covers to control access to the outer-side chambers. The test consisted of three consecutive stages of 5, 10 and 10 min each. During the 5-min first stage of habituation the mouse was allowed to freely explore the three chambers of the apparatus, detecting at this stage any innate side preference. After that the mouse was gently encouraged into the central chamber and confined there briefly by closing the side chamber doors. During the following 10-min stage sessions, a custom-made stainless-steel barred cup (6.5 × 15 cm) was placed upside down in one of the side chambers. A never-before-met intruder, previously habituated, was placed into an upside down identical cup in the other chamber. In the last session the subject encounters the first intruder as well as a second never-before-met intruder under another upside down cup in the “social novelty” session. The time spent sniffing each upside down cup, the time spent in each chamber, and the number of entries into each chamber were recorded. During the stages, the preference for sociability was defined as the time spent in the side chamber with the never-before-met-intruder compared to the chamber with the empty upside down cup or the already met intruder. The time spent in each chamber, the number of transitions between the chambers, the social interaction duration, the number of rearing and self-grooming were monitored.
In vivo microdialysis
Intracerebral in vivo microdialysis was performed in awake and freely moving mice. Mice were anesthetized with pentobarbital (50 mg/kg, i.p.), and the concentric microdialysis probes were stereotaxically implanted in the prefrontal cortex (PFC, AP +1.42 mm, L 0.5, and V 3 mm below the dura) according to the atlas of Franklin and Paxinos (1997). Microdialysis concentric probes were constructed as described by (Hutson et al. 1985) with 22G (0.4 mm I.D., 0.7 mm O.D.) stainless steel tubing: inlet and outlet cannulae (0.04 mm I.D., 0.14 mm O.D.) consisted of fused silica tubing (Scientific Glass Engineering, Melbourne, Australia). The microdialysis probe had a tubular dialysis membrane (Enka AG, Wuppertal, Germany) 0.8 mm in length. After a postoperative recovery period of approximately 24 h, dialysis probes were perfused with artificial cerebrospinal fluid (ACSF composition in mM: KCl, 2.5; NaCl, 125; MgCl2, 1.18; CaCl2, 1.26) (pH 7.2) at a rate of 1.0 μl/min using a Harvard Apparatus infusion pump (mod. 22). Following an initial 60 min equilibration period, five consecutive 30 min dialysate samples were collected. On completion of experiments, mice were anaesthetised with pentobarbital and their brains perfused–fixed via the left cardiac ventricle with heparinised paraformaldehyde saline (4 %). Brains were dissected out and fixed in a 10 % formaldehyde solution for 2 days. Each brain was cut in 40 μm thick slices and observed under a light microscope to identify the probe locations. Dialysates were analyzed for d-Asp and l-glutamate (l-Glu) content using a high-performance liquid chromatography coupled with fluorimetric detection method. The system comprised two Gilson pumps (model no. 303), a C18 reverse-phase column, and a Gilson fluorimetric detector (model no. 121). Dialysates were precolumn derivatized with o-pthaldialdehyde-N-acetylcysteine (OPA-NAC) (10 μl dialysate + 5 μl OPA-NAC + 10 μl borate buffer 10 %). The mobile phase consisted of two components: (A) 0.2 M Na2HPO4, 0.2 M citric acid and 20 % methanol and (B) 90 % acetonitrile. Gradient composition was determined using an Apple microcomputer installed with Gilson gradient management software. Data were collected using a Dell Corporation PC system 310 interfaced to the detector via a Drew data-collection unit. The mean dialysate concentration of amino acids in the five samples represents the basal release and the results were expressed as the mean ± SEM of the pmol in 10 µl of perfusate.
Western blotting
Medial prefrontal cortex contralateral to sciatic nerve ligation or sham surgery was dissected out and synaptic protein expression was assessed by western blotting analysis. The tissue samples were lysed in Syn-PER Synaptic Protein Extraction Reagent (Thermoscientific, USA) (10 ml of Syn-PER Reagent per gram of tissue), for synaptosome isolation. The buffer used for lysis contained a phosphatase and proteinase inhibitor mixture, Halt Protease Inhibitor Cocktail (Thermoscientific, USA), which was added immediately before use to avoid proteins degradation. Homogenization was carried out by mean dounce tissue grinder on ice and homogenate was centrifuged at 1200×g for 10 min at 4 °C. The pellet was removed and supernatant centrifuged at 15,000×g for 20 min at 4 °C. After removing supernatant, which contained cytosolic fraction, synaptosome pellet was suspended by adding 1–2 ml of Syn-PER Reagent per gram of sample. In each sample protein concentration was determined using Bradford (1976) assay and equal amounts of total proteins were loaded onto 10 % polyacrylamide gel for NR2A and NR2B, PSD-95, Shank1, and 15 % polyacrylamide gel for Homer 1a and Homer 1b. Proteins were separated by SDS-PAGE and transferred to nitrocellulose blotting membranes (GE Healthcare, UK). Following blocking in buffer solution including 5 % milk and 0.1 % Tween-20 in TBS (Tris HCl 25 mM, NaCl 137 mM), the membranes were immunoblotted with the following primary antibodies: NR2A (1:1000, Santa Cruz Biotechnology), NR2B (1:1000, Santa Cruz Biotechnology), Homer 1a (1:200; Santa Cruz Biotechnology), Homer 1b (1:1000, Santa Cruz Biotechnology), PSD-95 (1:1000, Santa Cruz Biotechnology) and Shank1 (1:1000, Santa Cruz Biotechnology). Blots were then incubated in species-appropriate horseradish peroxidase-conjugated secondary antibodies (1:2000, Santa Cruz Biotechnology) and proteins visualized by ECL detection (LiteAblot extend, Euroclone) at molecular weight target. Band intensity was quantified on scanned filters by Quantity one analysis software (Bio-Rad, USA). Background value was subtracted to minimize variability across membranes and each lane was normalized for the corresponding actin value for variation in loading and transfer.
Drugs
d-Asp was purchased from Sigma-Aldrich, Milan, Italy. d-Asp was delivered in drinking water at the concentration of 20 mM according to in vivo studies using the oral administration (Errico et al. 2008a, b, 2011a). These studies have shown that the dose of 20 mM of d-Asp is able to increase significantly the content of d-Asp in hippocampus (Errico et al. 2008a, 2011a), cortex, striatum and cerebellum (Errico et al. 2008b). Amitriptyline was purchased from Tocris Bioscience (Bristol, UK) and administered intraperitoneally at the dose of 10 mg/kg once a day starting the day of surgery (day 0). The dose and administration route of amitriptyline was chosen according to the literature (Benbouzid et al. 2008; Berrocoso et al. 2011). Water or d-Asp drinking solution were available ad libitum with all groups of mice consuming from 4 to 6 ml each 24 h.
