Abstract
The aim of this study was to assess the effects of the neurotoxin N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine (DSP4) on attention in rats as measured using the 5-choice-serial-reaction-time task (5CSRTT) and to investigate whether methylphenidate has effects on DSP4-treated rats. Methylphenidate is a noradrenaline and dopamine reuptake inhibitor and commonly used in the pharmacological treatment of individuals with attention deficit/hyperactivity disorder (ADHD). Wistar rats were trained in the 5CSRTT and treated with one of three doses of DSP4 or saline. Following the DSP4 treatment rats were injected with three doses of methylphenidate or saline and again tested in the 5CSRTT. The treatment with DSP4 caused a significant decline of performance in the number of correct responses and a decrease in response accuracy. A reduction in activity could also be observed. Whether or not the cognitive impairments are due to attention deficits or changes in explorative behaviour or activity remains to be investigated. The treatment with methylphenidate had no beneficial effect on the rats’ performance regardless of the DSP4 treatment. In the group without DSP4 treatment, methylphenidate led to a reduction in response accuracy and bidirectional effects in regard to parameters related to attention. These findings support the role of noradrenaline in modulating attention and call for further investigations concerning the effects of methylphenidate on attentional processes in rats.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Attention deficit hyperactivity disorder (ADHD) is one of the most common psychiatric disorders of childhood and adolescence and is characterised by the core symptoms of hyperactivity, inattentiveness, impulsivity and distractibility; other cognitive impairments may also be present (Barkley 2006; Biederman and Faraone 2005; Heal et al. 2008; Lange et al. 2007, 2010; Tucha and Lange 2001; Tucha et al. 2006, 2008).
Environmental, social, neurobiological and genetic aspects have been discussed in regard to the etiology and pathogenesis of ADHD (Barkley 2006; Biederman et al. 1992, 1995; Döpfner 1999; Wankerl et al. 2014). However, these approaches are still unable to sufficiently explain the etiology of ADHD. A dysregulation of catecholaminergic neurotransmission in the prefrontal cortex and its connections to striatal areas has been proposed as a possible neurobiological factor (Arnsten and Dudley 2005; Heal et al. 2008; Russell et al. 2005) and the characteristic deficiencies observed in ADHD have been proposed to be a dysfunction of the frontostriatal system (Davids et al. 2003; Sontag et al. 2008, 2010).
In addition to impaired dopaminergic neurotransmission in ADHD (for detail see Russell et al. 2005; Clements et al. 2003; Heal et al. 2008; Swanson et al. 1998), there is evidence suggesting that noradrenergic neurotransmission may also be affected in individuals with ADHD (Arnsten 2011; Heal et al. 2008; Russell et al. 2005). While some authors have suggested low noradrenaline activity in ADHD (Halperin et al. 1997; Heal et al. 2008; Oades 1987), others have proposed an increased noradrenaline activity in the prefrontal cortex of children with ADHD (Russell 2002; Russell et al. 2005; Solanto 1998).
Noradrenaline has been shown to be involved in the regulation of the signal-to-noise ratio (McGaugh 1989) and the facilitation of excitatory and inhibitory responses in the limbic system (Pichler and Kobinger 1985; Riekkinen et al. 1993). In addition, noradrenaline plays an important role in arousal, vigilance and responses to novel salient stimuli (Arnsten 1997; Aston-Jones et al. 1994; Clark et al. 1987; Harley 1987; Solanto 1998). Other processes, such as learning and attention, are also influenced by noradrenergic activity (Cole and Robbins 1989; Coull 1994; Jonge et al. 1981; Everitt et al. 2009; Micheletti et al. 1987). Since some of these functions are known to be affected in ADHD, it is of interest to investigate the involvement of noradrenaline in the etiology of this disorder.
A primarily noradrenergic dysfunction was discussed as one of the earliest models of ADHD (Mefford and Potter 1989). This was supported by the findings of animal studies, e.g., a rise in brain noradrenaline leads to an increase in focused behaviour, while a reduction in noradrenaline increases the reaction to irrelevant stimuli (Aston-Jones et al. 1997). Pliszka et al. (1996) postulated a dysfunction in both noradrenergic and dopaminergic neurotransmission and described a posterior system, in which a noradrenergic dysfunction results in attention deficits, and an anterior system, in which a hypofunctional dopamine system causes impaired executive functions. Another hypothesis suggests a reduced noradrenaline activity inducing a changed alpha-2 receptor activity in the prefrontal cortex and resulting in a dysfunctional inhibition control in children with ADHD (Arnsten et al. 1996; Arnsten and Dudley 2005). There are differences between the hypotheses attempting to explain the neurobiological basis of ADHD, but they have certain aspects in common, i.e. the important role of catecholamines, particularly noradrenaline, and the prefrontal cortex.
Noradrenergic functioning in the brain can be impaired by noradrenaline depletion induced by the administration of the neurotoxins 6-hydroxydopamine (6-OHDA) (Brozoski et al. 1979; Collins et al. 1998; Crofts et al. 2001; Roberts et al. 1994) or N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine (DSP4) (Cheetham et al. 1996; Fritschy and Grzanna 1991; Ross and Stenfors 2015). A depletion of noradrenaline in neonatal rats following the administration of 6-OHDA in combination with a selective dopamine transporter inhibitor (Teicher et al. 1986) has been shown to induce motor hyperactivity (Raskin et al. 1983), learning deficits (Roberts et al. 1976) and attention deficits (Carli et al. 1983). DSP4 can be administered systemically, is effective in a dose-dependent manner and selective for the noradrenergic terminals of the locus coeruleus (Fritschy and Grzanna 1991; Ross and Stenfors 2015).
Previous studies have assessed behavioural aspects of central noradrenergic depletion after administration of DSP4. Behavioural studies following DSP4 treatment were performed for various functions including working memory and reference memory (Ohno et al. 1993, 1997; Sontag et al. 2008, 2011), short-term memory and attention (Ruotsalainen et al. 1997), discrimination learning (Al Zahrani et al. 1997), and motor activity (Jones and Hess 2003). While some studies reported an impaired performance in these functions, others were unable to find any significant alterations. For example, some authors found impaired spatial working memory after DSP4 administration in rats (Compton et al. 1995; Wenk et al. 1987; Sontag et al. 2008), while others were unable to reveal any deficits (Al Zahrani et al. 1997; Benloucif et al. 1995; Langlais et al. 1993; Sontag et al. 2011). These inconsistent findings and the fact that the effect of a systemic noradrenergic depletion on attention has not sufficiently been investigated underline the need for further research.
