Abstract
Background
Symptomatic midline sacral meningeal cysts (MSMC) are rare, and, as a consequence, so are reports on the surgical techniques to address these lesions. Here we provide a description of the senior author’s (ATC) technique.
Method
A sacral laminectomy is performed. The cyst’s relation with the dural sac and sacral nerves is inspected; it is then opened and drained. Its lumen is explored for its point of communication with the dural sac, and this ostium is closed off with non-penetrating clips. A lumbar drain is inserted in select cases.
Conclusion
Cyst wall resection is unnecessary and closing the ostium is sufficient to treat MSMC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Relevant surgical anatomy
The sacrum constitutes a fused block of five vertebrae. In the anteroposterior view, it resembles a triangle with its apex pointing inferiorly. In the sagittal view, it is curved, with a convex dorsum. From midline to lateral, the fused posterior elements of the sacral vertebrae form the median sacral crest (fused rudimentary spinous processes), the sacral grooves (fused laminae), the intermediate sacral crests (fused articular processes) and the lateral sacral crests (fused transverse processes). The four pairs of posterior sacral neuroforamina lie just lateral to the intermediate sacral crests. Cranially, the intermediate sacral crests lead to the superior articular processes of S1, which articulate with the L5 vertebra, and caudally to the inferior articular processes of S5, palpable prominences on either side of the sacral hiatus also called the sacral cornua. The median sacral crest and sacral grooves are absent beyond the inferior portion of S4 and so form the sacral hiatus [1]. However, variations are not infrequent, and the hiatal apex can extend as high up as S2 [6]. Also, the presence of transitional vertebrae may be associated with a persistent S1/S2 interlaminar space.
The space located between the intermediate crests, capped by the fused laminae, constitutes the sacral spinal canal. It contains the caudal end of the dural sac—which usually terminates at the level of S2—five pairs of sacral and one pair of coccygeal nerve roots, epidural adipose tissue, a rich epidural venous plexus that is mostly ventrally situated and the filum terminale [7].
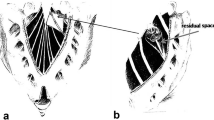
Midline sacral meningeal cysts (MSMC) are also referred to in the literature as sacral meningeal diverticula or sacral meningoceles. They are often confused with sacral perineurial cysts—also called Tarlov cysts—due to their sacral localisation, their cerebrospinal fluid (CSF) content and their tendency for bony erosion (Fig. 1). They are however distinct patho-anatomical entities [5, 8]. Whilst both are extradural cysts, in contrast to Tarlov cysts, MSMC do not incorporate nerve root fibres within their cyst wall or within their lumen (Fig. 1b; Fig. 2), which is at the base of their classification into type II and type Ib spinal meningeal cysts, respectively [5]. MSMC have fibrous walls (dural diverticula) with a pedicle leading to the terminal dural sac (Fig. 2; Fig. 3). The aim of surgery is to identify and close this communication [5].
a Sagittal T2-weighted lumbosacral MRI slice demonstrating a midline sacral meningeal cyst extending from mid S2 to the S4/S5 junction. b Axial slice from the same imaging study. The inset in the right upper corner indicates the level of the axial slice. The blue arrows indicate the S3 nerve roots pushed ventrally by the dorsally growing cyst, and the white arrows indicate the exited S2 nerve roots. Note the ventral and dorsal sacral bony erosion on both panels
Illustration demonstrating the patho-anatomical distinction between perineurial (Tarlov) and midline sacral meningeal cysts. A posterior view of the sacrum is shown, following complete laminectomies, with sections of dura removed to provide a window view within. The depicted S1 nerve roots exemplify normal anatomy. The S2 nerve roots are affected by Tarlov cysts, which are perineurial space dilatations of the nerve root sheaths (i.e. between the endoneurium and perineurium). The nerve root fibres are either splayed out in the cyst’s wall or run through the cyst’s lumen. In comparison, a midline sacral meningeal cyst—seen here overlying the tip of the thecal sac, with its ostium located on the thecal sac’s dorsal surface—does not contain nerve roots. This cyst type is a meningeal diverticulum and has a dural lining. Its trans-ostial communication with the subarachnoid space can cause it to progressively fill up with cerebrospinal fluid and build up pressure on the sacral nerves coursing around it