Treatment
The vehicle or drug chronic treatment started the same day of sham or SNI surgery (day 0) and was performed for 30 days. The SNI or sham mice receiving different treatments were divided as follows:
-
1.
Sham mice drinking water (n = 8–12).
-
2.
Sham mice drinking d-Asp water solution (20 mM) (n = 8–12).
-
3.
SNI mice drinking water (n = 8–12).
-
4.
SNI mice drinking d-Asp water solution (20 mM) (n = 8–12).
-
5.
Sham mice receiving amitriptyline (10 mg/kg, i.p.) (n = 8–12).
-
6.
SNI mice receiving amitriptyline (10 mg/kg, i.p.) (n = 8–12).
For western blotting analysis and motor coordination 4 or 19–24 mice per group, respectively, were used.
Data analysis and statistics
All data are given as mean ± SEM. Two-way ANOVA followed by the Newman–Keuls post hoc test have been used to analyze statistical differences between the different groups of mice. ANOVA or Student’s t test has been used for total brain d-Asp quantification. Turkey’s post hoc test was used as post hoc test in western blot analysis. P < 0.05 was considered as level of significance.
Results
Oral d-Asp treatment increases brain d-Asp level
We found that 1 month of d-Asp (20 mM) drinking solution, starting the same day of the SNI surgery (day 0) significantly increased the total brain content of d-Asp (F (3,32) = 11.69; p < 0.0001) in both SNI and sham mice compared with sham and SNI mice treated with water (Fig. 1; Table 1).
Typical HPLC determination of d-Asp by the OPA-N-acetyl-cysteine method. a HPLC separation of a standard mixture of amino acids (20 pmol each amino acid and 10 pmol of d-Asp) derivatized with OPA-NAC and fluorescence detection. Dashed line represents the HPLC gradient program. b Same sample as in a after treatment with DDO. The peak corresponding to d-Asp disappears because of oxidation by DDO. The arrow shows the elution of d-Asp
d-Asp drinking solution and amitriptyline alleviate mechanical allodynia in SNI mice
Dynamic plantar aesthesiometer
Sham mice did not show any change in MWT compared to naïve mice (data not shown). The 30 days d-Asp drinking solution (20 mM) treatment did not change the MWT in sham mice. SNI mice drinking water showed a significant decrease of the MWT in the ipsilateral hind paw compared to the sham mice drinking water (F (3,32) = 24.57; p < 0.001). The 1-month d-Asp treatment reverted mechanical allodynia as compared to the SNI mice receiving water for the same period (F (3,32) = 24.57; p < 0.001) (Fig. 2a).
Effects of d-Asp (20 mM) drinking solution on mechanical allodynia measured through dynamic plantar aesthesiometer (a), von Frey filaments (b) or motor coordination measured at the rotarod (c) 30 days after SNI or sham surgery. The effect of d-Asp on mechanical allodynia measured by von Frey was compared to that one of amitriptyline. Mechanical withdrawal threshold was significantly lower in the ipsilateral hind paw of SNI mice drinking water (SNI/water) (a, b). Chronic treatment with d-Asp drinking solution increased MWT in SNI mice (a, b). Chronic treatment with amitriptyline (10 mg/kg, i.p.) increased MWT in SNI mice without changing it in the shams (b). Motor coordination, determined as duration before fall down in s, was impaired in SNI mice and improved by 30 days treatment with d-Asp drinking solution (c). Each point represents the mean ± standard error of the mean (SEM) of the mechanical withdrawal threshold in grams or latency in s of 8–12 mice per group apart from motor coordination experiments where groups of 19–24 mice were considered. Open circles or asterisks indicate significant differences compared to sham/water or SNI/water, respectively. P < 0.05 was considered statistically significant
Von Frey
d-Asp drinking solution (20 mM) treatment did not alter the MWT in sham mice. SNI mice drinking water showed a significant reduction of the MWT in the ipsilateral hind paw compared to the sham mice drinking water (F (5,42) = 10.77; p < 0.001). The 1-month d-Asp treatment reverted mechanical allodynia, compared to the SNI mice receiving water for the same period (F (5,42) = 10.77; p < 0.001) (Fig. 2b). The 30 days amitriptyline treatment (10 mg/kg, i.p.) significantly reverted mechanical allodynia in SNI mice (F (5,42) = 10.77; p < 0.001) without changing the MWT in the shams (Fig. 2b).
d-Asp drinking solution improves motor coordination in SNI mice
The effect of 1 month d-Asp drinking solution on motor coordination was evaluated using the Rota-rod apparatus. Sham mice did not show any change in motor activity as compared to naïve mice (not shown). d-Asp (20 mM) did not change the latency to fall down on the rotating rod in sham mice. The SNI mice drinking water showed a significant decrease in the latency to fall down compared to the sham mice drinking water (F (3,87) = 11.26; p < 0.001). One month d-Asp drinking solution (20 mM) increased the duration of the time mice spent walking on the rotating rod (F (3,87) = 11.26; p < 0.01) (Fig. 2c).
d-Asp drinking solution or amitriptyline ameliorate cognition in SNI mice
Y maze
d-Asp drinking solution treatment (20 mM) did not change the percentage of alternations in sham mice. SNI mice drinking water showed memory impairments, as indicated by a significant decrease in the percentage of alternations (F (5,42) = 6.83; p < 0.05) compared with sham mice drinking water. In SNI mice, the percentage of alternation was significantly increased by a month d-Asp drinking solution treatment (F (5,42) = 6.83; p < 0.001) (Fig. 3a). Amitriptyline treatment (10 mg/kg, i.p.) significantly increased the percentage of alternation in the SNI mice (F (5,42) = 6.83; p < 0.001) (Fig. 3a).