In this context, the 5-choice-serial-reaction-time-task (5CSRTT) appears to be a suitable paradigm to investigate the effects of DSP4 and methylphenidate on attention. The 5CSRTT is a preclinical analogue to the continuous performance task (CPT), which is sensitive in detecting attention deficits in several disorders including ADHD (Banaschewski et al. 2003; Navarra et al. 2008; Robbins 2002; Teicher et al. 2004). In the CPT, human volunteers are required to detect correct letters and react by pressing a button, while in the 5CSRTT, rats are trained with a food reward to react correctly by discriminating visually presented brief-duration stimuli which occur randomly in one of five locations (Navarra et al. 2008). In both tasks, the CPT for humans and the 5CSRTT for rodents, the same parameters can be quantified (Navarra et al. 2008). In the 5CSRTT, missed or false responses to the stimuli are interpreted as attention deficits, while responses between the stimulus presentations are interpreted as impulsive reactions. The 5CSRTT therefore allows to distinguish between inattention and impulsiveness.
The aim of this study was to investigate the role of central noradrenaline regarding attentional aspects of cognitive performance in rats using an established test paradigm (5CSRTT) for the assessment of attention in rodents. We have assessed attentional processes following a systemic administration of DSP4 and the influence of methylphenidate on DSP4-treated rats.
Methods
Animals and feeding procedure
Forty-eight male Wistar rats (Charles River Laboratories, Sulzbach, Germany) aged seven weeks (body weight approximately 330 g at the beginning of the experiment) were used. The experiment consisted of three phases: (1) At the beginning of the experiment the seven-week-old rats were trained for 8 weeks, (2) after the training phase rats were treated with DSP4, followed by a two-week recovery period, and (3) finally rats were tested for four weeks under various methylphenidate conditions. At the beginning of the testing phase the rats were 17 weeks old, the experiment ended at the age of 21 weeks. The rats were housed in standard cages and under standard animal laboratory conditions (12:12 h light/dark cycle, room temperature 22 °C, humidity 50%) in the animal laboratories of the University of Regensburg. Access to food was restricted since the behavioural paradigm used in this study (i.e. 5CSRTT) is based on food reinforcement. Water was provided ad libitum. After the training or testing procedures the rats had free access to food for 1 h a day. The rats’ weight was carefully controlled and a weight reduction of more than 10–15% was avoided to prevent stress (Bear 1999; Deroche et al. 1995) and subsequent changes in the dopaminergic system (Pothos et al. 1995).
Ethics
All experiments were performed in accordance with the national laws (German law on Protection of Animals) and the principles of laboratory animal care (NIH publication No. 86–23, revised 1996). The rats were handled according to the guidelines of the Federation for European Laboratory Animal Science Associations (FELASA). All efforts were used to reduce the number of animals and to minimize animal suffering during experiments. After DSP4 administration, the rats were given a recovery period of two weeks, during which time they were monitored daily for health concerns and body weight. Body weight was assessed to avoid its reduction following the administration of DSP4. In case of weight loss, the rats were fed individually with food mash. No experiments or tests were conducted during this recovery period. After the experiments, rats were sacrificed using carbon dioxide.
The 5-choice-serial-reaction-time-task
The experiment was performed using four ventilated wooden chambers (Campden Instruments, Loughborough, Leicestershire, England) containing a stainless steel chamber (26 cm × 26 cm × 30 cm height). The steel chambers were lightened by 3-Watt light bulbs. Each chamber was equipped with five holes, which were arranged horizontally in the curved rear wall. Each hole had a diameter of 2 cm, adjacent holes were 6 cm apart and the holes were 2 cm above the chamber floor (stainless steel grid). In each hole, an infrared photocell was installed to detect a nose poke response of the rat to the hole. In addition, each hole was equipped with a standard light bulb (3 W). The animals were required to respond correctly to a stimulus by a nose poke into one of the five holes. A stimulus was defined as the illumination of a hole by flashing the light bulb, only one hole at a time could be illuminated. A correct response was rewarded with a food pellet (45 mg dustless sucrose pellets, Bio-Serv, Frenchtown, New Jersey, USA), which was dispensed into a food tray at the front wall (opposite the holes), false or missed responses were punished with a 5-s period of darkness.
The behavioural paradigm consisted of three phases. In the habituation phase, the ambient light was permanently turned on, 10 pellets were baited in the food tray and one pellet was placed in each illuminated hole. The rats were required to habituate to the boxes for 30 min a day. This phase was finished when all pellets were found and collected, which was accomplished within two consecutive days. In the training phase, the rats were required to learn to respond correctly to the stimulus (i.e. random illumination of a hole, once per trial) to obtain a food pellet. The stimulus duration was gradually reduced when a rat responded correctly—within one training session of 30 min—in at least 80% of the trials (number of correct trials/total number of trials responded to) and the omission rate was less than 20% (number of trials missed/number of trials presented). The stimulus duration lasted from 60 s (training level 1) to 1.5 s (final training level). All other parameters were kept constant during the training phase (inter-trial interval ITI of 5 s). In the final (testing) phase, the stimulus duration was 1.5 s and the test sessions were similar to the training sessions except that the ITIs varied randomly between 0.5, 1.5, 3 and 4.5 s. Each ITI occurred 25 times within one session. The order in which the groups were tested was randomised in all phases.
Training and DSP4 administration
The training was performed with untreated rats in order to avoid any effects of the DSP4 treatment on learning. The animals were trained on five consecutive days per week for 8 weeks (40 sessions). After the training phase the rats were divided randomly into four groups of 12 rats each and were treated with one dose of DSP4 (10, 20 or 50 mg/kg body weight) or saline. The DSP4 doses used in this study are based on the findings of Cheetham et al. (1996). According to the authors these DSP4 doses cause a noradrenergic depletion in the rat brain in a linear fashion by 51, 73 or 100%, induced by the selective destruction of the noradrenergic terminals originating in the locus coeruleus. DSP4 (Sigma-Aldrich, Schnelldorf, Germany) was dissolved in saline, the volume injected was calculated individually (1 ml/kg body weight), both DSP4 and saline were injected intraperitoneally.
A period of two weeks followed the DSP4 administration to allow for the recovery of the peripheral noradrenaline levels. During this period, the rats were checked for body weight and general health once a day. Feeding was less restrictive to prevent any additional stress.
