Abstract
The development of cavernous malformations many years following conventionally fractionated brain irradiation is well recognized and commonly reported. However, cavernous malformation induction following stereotactic radiosurgery (SRS) is largely unreported. Herein, we describe two cases of cavernous malformation formation years following SRS for brain metastases. A 20-year-old woman with breast cancer brain metastases received treatment with whole brain radiotherapy (WBRT), then salvage SRS 1.4 years later for progression of a previously treated metastasis. This lesion treated with SRS had hemorrhagic enlargement 3.0 years after SRS. Resection revealed a cavernous malformation. A 25-year-old woman had SRS for a brain metastasis from papillary thyroid carcinoma. Resection of a progressive, hemorrhagic lesion within the SRS field 2 years later revealed both recurrent carcinoma as well as cavernous malformation. As patients with brain metastases live longer following SRS, our cases highlight that the differential diagnosis of an enlarging enhancing lesion within a previous SRS field includes not only cerebral necrosis and tumor progression but also cavernous malformation induction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cavernous malformations (CM) are angiographically occult vascular malformations consisting of thin-walled, dilated, vascular channels without supporting vascular smooth muscle or intervening brain parenchyma [9]. Although most cases are congenital [9], delayed CM development occurs in familial forms [23] or following radiotherapy [20]. Most radiation-associated CMs occur after conventionally fractionated radiotherapy or whole brain radiation [20]; however, they have also been reported following stereotactic radiosurgery (SRS) for arteriovenous malformations (AVM) [18, 30]. CM induction following SRS, defined as stereotactic treatment in 1 to 5 days [2], for a brain metastasis has been rarely reported [11, 14] despite brain metastases being the most common indication for SRS. The rarity of this event is likely due to the long latency required for CM induction and the limited lifespan of patients with metastatic cancer. As patients with metastatic cancer survive longer and the role for SRS is supported for up to 10 brain metastases [31], SRS-induced CM may become more prevalent. The clinician should be aware that the differential diagnosis of a new enhancing lesion within a prior SRS field includes not only cerebral necrosis or tumor progression but also CM induction. We report two cases of CM acquired following SRS for brain metastases.
Case reports
Case #1
Patient case #1 highlights the differential diagnosis of an enlarging enhancing lesion following SRS: radiation necrosis, tumor recurrence, and cavernous malformation all in the same patient.
Initial presentation and whole brain radiotherapy
A 20-year-old woman with a family history of breast cancer was diagnosed with a grade 3 infiltrating ductal carcinoma (hormone receptor positive, Her-2 negative, BRCA 1 and 2 negative). At diagnosis, she had metastases to the liver, lungs, bones, and brain. As she had five large brain metastases, she received whole brain radiotherapy (WBRT), 30 Gy in 10 fractions via standard opposed lateral fields. She tolerated the treatment well with resolution of her pre-treatment headaches and nausea. A post-WBRT MRI 3 months later revealed dramatic shrinkage of her dominant left frontal metastasis (Fig. 1a, b). This lesion ultimately developed a cavernous malformation, as below. She subsequently received multiple courses of chemotherapy, including adriamycin, cyclophosphamide, taxol, and tamoxifen.
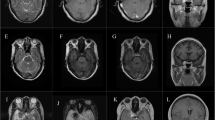
An enlarging hemorrhagic mass following stereotactic radiosurgery and whole brain radiotherapy: cavernous malformation. A 20-year-old woman had a left frontal breast cancer brain metastasis (a) treated with WBRT with significant response (b). This same site progressed 1.4 years later, treated with SRS (c). After being controlled for 2.9 more years, it became hemorrhagic (d), with resection revealing a cavernous malformation (e)
1.4 years post-WBRT: salvage SRS to progressive metastases
Due to progression in multiple metastases previously treated with WBRT, 1.4 years later, she had three enlarging lesions treated with CyberKnife robotic linear accelerator SRS, as previously described [25]: 24 Gy in 3 fractions prescribed to the 75% isodose line covering the periphery of the dominant left frontal lesion (5.2 cm3 volume, conformity index 1.3, maximum dose 32 Gy, collimator sizes of 7.5 mm and 10 mm) (Fig. 1c), and 22 Gy to multiple tumors, notably a left cerebellar metastasis (Fig. 2a).