Effects of a month d-Asp (20 mM) drinking solution or amitriptyline (10 mg/kg, i.p.) on the percentage of alternation in the Y maze (a) or the recognition index in the novel object recognition task (b) in sham and SNI mice. Values are mean ± SEM of 8–12 animals per group. Open circles or asterisks indicate significant differences compared to sham/water or SNI/water, respectively. P < 0.05 was considered statistically significant
Novel object recognition
In the novel object recognition test sham mice did not show any change in recognition index compared to naïve mice (data not shown). One month d-Asp drinking solution treatment (20 mM) did not change the recognition index in sham mice. SNI mice drinking water showed a significant reduction in the recognition index as compared to sham mice drinking water (F (5,42) = 6.80; p < 0.001). The 1 month d-Asp drinking solution significantly increased the recognition index in the SNI mice (F (5,42) = 6.80; p < 0.001) (Fig. 3b). Amitriptyline treatment (10 mg/kg, i.p.) significantly increased the recognition index in the SNI mice (F (5,42) = 6.80; p < 0.05) (Fig. 3b).
d-Asp drinking solution does not ameliorate depression-like behavior in SNI mice
Tail suspension
Sham mice did not show any change in the duration of immobility compared to naïve mice (not shown). The d-Asp drinking solution (20 mM) did not change the immobility time in sham mice. SNI mice drinking water showed a significant increase in the duration of immobility 30 days post SNI (F (5,42) = 17.17; p < 0.001). The treatment with d-Asp (20 mM) drinking solution did not modify significantly the duration of the immobility in SNI mice (Fig. 4a). The treatment with amitriptyline (10 mg/kg, i.p.) significantly reduced the duration of the immobility in SNI mice (F (5,42) = 17.17; p < 0.001) (Fig. 4a).
Effects of a month d-Asp (20 mM) drinking solution or amitriptyline (10 mg/kg, i.p.) on the duration of immobility in the tail suspension (a) or in the forced swimming (b) tests in sham and SNI mice. Values are mean ± SEM of 8–12 animals per group. Open circles or asterisks indicate significant differences compared to sham/water or SNI/water, respectively. P < 0.05 was considered statistically significant
Forced swimming
Sham mice did not show differences in the duration of immobility compared to naïve mice (not shown). The d-Asp drinking solution (20 mM) did not alter the duration of immobility time in sham mice. SNI mice drinking water showed a significant increase in the duration of immobility 30 days post SNI (F (5,42) = 119.6; p < 0.001). The treatment with d-Asp (20 mM) drinking solution did not modify significantly the duration of the immobility in SNI mice (Fig. 4a). The treatment with amitriptyline (10 mg/kg, i.p.) significantly reduced the duration of the immobility in SNI mice (F (5,42) = 119.6; p < 0.001) (Fig. 4b).
d-Asp drinking solution does not affect anxiety-like behaviour in SNI mice
Light/dark box
Sham mice did not show any change in time spent in the illuminated compartment of the light dark box compared to naive animals (data not shown). Sham mice drinking d-Asp solution (20 mM) or treated with amitriptyline (10 mg/kg, i.p.) did not show any significant change in the time spent in the illuminated compartment of the light/dark box. SNI mice drinking d-Asp solution (20 mM) did not show any change in the time spent in the illuminated compartment of the light/dark box (Fig. 5a). SNI mice treated with amitriptyline showed a significant increase in the time spent in the illuminated compartment of the light/dark box (p < 0.001) (Fig. 5a). Sham mice drinking d-Asp solution (20 mM) show a significant increase in the number of transitions in the two compartments (F (5,42) = 22.40; p < 0.001) while amitriptyline (10 mg/kg, i.p.) caused the opposite effect (F (5,42) = 22.40; p < 0.05) compared to sham mice drinking water. The SNI mice drinking water showed a significant decrease in the number of transitions in the two compartments (F (5,42) = 22.40; p < 0.01) compared to the shams drinking water. The treatment with d-Asp (20 mM) did not show any significant change while amitriptyline increased significantly the number of transitions (F (5,42) = 22.40; p < 0.001) in the SNI mice (Fig. 5b).
Effects of a month d-Asp (20 mM) drinking solution or amitriptyline (10 mg/kg, i.p.) on the time spent in the light box (a), and the number of transitions (b) in the light-dark box, or the number of digging events (c) and the number of marbles buried (d) in the marble burying in sham and SNI mice. Values are mean ± SEM of 8–12 animals per group. Open circles or asterisks indicate significant difference compared to sham/water or SNI/water, respectively. P < 0.05 was considered statistically significant
Murble burying
No difference in the number of marbles buried and digging events were observed in sham mice compared to naïve mice (data not shown). The d-Asp drinking solution (20 mM) did not change the number of marbles buried and digging events in sham mice. SNI mice drinking water showed an increase in the number of digging events (F (3,44) = 6.46; p < 0.01) (Fig. 5c) and marbles buried (F (3,44) = 26.83; p < 0.001) (Fig. 5d) and a decrease of latency time to dig (not shown) compared to sham mice drinking water. The d-Asp drinking solution (20 mM) did not change the number of digging events and marbles buried in the SNI group of mice (Fig. 5c, d).
d-Asp drinking solution rescues the social interaction and social recognition in SNI mice
In the three-chamber paradigm sham mice did not show any change in social affiliation and the social recognition memory compared to naïve mice (data not shown). The 1 month d-Asp drinking solution (20 mM) did not change the social affiliation and the social recognition in sham mice (not shown). SNI mice drinking water for a month showed a significant impairment in sociability and social recognition memory compared to the sham mice (F (5,42) = 35.22 and F (5,42) = 17.34, respectively with p < 0.001 for both) (Fig. 6c, d). The 1 month d-Asp drinking solution (20 mM) produced a normalization of social affiliation and social memory behavior (F (5,42) = 35.22 and F (5,42) = 17.34, respectively with p < 0.0001 for both) (Fig. 6c, d).
Effects of 1 month water or d-Asp drinking solution on the social interaction paradigm in sham and SNI mice. a, b The time of permanence of the different groups of mice in each chamber in the first and second session, respectively. c The total social interaction time and d social recognition memory. Values are mean ± SEM of 8–12 animals per group. Open circles indicates significant difference compared to sham/water. P < 0.05 was considered statistically significant
d-Asp affects amino acids release in the mPFC of SNI mice
The d-Asp and l-Glu content in the mPFC of sham and SNI mice drinking d-Asp solution or treated with amitriptyline (10 mg/kg, i.p.) was measured by in vivo microdialysis associated with HPLC. A significant increase of d-Asp was observed in sham mice drinking d-Asp (20 mM) solution whereas sham mice treated with amitriptyline (10 mg/kg, i.p.) did not show changes in d-Asp level (Fig. 7a). SNI mice drinking water showed a significant increase in d-Asp content compared to sham mice drinking water. The increase of extracellular d-Asp in the SNI mice was significantly reverted by d-Asp (20 mM) drinking solution (F (5,48) = 3.74; p < 0.05) though not by amitriptyline (10 mg/kg, i.p.). Level of l-Glu was significantly reduced in sham mice drinking d-Asp (F (5,48) = 5.98; p < 0.05) or treated with amitriptyline (F (5,48) = 5.98; p < 0.05) or in SNI mice drinking water (F (5,48) = 5.98; p < 0.05). The decrease of l-Glu level in SNI mice was not modified after d-Asp (20 mM) drinking solution or amitriptyline (10 mg/kg, i.p.) (Fig. 7b).