Testing and treatment with methylphenidate
During the testing phase, the rats were tested twice a week (Tuesday and Friday). On the day prior to testing, rats were trained using the stimulus duration of the final training level to guarantee optimal performance of the animals. In addition to DSP4 administration, the rats were treated with methylphenidate. On testing days the rats were injected with saline (control) or methylphenidate at a dose of 1, 3 or 12 mg/kg body weight (Sigma-Aldrich, Schnelldorf, Germany) 20 min prior to testing because the maximum brain concentration can be measured within the first 20 min following administration (Huff and Davies 2002). Methylphenidate was dissolved in saline, the volume injected was determined individually (1 ml/kg body weight). Both methylphenidate and saline were administered intraperitoneally. Each rat received one dose of methylphenidate twice a week and was exposed to all doses and the control condition. The methylphenidate doses were chosen on the basis of previous findings which have demonstrated their effectiveness (Yang et al. 2006; Pattij et al. 2012; Koda et al. 2010; Slezak and Anderson 2011). The testing period lasted four weeks. Saline and the three methylphenidate doses were administered in a balanced manner to avoid systematic carry-over effects. In addition, a wash-out period of 3 days was used (see also Yang et al. 2003).
Statistical analysis
The following parameters were analysed: (1) number of correct responses, (2) number of incorrect responses, (3) number of omissions, (4) percentage of correct responses (i.e. number of correct responses/total correct and incorrect responses as percent), (5) percentage of omissions (i.e. total number of omissions/total trials completed as percent) and (6) number of trials completed. In addition, the following parameters for the comparisons between the DSP4 groups and saline-treated group were analysed: (7) number of front beam breaks and (8) number of back beam breaks. A mean value for each animal was calculated in order to compare the differences between the DSP4 groups and between the methylphenidate conditions. All findings concerning group differences and comparisons between the methylphenidate conditions within each group are expressed as means ± standard errors. The statistical analysis of differences between DSP4 groups was performed using the Mann-Whitney U-test (between-subject design). The methylphenidate doses within each group were compared using the Wilcoxon test. An alpha level of 0.05 was applied. All statistical analyses were performed using the Statistical Package for Social Sciences 19.0 (SPSS) for Windows.
Results
Comparisons between the DSP4 groups
All means (M) and standard errors (SE) regarding the performance (i.e. the number of correct and incorrect responses, the numbers of omissions and trials completed, and the percentage of correct responses and omissions) of the DSP4 treated groups are presented in Table 1. Statistically significant differences were found regarding the numbers of correct responses, omissions, trials completed as well as the percentages of correct responses and omissions. The DSP4_50 mg/kg group made significantly fewer correct responses than the DSP4_control group (p < 0.001; Z = −4.311), DSP4_10 mg/kg group (p < 0.001; Z = −3.869) and the DSP4_20 mg/kg group (p = 0.003; Z = −2.945). Furthermore, the DSP4_20 mg/kg group missed significantly more signals (i.e. numbers of omissions) in comparison with the DSP_4 control group (p = 0.005; Z = −2.793), the DSP4_10 mg/kg group (p = 0.001; Z = −3.274) and the DSP4_50 mg/kg group (p = 0.049; Z = −1.965). The DSP4_50 mg/kg group completed significantly fewer trials in comparison to the DSP4_control group (p < 0.001; Z = −3.537) and the DSP4_20 mg/kg group (p < 0.001; Z = −3.973). Both the DSP4_20 mg/kg group and the DSP4_50 mg/kg group made significantly more omissions given as percentage compared to the DSP4_control group (DSP4_20 mg/kg compared to DSP4_control: p = 0.035; Z = −2.111; DSP4_50 mg/kg compared to DSP4_control: p = 0.001; Z = −2.452). None of the remaining comparisons between the DSP4 groups reached statistical significance (for means and standard errors see Table 1, p values and Z values are not shown).
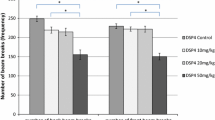
As shown in Fig. 1, the administration of DSP4_10 mg/kg, DSP4_20 mg/kg and DSP4_50 mg/kg significantly reduced the numbers of back beam breaks compared to the control group [DSP4_control (M ± SE 269.2 ± 12.1) compared with DSP4_10 mg/kg (M ± SE 194.1 ± 23.1): p < 0.001; Z = −4.213; DSP4_control compared with DSP4_20 mg/kg (M ± SE 207.0 ± 17.0): p = 0.001; Z = −3.442; DSP4_control compared with DSP4_50 mg/kg (M ± SE 181.3 ± 13.7): p < 0.001; Z = −5.673]. In addition, the difference between the DSP4_20 mg/kg group and the DSP4_50 mg/kg group was statistically significant (p = 0.02; Z = −2.325). All the remaining comparisons between the DSP4 groups concerning the number of back beam breaks failed statistical significance.
The treatment with 10 mg/kg DSP4 and 50 mg/kg DSP4 caused a significant decrease in the numbers of front beam breaks in comparison to the saline-treated group [DSP4_control (M ± SE 232.2 ± 13.5) compared with DSP4_10 mg/kg (M ± SE 165.8 ± 17.2): p < 0.001; Z = −4.431; DSP4_control compared with DSP4_50 mg/kg (M ± SE 176.7 ± 9.1): p < 0.001; Z = −5.662]. Furthermore, the DSP4_ 20 mg/kg group made significantly more front beam breaks than the DSP4_10 mg/kg and the DSP4_50 mg/kg group (DSP4_20 mg/kg (M ± SE 193.4 ± 18.7) compared with DSP4_10 mg/kg: p = 0.036; Z = −2.096; DSP4_20 mg/kg compared with DSP4_50 mg/kg: p = 0.001; Z = −3.309). All other comparisons concerning the number of back beam breaks were not statistically significant. The numbers of front and back beam breaks are presented in Fig. 1.
Differences between methylphenidate doses within the DSP4 groups (for details see Tables 2, 3, 4 and 5)
Within the DSP4_control group (see also Table 2) the administration of 3 mg/kg methylphenidate led to a significant decrease in the parameters number of correct responses (p = 0.033; Z = −2.134), number of trials completed (p = 0.011; Z = −2.547) and the percentage of correct responses (p = 0.034; Z = −2.118), compared to the MPH_control condition. In addition, the animals of the DSP4_control group made statistically significant fewer omissions in the MPH_12 mg/kg condition compared to the treatment with 1 mg/kg methylphenidate (p = 0.014; Z = −2.452).
The DSP4_10 mg/kg group (Table 3) showed significantly fewer omissions when treated with 3 mg/kg methylphenidate and compared to the MPH_control condition (p = 0.034; Z = −2.121). In the DSP4_10 mg/kg group, no other difference between the methylphenidate doses reached statistical significance.
In the DSP4_20 mg/kg group (see Table 4), only one comparison between the methylphenidate conditions reached statistical significance. The rats made significantly more incorrect responses when treated with 12 mg/kg MPH compared to the MPH_1 mg/kg condition (p = 0.027; Z = −2.214). In this group, the other comparisons failed statistical significance.
As shown in Table 5, in the DSP4_50 mg/kg group none of the comparisons between the methylphenidate conditions were statistically significant.
