Summary
Background
Optic disc drusen (ODD) are calcified deposits potentially caused by disturbances in axonal metabolism. The clinical course and visual impairment of ODD is usually mild; however, significant ocular morbidity may occur, such as visual field defects and retinal haemorrhages. Optic disc drusen may pose a diagnostic dilemma and differentiating these from other entities that can lead to similar compressive axonal distress is imperative. We present a novel technique for three-dimensional (3D) characterisation of superficial ODD based on 3D volume rendering of optical coherence tomography angiography (3DOCTA) scans.
Material and methods
Optical coherence tomography (Cirrus™ HD-OCT Model 5000 with AngioPlex, Carl Zeiss Meditec, Inc., Dublin, USA) scans were obtained from the optic nerve head of a healthy 22-year-old female. Consequently, 3D structural OCT data and OCTA were analysed, enabling ODD segmentation and spatial characterization.
Results
Volumetric analysis of superficial ODD showed a maximal drusen horizontal diameter of 223 μm, maximal vertical diameter of 268 μm, surface area of 6617 μm2 and volume measurement of 12,875 μm3. The drusen were characterised by a connected network of multiple drusen islands instead of forming a dense mass. Multiple vascular channels with perforating vessels were found across the drusen.
Conclusion
Three-dimensional volume rendering of OCTA scans provided new insight on the spatio-anatomical features of superficial ODD. The new features herein described, namely multilobulated drusen islands and intradrusen channels, may directly contribute to the pathogenic events leading to transient non-embolic visual loss and small vessel occlusion secondary to ODD.
Zusammenfassung
Hintergrund
Drusen des Sehnerven (Drusenpapille, „optic disc drusen“) stellen sich als verkalkte Ablagerungen dar, die möglicherweise durch Störungen im axonalen Stoffwechsel verursacht werden. Obwohl diese Drusen meist einen milden klinischen Verlauf zeigen und Sehbehinderungen selten sind, können sie mit Gesichtsfelddefekten und Netzhautblutungen assoziiert sein. Drusenpapillen können diagnostische Schwierigkeiten verursachen im Vergleich mit anderen Pathologien, die mit kompressiven axonalen Störungen verbunden sind, wie Tumoren des Sehnerven. Daher werden neue bildgebende Modalitäten wie die dreidimensionale Volumendarstellung der optischen Kohärenztomographie-Angiographie (3DOCTA) für eine bessere Visualisierung und das Verständnis von Drusenpapillen angewendet.
Material und Methode
Bei einer 22-jährigen gesunden Frau wurden oberflächliche Drusenpapillen mittels optischer Kohärenztomographie-Angiographie (OCTA, Zeiss Cirrus™ HD-OCT Model 5000 mit AngioPlex) abgebildet. Die dreidimensionale Struktur-OCT und die OCTA wurden überlagert, die Drusen segmentiert und vermessen.
Ergebnisse
Die oberflächlichen Drusenpapillen zeigten einen maximalen horizontalen Durchmesser von 223 µm, einen maximalen vertikalen Durchmesser von 268 µm, die Oberfläche betrug 6617 µm2 und die Volumenmessung zeigte 12.875 µm3. Die Drusen zeigten keine ganz dichte Masse, sondern stellten sich als ein verbundenes Netzwerk von Druseninseln dar. Mehrere offene Kanäle wurden innerhalb der Drusen gefunden, durch welche Gefäße eindringen konnten.
Schlussfolgerung
Die 3DOCTA bei Drusenpapillen zeigte neue morphologische Merkmale, wie multilobulierte Druseninseln und Kanäle innerhalb der Drusen, die für die Netzhautperfusion im Fall einer Gefäßokklusion kritisch sein können.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Optic disc drusen (ODD) are acellular deposits composed of mucoproteins, mucopolysaccharides and iron which undergo progressive calcification [1]. The size and number of ODD vary considerably and they can be found at all prelaminar levels. Smaller ODD tend to be located more proximal to the lamina cribrosa [2]. The clinical course of ODD is usually benign, although they can be associated with visual field defects [3,4,5,6], retinal haemorrhages [7,8,9], optic nerve fibre compression and partial optic nerve atrophy, and juxtapapillary retinal scarring [2]. Diagnosis of ODD is usually based on funduscopic appearance, ultrasonography and fundus autofluorescence. Visual field testing or spectral domain optical coherence tomography (SDOCT) [10,11,12,13] may also be of utility. In SDOCT, ophthalmoscopically visible ODD appear as multiple lumps mostly inside the disc with highly reflective borders and internal spaces. Buried ODD appear as a C-shaped mass outside the disc with relatively less distinct borders [12].
