Abstract
Actigraphy is a non-invasive method of monitoring circadian rhythms and motor activity. We systematically reviewed extant evidence until September 2018 pertaining to actigraphy use in schizophrenia, its clinical/biological correlates and posit future research directions. Within 38 included studies involving 2700 subjects, patients with schizophrenia generally have lower motor activity levels, poorer sleep quality and efficiency, increased sleep fragmentation and duration compared with healthy controls. Lowered motor activity and longer sleep duration in patients were associated with greater severity of negative symptoms. Less structured motor activity and decreased sleep quality were associated with greater severity of positive symptoms, worse cognitive functioning involving attention and processing speed, illness chronicity, higher antipsychotic dose, and poorer quality of life. Correlations of actigraphic measures with biological factors are sparse with inconclusive results. Future studies with larger sample sets may adopt a multimodal, longitudinal approach which examines both motor and sleep activity, triangulates clinical, actigraphic and biological measures to clarify their inter-relationships and inform risk prediction of illness onset, course, and treatment response over time.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Schizophrenia is a major psychiatric disorder which affects not only the thoughts and cognition, but also the emotions, behaviour, daily functioning, and quality of life of those afflicted (Costa et al. 2018; García et al. 2018; Strassnig et al. 2018). Sleep disturbance is a common symptom associated with the disorder (Chouinard et al. 2004) and can be a significant predisposing, precipitating or perpetuating factor affecting the non-remission or relapse of the illness (Afonso et al. 2014; Reeve et al. 2015; Van Kammen et al. 1986). In addition, positive and negative symptoms experienced in schizophrenia have been associated with motor and sleep activity in schizophrenia (Baandrup and Jennum 2015; Kurebayashi and Otaki 2017; Mulligan et al. 2016; Shin et al. 2016). Hence, there has been continuous interest in better evaluating sleep patterns and motor activity of patients as clinical markers of treatment outcomes and illness course over time (Birkhofer et al. 2013; Walther et al. 2015a, b).
Actigraphy is a non-invasive method of monitoring human rest and activity cycles including circadian rhythms and motor activity trends (Teicher 1995). It can measure a range of sleep-related parameters (for example, total sleep duration, sleep onset latency, wake after sleep onset, sleep efficiency, fragmentation) as well as motor activity variables (for example, activity counts, duration of activity, periods of inactivity, movement index). This facilitates assessment of these different parameters and their relationships with other clinical factors such as phenomenology, personal functioning, treatment response, illness trajectory and even biobehavioural markers such as serum and imaging parameters (Docx et al. 2017; Kluge et al. 2018). While most actigraphy monitors are watch-like devices worn on the wrist, some are worn as patches or electrodes on the chest, hip or limbs (Brown et al. 1990).
Since the accelerated use of clinical actigraphy in the 1990s (Brown et al. 1990; Sadeh et al. 1991), its application has been extended to neuropsychiatric disorders including affective disorders, attention deficit hyperactivity disorder (De Crescenzo et al. 2016; Scott et al. 2017; Teicher et al. 1997) and dementia (Khan et al. 2018). Initially, actigraphy has been relatively less applied to the examination of activity patterns in psychotic disorders, as seen in an earlier systematic review of 14 studies (Tahmasian et al. 2013). Since then, there has been increasing efforts to assess these actigraphic measures in the context of schizophrenia, and combining them with other biological and clinical parameters, including treatment.
In view of the clinical importance of better evaluating activity patterns and biobehavioural correlates in schizophrenia, as well as recent advances in this field since the last review of published studies until 2011 (Tahmasian et al. 2013), we conducted a systematic review to synthesise the extant evidence pertaining to the utility of actigraphy in schizophrenia and their clinical and biological correlates. In addition, we posited current limitations and future research implications and directions.
Methods
Literature search
We searched the National Centre of Biotechnology Information (NCBI), Pubmed/Medline and Cochrane databases for empirical studies related to the use of actigraphy in schizophrenia subjects, reported until September 2018. Potentially useful reports were screened as abstracts based on the inclusion criteria listed below. We reviewed promising studies as full reports and screened their bibliographies for additional references. Keywords for the literature search included “schizophrenia”, “psychotic”, “actigraphy”, “actimetry”, “activity sensors”, either alone or as a combination of terms.
Inclusion/exclusion criteria
Reports were selected for inclusion if: (a) they involved empirical studies adopting actigraphy assessment and involved subjects diagnosed with schizophrenia by standard international criteria such as DSM or ICD; and (b) were in English.
Data extraction
For each individual study, we extracted variables including the number and type of subjects, socio-demographic characteristics, methods of actigraphy evaluation, other clinical or biological measures employed and salient findings.
Data synthesis
The preceding data were organised in digitalised spreadsheets and then summarised in tables to guide preparation of critical assessments included in this study as well as independent consideration by readers. We considered essential findings with respect to assessment of motor and/or sleep activity patterns using actigraphy and clinical and biobehavioural correlates in human subjects diagnosed with schizophrenia.
Results
Retrieved studies
A total of 65 studies were identified in the search of the databases, of which 16 were excluded as they did not meet inclusion criteria. Out of the remaining 49 full-text articles that were eventually assessed for eligibility, 11 were excluded due to reasons as follows: one was an earlier systematic review (Tahmasian et al. 2011), two were case studies (Haug et al. 2000; Wulff et al. 2006), seven did not include relevant patients (Cosgrave et al. 2018; Gonçalves et al. 2016; Kiang et al. 2003; Lunsford-Avery et al. 2015, 2017; Mittal et al. 2013; Reeve et al. 2017), and one did not perform subgroup analyses of schizophrenia patients from among patients with diagnoses of other mental disorders (Baandrup et al. 2016). This resulted in an overall total of 38 empirical studies being included for our synthesis. Figure 1 displays the PRISMA flowchart detailing the selection of relevant publications for inclusion in this review.