Discussion
The first aim of this study was to investigate the role of noradrenaline in attentional processes in rats using the 5CSRTT. The results showed a decline of performance in regard to attention parameters in the 5CSRTT. The number of correct responses decreased in rats treated with DSP4. This effect was dose dependent with statistical significance for the high DSP4 dose compared to saline-treated animals and the small and middle DSP4 doses. The animals treated with DSP4_20 mg/kg missed significantly more signals in comparison to the other groups (i.e. saline, DSP4_10 mg/kg and DSP_50 mg/kg groups). This finding may suggest an inverted U curve for the DSP4 doses. In addition, the percentage of omissions was increased in all DSP4 groups compared to the saline-treated group reaching statistical significance for the middle and high DSP4 group compared to the saline-treated animals. These parameters indicate that attentional processes were negatively affected by the treatment with DSP4. Moreover, the rats of all DSP4 groups made more incorrect responses compared to the saline group. This effect was dose dependent. However, these comparisons failed to be statistically significant. This indicates a detrimental effect of DSP4 and is in line with previous findings showing some cognitive impairment following noradrenergic manipulations (Brozoski et al. 1979; Carli et al. 1983; Compton et al. 1995; Coull 1994; Roberts et al. 1976; Wenk et al. 1987).
The percentage of correct responses 5CSRTT has previously been used as a parameter of choice accuracy (Carli et al. 1983; Ruotsalainen et al. 1997). In this study, the percentage of correct responses was unaltered by the administration of DSP4, which is in line with previous findings based on the 5CSRTT. A study by Ruotsalainen et al. (1997) reported that the noradrenergic depletion induced by DSP4 did not affect the performance of rats, i.e. choice accuracy was not altered after DSP4 administration. Liu et al. (2015) have recently investigated the effects of noradrenergic depletion by DSP4 on attention and impulsivity, as measured by the 5CSRTT, in rats treated with atomoxetine or saline (Liu et al. 2015). These authors found that the pre-treatment with DSP4 had no influence on parameters measuring impulsivity (Liu et al. 2015). In regard to the parameters associated with attention (i.e. percentage of correct responses, percentage of omissions), the authors found no differences between the DSP4-treated and control groups (Liu et al. 2015). The behavioural paradigm used in this experiment was the same as in previous studies. However, there are some differences. Beside the fact that three different doses of DSP4 were used in this experiment in contrast to one dose in the study by Ruotsalainen et al. (1997), there is another important difference between these studies. Ruotsalainen et al. (1997) used a fixed ITI of 5 s, while variable ITIs were used in the present experiment. Carli et al. (1983) showed that an unpredictable stimulus presentation is important in the assessment of attention deficits. Consequently, Liu et al. (2015) used variable ITIs, but they used only the maximum dose of DSP4 (i.e. 50 mg/kg) in comparison to sham-treated rats (Liu et al. 2015). In regard to the inconsistent results of the present work and the study performed by Liu et al. (2015), one can only speculate about possible explanations, particularly in view of the fact that there are several similarities between the studies, i.e. the high dose of DSP4 used in this study and the pre-treatment with DSP4 used by Liu et al. (2015). In both studies, the rats were trained before the treatment with DSP4. Although variable ITIs were used in both studies, there was a remarkable difference which could explain the inconsistent findings, i.e. the ITIs varied from 0.5 to 4.5 s in this study, while Liu et al. (2015) used longer ITIs (i.e. 3, 5 and 7 s), which are more suitable to assess impulsivity. Moreover, in the present experiment a stimulus duration of 1.5 s was used in the testing phase. Liu et al. (2015) used variable stimulus durations (i.e. 0.5 and 1 s) in their testing sessions. All these experimental differences might explain the inconsistencies between previous studies and the present findings.
The present findings suggest both some impairment induced by the administration of DSP4 (as shown by a decrease in the number of correct responses, mixed results in the number of omissions, an increase in the percentage of omissions) and the absence of significant effects of DSP4 (as shown by the unaltered percentage of correct responses). These results are in agreement with previous findings (Carli et al. 1983; Ruotsalainen et al. 1997; Liu et al. 2015) and indicate that the administration of DSP4 can cause attention deficits as measured in the 5CSRTT.
However, there may be another plausible explanation for the present results. On closer examination these data show that the number of total responses (i.e. correct responses and incorrect responses) in the DSP4-treated groups in general, and in the high DSP4 dose in particular, was lower than in the saline-treated rats. This is supported by the fact that the DSP4-treated groups completed fewer trials than the saline-treated rats (number of trials completed). These effects were statistically significant for the high DSP4 dose compared to both saline-treated animals and the middle DSP4 dose. In addition, the DSP4 groups made more omissions in relation to the completed trials (percentage of omissions, significant for the high and middle DSP4 doses compared to saline). The changes in these parameters indicate a reduction in general activity rather than an impairment of attentional functions. This is supported by the finding that following DSP4 administration there were no significant changes in choice accuracy (as measured by the percentage of correct responses) which has been discussed as an important parameter of attentional processes (Carli et al. 1983; Ruotsalainen et al. 1997). In this context, one may also consider another aspect discussed by Robbins (2002) who argues that omission errors may reflect altered attention but can also be a consequence of changes in sensory, motor or motivational characteristics (Robbins 2002). It has also been suggested that the number of omissions typically increases with drug exposure, irrespective of the pharmacological target (Robbins 2002; Paine et al. 2007).
Although the 5CSRTT does not provide a special parameter of motor activity, the overall activity can be measured by the numbers of front beam breaks and back beam breaks. Interestingly, the DSP4-treated groups showed a reduction in the numbers of both front beam breaks and back beam breaks (most of the comparisons between the saline group and the DSP4 doses reached statistical significance, for details see Fig. 1), indicating a decrease in overall activity following DSP4 administration. This explanation is supported by the findings of Jones and Hess (2003) who investigated the role of noradrenaline with regard to the hyperactivity of the coloboma mutant mouse, an animal model of ADHD, by treating the animals with DSP4. The depletion of noradrenergic terminals by DSP4 reduced the motor hyperactivity in coloboma mutant mice. This is in accord with the observations of other authors who reported reduced explorative behaviour as a consequence of DSP4 administration (Delini-Stula et al. 1984; Harro et al. 1995; Skrebuhhova et al. 1999) and reduced hyperactivity as described above (Bruno et al. 2007). The question of whether or not the depletion of noradrenaline following the administration of DSP4 induces attention deficits or is responsible for a decrease in activity in general, possibly associated with altered motivation, cannot be answered sufficiently and therefore calls for further investigation.