An enlarging hemorrhagic mass following stereotactic radiosurgery and whole brain radiotherapy: recurrent tumor. A left cerebellar breast cancer metastasis was recurrent 1.4 years following WBRT and treated with SRS (a). After control for 3.6 years, it had hemorrhagic enlargement (b), with an identical imaging appearance to the patient’s prior cavernous malformation (Fig. 1d). However, pathology revealed viable breast adenocarcinoma (c), highlighting that recurrent tumor, although rare after years of local control, is always on the differential diagnosis
4.4 years post-WBRT, 2.9 years post-SRS: left frontal SRS-associated cavernous malformation
The dominant left frontal tumor, treated with WBRT and salvage SRS, was stable for 2.9 years until progression with hemorrhage and fluid-fluid levels (Fig. 1d). Surgical resection was performed for presumed hemorrhagic tumor progression. However, histology revealed radiation necrosis, with no viable tumor, surrounding a cavernous malformation (Fig. 1e).
5 years post-WBRT, 3.6 years post-SRS: left cerebellar hemorrhagic tumor recurrence mimicking a cavernous malformation
Seven months later, the left cerebellar lesion previously treated with salvage SRS (Fig. 2a) had hemorrhagic enlargement with fluid-fluid levels. Given the similar imaging appearance to the left frontal SRS-associated cavernous malformation (Figs. 1d and 2b), the presumed diagnosis was another cavernous malformation. However, surgical resection revealed breast adenocarcinoma (Fig. 2c), recurrent despite being controlled for 3.6 years following SRS.
This example highlights that tumor progression, although unexpected after 3.6 years of local control, is always on the differential diagnosis.
She unfortunately died of progressive systemic disease, 7 years following her initial WBRT.
Case report #2
A 4-year-old girl had papillary thyroid carcinoma treated with thyroidectomy. From the ages of 7 to 15, she had five total treatments of I-131 radioactive iodine ablation for pulmonary metastases. When she was 25 years old, an MRI demonstrated brain metastases, histologically confirmed as metastatic papillary thyroid carcinoma. An intact left frontal metastasis was treated at an outside institution with an MLC (multi-leaf collimator)-based linear accelerator SRS plan, with 9 non-coplanar fields, with a 3-mm margin, 20 Gy in 1 fraction to the 80% isodose line covering the periphery of a 3.8 cm3 tumor (conformity index 1.4, maximum dose 25 Gy) (Fig. 3a).
An enlarging hemorrhagic mass following stereotactic radiosurgery: cavernous malformation. A 25-year-old woman had a left frontal thyroid carcinoma brain metastasis treated with SRS (a). After 2.0 years of local control, a hemorrhagic enlarging lesion (b) was resected. Histology displayed both a cavernous malformation (c) admixed with recurrent metastatic papillary thyroid carcinoma
2.0 years following post-resection cavity SRS: SRS-induced cavernous malformation
This lesion progressed 2 years later as a hemorrhagic mass with fluid-fluid levels (Fig. 3b). Surgical resection revealed both a cavernous malformation (Fig. 3c) and metastatic papillary thyroid carcinoma; whether the cells represented viable tumor could not be determined. Follow-up imaging 11 months later revealed no tumor recurrence within the brain.
Discussion
Currently, brain metastases are the most common indication for SRS. The prevalence of SRS treatment is likely to increase, as WBRT use is decreasing [1] and prospective data supports SRS alone for up to 10 metastases [31]. Approximately 60% of patients treated with SRS will develop additional brain metastases at some point in the course of their disease, necessitating frequent surveillance brain MRIs. Given that patients with metastatic disease are living longer, post-SRS imaging changes will become increasingly prevalent. Historically, the main differential diagnosis of an enlarging lesion consisted of recurrent tumor or radiation necrosis [27], with both events potentially occurring many years following SRS [7]. Our cases highlight that clinicians should also consider SRS-associated cavernous malformations. Similarly, SRS is associated with other rare, acquired vascular entities such as extravascular papillary endothelial hyperplasia (i.e., Masson’s tumor) [5, 12].
Radiotherapy is a known risk factor for acquired or de novo cavernous malformations. In 1994, Ciricillo et al. first proposed the induction of CMs by irradiation, but through an unknown mechanism [4]. In 1998, Larson et al. suggested two mechanisms for CMs associated with radiotherapy [17]: acquired de novo as a result of radiation or radiographically occult prior to irradiation with subsequent appearance due to radiotherapy-induced hemorrhage. Overall, causation is unknown and further research is needed to determine the relationship between CM appearance and radiation dosimetry, time latency, and patient characteristics.
The baseline risk of CM in the general population is largely uncharacterized but may be up to 0.5% in autopsy and MRI series [9, 26]. Similarly, the incidence of radiotherapy-associated CM is unknown. The largest retrospective studies on CMs induced by conventionally fractionated radiotherapy included 108, 89, and 76 patients respectively [6, 13, 20]. The available data are not consistent between reports [6], but there is a suggestion that CM induction may be higher with greater irradiation dose [15], younger age [10, 20, 22], or with a shorter latency with younger age [10].