We present a novel technique characterised by three-dimensional volume-rendering of OCTA scans (3DOCTA) of superficial ODD. This approach enhances the diagnostic abilities of non-invasive angiography whilst giving new insight on the spatio-anatomical features and how these integrate with angiographic findings of superficial ODD.
Methods
Our technique was applied to OCT scans obtained in a 22-year-old Caucasian female. Informed consent was obtained from her for being included in the study. On ophthalmologic examination, best corrected visual acuity was 20/15 and 20/15 in the right and left eyes, respectively. There was no relative afferent pupillary defect. Intraocular pressure was within normal range and colour vision was normal (Ishihara test plates). The anterior chambers and vitreous were clear in both eyes. Fundus examination revealed bilateral superficial ODD with corresponding hyperautofluorescence on autofluorescence imaging.
Autofluorescence and OCT imaging were performed using the Heidelberg Spectralis (Heidelberg Engineering, Heidelberg, Germany). The following OCT parameters were used: 15 ° scan angle with a scan area 4.4 mm × 4.4 mm × 1.9 mm, Enhanced Depth Imaging on, 73 B-scans with an interslice distance of 61 μm. Imaging with OCT was averaged for nine scans using the automatic averaging and tracking feature.
The OCTA measurements (3 mm × 3 mm scan area, 245 × 245 pixel) were performed with the Zeiss Cirrus™ HD-OCT Model 5000 with AngioPlex (Review software 9.0.0.281, Carl Zeiss Meditec, Jena, Germany). All en-face OCTA cutting planes were exported into a 3DOCTA stack. In addition, all structural OCT en-face images were exported into a 3D structural OCT stack (3DOCT). Both image stacks comprised of structural OCT volume (3DOCT) and flow information volume (3DOCTA) were aligned and overlayed. The spaces occupied by ODD were manually segmented by thresholding of pixel intensity. A threshold from 0 to 70 grey-scale units (scale 0 to 255) enabled delineation and separation of the individual lesions. Image artefact from surrounding vessel shadowing was excluded. After segmentation, volume and surface measurements were performed. Finally, all compartments were integrated into one combined volume of structural OCT, OCTA and segmented drusen to study the topographic relationships between vessels and drusen in 3D.
Results
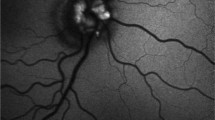
The clinical diagnosis of ODD was confirmed on autofluorescence imaging. Two large drusen were identified in the nasal superior quadrant of the left optic nerve head (ONH; Fig. 1).
Fundus imaging of optic disc drusen (ODD) in a 22-year-old healthy female. a Scanning laser ophthalmoscopy scan of the right optic nerve shows an elevated optic disc with no features of optic disc oedema. Multiple pinpoint-like lesions are seen in the nasal superior quadrant. b Corresponding autofluorescence scan of the right eye showing hyperautofluorescence and confirming the diagnosis of ODD
Segmentation of the drusen revealed a maximal drusen horizontal diameter of 223 μm, maximal vertical diameter of 268 μm, surface area of 6617 μm2 and volume measurement of 12,875 μm3. A close relationship of the ODD with the surrounding vessels was identified on 3DOCTA (Fig. 2), which in addition revealed the presence of several notches on the surface of the ODD (“drusen indentations”). In the structural OCT, superficial drusen had a round appearance and appeared hyperreflective with a granular aspect.
Image processing for three-dimensional optical coherence tomography (OCT) volume rendering of OCT angiography (3DOCTA). a En-face view of optic disc drusen (ODD; arrow) which demonstrates a hyperreflective area between two hyporeflective vessels (arrow head). b Corresponding en-face OCTA scan of the optic disc depicted in (a) at an identical level. In the area of superficial ODD, an ovaloid area of poor signal is seen (arrow). Minor signals are detected inside the ovoid space. The two hyporeflective vessels around the drusen appear to be pushed outwards. Larger optic disc vessels are separated from smaller vessels by their calibre (arrow heads). c 3DOCTA of the same eye shows an elevated vascular network which is a typical feature of ODD. d En-face 3D volume rendering shows a signal void area of drusen with visualisation of the white image background. Clearly, the adjacent large vessels (arrow head) are dislocated by the drusen. e Drusen are not depicted in the 3D volume rendering of structural OCT. The white spot reflects condensation of posterior vitreous. f Combined volume rendering of structural and OCTA of the same optic disc
On the OCTA, the vessel signal density, though reduced, was not entirely devoid of signal. A fainter signal stemming from small vessels could be detected in the hyporeflective areas occupied by the ODD (Fig. 2b). The drusen appeared as islands encroaching on these smaller vessels, forming interconnecting vascular channels (“drusen channels”).