The second aim of this experiment was to investigate the influence of the psychostimulant methylphenidate on DSP4-treated rats, which is commonly used in the pharmacological treatment of individuals with ADHD. The administration of methylphenidate had no beneficial effect on the performance of the saline-treated rats. Following the treatment with methylphenidate, the rats of the DSP4_control group showed fewer correct responses, fewer completed trials, a decrease in choice accuracy (i.e. percentage of correct responses; all significant for the comparison between the saline treatment and the middle dose of methylphenidate), mixed results in the number of omissions (significant for the comparison between the small and the high dose of methylphenidate) and the percentage of omissions (not significant) and a slight increase in the number of incorrect responses (no statistical significance). In all DSP4-treated groups, only two of the comparisons between the methylphenidate conditions reached statistical significance (the comparison between the MPH_control and the MPH_3 mg/kg dose for the number of omissions in the DSP4_10 mg/kg group and the comparison between the MPH_1 mg/kg and the MPH_12 mg/kg condition for the number of incorrect responses in the DSP4_20 mg/kg group). The treatment with methylphenidate in the DSP4-treated groups showed mixed results in the number of correct responses, the number of omissions and their percentage values and mixed results in the number of incorrect responses with only a few comparisons reaching statistical significance (see above). In regard to the number of completed trials all comparisons of the methylphenidate conditions failed statistical significance and no linear dose-response effects were found. These results are remarkable because well established and effective doses of methylphenidate were used (Koda et al. 2010; Koffarnus and Katz 2011; Navarra et al. 2008; Pattij et al. 2012; Slezak and Anderson 2011). The main difference between previous findings and the present results is the method used to induce noradrenergic depletion. With the exception of one study (Sontag et al. 2011), methylphenidate has not previously been used in DSP4-treated animals. In this study, the authors investigated the effects of methylphenidate on DSP4 pre-treated rats using a holeboard test to measure spatial memory performance (Sontag et al. 2011). Although the authors found changes (i.e. increased reference memory error in DSP4-treated rats) in the spatial memory performance after methylphenidate treatment (Sontag et al. 2011), the major difference between these findings and this experiment is the use of different behavioural test paradigms. This makes the comparison between the two studies difficult. These results may be explained in several ways: (1) on the basis of the findings of Cheetham et al. (1996) one can assume that the DSP4 doses used in this study reduced noradrenaline in the cortex of rats in a linear fashion by 51, 73 and 100%, respectively. Based on these data, one could conclude that the depletion by DSP4 was too severe to be compensated for by the methylphenidate doses used, although methylphenidate is a noradrenaline and dopamine reuptake inhibitor and affects, therefore, not only noradrenaline neurotransmission (Koda et al. 2010; Paterson et al. 2012). (2) It is possible that the methylphenidate doses used were too small to be effective in DSP4-treated rats. Interestingly, Bizarro et al. (2004) found significant improvements in the 5CSRTT including an increase in response accuracy when using similar methylphenidate doses (2.5–10 mg/kg). The main difference between this study and the experiment by Bizarro et al. (2004) is that methylphenidate was used in rats pre-treated with DSP4. Under the assumption that the noradrenergic depletion caused by DSP4 was too severe to be compensated for by the methylphenidate doses used, a reduction in the DSP4 doses may be more promising than an increase of the methylphenidate doses. It will, therefore, be necessary to find adequate doses of DSP4 and methylphenidate by assessing the dose-effect relationships of the two compounds. The present experiment could then be replicated using the new methylphenidate doses. (3) Finally, one could conclude that no functional relationship exists between central noradrenaline/dopamine and the behaviour measured in this study. The results discussed above concerning treatment with DSP4 and methylphenidate in the DSP4 control group as well as previous findings regarding DSP4 and methylphenidate indicate that a manipulation of noradrenergic and dopaminergic systems affects behavioural aspects as measured in this experiment (Carli et al. 1983; Compton et al. 1995; Coull 1994; Koda et al. 2010; Koffarnus and Katz 2011; Levin et al. 2011; Navarra et al. 2008; Pattij et al. 2012; Roberts et al. 1976; Slezak and Anderson 2011; Sontag et al. 2008, 2011; Wenk et al. 1987). Therefore, a functional relationship between noradrenaline and the behavioural aspects measured here is apparent.
In the DSP4_control group in particular, an improvement in performance was expected, not least because previous reports suggest a beneficial effect of methylphenidate (Koffarnus and Katz 2011; Navarra et al. 2008; Pattij et al. 2012; Slezak and Anderson 2011). Interestingly, no beneficial effect could be observed in the DSP4_control group without noradrenergic depletion. In this group, the rats made fewer correct responses (statistically significant for the middle methylphenidate dose), showed a decreased choice accuracy (percentage of correct responses; significant for the middle methylphenidate dose compared to the saline condition) and made more incorrect responses in all methylphenidate conditions compared to the saline treatment (comparisons failed statistical significance). Moreover, methylphenidate treatment caused an increase in the number of omissions for the comparison between the small methylphenidate dose and the saline condition (not significant), and a decrease in the middle and high methylphenidate doses compared to the other conditions (statistical significance only for the comparison between the small methylphenidate dose and the treatment with the high methylphenidate dose, the comparison with the saline condition failed statistical significance). Taken together, these data suggest a detrimental effect of methylphenidate on parameters associated with attentional functions (represented by a decrease in correct responses and choice accuracy with a concomitant increase of incorrect responses and mixed results in the number and percentage of omissions). Some authors have previously reported an increase in correct responses using the 5CSRTT (Koffarnus and Katz 2011; Navarra et al. 2008; Paterson et al. 2012), while others found a detrimental effect following methylphenidate administration (Pattij et al. 2012). Therefore, the present findings disagree with previous results suggesting a beneficial effect of methylphenidate on correct responses (Koffarnus and Katz 2011; Navarra et al. 2008; Paterson et al. 2012) and are in line with the findings of Pattij et al. (2012) who also found a reduction in correct responses.
In conclusion, the present findings showed that treatment with DSP4 leads to marked impairments in cognitive performance as measured with the 5CSRTT. Whether or not these impairments are due to attention deficits or changes in general explorative activity remains to be investigated. Taken together, the administration of methylphenidate had no effect on the performance of DSP4-treated rats, regardless of the DSP4 doses used. The most likely explanation for this phenomenon is that the methylphenidate doses were too small. It therefore would be useful to investigate the effects of more suitable doses of methylphenidate following noradrenergic depletion, or to use lower DSP4 doses. Finally, the administration of methylphenidate to drug-naive rats caused detrimental effects on parameters associated with attention.
Change history
07 August 2017
An erratum to this article has been published.