Effects of a month d-Asp (20 mM) drinking solution or amitriptyline (10 mg/kg, i.p.) on the d-Asp (a) or glutamate (b) extracellular concentration in the mPFC of sham or SNI mice. a, b The pmol in 10 μl of microdialysis perfusate. Values are mean ± SEM of 8–12 animals per group. Open circles or asterisks indicate significant difference compared to sham/water or SNI/water, respectively. P < 0.05 was considered statistically significant
d-Asp drinking solution restores postsynaptic protein expression in SNI mice
Western blotting was carried on mPFC synaptosome fractions obtained from sham or SNI mice after 1 month water, d-Asp drinking solution or amitriptyline (10 mg/kg, i.p.). SNI of the sciatic nerve induced a significant increase in Homer 1a (F (4,15) = 28.26; p < 0.05) (Fig. 8) and NR2B subunit of NMDA receptor (Fig. 9) (F (4.12) = 6.39; p < 0.05) as compared with sham mice drinking water. No changes in the Shank1 and PSD-95 protein levels were observed in SNI mice drinking water as compared to sham mice drinking water (Fig. 8). One month d-Asp (20 mM) drinking solution completely reverted the Homer 1a (F (4,15) = 28.26; p < 0.05) and NR2B subunit (F (4.12) = 6.39; p < 0.05) increase in the SNI group of mice (Fig. 7). Moreover, 1 month d-Asp drinking solution (20 mM) significantly decreased Shank1 (F (4.15) = 11.12; p < 0.05) and PSD-95 (F (4.15) = 107.3; p < 0.05) protein levels as compared to SNI mice drinking water (Fig. 9). One month amitriptyline treatment (10 mg/kg, i.p.) increased significantly Homer 1a, Homer 1b, Shank1 and PSD-95 protein levels in the SNI group of mice (Fig. 9).
Effects of a month d-Asp (20 mM) drinking solution or amitriptyline (10 mg/kg, i.p.) on the expression levels of Homer 1A (a), Homer 1B (b), Shank1 (c) or PSD-95 (d) normalized to β-actin in the mPFC of sham or SNI mice. Values are mean ± SEM of four animals per group. Open circles or asterisks indicate significant difference compared to sham/water or SNI/water, respectively. P < 0.05 was considered statistically significant
Effects of a month d-Asp (20 mM) drinking solution on the expression levels of NMDA receptor subunits NR2A (a) or NR2B (b) normalized to β-actin in the mPFC of sham or SNI mice. Values are mean ± SEM of four animals per group. Open circles or asterisks indicates significant difference compared to sham/water or SNI/water, respectively. P < 0.05 was considered statistically significant
Discussion
The SNI of the sciatic nerve reproduces the multiple symptoms related to neuropathic pain in rodents, ranging from mechanical allodynia and thermal hyperalgesia to affective and cognitive impairments mimicking human experience (Neugebauer et al. 2009). Indeed in our study the SNI of the sciatic nerve apart from mechanical allodynia drove to anxiety- and depression-like behavior, cognitive impairment and social isolation in mice.
Altered glutamate transmission is found in chronic pain conditions (al-Ghoul et al. 1993; Hudson et al. 2002) and psychiatric disorders (Campbell and MacQueen 2006; Konarski et al. 2008; Koolschijn et al. 2009; Coyle 2012). The NMDA receptor plays a major role in pain sensitization (Woolf and Salter 2000), schizophrenia (Coyle et al. 2003), depression (Duman 2014) and cognition (Kullmann et al. 2000). d-Asp has been suggested to orthosterically activate NMDA receptor (Fagg and Matus 1984; Monahan and Michel 1987; Shibata et al. 2011) and be the endogenous precursor of NMDA produced by means of a d-aspartate methyl transferase (NMDA-synthetase) in the rat nervous tissue (D’Aniello et al. 2000a, b). The first finding of this study was that 1-month treatment with d-Asp drinking solution causes a twofold increases of d-Asp level in the brain in both sham and SNI mice according to Errico et al. (2011a) and alleviated mechanical allodynia in SNI mice. These findings suggest that d-Asp may revert mechanical allodynia by stimulating NMDA receptors at supraspinal level. Indeed, NMDA receptor activation in the medial prefrontal cortex (mPFC), has proven to inhibit pain (Millecamps et al. 2007). mPFC is one of the main brain region implicated in the neural network involved in the affective/cognitive consequences of chronic pain (Apkarian et al. 2005). Conversely, at spinal level high amount of d-Asp leads to increased evoked activity of nociceptive specific neurons and a decrease of mechanical and thermal pain thresholds (Boccella et al. 2015). Microdialysis studies found increased extracellular levels of d-Asp in the mPFC of SNI mice, and a decrease of extracellular glutamate. Intriguingly the d-Asp treatment was able to increase the level of d-Asp in mPFC in sham mice while normalizing d-Asp concentration to levels similar to the controls in SNI mice. The increased levels of d-Asp in neuropathic pain condition which happens concurrently to a glutamate decrease in the mPFC is consistent with the finding of Guida et al. (2015). Therefore an opposite modulation of d-Asp and glutamate may occur in the SNI mice as consequence of abnormal glutamate transmission following long lasting SNI, which in turn may drive changes in the NMDA receptor and PSD proteins expression (see afterwards). Moreover, the effect of d-Asp drinking solution on the d-Asp levels in the SNI mice in the mPFC appears different from that on total brain, where the d-Asp content was significantly increased. The d-Asp drinking solution may thus cause changes in d-Asp level which apart from being site-specific may depend on the physiological/pathological preexisting condition. Indeed in this study the treatment with d-Asp drinking solution normalized the d-Asp content in the mPFC in neuropathic pain condition. Even if a further increase of d-Asp content in mPFC in SNI mice treated with d-Asp drinking solution was expected it may be that compensatory mechanisms occur for preventing that high levels of d-Asp by increasing NMDA receptor-mediated currents (Krashia et al. 2015) and changing PSD proteins (Krashia et al. 2015 and this study) may switch to excite-toxicity (Fan et al. 2014). Indeed, d-Asp oral treatment reverted the sensory and cognitive symptoms in the SNI mice. Even if thermal hyperalgesia develops in the SNI model of neuropathic pain (Decosterd and Woolf 2000) in the current study mechanical allodynia was considered as the only evoked sensory symptom since it is the most troublesome with suffering often persisting after cessation of stimulation and highly affecting quality of life (Gottrup et al. 2003).