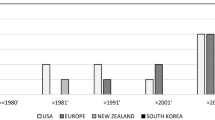
The summaries of main findings of the included studies are found in Table 1. Of these included studies, the majority (31 out of 38 studies) were conducted in Europe and USA. Overall, the total number of subjects was 2700, out of which 1803 (66.7%) were patients with schizophrenia, 341 (12.6%) were patients with other psychiatric disorders and 556 (20.6%) were healthy controls. The number of subjects in each study ranged from 11 to 199, with 52.6% (N = 20) of the included studies having 50 subjects and below. With the exclusion of two studies which did not provide gender data, 53.8% (N = 1452) of the overall subjects were male. The overall mean age of the subjects was 38.4 years, ranging between 22.5 and 58.3 years. Of the included studies, 24 (63.2%) assessed motor activity, 12 (31.6%) assessed sleep and two (5.3%) assessed both.
Actigraphy in relation to motor activity and sleep parameters
In terms of motor activity, studies using actigraphic motor parameters have generally observed that patients with schizophrenia have a lower total motor activity level compared to healthy controls (Bracht et al. 2012; Fasmer et al. 2016) and patients with depression (Berle et al. 2010). In a study performed by Berle et al. (2010) comparing the motor activity of 23 schizophrenia patients, 23 major depressive patients and 32 healthy controls, the schizophrenia group was found to have higher inter-daily stability and lower intra-daily variability than controls, especially in patients treated with clozapine. This suggests that schizophrenia patients have a more structured and monotonous motor behavioural pattern.
In terms of sleep parameters, patients with schizophrenia tended to have poorer sleep quality and efficiency as well as increased sleep fragmentation and sleep duration compared with healthy controls (Hofstetter et al. 2005; Mulligan et al. 2016; Robillard et al. 2015). A study performed by Robillard et al. (2015) compared measures of sleep–wake and activity–rest patterns among five groups of participants, namely healthy controls and patients with anxiety disorders, unipolar depression, bipolar disorder and psychotic disorders, using actigraphy and sleep diaries. Patients with psychotic disorders were found to have more prolonged sleep and irregular circadian rhythms compared to healthy controls, as well as the most unstable sleep schedules amongst all groups (Robillard et al. 2015).
Afonso et al. (2014) compared the sleep patterns of 34 schizophrenia patients with that of 34 healthy controls. An actimetry sensor was worn for 7 consecutive days while participants carried out their usual activities. Bedtime, wake time, night awakenings and day naps were recorded in a diary to aid researchers in distinguishing sleep from sedentary activities on the sensor-recorded patterns. They found that schizophrenia patients had higher sleep latency, more night awakenings and poorer sleep efficiency. In addition, three patients were found to have advanced sleep-phase syndrome and another three had irregular sleep–wake rhythms.
Actigraphy in relation to clinical correlates (psychopathology, cognition, clinical course, functioning, treatment)
In terms of psychopathology, lower motor activity and longer total sleep time have been associated with greater severity of negative symptoms (Docx et al. 2012, 2013, 2017; Shin et al. 2016; Walther et al. 2009b, 2014; Wichniak et al. 2011) including apathy (Kluge et al. 2018) and avolition (Docx et al. 2013) (see Fig. 2). Conversely, increased (Shin et al. 2016) but less structured motor activity (Walther et al. 2014), and decreased sleep quality (Afonso et al. 2011a, 2014) were associated with greater severity of positive symptoms (see Fig. 3). Walther et al. (2014) used actigraphy to measure mean activity level in counts/min and the Positive and Negative Syndrome Scale (PANSS) to assess symptom severity in 100 subjects with schizophrenia. They analysed the time series of movement counts over 60 min to establish if the amount of movement at one time point would be associated with that of subsequent time points. Reduced number of lags, indicating less structured movement patterns, was found to be associated with higher positive syndrome scores, while higher negative syndrome scores was associated with lower mean motor activity.
Afonso et al. (2014) divided 23 schizophrenia patients into predominantly positive and negative groups and along with actigraphy, used the Pittsburgh Sleep Quality Index to assess sleep patterns. They found that 11 out of the 23 patients studied had irregular sleep–wake cycles with daytime napping and night-time fragmentation. In addition, patients with predominantly positive symptoms had more disrupted sleep–wake patterns compared to the predominant negative symptom group. Poor sleep quality is also predictive of positive psychotic symptoms the following day, such as delusions and hallucinations (Mulligan et al. 2016). Regarding illness subtypes, it has been suggested that lower motor activity differentiated patients with schizophrenia, especially the catatonic subtype (Walther et al. 2009b), from other psychotic spectrum conditions such as cycloid psychosis (Walther et al. 2009a).
Furthermore, decreased motor activity and sleep quality were associated with poorer cognitive functioning involving attention and processing speed (Chen et al. 2016), reduced employment of positive reappraisal (Hofstetter et al. 2005) with implication of frontal lobe function (Bromundt et al. 2011). Chen et al. (2016) examined attention and concentration, and used the Grooved Pegboard Test to test for manual dexterity, upper-limb motor speed, hand–eye coordination and processing speed, in order to compare the cognitive functioning of 199 schizophrenia patients with 60 healthy controls. They found that the schizophrenia group was less engaged in light and moderate–vigorous activity and had poorer attention, concentration and processing speed than the control group. Participating in more light physical activity or moderate–vigorous activity was associated with better cognitive performance among patients with schizophrenia, with the influence of light activity being stronger than that of moderate–vigorous activity. Patients with schizophrenia who spent more time doing light activity had better performance on cognitive measures. Bromundt et al. (2011) evaluated frontal lobe functioning using Trail Making Test, Stroop and Supermarket Item Tests and observed that normal rest–activity circadian rhythm cycles correlated with significantly better cognitive functioning involving the frontal lobe (see Fig. 4).