References
Al Zahrani SS, Al Ruwaitea AS, Ho MY, Bradshaw CM, Szabadi E (1997) Destruction of central noradrenergic neurones with DSP4 impairs the acquisition of temporal discrimination but does not affect memory for duration in a delayed conditional discrimination task. Psychopharmacology 130(2):166–173
Arnsten AF (1997) Catecholamine regulation of the prefrontal cortex. J Psychopharmacol 11(2):151–162
Arnsten AF (2011) Catecholamine influences on dorsolateral prefrontal cortical networks. Biol Psychiatry 69(12):e89–e99
Arnsten AF, Dudley AG (2005) Methylphenidate improves prefrontal cortical cognitive function through alpha2 adrenoceptor and dopamine D1 receptor actions: relevance to therapeutic effects in attention deficit hyperactivity disorder. Behav Brain Funct 1(1):2
Arnsten AF, Steere JC, Hunt RD (1996) The contribution of alpha 2-noradrenergic mechanisms of prefrontal cortical cognitive function. Potential significance for attention-deficit hyperactivity disorder. Arch Gen Psychiatry 53(5):448–455
Aston-Jones G, Rajkowski J, Kubiak P, Alexinsky T (1994) Locus coeruleus neurons in monkey are selectively activated by attended cues in a vigilance task. J Neurosci 14(7):4467–4480
Aston-Jones G, Rajkowski J, Kubiak P (1997) Conditioned responses of monkey locus coeruleus neurons anticipate acquisition of discriminative behavior in a vigilance task. Neuroscience 80(3):697–715
Banaschewski T, Brandeis D, Heinrich H, Albrecht B, Brunner E, Rothenberger A (2003) Association of ADHD and conduct disorder–brain electrical evidence for the existence of a distinct subtype. J Child PsycholPsychiatry 44(3):356–376
Barkley RA (2006) Attention-deficit hyperactivity disorder: a handbook for diagnosis and treatment, vol 3rd. Guilford Press, New York
Bear MF (1999) Homosynaptic long-term depression: a mechanism for memory? Proc Natl Acad Sci USA 96(17):9457–9458
Benloucif S, Bennett EL, Rosenzweig MR (1995) Norepinephrine and neural plasticity: the effects of xylamine on experience-induced changes in brain weight, memory, and behavior. Neurobiol Learn Mem 63(1):33–42
Biederman J, Faraone SV (2005) Attention-deficit hyperactivity disorder. Lancet 366(9481):237–248
Biederman J, Faraone SV, Keenan K, Benjamin J, Krifcher B, Moore C, Sprich-Buckminster S, Ugaglia K, Jellinek MS, Steingard R (1992) Further evidence for family-genetic risk factors in attention deficit hyperactivity disorder. Patterns of comorbidity in probands and relatives psychiatrically and pediatrically referred samples. Arch Gen Psychiatry 49(9):728–738
Biederman J, Faraone SV, Mick E, Spencer T, Wilens T, Kiely K, Guite J, Ablon JS, Reed E, Warburton R (1995) High risk for attention deficit hyperactivity disorder among children of parents with childhood onset of the disorder: a pilot study. Am J Psychiatry 152(3):431–435
Bizarro L, Patel S, Murtagh C, Stolerman IP (2004) Differential effects of psychomotor stimulants on attentional performance in rats: nicotine, amphetamine, caffeine and methylphenidate. Behav Pharmacol 15(3):195–206
Brozoski TJ, Brown RM, Rosvold HE, Goldman PS (1979) Cognitive deficit caused by regional depletion of dopamine in prefrontal cortex of rhesus monkey. Science 205(4409):929–932
Bruno KJ, Freet CS, Twining RC, Egami K, Grigson PS, Hess EJ (2007) Abnormal latent inhibition and impulsivity in coloboma mice, a model of ADHD. Neurobiol Dis 25(1):206–216
Carli M, Robbins TW, Evenden JL, Everitt BJ (1983) Effects of lesions to ascending noradrenergic neurones on performance of a 5-choice serial reaction task in rats; implications for theories of dorsal noradrenergic bundle function based on selective attention and arousal. Behav Brain Res 9(3):361–380
Cheetham SC, Viggers JA, Butler SA, Prow MR, Heal DJ (1996) [3H]nisoxetine–a radioligand for noradrenaline reuptake sites: correlation with inhibition of [3H]noradrenaline uptake and effect of DSP-4 lesioning and antidepressant treatments. Neuropharmacology 35(1):63–70
Clark CR, Geffen GM, Geffen LB (1987) Catecholamines and attention. I: animal and clinical studies. Neurosci Biobehav Rev 11(4):341–352
Clements KM, Girard TA, Xing HC, Wainwright PE (2003) Spontaneously hypertensive and Wistar Kyoto rats differ in delayed matching-to-place performance and response to dietary long-chain polyunsaturated fatty acids 11. Dev Psychobiol 43(1):57–69
Cole BJ, Robbins TW (1989) Effects of 6-hydroxydopamine lesions of the nucleus accumbens septi on performance of a 5-choice serial reaction time task in rats: implications for theories of selective attention and arousal. Behav Brain Res 33(2):165–179
Collins P, Roberts AC, Dias R, Everitt BJ, Robbins TW (1998) Perseveration and strategy in a novel spatial self-ordered sequencing task for nonhuman primates: effects of excitotoxic lesions and dopamine depletions of the prefrontal cortex. J Cogn Neurosci 10(3):332–354
Compton DM, Dietrich KL, Smith JS, Davis BK (1995) Spatial and non-spatial learning in the rat following lesions to the nucleus locus coeruleus. Neuro Report 7(1):177–182
Coull JT (1994) Pharmacological manipulations of the alpha 2-noradrenergic system. Effects on cognition. Drugs Aging 5(2):116–126
Crofts HS, Dalley JW, Collins P, Van Denderen JC, Everitt BJ, Robbins TW, Roberts AC (2001) Differential effects of 6-OHDA lesions of the frontal cortex and caudate nucleus on the ability to acquire an attentional set. Cereb Cortex 11(11):1015–1026
Davids E, Zhang K, Tarazi FI, Baldessarini RJ (2003) Animal models of attention-deficit hyperactivity disorder. Brain Res Brain ResRev 42(1):1–21
Delini-Stula A, Mogilnicka E, Hunn C, Dooley DJ (1984) Novelty-oriented behavior in the rat after selective damage of locus coeruleus projections by DSP-4, a new noradrenergic neurotoxin. Pharmacol Biochem Behav 20(4):613–618
Deroche V, Marinelli M, Maccari S, Le Moal M, Simon H, Piazza PV (1995) Stress-induced sensitization and glucocorticoids. I. Sensitization of dopamine-dependent locomotor effects of amphetamine and morphine depends on stress-induced corticosterone secretion. J Neurosci 15(11):7181–7188