The reason why cavernous malformations are associated with fractionated radiotherapy rather than SRS is unknown, but factors may include underreporting, differences in dosimetry, radiobiology, irradiation volume, and, specific to our brain metastases cases, latency of induction. In 108 children, the mean latency between cranial radiotherapy and CM development was 5 years for patients irradiated in the first decade of life and 3.3 years for the second decade of life [6]. Prognostic indices for brain metastases vary widely, suggesting a median maximum survival of 7–25 months [8, 28]. Most patients with brain metastases simply do not live long enough to develop SRS-associated CMs. To our knowledge, the only other cases of SRS-associated cavernous malformation for brain metastases were reported by Kleinschmidt-DeMasters et al. [14] and Iwai et al. [11]. Similar to our case #1, Iwai’s patient underwent 2 courses of irradiation for a recurrent lung cancer brain metastasis and developed a CM 2.9 years following the first course of SRS. In our first patient, the combination of WBRT and SRS may have contributed to CM induction; however, our second patient had only received one course of SRS.
Other reported cases of SRS-associated CMs occurred in non-malignant diseases such as vestibular schwannoma [19, 24] and AVM [18, 30], where patients survive for the expected latency period (Table 1). Additionally, a de novo CM following SRS for a CM has been reported [32], highlighting the possible interplay between intrinsic biologic risk and environmental factors such as irradiation.
We did not include a case of a pre-existing CM treated with SRS [22], nor reports of post-SRS chronic encapsulated hematoma [16, 21, 29] or “radiation-induced organizing hematoma” characterized by organizing hematoma rather than the vascular proliferation of cavernous malformations [3]. We acknowledge that the histology and terminology of these post-SRS vascular lesions may not be standardized amongst neuropathologists. As highlighted by the thoughtful report and review of radiation-induced cavernous hemangiomas or “RICHs,” Kleinschmidt-DeMasters et al. [14] found two histological subtypes following cranial irradiation: cavernoma-like RICHs and coagulum-like RICHs consisting of “amorphous coagulum and fibrinous deposits with recanalization and formation of aggregates of vascular spaces.” They did not find a correlation between type of irradiation (i.e., radiotherapy or SRS) and histologic type of post-irradiation malformation. It is possible that there may be overlap in the histopathology of these entities. Our report adds to this literature to allow more definitive classification in the future.
Conclusion
Our patients highlight that an enlarging lesion following SRS may represent the typical findings of recurrent tumor (at 3.6 years post-SRS in case #1) or radiation necrosis (at 2.9 years post-SRS in case #1). However, we highlight that the differential diagnosis should also include a hemorrhagic cavernous malformation (with a 2.9-year latency after SRS in case 1) or a mixed histology of both recurrent tumor and cavernous malformation (with a 2.0-year latency as in case #2). As patients live longer following SRS for brain metastases, the incidence of SRS-associated cavernous malformations may increase. We encourage future reports documenting the long-term side effects of SRS and characterizing post-treatment imaging changes.
References
American Society for Radiation Oncology: Choosing Wisely 2014. https://www.astro.org/Patient-Care/Patient-Education/2014-Choosing-Wisely-List/. Accessed 1 Nov 2017
Barnett GH, Linskey ME, Adler JR, Cozzens JW, Friedman WA, Heilbrun MP, Lunsford LD, Schulder M, Sloan AE, American Association of Neurological S, Congress of Neurological Surgeons Washington Committee Stereotactic Radiosurgery Task F (2007) Stereotactic radiosurgery--an organized neurosurgery-sanctioned definition. J Neurosurg 106:1–5. https://doi.org/10.3171/jns.2007.106.1.1