In terms of illness trajectory, there is some evidence that lower motor activity is associated with chronicity of schizophrenia (Walther et al. 2015b) which needs validation in other samples. Actigraphy has shown potential in the precise detection of Neurological Soft Signs (NSS), known minor neurological signs more prevalent in individuals with schizophrenia than in a healthy population (Heinrichs and Buchanan 1988). This was observed in a study by Le Seac’h et al. (2012) using actigraphy paired with in-built inertial sensors capable of accurately calculating fine motor movements. They found that actigraphy detection of specific fine movements while performing neurological tasks assessing balance, alternative movements and extrapyramidal rigidity was positively correlated with the presence of NSS on clinical examination. Given that studies have demonstrated the increasing potential of NSS as both markers of early stages of schizophrenia and prognosticators of disease and treatment outcomes (Chan et al. 2016; Tamagni et al. 2013), the ability to objectively quantify and precisely detect them via actigraphy would be clinically valuable.
In terms of treatment and functioning, data are sparse. Decreased motor activity has been associated with higher antipsychotic dose, especially in early onset cases (Walther et al. 2015b), while increased motor activity has been associated with greater neuroleptic-induced extrapyramidal side effects (Poyurovsky et al. 2000). Poor sleep quality has been associated with poorer quality of life for patients with schizophrenia (Afonso et al. 2014) and working has been correlated with better sleep and circadian rhythms (Martin et al. 2005).
Actigraphy and biological and neuroimaging correlates
Based on extant data, poor sleep quality has been associated with various biological factors such as raised inflammatory marker counts like neutrophil concentration (Fang et al. 2016) and decreased melatonin (Afonso et al. 2011b). Decreased motor activity has been associated with reduced cerebral perfusion of cortical (frontal, temporal) and subcortical (thalamus, hippocampal) brain regions (Walther et al. 2011a), decreased frontal–striatal activation on functional magnetic resonance imaging and lower anterior cingulate volume (Farrow et al. 2005). Shamir et al. (2000) demonstrated that patients with chronic schizophrenia tended to have low melatonin output based on nocturnal urinary excretion of 6-sulfatoxymelatonin, a key melatonin metabolite. Hence, melatonin treatment could improve sleep quality in low-efficiency sleepers, including increase in sleep efficiency, duration and shortened latency (Shamir et al. 2000). Subsequently, Afonso et al. (2011b) demonstrated using actigraphy and nocturnal salivary melatonin radioimmunoassay that concentrations of endogenous nocturnal melatonin between schizophrenia patients and healthy controls were not statistically different. However, the sleep-promoting action of melatonin may be compromised in schizophrenia as the schizophrenia patient group had worse sleep quality with higher latency, lower efficiency and more night-time awakenings.
Docx et al. (2017) performed diffusion kurtosis imaging (DKI) on 20 schizophrenia patients and 16 controls and examined correlations and group differences with regard to fractional anisotropy (FA), mean diffusivity (MD), mean kurtosis (MK) and motor activity level. In the schizophrenia group, motor activity levels were positively correlated with MK in the inferior, medial and superior longitudinal fasciculus, corpus callosum, posterior fronto-occipital fasciculus and posterior cingulum. Walther et al. (2011b) found lower FA levels in the prefrontal and left temporal regions in schizophrenia patients. They also observed a negative correlation between motor activity levels and white matter integrity beneath the right supplementary motor area. Bracht et al. (2013) reported that increased probability index values, an indication that a voxel is part of the connecting fibre bundle of interest (PIBI), were correlated with motor activity in the left pre-SMA–SMA-proper connection for patients with schizophrenia. With regard to functional magnetic resonance imaging (fMRI), Kluge et al. (2018) study, which involved functional magnetic resonance imaging with reward anticipation tasks, found that lower motor activity level was associated with inferior frontal gyrus hypoactivation, while interview-based apathy was associated with ventral striatum hypoactivation. Additionally, resting state fMRI revealed that spontaneous motor activity was correlated with functional connectivity between left M1 and right cerebellum in patients (Walther et al. 2017).
Discussion
Clinical implications of findings
Our review reveals that most studies pertaining to the use of actigraphy in schizophrenia were conducted in Europe and the USA over the last two decades. While all have incorporated either motor activity or sleep trends, few have combined both major parameters or applied them to the comparison of different psychiatric conditions. In addition, there have been more studies correlating changes in motor activity or sleep quality with psychotic psychopathology as compared to cognitive functioning, illness trajectory, treatment factors, daily function or quality of life. Correlations of actigraphic measures with biological factors are yet sparser and have yielded less conclusive results.
Actigraphy studies have captured patterns that may distinguish schizophrenia patients from healthy controls, including monotonous motor activity patterns (Berle et al. 2010), lower mean levels of activity (Sano et al. 2012), as well as sleep irregularities and disturbances (Afonso et al. 2014; Robillard et al. 2015). Only two studies (Fasmer et al. 2016; Robillard et al. 2015) compared the actigraphic patterns between schizophrenia and non-psychotic disorders including affective and anxiety disorders, demonstrating a pattern of prolonged sleep duration, unstable sleep schedules and greater motor inactivity in the schizophrenia group. Another two studies by Walther et al. (2009a, b) examined subtypes of psychotic spectrum disorders (such as cycloid psychosis vs. paranoid/disorganised/catatonic schizophrenia) and attempted to delineate clinical presentations of these conditions based on motor activity. Several studies have also begun to explore the use of actigraphic measures in participants with at-risk mental state (ARMS). ARMS has been associated with the development of psychosis over a 10-year period (Fusar-Poli et al. 2012; Nelson et al. 2013; Yung et al. 1996). Similar to findings in schizophrenia patients, actigraphic measures showed less regular circadian rest–activity rhythms and more nap time in the day for ARMS individuals as compared to healthy controls (Castro et al. 2015). Further evaluation may proffer better clinical markers that can supplement extant clinical evaluation and corroborative information from family members, to distinguish between different psychiatric disorders and even subtypes of psychotic disorders.