Discussion
The use of 3DOCTA enabled the identification of new morphological features and interactions in ODD. This new technique facilitated the diagnosis, visualisation and volumetric characterisation of ODD. In contrast to other imaging methods, namely two-dimensional standard OCT [11, 14, 15] and autofluorescence imaging [13, 16], a much more detailed drusen microanatomy with the presence of multilobulated, interconnected drusen lobes was found on combined three dimensional structural OCT and 3DOCTA. Several drusen were organised into a “drusen cluster”, whereas others were singular. A new metric approach was provided to measure not only the vertical and horizontal drusen diameter, but to define single drusen surface and volume as well. Potentially, this technique can guide to a specific “drusen cluster matrix”. This could be helpful to document a drusen’s state of equilibrium or express characteristics for drusen dynamics, e. g. fusing of drusen into a cluster. Distinct parameters can be used to monitor the interconnections with other clusters and to classify the clusters and lobules, which in turn could be used to document growth and assess mechanical properties. The ability to visualise and perform volumetric and surface measurements in ODD serves as a potential diagnostic and follow-up tool. It may also provide new insight into the pathogenic mechanisms of ODD-related ischaemic events such as retinal occlusion, choroidal neovascularisation [17,18,19], non-arteritic ischaemic optic neuropathy and peripapillary subretinal haemorrhages [7, 20,21,22].
Our method revealed another interesting finding: ODD are not a compact circumscribed mass but show several openings (“drusen channels”), through which persisting vessels penetrate (Fig. 3).
Combined volume rendering optical coherence tomography (OCTA) and segmented optic disc drusen (ODD). a Two drusen are visible on the surface of optic disc OCTA (arrows). Smaller, persisting vessels are seen within the ODD, corresponding to the granular OCTA signals in Fig. 2b. Larger vessels wind around the ODD (arrow head). b Same volumetric analysis as in (a) in a more posterior cutting plane demonstrates that the ODD are not a uniform corpuscle, but are composed of at least four islands of interconnected extravascular material. c After segmentation, the ODD are shown to consist of several smaller lobules partly connected by fine bridges, contrary to their appearance on autofluorescence as a dense compact mass (Fig. 1b). d Magnification of the upper left ODD in (c). 3D volume rendering enabled the identification of small openings from within the ODD through which persisting vessels emerge (“drusen channels”, arrow head). The surface of the ODD is irregular and several notches are visible (“drusen indentations”, arrows)
Calcification of ODD and aggregation formation may potentially contribute to obstruction of the capillary network with consecutive anatomical and functional damage of the retinal nerve fibre layer (RNFL) [23]. It may be a seemingly simple question of what governs and adjusts the geometry of these drusen channels. However, it may be a challenge to develop new morphodynamic models that should be addressed by future research. Implementation of this technique in clinical practice will depend on further temporal analysis of ODD.
In this patient, the ODD showed a very close relationship to the ONH vessels. Arguably, depending on the nature and consistency of the ODD material, the elasticity of the surrounding vessels could be restricted or endovascular changes such as inflammatory effects originated on the drusen surface could be triggered [2], which could lead to a transient or permanent restriction of the vessel perfusion.
There are several limitations to our study. Only superficial drusen were examined and the number of drusen was too low for us to extrapolate our findings to all superficial ODD. The segmentation of the drusen was performed manually with threshold filtering, which may lead to artefacts depending on the levels of the threshold and the speckle noise signal. Furthermore, a generally accepted threshold level has not yet been defined. As OCTA is based on the motion contrast to show blood flow, the visualisation of the vessel signal may be limited because of artefacts due to OCT image acquisition, eye motion or image processing [24].
This study describes the first use of a new imaging technique, 3DOCTA, applied to superficial ODD. This technique enabled the characterisation of new morphological features of superficial ODD which, to the best of our knowledge, had never been reported. This technique may provide new insight on the pathogenic mechanisms associated with symptomatic ODD.
References
Tso MO. Pathology and pathogenesis of drusen of the optic nervehead. Ophthalmology. 1981;88(10):1066–80.
Friedman AH, Henkind P, Gartner S. Drusen of the optic disc. A histopathological study. Trans Ophthalmol Soc U K. 1975;95(1):4–9.
Wilkins JM, Pomeranz HD. Visual manifestations of visible and buried optic disc drusen. J Neuroophthalmol. 2004;24(2):125–9.
Duncan JE, Freedman SF, El-Dairi MA. The incidence of neovascular membranes and visual field defects from optic nerve head drusen in children. J AAPOS. 2016;20(1):44–8.