a–j High-resolution T2-weighted sacral MRI in patient from Fig. 1. a, c, e, g, i Oblique coronal slices through the midline sacral meningeal cyst (MSMC) and b, d, f, h, j sagittal slice through the midline, with blue line indicating the level of each oblique slice above it. The laterality of the oblique slices has been inversed to match the intraoperative view (k) so that the left side of each panel corresponds to the patient’s left (L: left; R: right). k The intraoperative view is within the MSMC’s lumen, looking at the region of its ostium. The tip of the dural sac communicates with the MSMC through a pedicle with complex septations: A fold in the MSMC’s wall forms a septum (a, k; hollow arrowhead). Towards its base, the septum splits into two smaller septations, between which a small CSF-filled cyst is found. This small structure is in communication with the lumbosacral cistern (g, i). k It is seen between the two branches of the forceps. It has an arachnoid lining and is covered by a thin membrane, continuous with the thicker fibrous wall of the MSMC. This covering is only partial so that a portion of the small cyst’s arachnoid lining effectively protrudes into the lumen of the MSMC, likely constituting a ball-and-valve mechanism that opens when the lumbar cistern’s hydrostatic pressure is greater than that within the MSMC (e.g. when standing or straining). CSF can be seen pearling during the dissection of the thin membrane of the dome of the small cyst. a–j Note a sedimentation level, corresponding to haematic contamination from a CT-guided drainage performed 2 weeks earlier (see Video legend for patient history)
Description of the technique
After prone positioning, a fluoroscopy identifies the projection to the skin of the L5/S1 segment and of the level of the diverticulum’s ostium. A midline incision is made, the thoracolumbar fascia is incised on either side of the midline and the underlying musculature is stripped laterally to expose the L5/S1 interlaminar space and the dorsal sacral surface (Fig. 4a). The L5/S1 midline ligamentous structures are resected, and a multilevel sacral laminectomy is performed by drilling cranio-caudally at the interface between the sacral groove and the intermediate crest, and transversally through the caudally exposed sacral grooves and median crest.
Intraoperative views through the surgical microscope, in the patient from Figs. 1 and 3, following a a midline approach to the L5/S1 interlaminar window and dorsal sacrum (L, lumbar; S, sacrum; the patient’s left is to the bottom of the panel) and b sacral laminectomy from S1 to S3. The asterisk indicates the dural sac. The hollow arrow indicates the epidural fat pad at the junction between the dural sac and the MSMC, seen on the sagittal 2-weighted MRI inset. c–f View within the cyst’s lumen: c The ostium presents as a thinning in the MSMC’s wall. Vessels are seen entering through it. d The ostium is widened with a blunt hook, e revealing a CSF-filled cyst in continuity with the lumbosacral cistern. The arachnoid lining of this small cyst is incised (f) and this opening is enlarged with a blunt hook allowing to bring down arachnoid septations within. This allows to incorporate the sheets of arachnoid into the ostial closure along with the opposing sheets of the diverticular wall. g For this, we use non-penetrating clips (AnastoClip®; LeMaitre Vascular, Inc., Burlington, MA, USA). h The dural repair is reinforced with alternately applied layers of collagen-based dural regeneration matrix (shown here, DuraGen®; Integra®, Plainsboro, NJ, USA) and fibrin sealant (Evicel®; Ethicon™, Somerville, NJ, USA)
The exposed contents of the sacral canal are inspected. The cyst’s relation to the dural sac and nerve roots is explored (Fig. 4b). MSMC can either grow dorsally to the sacral nerve roots (Fig. 1; Fig. 4b) or ventrally to them, in which case the nerve roots are displaced dorsally and may need to be dissected off the cyst’s dome. The cyst is incised and its lumen is explored for its communication with the terminal dural sac (Fig. 4c). The ostium may not be readily identifiable. This may be due to complex septations within the pedicle leading to the dural sac (Fig. 3; Fig. 4c–f). A Valsalva manoeuvre may help to identify the location of the ostium, as it begins to pearl with CSF. It may also present with vessels passing through it and leading to the cyst wall (Fig. 4c) [5]. An intraoperative fluoroscopy is useful to confirm the level of interest identified on preoperative MRI.