Furthermore, actigraphic patterns hold promise as surrogate clinical markers of symptom status in patients with schizophrenia, given their close correlation with psychopathology. Increased positive symptoms are associated with less structured movement patterns (Walther et al. 2014) and disrupted sleep–wake patterns (Afonso et al. 2011a) whilst increased negative symptoms are associated with less motor activity (Kluge et al. 2017; Walther et al. 2014), prolonged sleep, more night-time awakenings and daytime sleepiness (Wichniak et al. 2011). These correlations with psychopathology are mirrored in studies involving ARMS individuals and healthy controls as well. Lunsford-Avery et al. (2015) noted that reduced sleep efficiency (SE), more wake upon sleep onset (WASO), and night-time movement correlated with positive symptom severity in an ultra-high-risk population. They also found that reduced SE, increased WASO, and reduced total sleep time predicted greater positive symptom severity 1 year later. In a sample of healthy controls, participants were more likely to endorse a greater number of psychotic experiences when fewer hours of sleep as captured by actigraphy was coupled with poor perceived sleep quality (Cosgrave et al. 2018).
Next-day functioning has been found to be significantly affected by sleep disturbance and the presence of auditory hallucinations, while delusions of control can be predicted by different aspects of sleep disturbance (Mulligan et al. 2016). However, there are still comparatively fewer studies of correlations with cognitive and psychosocial functioning including quality of life (Afonso et al. 2011a, 2014; Bracht et al. 2012; Chen et al. 2016). Such studies would complement understanding of the inter-relationship between symptomatology, neuropsychological performance and impact on daily functioning in individuals with schizophrenia. Notwithstanding the fewer studies, available data from Afonso et al. (2011a, 2014) suggest that patients with schizophrenia, and in particular those with more positive symptoms, tend to report poorer sleep quality and lower quality of life and Hofstetter et al. (2005) also reported reduced preference for positive cognitive appraisal of circumstances around them and more avoidant coping compared to healthy controls. This behoves the need to better optimise the clinical management of these patients, especially positive symptomatology, by employing pharmacological and non-pharmacological interventions.
Early detection of schizophrenia is crucial for early intervention and educating patients on symptom management. One possible marker present in early stages of schizophrenia is Neurological Soft Signs, which can potentially be more reliably detected using actigraphy (Le Seac’h et al. 2012). Additionally, low level of motor activity has been correlated with the chronicity of the disorder as well as severity of avolition (Walther et al. 2015b). Hence, such objective measures could possibly be used to alert clinicians regarding possible onset of illness, deterioration of condition or to approximate the duration of illness. There are currently mixed findings regarding the effect of antipsychotic dosage on motor activity levels (Docx et al. 2017; Walther et al. 2009c, 2015b) and further research is required to tease apart the relative influence of medication and other disorder-related factors in contributing to lowered activity levels observed in schizophrenia patients. Antipsychotics have been associated with increased cardiovascular mortality (Birkhofer et al. 2013) and akathisia (Poyurovsky et al. 2000), highlighting the importance of close patient monitoring, which can be achieved through the use of actigraphic tools.
Studies correlating actigraphic measures with clinical and biological correlates have the potential to shed light on the underlying biological basis for clinical manifestations of schizophrenia. Poorer sleep quality in schizophrenia patients has been associated with increased inflammation (Fang et al. 2016), which could either reflect inherent biological state or further contribute to other comorbid health conditions experienced by patients with schizophrenia. Neuroimaging studies have observed decreased motor activity to be associated with decreased volume of anterior cingulate (Farrow et al. 2005) and reduced cerebral perfusion to cortical–subcortical brain regions (Walther et al. 2011a) which are implicated in schizophrenia. In addition, cerebral white matter changes (Bracht et al. 2013; Docx et al. 2017; Walther et al. 2011b) are associated with lower motor activity levels, which is consistent with earlier data linking deficit subtype of schizophrenia (with greater inactivity) with such brain white matter changes (Voineskos et al. 2013). Higher probability indices of cortical pathways involving supplementary motor areas may suggest possible compensatory mechanisms towards underlying basal ganglia dysfunction in patients (Bracht et al. 2013; Walther et al. 2011a).
Future directions
Several future directions can be posited. First, future studies may want to combine studies of motor with sleep activity to enable deeper phenotyping of the clinical features observed in patients with schizophrenia. Second, trans-diagnostic investigations involving different psychiatric conditions and subtypes of these conditions may allow better clinical understanding of underlying motor and circadian rhythm changes. Third, a multimodal approach allowing elucidation of interactive relationships between clinical, actigraphic and biological parameters (such as physiological, imaging measures) would enable more extensive correlations between these factors. Fourth, longitudinal studies are warranted to examine the utility of these actigraphic measures for prediction of relapse and prognostication. In the areas above, studies in this field could benefit from ‘big data’ approaches and cross-country collaborations which aggregate data from patients with various psychiatric conditions to examine clinical correlates of these actigraphic measures. Fifth, once better proven, actigraphy can be used as a potential objective monitor of treatment response, and feedback for different interventions including lifestyle changes. Patients can use actigraphy as a form of biofeedback to track their own activity and sleep, which in turn empower them to make further lifestyle changes (Shin et al. 2016). Correlations between actigraphic measures and symptoms can be charted over time using mobile applications or software which can be viewed by both the patient and clinician. Such monitoring techniques highlight specific areas which each patient could alter to bring about symptom alleviation and increased functionality. This can motivate patients to play an active role in managing their illness. Clinicians can also harness this technology to track patients’ progress or make individualised suggestions for lifestyle modification based on their baseline activity levels. Additionally, actigraphy is no longer limited to external wearables, with accelerometers and sleep tracking applications readily available on most mobile devices. Despite limitations in degree of accuracy, it may be an ideal option to introduce patients to the benefits of actigraphy even for those unable or unwilling to use wearables. Lastly, actigraphy also holds promise in the field of translational neuroscience, because motor activity alterations have trans-species value as demonstrated in studies such as that by Perry et al. (2009). They noted that mouse models with inhibited or lack of dopamine active transporter (DAT) function showed similar spatial patterns of locomotion as that observed in bipolar mania. This suggests potential neurobiological underpinnings and therapeutic targets for bipolar disorder, and such trans-species applications could be extended to schizophrenia as well.