Cha YJ, Nahm JH, Ko JE, Shin HJ, Chang JH, Cho NH, Kim SH (2015) Pathological evaluation of radiation-induced vascular lesions of the brain: distinct from de novo cavernous hemangioma. Yonsei Med J 56:1714–1720. https://doi.org/10.3349/ymj.2015.56.6.1714
Ciricillo SF, Cogen PH, Edwards MS (1994) Pediatric cryptic vascular malformations: presentation, diagnosis and treatment. Pediatr Neurosurg 20:137–147
Crocker M, deSouza R, Epaliyanage P, Bodi I, Deasy N, Selway R (2007) Masson’s tumour in the right parietal lobe after stereotactic radiosurgery for cerebellar AVM: case report and review. Clin Neurol Neurosurg 109:811–815. https://doi.org/10.1016/j.clineuro.2007.07.005
Di Giannatale A, Morana G, Rossi A, Cama A, Bertoluzzo L, Barra S, Nozza P, Milanaccio C, Consales A, Garre ML (2014) Natural history of cavernous malformations in children with brain tumors treated with radiotherapy and chemotherapy. J Neuro-Oncol 117:311–320. https://doi.org/10.1007/s11060-014-1390-9
Fujimoto D, von Eyben R, Gibbs IC, Chang SD, Li G, Harsh GR, Hancock S, Fischbein N, Soltys SG (2018) Imaging changes over 18 months following stereotactic radiosurgery for brain metastases: both late radiation necrosis and tumor progression can occur. J Neuro-Oncol 136:207–212. https://doi.org/10.1007/s11060-017-2647-x
Gaspar LE, Scott C, Murray K, Curran W (2000) Validation of the RTOG recursive partitioning analysis (RPA) classification for brain metastases. Int J Radiat Oncol Biol Phys 47:1001–1006
Gross BA, Batjer HH, Awad IA, Bendok BR (2009) Brainstem cavernous malformations. Neurosurgery 64:E805–E818; discussion E818. https://doi.org/10.1227/01.NEU.0000343668.44288.18
Heckl S, Aschoff A, Kunze S (2002) Radiation-induced cavernous hemangiomas of the brain: a late effect predominantly in children. Cancer 94:3285–3291. https://doi.org/10.1002/cncr.10596
Iwai Y, Yamanaka K, Yoshimura M (2007) Intracerebral cavernous malformation induced by radiosurgery. Case report. Neurol Med Chir (Tokyo) 47:171–173
Karamchandani J, Vogel H, Fischbein N, Gibbs I, Edwards MS, Griffith H (2012) Extravascular papillary endothelial hyperplasia mimicking neoplasm after radiosurgery: case report. Neurosurgery 70:E1043–E1048; discussion E1048. https://doi.org/10.1227/NEU.0b013e31822e81f9
Keezer MR, Del Maestro R (2009) Radiation-induced cavernous hemangiomas: case report and literature review. Can J Neurol Sci 36:303–310
Kleinschmidt-DeMasters BK, Lillehei KO (2016) Radiation-induced cerebral vascular “malformations” at biopsy. J Neuropathol Exp Neurol. https://doi.org/10.1093/jnen/nlw085
Koike T, Yanagimachi N, Ishiguro H, Yabe H, Yabe M, Morimoto T, Shimizu T, Takakura H, Kato S (2012) High incidence of radiation-induced cavernous hemangioma in long-term survivors who underwent hematopoietic stem cell transplantation with radiation therapy during childhood or adolescence. Biol Blood Marrow Transplant 18:1090–1098. https://doi.org/10.1016/j.bbmt.2011.12.582
Kurita H, Sasaki T, Kawamoto S, Taniguchi M, Kitanaka C, Nakaguchi H, Kirino T (1996) Chronic encapsulated expanding hematoma in association with gamma knife stereotactic radiosurgery for a cerebral arteriovenous malformation. Case report. J Neurosurg 84:874–878. https://doi.org/10.3171/jns.1996.84.5.0874
Larson JJ, Ball WS, Bove KE, Crone KR, Tew JM Jr (1998) Formation of intracerebral cavernous malformations after radiation treatment for central nervous system neoplasia in children. J Neurosurg 88:51–56. https://doi.org/10.3171/jns.1998.88.1.0051
Motegi H, Kuroda S, Ishii N, Aoyama H, Terae S, Shirato H, Iwasaki Y (2008) De novo formation of cavernoma after radiosurgery for adult cerebral arteriovenous malformation--case report. Neurol Med Chir (Tokyo) 48:397–400
Murakami K, Jokura H, Kawagishi J, Watanabe M, Tominaga T (2011) Development of intratumoral cyst or extratumoral arachnoid cyst in intracranial schwannomas following gamma knife radiosurgery. Acta Neurochir 153:1201–1209. https://doi.org/10.1007/s00701-011-0972-y
Nimjee SM, Powers CJ, Bulsara KR (2006) Review of the literature on de novo formation of cavernous malformations of the central nervous system after radiation therapy. Neurosurg Focus 21:e4