Döpfner M (1999) Hyperkinetische Störungen. In: Petermann F (ed) Lehrbuch der klinischen Kinderpsychologie und -psychotherapie, 4th edn. Hogrefe Verlag, Göttingen, pp 151–186
Everitt BJ, Robbins TW, Selden NR (2009) Functions of the locus coeruleus noradrenergic system: a neurobiological and behavioural synthesis. In: Heal DJ, Marsden CA (eds) The pharmacology of noradrenaline in the central nervous system. Oxford University Press, Oxford, pp 349–378
Fritschy JM, Grzanna R (1991) Experimentally-induced neuron loss in the locus coeruleus of adult rats. Exp Neurol 111(1):123–127
Halperin JM, Newcorn JH, Schwartz ST, Sharma V, Siever LJ, Koda VH, Gabriel S (1997) Age-related changes in the association between serotonergic function and aggression in boys with ADHD. Biol Psychiatry 41(6):682–689
Harley CW (1987) A role for norepinephrine in arousal, emotion and learning?: limbic modulation by norepinephrine and the Kety hypothesis. Prog Neuropsychopharmacol Biol Psychiatry 11(4):419–458
Harro J, Oreland L, Vasar E, Bradwejn J (1995) Impaired exploratory behaviour after DSP-4 treatment in rats: implications for the increased anxiety after noradrenergic denervation. Eur Neuropsychopharmacol 5(4):447–455
Heal DJ, Smith SL, Kulkarni RS, Rowley HL (2008) New perspectives from microdialysis studies in freely-moving, spontaneously hypertensive rats on the pharmacology of drugs for the treatment of ADHD. Pharmacol Biochem Behav 90(2):184–197
Huff JK, Davies MI (2002) Microdialysis monitoring of methylphenidate in blood and brain correlated with changes in dopamine and rat activity. J Pharm Biomed Anal 29(5):767–777
Jones MD, Hess EJ (2003) Norepinephrine regulates locomotor hyperactivity in the mouse mutant coloboma. Pharmacol Biochem Behav 75(1):209–216
Jonge AD, van Meel JC, Timmermans PB, van Zwieten PA (1981) A lipophilic, selective alpha1 -adrenoceptor agonist: 2-(2-chloro-5-trifluoromethylphenylimino)imida-zolidine (St 587). Life Sci 28(18):2009–2016
Koda K, Ago Y, Cong Y, Kita Y, Takuma K, Matsuda T (2010) Effects of acute and chronic administration of atomoxetine and methylphenidate on extracellular levels of noradrenaline, dopamine and serotonin in the prefrontal cortex and striatum of mice. J Neurochem 114(1):259–270
Koffarnus MN, Katz JL (2011) Response requirement and increases in accuracy produced by stimulant drugs in a 5-choice serial reaction-time task in rats. Psychopharmacology 213(4):723–733
Lange KW, Tucha L, Walitza S, Gerlach M, Linder M, Tucha O (2007) Interaction of attention and graphomotor functions in children with attention deficit hyperactivity disorder. J Neural Transm Suppl 72:249–259
Lange KW, Reichl S, Lange KM, Tucha L, Tucha O (2010) The history of attention deficit hyperactivity disorder. Atten Defic Hyperact Disord 2(4):241–255
Langlais PJ, Connor DJ, Thal L (1993) Comparison of the effects of single and combined neurotoxic lesions of the nucleus basalis magnocellularis and dorsal noradrenergic bundle on learning and memory in the rat. Behav Brain Res 54(1):81–90
Levin ED, Bushnell PJ, Rezvani AH (2011) Attention-modulating effects of cognitive enhancers. Pharmacol Biochem Behav 99(2):146–154
Liu YP, Huang TS, Tung CS, Lin CC (2015) Effects of atomoxetine on attention and impulsivity in the five-choice serial reaction time task in rats with lesions of dorsal noradrenergic ascending bundle. Prog Neuropsychopharmacol Biol Psychiatry 56:81–90
McGaugh JL (1989) Involvement of hormonal and neuromodulatory systems in the regulation of memory storage. Annu Rev Neurosci 12:255–287
Mefford IN, Potter WZ (1989) A neuroanatomical and biochemical basis for attention deficit disorder with hyperactivity in children: a defect in tonic adrenaline mediated inhibition of locus coeruleus stimulation. Med Hypotheses 29(1):33–42
Micheletti G, Warter JM, Marescaux C, Depaulis A, Tranchant C, Rumbach L, Vergnes M (1987) Effects of drugs affecting noradrenergic neurotransmission in rats with spontaneous petit mal-like seizures. Eur J Pharmacol 135(3):397–402
Navarra R, Graf R, Huang Y, Logue S, Comery T, Hughes Z, Day M (2008) Effects of atomoxetine and methylphenidate on attention and impulsivity in the 5-choice serial reaction time test. Prog Neuropsychopharmacol Biol Psychiatry 32(1):34–41
Oades RD (1987) Attention deficit disorder with hyperactivity (ADDH): the contribution of catecholaminergic activity. Prog Neurobiol 29(4):365–391
Ohno M, Yamamoto T, Kobayashi M, Watanabe S (1993) Impairment of working memory induced by scopolamine in rats with noradrenergic DSP-4 lesions. Eur J Pharmacol 238(1):117–120
Ohno M, Yoshimatsu A, Kobayashi M, Watanabe S (1997) Noradrenergic DSP-4 lesions aggravate impairment of working memory produced by hippocampal muscarinic blockade in rats. Pharmacol Biochem Behav 57(1–2):257–261
Paine TA, Tomasiewicz HC, Zhang K, Carlezon WA Jr (2007) Sensitivity of the five-choice serial reaction time task to the effects of various psychotropic drugs in Sprague–Dawley rats. Biol Psychiatry 62(6):687–693. doi:10.1016/j.biopsych.2006.11.017
Paterson NE, Wetzler C, Hackett A, Hanania T (2012) Impulsive action and impulsive choice are mediated by distinct neuropharmacological substrates in rat. Int J Neuropsychopharmacol 15(10):1473–1487
Pattij T, Schetters D, Schoffelmeer AN, van Gaalen MM (2012) On the improvement of inhibitory response control and visuospatial attention by indirect and direct adrenoceptor agonists. Psychopharmacology 219(2):327–340
Pichler L, Kobinger W (1985) Possible function of alpha 1-adrenoceptors in the CNS in anaesthetized and conscious animals. Eur J Pharmacol 107(3):305–311
Pliszka SR, McCracken JT, Maas JW (1996) Catecholamines in attention-deficit hyperactivity disorder: current perspectives. J Am Acad Child Adolesc Psychiatry 35(3):264–272