Limitations of studies
There are several limitations of the studies reviewed. First, as a cross-sectional design was employed for most of the studies, causal relationships could not be established. Second, findings of studies with smaller sample sizes should be replicated in larger sample sets. Third, future studies may want to investigate the impact of different psychotropic medications on actigraphic patterns or include other confounders including gender, age, comorbid psychiatric disorders to better understand such actigraphic patterns in naturalistic real-life settings. Fourth, the presence of comorbid medical conditions such as sleep disorders can confound findings of poor sleep quality and daytime somnolence (Afonso et al. 2014) or increased inflammation (Fang et al. 2016). Germane to this, it has been noted that patients on antipsychotics have increased risk of developing metabolic syndrome and obstructive sleep apnoea (Lieberman 2004; Rishi et al. 2010). Fifth, inpatients had structured institutional schedules and ward routines, which may have affected actigraphically recorded inter-daily stability of motor activity (Manoach et al. 2010). The potential for these findings to be applied to outpatients who conversely, have greater independent control of their daily activities, is less certain.
Limitations of actigraphy
As a measure of sleep, sleep/wake timings are estimated by movement detected on actigraphy and not based on cortical activity levels (Robillard et al. 2015). Hence, sleep parameters provided by the device are only an approximate reflection of participants’ sleep/wake states even with the use of sleep logs or diaries. This makes it liable to overestimating total sleep time and sleep efficiency. This is especially so in sedentary or bedbound patients who may be logged as being asleep during periods of akinesia (Manoach et al. 2010). Patients lying still in bed but remain vigilant may be logged as having shorter sleep onset latency than is the case (Manoach et al. 2010). Additionally, current actigraphy devices are not validated for measuring sleep stages (Martin and Hakim 2011). It is also not possible to log brief awakenings or periods of sleep, which can affect the accuracy of data collected in patients with severely disturbed sleep patterns. While devices with electroencephalographic sensors can be employed in tandem for these purposes (Ancoli-Israel et al. 2002), it may not be as conducive (Martin et al. 2005). Other modalities such as polysomnography, sleep diaries and sleep questionnaires can complement actigraphy to provide direct additional information regarding sleep duration, as well as derive subjective information on sleep quality.
As a measure of motor activity, the motion sensors employed within individual actigraph devices are currently not yet sufficiently adequate and precise for evaluating complex movements (Bracht et al. 2012). Examples of these include local movements of restricted muscle groups, coordination, movement sequence and symmetry. Similarly, only a limited number of Neurological Soft Signs can be picked up by current inertial sensors, limiting actigraphy’s use in the identification of these clinical features (Krebs et al. 2000; Le Seac’h et al. 2012). Measurement accuracy may also be affected by movement sleep disorders like Periodic Limb Movement Disorder and REM Sleep Behaviour Disorder (Markkula and Lauerma 1997). It has been recommended that multiple devices be placed concurrently at different body parts or simultaneous video recording be done to overcome these limitations (Bracht et al. 2012; Le Seac’h et al. 2012; Poyurovsky et al. 2000). However, these methods will indubitably impact user’s willingness for its wearability.
Evaluating the feasibility of actigraphy in the setting of long-term monitoring of schizophrenia patients with acute or residual active psychotic symptoms, patients may harbour persecutory and/or perceptual delusions about the wearable actigraphy devices. In one study, several participants believed that the device was making surveillance of their lives, tracking their location, or that they were being experimented upon (Shin et al. 2016). This has the potential to precipitate a psychotic relapse or worsen psychotic psychopathology. Patients may also discard these devices secondary to their psychotic experiences. Furthermore, disorganised thinking can potentiate unpredictable and erratic behaviour deviating from usual motor and sleep patterns and levels. For instance, as reported by Shin et al. (2016), some participants with known baseline of low motor activity displayed abnormally increased activity levels only during the recording period, leading to concerns about consistency of the data logged.
Conclusion
In conclusion, this review found lower motor activity and poorer sleep quality in patients with schizophrenia compared with healthy controls which are associated with clinical features (such as psychotic phenomenology, cognitive functioning, subtypes, quality of life, illness chronicity, medication dose) and less conclusively with biological features (inflammatory blood markers, structural and functional MRI features). Notwithstanding inherent limitations of studies reviewed and actigraphic tools, actigraphy holds promise as a surrogate objective monitor of clinical status, functioning, treatment response. It also shows potential to be a means of feedback for different interventions including lifestyle changes for patients with such a crippling psychiatric condition.