If the ostium is evident, it can simply be closed off. For this, we use non-penetrating clips (AnastoClip®; LeMaitre Vascular, Inc., Burlington, MA, USA). If the ostium is not evident, it may be necessary to open its septations and search for the communication with the subarachnoid space (Fig. 4d–f), before closing it off (Fig. 4g). A lumbar drain (LD) is advised in cases of significant leakage of CSF and is inserted intraoperatively through the L5/S1 interlaminar window. Closure involves epidural layers of collagen-based dural regeneration matrix and fibrin sealant (Fig. 4h).
Indications
Invalidating symptoms attributable to MSMC that have failed conservative management and for which alternative causes have been explored.
Symptoms include lumbosacral pain, radicular pain, sensorimotor disturbance in the perineum and lower limbs, and uro-anal sphincter disturbance. Symptoms may worsen with standing or Valsalva manoeuvres [2, 4, 5, 8].
Limitations
In MSMC growing ventrally to the dural sac, the ostium will likely be in relation to the dural sac’s ventral surface. It is therefore hidden from the surgical line of view and so requires the sac to be rotated to a degree [3].
How to avoid complications
-
Detailed study of preoperative imaging to avoid neural injury intraoperatively:
-
Variation of bony anatomy (e.g. persistent S1/S2 interlaminar space, high apex of sacral hiatus);
-
MSMC-induced thinning of dorsal sacral bone;
-
Ventrally growing MSMC relative to the dural sac, causing dorsal displacement of nerve roots against the sacral laminae [3].
-
-
Copious disinfection due to incision’s proximity with the gluteal region.
-
Drape wide in anticipation of an eventual LD (lateral exit point) so that it does not pass through the drapes during tunnelling, as this carries a risk of pull out during undraping.
-
Seeing that aggressive resection of the MSMC’s walls is not necessary, the caudal extent of the skin incision and sacral laminectomy can be limited to exposing only the portion of the cyst harbouring the ostium.
-
Avoid passing the LD through the fascial incision, as it may get caught into the suture or interfere with adequate fascial closure. Instead, tunnel the LD at a distance from the fascial incision.
-
Watertight closure of fascia.
Specific information for the patient
-
If LD is inserted, 3–5 day flat bed rest before LD removal.
-
Risk of cyst recurrence (dependent on the solidity of ostial closure).
Data availability
Not applicable.
Abbreviations
- CSF:
-
Cerebrospinal fluid
- LD:
-
Lumbar drain
- MSMC:
-
Midline sacral meningeal cyst
- MRI:
-
Magnetic resonance imaging
References
Cheng JS, Song JK (2003) Anatomy of the sacrum. Neurosurg Focus 15:E3. https://doi.org/10.3171/foc.2003.15.2.3
Feigenbaum F, Henderson F (2006) Surgical management of meningeal cysts, including perineural (Tarlov) cysts and meningeal diverticula. Semin Spine Surg 18:154–160. https://doi.org/10.1053/j.semss.2006.06.004
Feigenbaum F, Henderson FC (2006) Giant sacral meningeal diverticula: surgical implications of the “thecal tip” sign. Report of two cases. J Neurosurg Spine 5:443–446. https://doi.org/10.3171/spi.2006.5.5.443
Fogel GR, Cunningham PY 3rd, Esses SI (2004) Surgical evaluation and management of symptomatic lumbosacral meningeal cysts. Am J Orthop (Belle Mead NJ) 33:278–282
Nabors MW, Pait TG, Byrd EB, Karim NO, Davis DO, Kobrine AI, Rizzoli HV (1988) Updated assessment and current classification of spinal meningeal cysts. J Neurosurg 68:366–377. https://doi.org/10.3171/jns.1988.68.3.0366
Nastoulis E, Karakasi MV, Pavlidis P, Thomaidis V, Fiska A (2019) Anatomy and clinical significance of sacral variations: a systematic review. Folia Morphol (Warsz) 78:651–667. https://doi.org/10.5603/FM.a2019.0040
Sakka L, Gabrillargues J, Coll G (2016) Anatomy of the spinal meninges. Oper Neurosurg (Hagerstown) 12:168–188. https://doi.org/10.1227/NEU.0000000000001048
Tarlov IM (1970) Spinal perineurial and meningeal cysts. J Neurol Neurosurg Psychiatry 33:833–843. https://doi.org/10.1136/jnnp.33.6.833
Author information
Authors and Affiliations
Contributions
All authors contributed to the manuscript’s conception and design. The first draft was written by Ivan Cabrilo and critically revised by all three authors. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Key points
▪ Although rare, the intracystic buildup of CSF within MSMC can cause symptomatic compression of the sacral nerve roots.