Pozzati E, Giuliani G, Gaist G, Piazza G, Vergoni G (1986) Chronic expanding intracerebral hematoma. J Neurosurg 65:611–614. https://doi.org/10.3171/jns.1986.65.5.0611
Pozzati E, Acciarri N, Tognetti F, Marliani F, Giangaspero F (1996) Growth, subsequent bleeding, and de novo appearance of cerebral cavernous angiomas. Neurosurgery 38:662–669 discussion 669-670
Rigamonti D, Hadley MN, Drayer BP, Johnson PC, Hoenig-Rigamonti K, Knight JT, Spetzler RF (1988) Cerebral cavernous malformations. Incidence and familial occurrence. N Engl J Med 319:343–347. https://doi.org/10.1056/NEJM198808113190605
Sasagawa Y, Akai T, Itou S, Iizuka H (2009) Gamma knife radiosurgery-induced cavernous hemangioma: case report. Neurosurgery 64:E1006–E1007; discussion E1007. https://doi.org/10.1227/01.NEU.0000344005.23146.CC
Shultz DB, Modlin LA, Jayachandran P, Von Eyben R, Gibbs IC, Choi CY, Chang SD, GRt H, Li G, Adler JR, Hancock SL, Soltys SG (2015) Repeat courses of stereotactic radiosurgery (SRS), deferring whole-brain irradiation, for new brain metastases after initial SRS. Int J Radiat Oncol Biol Phys 92:993–999. https://doi.org/10.1016/j.ijrobp.2015.04.036
Smith ER, Scott RM (2010) Cavernous malformations. Neurosurg Clin N Am 21:483–490. https://doi.org/10.1016/j.nec.2010.03.003
Sneed PK, Mendez J, Vemer-van den Hoek JG, Seymour ZA, Ma L, Molinaro AM, Fogh SE, Nakamura JL, McDermott MW (2015) Adverse radiation effect after stereotactic radiosurgery for brain metastases: incidence, time course, and risk factors. J Neurosurg 123:373–386. https://doi.org/10.3171/2014.10.JNS141610
Sperduto PW, Kased N, Roberge D, Xu Z, Shanley R, Luo X, Sneed PK, Chao ST, Weil RJ, Suh J, Bhatt A, Jensen AW, Brown PD, Shih HA, Kirkpatrick J, Gaspar LE, Fiveash JB, Chiang V, Knisely JP, Sperduto CM, Lin N, Mehta M (2012) Summary report on the graded prognostic assessment: an accurate and facile diagnosis-specific tool to estimate survival for patients with brain metastases. J Clin Oncol 30:419–425. https://doi.org/10.1200/JCO.2011.38.0527
Takei J, Tanaka T, Yamamoto Y, Teshigawara A, Tochigi S, Hasegawa Y, Murayama Y (2016) Chronic encapsulated expanding thalamic hematoma associated with obstructive hydrocephalus following radiosurgery for a cerebral arteriovenous malformation: a case report and literature review. Case Rep Neurol Med 2016:5130820. https://doi.org/10.1155/2016/5130820
Wang X, Hui XH, Liu JP, Mao Q (2012) Radiation-induced cavernous malformation at the site of arteriovenous malformation following gamma knife radiosurgery: case report. Clin Neurol Neurosurg 114:1287–1289. https://doi.org/10.1016/j.clineuro.2012.03.005
Yamamoto M, Serizawa T, Shuto T, Akabane A, Higuchi Y, Kawagishi J, Yamanaka K, Sato Y, Jokura H, Yomo S, Nagano O, Kenai H, Moriki A, Suzuki S, Kida Y, Iwai Y, Hayashi M, Onishi H, Gondo M, Sato M, Akimitsu T, Kubo K, Kikuchi Y, Shibasaki T, Goto T, Takanashi M, Mori Y, Takakura K, Saeki N, Kunieda E, Aoyama H, Momoshima S, Tsuchiya K (2014) Stereotactic radiosurgery for patients with multiple brain metastases (JLGK0901): a multi-institutional prospective observational study. Lancet Oncol 15:387–395. https://doi.org/10.1016/S1470-2045(14)70061-0
Yeon JY, Suh YL, Kim JH, Lee JI (2010) Development of de novo cavernous hemangioma after radiosurgery for cavernous hemangioma. J Korean Neurosurg Soc 48:532–533. https://doi.org/10.3340/jkns.2010.48.6.532
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The patients have consented to this IRB-approved review and submission of this case report to the journal.
Conflict of interest
Scott G Soltys – Consultant – Inovio Pharmaceuticals, Inc.
The remaining authors declare no other conflicts of interest.
Additional information
This article is part of the Topical Collection on Brain Tumors
Rights and permissions
About this article
Cite this article
Seiger, K., Pendharkar, A.V., Samghabadi, P. et al. Cavernous malformations are rare sequelae of stereotactic radiosurgery for brain metastases. Acta Neurochir 161, 43–48 (2019). https://doi.org/10.1007/s00701-018-3701-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-018-3701-y