The effect of d-Asp on mechanical allodynia was compared and it has proven to be similar to that of amitriptyline, a first line antidepressant used against neuropathic pain (Sindrup et al. 2005). Amitriptyline relieves neuropathic pain by inhibiting presynaptic reuptake of serotonin and noradrenalin enhancing monoamine role as endogenous inhibitors of the pain gate at the dorsal horn of the spinal cord level. Other plausible analgesic mechanisms of amitriptyline are: (1) opioid receptor stimulation (Hall and Ogren 1981), (2) NMDA receptor blockade (McCaslin et al. 1992; Watanabe et al. 1993; Eisenach and Gebhart 1995), (3) blockade of sodium (Ishii and Sumi 1992; Deffois et al. 1996; Pancrazio et al. 1998) or (4) calcium channels (Lavoie et al. 1990; Shimizu et al. 1992). Apart from relieving mechanical allodynia, both d-Asp and amitriptyline restored cognitive performance in the SNI mice showing cognitive deficit. This outcome is in line with studies demonstrating that increased d-Asp level in the hippocampus was able to enhance NMDA receptor-dependent LTP (Errico et al. 2008a, 2011a, b), the cellular mechanism at the base of learning and memory (Bliss and Collingridge 1993). The effect of amitriptyline in improving cognition has been already described and will be not discussed here (Orsetti et al. 2007; Hu et al. 2010). The 1 month d-Asp drinking solution normalized also the social affiliation and sociability, which were deeply disrupted in SNI mice. Asociality (i.e., withdrawal from social contact) has been demonstrated to be associated with NMDA receptor hypo-function (Coyle 2012; Errico et al. 2014) and down-regulation of d-Asp binding subunit of NMDA receptor (Errico et al. 2014). Thus the d-Asp drinking solution by activating NMDA receptors may have counteracted the development of NMDA hypo-function. In our study the affective behavior such as anxiety and depression were unaffected after the oral d-Asp treatment. A large body of evidence is now showing that the NMDA receptor blockade on GABAergic interneurons and the following dis-inhibition of glutamate outflow is the molecular mechanism possibly responsible of the rapid and potent antidepressant effect of ketamine (Paul and Skolnick 2003; Skolnick et al. 2009; Sanacora et al. 2012; Pilc et al. 2013; Duman 2014). The increased glutamate outflow may in turn stimulate synaptogenesis via mammalian target of rapamycin (mTOR) signaling and brain derived neurotrophic factor (BDNF) release (Duman 2014). Further investigations examining the role of d-Asp oral treatment on brain BDNF or mTOR signaling in hippocampus or prefrontal cortex, together with ultra-structural studies showing synaptogenesis, would be useful to elucidate the lack of anxiolytic/antidepressant action of d-Asp oral treatment. As expected, amitriptyline, which among other thing block NMDA receptor such as ketamine (Reynolds and Miller 1988; Cai and McCaslin 1992; McCaslin et al. 1992; Watanabe et al. 1993), was effective in ameliorating anxiety and depression-like behavior.
In SNI mice was detected an increased expression of Homer 1a, the inducible Homer isoform acting as dominant negative on Homer 1 constitutive forms and therefore disrupting the anatomical and functional network between glutamatergic receptors, PSD scaffolding proteins and intracellular effectors (de Bartolomeis et al. 2014). NR2B subunit of NMDA receptor was also increased in SNI group of mice. NR2B-containing receptors, compared with other NMDA receptor subtypes, appears to contribute preferentially to pathological processes linked to over-excitation of glutamate transmission. Shank1 and PSD-95, postsynaptic proteins involved in signal transduction and psychiatric disorders (Helyes et al. 2003) were not modified in SNI mice. The 1 month d-Asp drinking solution normalized the expression levels of Homer 1a and NR2B subunit of NMDA receptors. It is worthy of note that compounds able to antagonize NR2B subunit selectively are effective in relieving neuropathic pain sensory symptoms in line with the analgesic effect of d-Asp chronic treatment (Wu and Zhuo 2009). d-Asp treatment decreased Shank1 and PSD-95 protein levels whereas no changes were detected for Homer 1b expression in the SNI mice. Previous studies have indeed demonstrated that altered glutamate transmission and synaptic maladaptive changes in the postsynaptic density at this level may contribute to central sensitization associated with neuropathic pain and consequent neurological/psychiatric disorders (Wei et al. 2001; Neugebauer et al. 2009; Jernigan et al. 2011; Giordano et al. 2012; Nithianantharajah and Hannan 2013; Hung et al. 2014; Iasevoli et al. 2014; Guida et al. 2015). Accordingly, the effect of d-Asp in reducing NR2B subunit of the NMDA receptor, Homer 1a, Shank1 and PSD-95 expression may be interpreted as a correction of “hyperglutamatergic state” occurring in pathological neural conditions such as neuropathic pain (Herrero et al. 2000) suggesting that restoring of glutamate postsynaptic signaling, which is altered in SNI mice, may drive to a relief of sensorial, cognitive and social impairments as observed in the SNI group of mice. Chronic treatment with amitriptyline also changed the expression level of PSD proteins which occurred concurrently to its effectiveness in ameliorating neuropathic pain symptoms from sensorial to affective/cognitive ones. In this case, however, an increase of Homer 1a, Homer 1b, Shank1 and PSD-95 was observed. The observed discrepancy among the effect of amytriptyline and d-Asp treatment on PSD protein expression level is in line with the opposite action of the two drugs on NMDA receptor: antagonist or agonist activity, respectively. This result is consistent also with an agonist-induced down-regulation of NR2B subunit of NMDA receptor as it was found in the current study. While it is hard to explain the difference of efficacy of amitriptyline or d-Asp in the depression/anxiety behavioural test and on PSD protein levels this study suggests that targeting altered glutamate neurotransmission may ameliorate abnormal sensory, motor, cognitive and social behavior associated with neuropathic pain.