References
Afonso P, Brissos S, Figueira ML, Paiva T (2011a) Schizophrenia patients with predominantly positive symptoms have more disturbed sleep-wake cycles measured by actigraphy. Psychiatry Res 189(1):62–66
Afonso P, Figueira ML, Paiva T (2011b) Sleep-promoting action of the endogenous melatonin in schizophrenia compared to healthy controls. Int J Psychiatry Clin Pract 15(4):311–315
Afonso P, Figuiera ML, Paiva T (2014) Sleep-wake patterns in schizophrenia patients compared to healthy controls. World J Biol Psychiatry 15(7):517–524
Ancoli-Israel S, Cole R, Alessi CA, Chambers M, Moorcroft WH, Pollak C (2002) The role of actigraphy in the study of sleep and circadian rhythms. Sleep 26:342–392
Ancoli-Israel S, Kripke DF, Klauber MR, Mason WJ, Fell R, Kaplan O (1991) Sleep-disordered breathing in community-dwelling elderly. Sleep 14(6):486–495
Baandrup L, Jennum PJ (2015) A validation of wrist actigraphy against polysomnography in patients with schizophrenia or bipolar disorder. Neuropsychiatr Dis Treat 11:2271–2277
Baandrup L, Fasmer OB, Glenthøj BY, Jennum PJ (2016) Circadian rest-activity rhythms during benzodiazepine tapering covered by melatonin versus placebo add-on: data derived from a randomized clinical trial. BMC Psychiatry 16:348
Berle JO, Hauge ER, Oedegaard KJ, Holsten F, Fasmer OB (2010) Actigraphic registration of motor activity reveals a more structured behavioural pattern in schizophrenia than in major depression. BMC Res Notes 3:149
Birkhofer A, Geissendoerfer J, Alger P, Mueller A, Rentrop M, Strubel T, Leucht S, Förstl H, Bär KJ, Schmidt G (2013) The deceleration capacity – a new measure of heart rate variability evaluated in patients with schizophrenia and antipsychotic treatment. Eur Psychiatry 28(2):81–86
Bracht T, Heidemeyer K, Koschorke P, Horn H, Razavi N, Wopfner A, Strik W, Walther S (2012) Comparison of objectively measured motor behaviour with ratings of the motor behaviour domain of the Bern Psychopathology (BPS) in schizophrenia. Psychiatry Res 198(2):224–229
Bracht T, Schnell S, Federspiel A, Razavi N, Horn H, Strik W, Wiest R, Dierks T, Müller TJ, Walther S (2013) Altered cortico-basal ganglia motor pathways reflect reduced volitional motor activity in schizophrenia. Schizophr Res 143(2–3):269–276
Bromundt V, Köster M, Georgiev-Kill A, Opwis K, Wirz-Justice A, Stoppe G, Cajochen C (2011) Sleep-wake cycles and cognitive functioning in schizophrenia. Br J Psychiatry 198(4):269–276
Brown A, Smolensky M, D’Alonzo G, Redmond D, Conrad E, Hsi B (1990) Circadian rhythm in human activity objectively quantified by actigraphy. Prog Clin Biol Res 341A:77–83
Castro J, Zanini M, Gonçalves B, da SB, Coelho, Bressan FMS R, et al (2015) Circadian rest-activity rhythm in individuals at risk for psychosis and bipolar disorder. Schizophr Res 168:50–55
Chan RC, Xie W, Geng FL, Wang Y, Lui SS, Wang CY, Yu X, Cheung EF, Rosenthal R (2016) Clinical utility and lifespan profiling of neurological soft signs in schizophrenia spectrum disorders. Schizophr Bull 42(3):560–570
Chen LJ, Steptoe A, Chung MS, Ku PW (2016) Association between actigraphy-derived physical activity and cognitive performance in patients with schizophrenia. Psychol Med 46(11):2375–2384
Chouinard S, Poulin J, Stip E, Godbout R (2004) Sleep in untreated patients with schizophrenia: a meta-analysis. Schizophr Bull 30(4):957–967
Cosgrave J, Haines R, van Heugten-van der Kloet D, Purple R, Porcheret K, Foster R, Wulff K (2018) The interaction between subclinical psychotic experiences, insomnia and objective measures of sleep. Schizophr Res 193:204–208
Costa R, Bastos T, Probst M, Seabra A, Abreu S, Vilhena E, Rosenbaum S, Ward PB, Corredeira R (2018) Association of lifestyle-related factors and psychological factors on quality of life in people with schizophrenia. Psychiatry Res 267:382–393
De Crescenzo F, Licchelli S, Ciabattani M, Menghini D, Armando M, Alfieri P, Mazzone L, Pontrelli G, Livadiotti S, Foti F, Quested D, Vicari S (2016) The use of actigraphy in the monitoring of sleep and activity in ADHD: A meta-analysis. Sleep Med Rev 26:9–20
Docx L, Morrens M, Bervoets C, Hulstijn W, Fransen E, De Hert M. Baeken C, Audenaert K, Sabbe B (2012) Parsing the components of the psychomotor syndrome in schizophrenia. Acta Psychiatr Scand 126:256–265
Docx L, Sabbe B, Provinciael P, Merckx N, Morrens M (2013) Quantitative psychomotor dysfunction in schizophrenia: a loss of drive, impaired movement execution or both? Neuropsychobiology 68(4):221–227
Docx L, Emsell L, Van Hecke W, De Bondt T, Parizel PM, Sabbe B, Morrens M (2017) White matter microstructure and volitional motor activity in schizophrenia: A diffusion kurtosis imaging study. Psychiatry Res Neuroimaging 260:29–36
Fang SH, Suzuki K, Lim CL, Chung MS, Ku PW, Chen LJ (2016) Associations between sleep quality and inflammatory markers in patients with schizophrenia. Psychiatry Res 246:154–160
Farrow TF, Hunter MD, Wilkinson ID, Green RD, Spence SA (2005) Structural brain correlates of unconstrained motor activity in people with schizophrenia. Br J Psychiatry 187:481–482
Fasmer OB, Hauge E, Berle J, Dilsaver S, Oedegaard KJ (2016) Distribution of active and resting periods in the motor activity of patients with depression and schizophrenia. Psychiatry Investig 13(1):112–120
Fusar-Poli P, Bonoldi I, Yung AR, Borgwardt S, Kempton MJ, Valmaggia L et al (2012) Predicting psychosis: meta-analysis of transition outcomes in individuals at high clinical risk. Arch Gen Psychiatry 69:220–229