Calvo-Gonzalez C, et al. Optic nerve drusen and deep visual fields defects. Arch Soc Esp Oftalmol. 2006;81(5):269–73.
Moody TA, et al. Sudden visual field constriction associated with optic disc drusen. J Clin Neuroophthalmol. 1993;13(1):8–13, discussion 14.
Lee KM, Hwang JM, Woo SJ. Hemorrhagic complications of optic nerve head drusen on spectral domain optical coherence tomography. Retina (Philadelphia, Pa). 2014;34(6):1142–8.
Romero J, Sowka J, Shechtman D. Hemorrhagic complications of optic disc drusen and available treatment options. Optometry. 2008;79(9):496–500.
Kazimierz Rubinstein KM, Ali M. Retinal complications of optic disc drusen. Br J Ophthalmol. 1982;66(2):83–95.
Chiang J, et al. The usefulness of multimodal imaging for differentiating pseudopapilloedema and true swelling of the optic nerve head: a review and case series. Clin Exp Optom. 2015;98(1):12–24.
Lee KM, Woo SJ, Hwang JM. Differentiation of optic nerve head drusen and optic disc edema with spectral-domain optical coherence tomography. Ophthalmology. 2011;118(5):971–7.
Lee KM, Woo SJ, Hwang JM. Morphologic characteristics of optic nerve head drusen on spectral-domain optical coherence tomography. Am J Ophthalmol. 2013;155(6):1139–1147.e1.
Chang MY, Pineles SL. Optic disk drusen in children. Surv Ophthalmol. 2016;61(6):745–58.
Silverman AL, et al. Assessment of optic nerve head drusen using enhanced depth imaging and swept source optical coherence tomography. J Neuroophthalmol. 2014;34(2):198–205.
Slotnick S, Sherman J. Buried disc drusen have hypo-reflective appearance on SD-OCT. Optom Vis Sci. 2012;89(5):E704–E8.
Sato T, Mrejen S, Spaide RF. Multimodal imaging of optic disc drusen. Am J Ophthalmol. 2013;156(2):275–282.e1.
Chaudhry NA, et al. Photodynamic therapy for choroidal neovascular membrane secondary to optic nerve drusen. Ophthalmic Surg Lasers Imaging. 2005;36(1):70–2.
Delas B, et al. Bilateral choroidal neovascularization associated with optic nerve head drusen treated by antivascular endothelial growth factor therapy. Clin Ophthalmol. 2012;6:225–30.
Silva R, et al. Bilateral CNV associated with optic nerve drusen treated with photodynamic therapy with verteporfin. Eur J Ophthalmol. 2004;14(5):434–7.
Sanders TE, Gay AJ, Newman M. Hemorrhagic complications of drusen of the optic disk. Am J Ophthalmol. 1971;71(1 Pt 2):204–17.
Sibony P, et al. Asymptomatic peripapillary subretinal hemorrhage: a study of 10 cases. J Neuroophthalmol. 2008;28(2):114–9.
Wise GN, Henkind P, Alterman M. Optic disc drusen and subretinal hemorrhage. Trans Am Acad Ophthalmol Otolaryngol. 1974;78(2):212–9.
Gili P, et al. Anatomical and functional impairment of the nerve fiber layer in patients with optic nerve head drusen. Graefes Arch Clin Exp Ophthalmol. 2013;251(10):2421–8.
Spaide RF, Fujimoto JG, Waheed NK. Image artifacts in optical coherence tomography angiography. Retina (Philadelphia, Pa). 2015;35(11):2163–80.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
P.M. Maloca is a consultant of Zeiss. Catherine Egan and Adnan Tufail were supported by the National Institute for Health Research (NIHR) Biomedical Research Centre based at Moorfields Eye Hospital NHS Foundation Trust and UCL Institute of Ophthalmology. A. Tufail, C. Egan, S. Zweifel, P.W. Hasler, A. Petzold and J. E. Ramos de Carvalho declare that they have no competing interests. The views expressed are those of the authors and not necessarily those of the NEI, NHS, the NIHR, the Department of Health or Zeiss.
Ethical standards
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 (5).
Rights and permissions
About this article
Cite this article
Maloca, P.M., Tufail, A., Egan, C. et al. Volume rendering of superficial optic disc drusen. Spektrum Augenheilkd. 31, 288–293 (2017). https://doi.org/10.1007/s00717-017-0359-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00717-017-0359-4
Keywords
- Three-dimensional volume rendering
- Optical coherence tomography angiography
- Optic disc drusen
- Retinal vessels
- Perfusion