Conclusions
In conclusion 1 month exposure to d-Asp alleviates mechanical allodynia and cognitive impairments in SNI mice, whose pain reaction and learning behavior were proved to be highly compromised. d-Asp consumption did not alter the affective responses, as amitriptyline did, but was able to counteract the social isolation. These data highlights the perspective to targeting endogenous d-Asp/NMDA synthesis and metabolism leading to altered glutamate transmission for the treatment of chronic pain and its related cognitive and affective disorders.
References
al-Ghoul WM, Li Volsi G, Weinberg RJ, Rustioni A (1993) Glutamate immunocytochemistry in the dorsal horn after injury or stimulation of the sciatic nerve of rats. Brain Res Bull 30:453–459
Apkarian AV, Bushnell MC, Treede RD, Zubieta JK (2005) Human brain mechanisms of pain perception and regulation in health and disease. Eur J Pain 9:463–484
Benbouzid M, Choucair-Jaafar N, Yalcin I, Waltisperger E, Muller A, Freund-Mercier MJ, Barrot M (2008) Chronic, but not acute, tricyclic antidepressant treatment alleviates neuropathic allodynia after sciatic nerve cuffing in mice. Eur J Pain 12:1008–1017
Berrocoso E, Mico JA, Vitton O, Ladure P, Newman-Tancredi A, Depoortère R, Bardin L (2011) Evaluation of milnacipran, in comparison with amitriptyline, on cold and mechanical allodynia in a rat model of neuropathic pain. Eur J Pharmacol 655:46–51
Bliss TV, Collingridge GL (1993) A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361:31–39
Boccella S, Vacca V, Errico F, Marinelli S, Squillace M, Guida F, Di Maio A, Vitucci D, Palazzo E, de Novellis V, Maione S, Pavone F, Usiello A (2015) d-Aspartate modulates nociceptive-specific neuron activity and pain threshold in inflammatory and neuropathic pain condition in mice. Biomed Res Int 2015:905906
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cai Z, McCaslin PP (1992) Amitriptyline, desipramine, cyproheptadine and carbamazepine, in concentrations used therapeutically, reduce kainate- and N-methyl-d-aspartate-induced intracellular Ca2+ levels in neuronal culture. Eur J Pharmacol 219:53–57
Campbell S, MacQueen G (2006) An update on regional brain volume differences associated with mood disorders. Curr Opin Psychiatry 19:25–33
Coyle JT (2012) NMDA receptor and schizophrenia: a brief history. Schizophr Bull 38:920–926
Coyle JT, Tsai G, Goff D (2003) Converging evidence of NMDA receptor hypofunction in the pathophysiology of schizophrenia. Ann N Y Acad Sci 1003:318–327
D’Aniello A (2007) d-Aspartic acid: an endogenous amino acid with an important neuroendocrine role. Brain Res Rev 53:215–234
D’Aniello A, Di Fiore MM, Fisher GH, Milone A, Seleni A, D’Aniello S, Perna AF, Ingrosso D (2000a) Occurrence of d-aspartic acid and N-methyl-d-aspartic acid in rat neuroendocrine tissues and their role in the modulation of luteinizing hormone and growth hormone release. FASEB J 14:699–714
D’Aniello G, Tolino A, D’Aniello A, Errico F, Fisher GH, Di Fiore MM (2000b) The role of d-aspartic acid and N-methyl-d-aspartic acid in the regulation of prolactin release. Endocrinology 141:3862–3870
D’Aniello S, Somorjai I, Garcia-Fernandez J, Topo E, D’Aniello A (2011) d-Aspartic acid is a novel endogenous neurotransmitter. FASEB J 25:1014–1027
de Bartolomeis A, Latte G, Tomasetti C, Iasevoli F (2014) Glutamatergic postsynaptic density protein dysfunctions in synaptic plasticity and dendritic spines morphology: relevance to schizophrenia and other behavioral disorders pathophysiology, and implications for novel therapeutic approaches. Mol Neurobiol 49:484–511
Decosterd I, Woolf CJ (2000) Spared nerve injury: an animal model of persistent peripheral neuropathic pain. Pain 87:149–158
Deffois A, Fage D, Carter C (1996) Inhibition of synaptosomal veratridine-induced sodium influx by antidepressants and neuroleptics used in chronic pain. Neurosci Lett 220:117–120
Duman RS (2014) Pathophysiology of depression and innovative treatments: remodeling glutamatergic synaptic connections. Dialogues Clin Neurosci 16:11–27
Dunlop DS, Neidle A, McHale D, Dunlop DM, Lajtha A (1986) The presence of free d-aspartic acid in rodents and man. Biochem Biophys Res Commun 14:27–32
Eisenach JC, Gebhart GF (1995) Intrathecal amitriptyline acts as an N-methyl-d-aspartate receptor antagonist in the presence of inflammatory hyperalgesia in rats. Anesthesiology 83:1046–1054
Errico F, Nisticò R, Palma G, Federici M, Affuso A, Brilli E, Topo E, Centonze D, Bernardi G, Bozzi Y, D’Aniello A, Di Lauro R, Mercuri NB, Usiello A (2008a) Increased levels of d-aspartate in the hippocampus enhance LTP but do not facilitate cognitive flexibility. Mol Cell Neurosci 37:236–246
Errico F, Rossi S, Napolitano F, Catuogno V, Topo E, Fisone G, D’Aniello A, Centonze D, Usiello A (2008b) d-aspartate prevents corticostriatal long-term depression and attenuates schizophrenia-like symptoms induced by amphetamine and MK-801. J Neurosci 28:10404–10414
Errico F, Nisticò R, Napolitano F, Mazzola C, Astone D, Pisapia T, Giustizieri M, D’Aniello A, Mercuri NB, Usiello A (2011a) Increased d-aspartate brain content rescues hippocampal age-related synaptic plasticity deterioration of mice. Neurobiol Aging 32:2229–2243
Errico F, Nisticò R, Napolitano F, Oliva AB, Romano R, Barbieri F, Florio T, Russo C, Mercuri NB, Usiello A (2011b) Persistent increase of d-aspartate in d-aspartate oxidase mutant mice induces a precocious hippocampal age-dependent synaptic plasticity and spatial memory decay. Neurobiol Aging 32:2061–2074