García R, Aliste F, Soto G (2018) Cognición social en esquizofrenia: aspectos cognitivos y neurobiológicos. Revista Colombiana de Psiquiatría 47(3):170–176
Gonçalves B, Castro J, Zanini MA, Bittencourt L, Gadelha A, Cunha GR, Coelho FM, Tufik S, Bressan RA, Brietzke E (2016) Circadian rhythm disturbances and conversion to psychosis in ultra high-risk youth. Braz J Psychiatry 38(2):178–179
Haug HJ, Wirz-Justice A, Rössler W (2000) Actigraphy to measure day structure as a therapeutic variable in the treatment of schizophrenic patients. Acta Psychiatr Scand Suppl 407:91–95
Heinrichs DW, Buchanan RW (1988) Significance and meaning of neurological signs in schizophrenia. Am J Psychiatry 145(1):11–18
Hofstetter JR, Lysaker PH, Mayeda AR (2005) Quality of sleep in patients with schizophrenia is associated with quality of life and coping. BMC Psychiatry 3:5–13
Janney CA, Ganguli R, Tang G, Cauley JA, Holleman RG, Richardson CR, Kriska AM (2015) Physical activity and sedentary behavior measured objectively and subjectively in overweight and obese adults with schizophrenia or schizoaffective disorders. J Clin Psychiatry 76(10):1277–1284
Khan SS, Ye B, Taati B, Mihailidis A (2018) Detecting agitation and aggression in people with dementia using Sensors- a systematic review. Alzheimers Dement 14(6):824–832
Kiang M, Daskalakis ZJ, Christensen BK, Remington G, Kapur S (2003) Actigraphic measurement of the effects of single-dose haloperidol and olanzapine on spontaneous motor activity in normal subjects. J Psychiatr Neurosci 28(4):293–299
Kluge A, Kirschner M, Hager OM, Bischof M, Habermeyer B, Seifritz E, Walther S, Kaiser S (2018) Combining actigraphy, ecological momentary assessment and neuroimaging to study apathy in patients with schizophrenia. Schizophr Res 195:176–182
Krebs M, Gut-Fayand A, Bourdel M, Dischamp J, Olié J (2000) Validation and factorial structure of a standardized neurological examination assessing neurological soft signs in schizophrenia. Schizophr Res 45:245–260
Kurebayashi Y, Otaki J (2017) Correlations between physical activity and neurocognitive domain functions in patients with schizophrenia: a cross-sectional study. BMC Psychiatry 17(1):4
Le Seac’h A, Picard H, Gosane MA, Vidal PP, Amado I, Krebs MO (2012) A step toward an objective quantification of subtle neurological signs in schizophrenia. Psychiatry Res 198(2):230–234
Lieberman JA (2004) Metabolic changes associated with antipsychotic use. Prim Care Companion J Clin Psychiatry 6(suppl 2):8–13
Lunsford-Avery JR, LeBourgeois MK, Gupta T, Mittal VA (2015) Actigraphic measured sleep disturbance predicts increased positive symptoms in adolescents at ultra high-risk for psychosis: a longitudinal study. Schizophr Res 164:15e20
Lunsford-Avery JR, Goncalves BDSB, Brietzke E, Bressan RA, Gadelha A, Auerbach RP, Mittal VA (2017) Adolescents at clinical-high risk for psychosis: circadian rhythm disturbances predict worsened prognosis at 1-year follow up. Schizophr Res 189:37–42
Manoach DS, Thakkar KN, Stroynowski E, Ely A, McKinley SK, Wamsley E, Djonlagic I, Vangel MG, Goff DC, Stickgold R (2010) Reduced overnight consolidation of procedural learning in chronic medicated schizophrenia is related to specific sleep stages. J Psychiatr Res 44(2):112–120
Markkula J, Lauerma H (1997) Mianserin and restless legs. Int Clin Psychopharmacol 12:53–58
Martin JL, Hakim AD (2011) Wrist actigraphy. Chest 139(6):1514–1527
Martin JL, Jeste DV, Ancoli-Israel S (2005) Older schizophrenia patients have more disrupted sleep and circadian rhythms than age-matched comparison subjects. J Psychiatr Res 39(3):251–259
Mittal VA, Gupta T, Orr JM, Pelletier AL, Dean DJ, Lunsford-Avery JR, Smith AK, Robustelli BL, Leopold DR, Millman ZB (2013) Physical activity level and medial temporal health in youth at ultra high-risk for psychosis. J Abnorm Psychol 122(4):1101–1110
Mulligan LD, Haddock G, Emsley R, Neil ST, Kyle SD (2016) High resolution examination of the role of sleep disturbance in predicting functioning and psychotic symptoms in schizophrenia: a novel experience sampling study. J Abnorm Psychol 125(6):788–797
Nelson B, Yuen HP, Wood SJ, Lin A, Spiliotacopoulos D, Bruxner A et al (2013) Long-term follow-up of a group at ultra high risk (“prodromal”) for psychosis: the PACE 400 study. JAMA Psychiatry 70:793–802
Osipov M, Behzadi Y, Kane JM, Petrides G, Clifford GD (2015) Objective identification and analysis of psychological and behavioural signs of schizophrenia. J Ment Health 24(5):276–282
Perry W, Minassian A, Paulus MP, Young JW, Kincaid MJ, Ferguson EJ, Henry BL, Zhuang X, Masten VL, Sharp RF, Geyer MA (2009) A reverse-translational study of dysfunctional exploration in psychiatric disorders: from mice to men. Arch Gen Psychiatry 66(10):1072–1080
Poyurovsky M, Nave R, Epstein R, Tzischinsky O, Schneidman M, Barnes TR, Weizman A, Lavie P (2000) Actigraphic monitoring (actigraphy) of circadian locomotor activity in schizophrenic patients with acute neuroleptic-induced akathisia. Eur Neuropsychopharmacol 10(3):171–176
Reeve S, Sheaves B, Freeman D (2015) The role of sleep dysfunction in the occurrence of delusions and hallucinations: a systematic review. Clin Psychol Rev 42:96–115
Reeve S, Emsley R, Sheaves B, Freeman D (2017) Disrupting sleep: the effects of sleep loss on psychotic experiences tested in an experimental study with mediation analysis. Schizophr Bull 44(3):662–671
Rishi MA, Shetty M, Wolff A, Amoateng-Adjepong Y, Manthous CA (2010) Atypical antipsychotic medications are independently associated with severe obstructive sleep apnea. Clin Neuropharmacol 33(3):109–113