Pothos EN, Hernandez L, Hoebel BG (1995) Chronic food deprivation decreases extracellular dopamine in the nucleus accumbens: implications for a possible neurochemical link between weight loss and drug abuse. Obes Res 3(Suppl 4):525S–529S
Raskin LA, Shaywitz BA, Anderson GM, Cohen DJ, Teicher MH, Linakis J (1983) Differential effects of selective dopamine, norepinephrine or catecholamine depletion on activity and learning in the developing rat. Pharmacol Biochem Behav 19(5):743–749
Riekkinen P Jr, Lammintausta R, Ekonsalo T, Sirvio J (1993) The effects of alpha 2-adrenoceptor stimulation on neocortical EEG activity in control and 6-hydroxydopamine dorsal noradrenergic bundle-lesioned rats. Eur J Pharmacol 238(2–3):263–272
Robbins TW (2002) The 5-choice serial reaction time task: behavioural pharmacology and functional neurochemistry. Psychopharmacology 163(3–4):362–380
Roberts DC, Price MT, Fibiger HC (1976) The dorsal tegmental noradrenergic projection: an analysis of its role in maze learning. J Comp Physiol Psychol 90(4):363–372
Roberts AC, De Salvia MA, Wilkinson LS, Collins P, Muir JL, Everitt BJ, Robbins TW (1994) 6-Hydroxydopamine lesions of the prefrontal cortex in monkeys enhance performance on an analog of the Wisconsin Card Sort Test: possible interactions with subcortical dopamine. J Neurosci 14(5 Pt 1):2531–2544
Ruotsalainen S, Haapalinna A, Riekkinen PJ Sr, Sirvio J (1997) Dexmedetomidine reduces response tendency, but not accuracy of rats in attention and short-term memory tasks. Pharmacol Biochem Behav 56(1):31–40
Russell VA (2002) Hypodopaminergic and hypernoradrenergic activity in prefrontal cortex slices of an animal model for attention-deficit hyperactivity disorder—the spontaneously hypertensive rat. Behav Brain Res 130(1–2):191–196
Russell VA, Sagvolden T, Johansen EB (2005) Animal models of attention-deficit hyperactivity disorder. Behav Brain Funct 1:9
Skrebuhhova T, Allikmets L, Matto V (1999) [3H]-ketanserin binding and elevated plus-maze behavior after chronic antidepressant treatment in DSP-4 and P-CPA pretreated rats: evidence for partial involvement of 5-HT2A receptors. Methods Find Exp Clin Pharmacol 21(7):483–490
Slezak JM, Anderson KG (2011) Effects of acute and chronic methylphenidate on delay discounting. Pharmacol Biochem Behav 99(4):545–551
Solanto MV (1998) Neuropsychopharmacological mechanisms of stimulant drug action in attention-deficit hyperactivity disorder: a review and integration. Behav Brain Res 94(1):127–152
Sontag TA, Tucha O, Walitza S, Lange KW (2010) Animal models of attention deficit/hyperactivity disorder (ADHD): a critical review. Atten Defic Hyperact Disord 2(1):1–20
Sontag TA, Hauser J, Tucha O, Lange KW (2011) Effects of DSP4 and methylphenidate on spatial memory performance in rats. Atten Defic Hyperact Disord 3(4):351–358
Swanson JM, Sunohara GA, Kennedy JL, Regino R, Fineberg E, Wigal T, Lerner M, Williams L, LaHoste GJ, Wigal S (1998) Association of the dopamine receptor D4 (DRD4) gene with a refined phenotype of attention deficit hyperactivity disorder (ADHD): a family-based approach. Mol Psychiatry 3(1):38–41
Teicher MH, Barber NI, Reichheld JH, Baldessarini RJ, Finklestein SP (1986) Selective depletion of cerebral norepinephrine with 6-hydroxydopamine and GBR-12909 in neonatal rat. Brain Res 395(1):124–128
Teicher MH, Lowen SB, Polcari A, Foley M, McGreenery CE (2004) Novel strategy for the analysis of CPT data provides new insight into the effects of methylphenidate on attentional states in children with ADHD. J Child Adolesc Psychopharmacol 14(2):219–232
Tucha O, Lange KW (2001) Effects of methylphenidate on kinematic aspects of handwriting in hyperactive boys. J Abnorm Child Psychol 29(4):351–356
Tucha O, Prell S, Mecklinger L, Bormann-Kischkel C, Kubber S, Linder M, Walitza S, Lange KW (2006) Effects of methylphenidate on multiple components of attention in children with attention deficit hyperactivity disorder. Psychopharmacology 185(3):315–326. doi:10.1007/s00213-006-0318-2
Tucha L, Tucha O, Laufkotter R, Walitza S, Klein HE, Lange KW (2008) Neuropsychological assessment of attention in adults with different subtypes of attention-deficit/hyperactivity disorder. J Neural Transm (Vienna) 115(2):269–278. doi:10.1007/s00702-007-0836-z
Wankerl B, Hauser J, Makulska-Gertruda E, Reissmann A, Sontag TA, Tucha O, Lange KW (2014) Neurobiology of attention deficit hyperactivity disorder. Fortschr Neurol Psychiatr 82(1):9–29
Wenk G, Hughey D, Boundy V, Kim A, Walker L, Olton D (1987) Neurotransmitters and memory: role of cholinergic, serotonergic, and noradrenergic systems. Behav Neurosci 101(3):325–332
Ross SB, Stenfors C (2015) DSP4, a selective neurotoxin for the locus coeruleus noradrenergic system. A review of its mode of action. Neurotox Res 27(1):15–30
Sontag TA, Hauser J, Kaunzinger I, Gerlach M, Tucha O, Lange KW (2008) Effects of the noradrenergic neurotoxin DSP4 on spatial memory in the rat. J Neural Transm (Vienna) 115(2):299–303
Yang PB, Amini B, Swann AC, Dafny N (2003) Strain differences in the behavioral responses of male rats to chronically administered methylphenidate. Brain Res 971(2):139–152
Yang PB, Swann AC, Dafny N (2006) Dose-response characteristics of methylphenidate on locomotor behavior and on sensory evoked potentials recorded from the VTA, NAc, and PFC in freely behaving rats. Behav Brain Funct 2:3. doi:10.1186/1744-9081-2-3
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article is available at https://doi.org/10.1007/s00702-017-1755-2.
Rights and permissions
About this article
Cite this article
Hauser, J., Reissmann, A., Sontag, TA. et al. Effects of methylphenidate on attention in Wistar rats treated with the neurotoxin N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine (DSP4). J Neural Transm 124, 643–654 (2017). https://doi.org/10.1007/s00702-017-1679-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-017-1679-x