Errico F, Nisticò R, Di Giorgio A, Squillace M, Vitucci D, Galbusera A, Piccinin S, Mango D, Fazio L, Middei S, Trizio S, Mercuri NB, Teule MA, Centonze D, Gozzi A, Blasi G, Bertolino A, Usiello A (2014) Free d-aspartate regulates neuronal dendritic morphology, synaptic plasticity, gray matter volume and brain activity in mammals. Transl Psychiatry 4:e417
Fagg GE, Matus A (1984) Selective association of N-methyl aspartate and quisqualate types of l-glutamate receptor with brain postsynaptic densities. Proc Natl Acad Sci USA 81:6876–6880
Fan X, Jin WY, Wang YT (2014) The NMDA receptor complex: a multifunctional machine at the glutamatergic synapse. Front Cell Neurosci 8:160
Franklin KBJ, Paxinos G (1997) The mouse brain in stereotaxic coordinates. Academic Press, San Diego
Furuchi T, Homma H (2005) Free d-aspartate in mammals. Biol Pharm Bull 28:1566–1570
Giordano C, Cristino L, Luongo L, Siniscalco D, Petrosino S, Piscitelli F, Marabese I, Gatta L, Rossi F, Imperatore R, Palazzo E, de Novellis V, Di Marzo V, Maione S (2012) TRPV1-dependent and -independent alterations in the limbic cortex of neuropathic mice: impact on glial caspases and pain perception. Cereb Cortex 22:2495–2518
Gottrup H, Kristensen AD, Bach FW, Jensen TS (2003) Aftersensations in experimental and clinical hypersensitivity. Pain 103:57–64
Guida F, Luongo L, Marmo F, Romano R, Iannotta M, Napolitano F, Belardo C, Marabese I, D’Aniello A, De Gregorio D, Rossi F, Piscitelli F, Lattanzi R, de Bartolomeis A, Usiello A, Di Marzo V, de Novellis V, Maione S (2015) Palmitoylethanolamide reduces pain-related behaviors and restores glutamatergic synapses homeostasis in the medial prefrontal cortex of neuropathic mice. Mol Brain 8:47
Hall H, Ogren SO (1981) Effects of antidepressant drugs on different receptors in the brain. Eur J Pharmacol 70:393–407
Hashimoto A, Kumashiro S, Nishikawa T, Oka T, Takahashi K, Mito T, Takashima S, Doi N, Mizutani Y, Yamazaki T et al (1993) Embryonic development and postnatal changes in free d-aspartate and d-serine in the human prefrontal cortex. J Neurochem 61:348–351
Hashimoto A, Oka T, Nishikawa T (1995) Anatomical distribution and postnatal changes in endogenous free d-aspartate and d-serine in rat brain and periphery. Eur J Neurosci 7:1657–1663
Helyes Z, Németh J, Thán M, Bölcskei K, Pintér E, Szolcsányi J (2003) Inhibitory effect of anandamide on resiniferatoxin-induced sensory neuropeptide release in vivo and neuropathic hyperalgesia in the rat. Life Sci 73:2345–2353
Herrero JF, Laird JM, López-García JA (2000) Wind-up of spinal cord neurons and pain sensation: much ado about something? Prog Neurobiol 61:169–203
Hu Y, Yang J, Hu Y, Wang Y, Li W (2010) Amitriptyline rather than lornoxicam ameliorates neuropathic pain-induced deficits in abilities of spatial learning and memory. Eur J Anaesthesiol 27:162–168
Huang AS, Lee DA, Blackshaw S (2008) d-Aspartate and d-aspartate oxidase show selective and developmentally dynamic localization in mouse retina. Exp Eye Res 86:704–709
Hudson LJ, Bevan S, McNair K, Gentry C, Fox A, Kuhn R, Winter J (2002) Metabotropic glutamate receptor 5 upregulation in A-fibers after spinal nerve injury: 2-methyl-6-(phenylethynyl)-pyridine (MPEP) reverses the induced thermal hyperalgesia. J Neurosci 22:2660–2668
Hung KL, Wang SJ, Wang YC, Chiang TR, Wang CC (2014) Upregulation of presynaptic proteins and protein kinases associated with enhanced glutamate release from axonal terminals (synaptosomes) of the medial prefrontal cortex in rats with neuropathic pain. Pain 155:377–387
Hutson PH, Sarna GS, Kantamaneni BD, Curzon G (1985) Monitoring the effect of a tryptophan load on brain indole metabolism in freely moving rats by simultaneous cerebrospinal fluid sampling and brain dialysis. J Neurochem 44:1266–1273
Iasevoli F, Tomasetti C, Buonaguro EF, de Bartolomeis A (2014) The glutamatergic aspects of schizophrenia molecular pathophysiology: role of the postsynaptic density, and implications for treatment. Curr Neuropharmacol 12:219–238
Ishii Y, Sumi T (1992) Amitriptyline inhibits striatal efflux of neurotransmitters via blockade of voltage-dependent Na+ channels. Eur J Pharmacol 221:377–380
Jernigan CS, Goswami DB, Austin MC, Iyo AH, Chandran A, Stockmeier CA, Karolewicz B (2011) The mTOR signaling pathway in the prefrontal cortex is compromised in major depressive disorder. Prog Neuropsychopharmacol Biol Psychiatry 35:1774–1779
Kim PM, Duan X, Huang AS, Liu CY, Ming GL, Song H, Snyder SH (2010) Aspartate racemase, generating neuronal d-aspartate, regulates adult neurogenesis. Proc Natl Acad Sci USA 107:3175–3179
Konarski JZ, McIntyre RS, Kennedy SH, Rafi-Tari S, Soczynska JK, Ketter TA (2008) Volumetric neuroimaging investigations in mood disorders: bipolar disorder versus major depressive disorder. Bipolar Disord 10:1–37
Koolschijn PC, van Haren NE, Lensvelt-Mulders GJ, Hulshoff Pol HE, Kahn RS (2009) Brain volume abnormalities in major depressive disorder: a meta-analysis of magnetic resonance imaging studies. Hum Brain Mapp 30:3719–3735
Krashia P, Ledonne A, Nobili A, Cordella A, Errico F, Usiello A, D’Amelio M, Mercuri NB, Guatteo E, Carunchio I (2015) Persistent elevation of d-aspartate enhances NMDA receptor-mediated responses in mouse substantia nigra pars compacta dopamine neurons. Neuropharmacology 103:69–78
Kullmann DM, Asztely F, Walker MC (2000) The role of mammalian ionotropic receptors in synaptic plasticity: LTP, LTD and epilepsy. Cell Mol Life Sci 57:1551–1561
Lavoie PA, Beauchamp G, Elie R (1990) Tricyclic antidepressants inhibit voltage-dependent calcium channels and Na(+)-Ca2+ exchange in rat brain cortex synaptosomes. Can J Physiol Pharmacol 68:1414–1418