Robillard R, Hermens DF, Naismith SL, White D, Rogers NL, Ip TK, Mullin SJ, Alvares GA, Guastella AJ, Smith KL, Rong Y, Whitwell B, Southan J, Glozier N, Scott EM, Hickie IB (2015) Ambulatory sleep-wake patterns and variability in young people with emerging mental disorders. J Psychiatry Neurosci 40(1):28–37
Sadeh A, Lavie P, Scher A, Tirosh E, Epstein R (1991) Actigraphic home-monitoring sleep-disturbed and control infants and young children: a new method for pediatric assessment of sleep-wake patterns. J Pediatr 87(4):494–499
Sano W, Nakamura T, Yoshiuchi K, Kitajima T, Tsuchiya A, Esaki Y, Yamamoto Y, Iwata N (2012) Enhanced persistency of resting and active periods of locomotor activity in schizophrenia. PLoS One 7(8):e43539
Scott J, Vaaler AE, Fasmer OB, Morken G, Krane-Gartiser K (2017) A pilot study to determine whether combinations of objectively measured activity parameters can be used to differentiate between mixed states, mania, and bipolar depression. Int J Bipolar Disord 5(1):5
Shamir E, Laudon M, Barak Y, Anis Y, Rotenberg V, Elizur A, Zisapel N (2000) Melatonin improves sleep quality of patients with chronic schizophrenia. J Clin Psychiatry 61(5):373–377
Shin S, Yeom CW, Shin C, Shin JH, Jeong JH, Shin JU, Lee YR (2016) Activity monitoring using a mHealth device and correlations with psychopathology in patients with chronic schizophrenia. Psychiatry Res 246:712–718
Strassnig M, Bowie C, Pinkham AE, Penn D, Twamley EW, Patterson TL, Harvey PD (2018) Which levels of cognitive impairments and negative symptoms are related to functional deficits in schizophrenia? J Psychiatr Res 104:124–129
Tahmasian M, Khazaie H, Golshani S, Avis KT (2013) Clinical application of actigraphy in psychotic disorders: a systemic review. Curr Psychiatry Rep 15(6):359
Tamagni C, Studerus E, Gschwandtner U, Aston J, Borgwardt S, Riecher-Rössler A (2013) Are neurological soft signs pre-existing markers in individuals with an at-risk mental state for psychosis? Psychiatry Res 210(2):427–431
Teicher MH (1995) Actigraphy and motion analysis: new tools for psychiatry. Harv Rev Psychiatry 3(1):18–35
Teicher MH, Glod CA, Magnus E, Harper D, Benson G, Krueger K, McGreenery CE (1997) Circardian rest-activity disturbances in seasonal affective disorder. Arch Gen Psychiatry 54(2):124–130
Van Kammen DP, Van Kammen WB, Peters JL (1986) CSF MHPG, sleep and psychosis in schizophrenia. Clin Neuropharmacol 9(4):575–577
Voineskos AN, Foussias G, Lerch J, Felsky D, Remington G, Rajji TK, Lobaugh N, Pollock BG, Mulsant BH (2013) Neuroimaging evidence for the deficit subtype of schizophrenia. JAMA Psychiatry 70(5):472–480
Walther S, Horn H, Koschorke P, Müller TJ, Strik W (2009a) Increased motor activity in cycloid psychosis compared to schizophrenia. World J Biol Psychiatry 10(4):746–751
Walther S, Horn H, Razavi N, Koschorke P, Müller TJ, Strik W (2009b) Quantitative motor activity differentiates schizophrenia subtypes. Neuropsychobiology 60(2):80–86
Walther S, Koschorke P, Horn H, Strik W (2009c) Objectively measured motor activity in schizophrenia challenges the validity of expert ratings. Psychiatry Res 169(3):187–190
Walther S, Federspiel A, Horn H, Razavi N, Wiest R, Dierks T, Strik W, Müller TJ (2011a) Resting state cerebral blood flow and objective motor activity reveal basal ganglia dysfunction in schizophrenia. Psychiatry Res 192(2):117–124
Walther S, Federspiel A, Horn H, Razavi N, Wiest R, Dierks T, Strik W, Müller TJ (2011b) Alterations of white matter integrity related to motor activity in schizophrenia. Neurobiol Dis 42(3):276–283
Walther S, Ramseyer F, Horn H, Strik W, Tschacher W (2014) Less structured movement patterns predict severity of positive syndrome, excitement, and disorganization. Schizophr Bull 40(3):585–591
Walther S, Stegmayer K, Horn H, Rampa L, Razavi N, Müller TJ, Strik W (2015a) The longitudinal course of gross motor activity in schizophrenia – within and between episodes. Front Psychiatry 6:10
Walther S, Stegmayer K, Horn H, Razavi N, Müller TJ, Strik W (2015b) Physical activity in schizophrenia is higher in the first episode than in subsequent ones. Front Psychiatry 5:191
Walther S, Stegmayer K, Federspiel A, Bohlhalter S, Wiest R, Viher PV (2017) Aberrant hyperconnectivity in the motor system at rest is linked to motor abnormalities in schizophrenia spectrum disorders. Schizophr Bull 43(5):982–992
Wichniak A, Skowerska A, Chojnacka-Wójtowicz J, Tafliński T, Wierzbicka A, Jernajczyk W, Jarema M (2011) Actigraphic monitoring of activity and rest in schizophrenic patients treated with olanzapine or risperidone. J Psychiatr Res 45(10):1381–1386
Wulff K, Joyce E, Middleton B, Dijk DJ, Foster RG (2006) The suitability of actigraphy, diary data, and urinary melatonin profiles for quantitative assessment of sleep disturbances in schizophrenia: a case report. Chronobiol Int 23(1–2):485–495
Yung AR, McGorry PD, McFarlane CA, Jackson HJ, Patton GC, Rakkar A (1996) Monitoring and care of young people at incipient risk of psychosis. Schizophr Bull 22:283–303
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No authors or any close family members have any current financial conflicts of interest related to this paper. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wee, Z.Y., Yong, S.W.L., Chew, Q.H. et al. Actigraphy studies and clinical and biobehavioural correlates in schizophrenia: a systematic review. J Neural Transm 126, 531–558 (2019). https://doi.org/10.1007/s00702-019-01993-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-019-01993-2