▪ MSMC are meningeal diverticula, connected to the tip of the dural sac through a pedicle that is thought to function as a ball-and-valve mechanism [5].
▪ Simple drainage is therefore usually associated with recurrence [2] and closure of the ostium is required.
▪ Closure of the ostium is usually sufficient to treat MSMC, and aggressive resection of the cyst’s walls is not necessary.
▪ The incision and approach are therefore limited to exposing the portion of the cyst where the ostium is located.
▪ Careful muscle stripping and drilling, to avoid inadvertently breaking into thinned down or absent dorsal sacral bone.
▪ The ostium may not be apparent.
▪ Valsalva manoeuvres may help to locate the ostium.
▪ Complex septations may need to be taken down before the ostium can be closed off.
▪ Non-penetrating clips are used for closure, to avoid suturing needle puncture holes.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Spine—Other
Supplementary Information
Below is the link to the electronic supplementary material.
Video. Surgical video, through operating microscope, of patient from Figures 1, 3-4. Patient history: 18-year-old female, previously fit and well, presented with pain in the lower back and lower abdomen associated with a progressive disturbance of bladder sensation and voiding, and ultimately with urinary retention that had initially been attributed to Fowler’s syndrome. Multiple trials without urinary catheter had failed. Shooting groin pain and gluteal region paraesthesia were also reported. Further radiological work-up revealed a large midline sacral meningeal cyst that was drained percutaneously under CT guidance. A near-immediate improvement of symptoms was reported and the patient was once more able to pass urine normally and was no longer catheter-dependent. 10 days later, her symptoms progressively re-appeared, and she was again found to be in urinary retention. An MRI demonstrated that the cyst had recollected, at which point she was referred to our hospital for consideration of surgical management, to which she and her family consented. Video commentary: The L5/S1 window and dorsal sacrum are exposed through a midline incision. The incision and laminectomy are limited to exposing the portion of the cyst where the ostium is located. Flowable haemostatic matrix (Surgiflo®; EthiconTM, Somerville, NJ, USA) is useful to control epidural venous bleeding (00:11 – 00:31). An intraoperative fluoroscopy (00:31 – 00:40) aids to orientate to the level of the cyst’s ostium (S2/S3 junction, see Figures 1 & 3). The absence of dorsally running nerve roots is confirmed (00:41 – 00:46). (00:46 – 01:23) The cyst is entered through a tear in its dome and its lumen is explored for the pedicle that connects it to the dural sac, without a clear communication found. A Valsalva manoeuvre causes CSF to pearl at the base of the intracystic septum, where the wall also appears thinner and where vessels are seen entering into the cyst (01:15, asterisk): these all represent give-away signs to the ostium’s location. (01:24 – 01:47) Blunt dissection of this region reveals a small spherical cyst with an arachnoid lining, effectively communicating with the lumbosacral CSF cistern (see Figure 3). (01:47 – 01:49) This cyst is incised and its arachnoid septa taken down with a blunt hook (01:50 – 01:56) so as to incorporate the arachnoid sheets into the ostial repair along with the dural layer, and so avoid an only partial closure of the ostium (01:57 – 02:06). For this, we use non-penetrating clips (AnastoClip®; LeMaitre Vascular, Inc., Burlington, MA, USA). (02:06 – 02:20) Closure is reinforced with alternating layers of fibrin sealant (shown here, Evicel®; EthiconTM, Somerville, NJ, USA) and collagen-based dural regeneration matrix (shown here, DuraGen®; Integra®, Plainsboro, NJ, USA; and DuraGen®; Integra®, Plainsboro, NJ, USA / DurepairTM; Medtronic, Minneapolis, MN, USA). A lumbar drain is inserted intraoperatively, followed by a watertight closure of the musculofascial and skin layers. Patient outcome: Sustained postoperative symptom resolution, including retrieved ability to control and pass urine. (MP4 342815 KB)
Rights and permissions
About this article
Cite this article
Cabrilo, I., Zaidman, N. & Casey, A.T. Midline sacral meningeal cyst decompression and repair. Acta Neurochir 163, 2777–2781 (2021). https://doi.org/10.1007/s00701-021-04948-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-021-04948-3