Lee JA, Homma H, Tashiro K, Iwatsubo T, Imai K (1999) d-Aspartate localization in the rat pituitary gland and retina. Brain Res 838:193–199
McCaslin PP, Yu XZ, Ho IK, Smith TG (1992) Amitriptyline prevents N-methyl-d-aspartate (NMDA)-induced toxicity, does not prevent NMDA-induced elevations of extracellular glutamate, but augments kainate-induced elevations of glutamate. J Neurochem 59:401–405
Millecamps M, Centeno MV, Berra HH, Rudick CN, Lavarello S, Tkatch T, Apkarian AV (2007) D-cycloserine reduces neuropathic pain behavior through limbic NMDA-mediated circuitry. Pain 132:108–123
Monahan JB, Michel J (1987) Identification and characterization of an N-methyl-d-aspartate-specific l-[3H]glutamate recognition site in synaptic plasma membranes. J Neurochem 48:1699–1708
Neidle A, Dunlop DS (1990) Developmental changes in free d-aspartic acid in the chicken embryo and in the neonatal rat. Life Sci 46:1517–1522
Neugebauer V, Galhardo V, Maione S, Mackey SC (2009) Forebrain pain mechanisms. Brain Res Rev 60:226–242
Nithianantharajah J, Hannan AJ (2013) Dysregulation of synaptic proteins, dendritic spine abnormalities and pathological plasticity of synapses as experience-dependent mediators of cognitive and psychiatric symptoms in Huntington’s disease. Neuroscience 251:66–74
Orsetti M, Colella L, Dellarole A, Canonico PL, Ghi P (2007) Modification of spatial recognition memory and object discrimination after chronic administration of haloperidol, amitriptyline, sodium valproate or olanzapine in normal and anhedonic rats. Int J Neuropsychopharmacol 10:345–357
Ota N, Shi T, Sweedler JV (2012) d-Aspartate acts as a signaling molecule in nervous and neuroendocrine systems. Amino Acids 43:1873–1886
Pancrazio JJ, Kamatchi GL, Roscoe AK, Lynch C 3rd (1998) Inhibition of neuronal Na+ channels by antidepressant drugs. J Pharmacol Exp Ther 284:208–214
Paul IA, Skolnick P (2003) Glutamate and depression: clinical and preclinical studies. Ann N Y Acad Sci 1003:250–272
Pilc A, Wierońska JM, Skolnick P (2013) Glutamate-based antidepressants: preclinical psychopharmacology. Biol Psychiatry 73:1125–1132
Reynolds IJ, Miller RJ (1988) Tricyclic antidepressants block N-methyl-d-aspartate receptors: similarities to the action of zinc. Br J Pharmacol 95:95–102
Sakai K, Homma H, Lee JA, Fukushima T, Santa T, Tashiro K, Iwatsubo T, Imai K (1998) Emergence of d-aspartic acid in the differentiating neurons of the rat central nervous system. Brain Res 808:65–71
Sanacora G, Treccani G, Popoli M (2012) Towards a glutamate hypothesis of depression: an emerging frontier of neuropsychopharmacology for mood disorders. Neuropharmacology 62(1):63–77
Shibata K, Sugaya N, Ono W, Abe K, Takahashi S, Kera Y (2011) Determination of d-aspartate N-methyltransferase activity in the starfish by direct analysis of N-methyl-d-aspartate with high-performance liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 879:3229–3234
Shimizu M, Nishida A, Yamawaki S (1992) Antidepressants inhibit spontaneous oscillations of intracellular Ca2+ concentration in rat cortical cultured neurons. Neurosci Lett 146:101–104
Sindrup SH, Otto M, Finnerup NB, Jensen TS (2005) Antidepressants in the treatment of neuropathic pain. Basic Clin Pharmacol Toxicol 96:399–409
Skolnick P, Popik P, Trullas R (2009) Glutamate-based antidepressants: 20 years on. Trends Pharmacol Sci 30:563–569
Spinelli P, Brown ER, Ferrandino G, Branno M, Montarolo PG, D’Aniello E, Rastogi RK, D’Aniello B, Baccari GC, Fisher G, D’Aniello A (2006) d-Aspartic acid in the nervous system of Aplysia limacina: possible role in neurotransmission. J Cell Physiol 206:672–681
Swanson CJ, Bures M, Johnson MP, Linden AM, Monn JA, Schoepp DD (2005) Metabotropic glutamate receptors as novel targets for anxiety and stress disorders. Nat Rev Drug Discov 4:131–144
Watanabe Y, Saito H, Abe K (1993) Tricyclic antidepressants block NMDA receptor-mediated synaptic responses and induction of long-term potentiation in rat hippocampal slices. Neuropharmacol 32:479–486
Wei H, Panula P, Pertovaara A (2001) Modulation of pain by [1DMe]NPYF, a stable analogue of neuropeptide FF, in neuropathic rats. Brain Res 900:234–243
Wolosker H, D’Aniello A, Snyder SH (2000) d-Aspartate disposition in neuronal and endocrine tissues: ontogeny, biosynthesis and release. Neuroscience 100:183–189
Woolf CJ, Salter MW (2000) Neuronal plasticity: increasing the gain in pain. Science 288:1765–1769
Wu LJ, Zhuo M (2009) Targeting the NMDA receptor subunit NR2B for the treatment of neuropathic pain. Neurotherapeutics 6(4):693–702
Acknowledgments
This work was financially supported by the Ministero Istruzione, Università e Ricerca, Project n° PRIN 2012WBSSY4_001.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors who took part in this study declare that they have nothing to disclose regarding competing interests or funding with respect to this manuscript.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed (Animal Ethics Committee of the Second University of Naples in compliance with the IASP and European Community (E.C. L358/1 18/12/86). All procedures were in accordance with the Animal Ethics Committee of the Second University of Naples. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Handling Editor: N. Singewald.
Rights and permissions
About this article
Cite this article
Palazzo, E., Luongo, L., Guida, F. et al. d-Aspartate drinking solution alleviates pain and cognitive impairment in neuropathic mice. Amino Acids 48, 1553–1567 (2016). https://doi.org/10.1007/s00726-016-2205-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-016-2205-4