Abstract
Background
Three different types of anti-siphon devices (ASDs) have been developed to counteract siphoning-induced overdrainage in upright posture. However, it is not known how the different ASDs affect CSF dynamics under the complex pressure environment seen in clinic due to postural changes. We investigated which ASDs can avoid overdrainage in upright posture best without leading to CSF accumulation.
Methods
Three shunts each of the types Codman Hakim with SiphonGuard (flow-regulated), Miethke miniNAV with proSA (gravitational), and Medtronic Delta (membrane controlled) were tested. The shunts were compared on a novel in vitro setup that actively emulates the physiology of a shunted patient. This testing method allows determining the CSF drainage rates, resulting CSF volume, and intracranial pressure in the supine, sitting, and standing posture.
Results
The flow-regulated ASDs avoided increased drainage by closing their primary flow path when drainage exceeded 1.39 ± 0.42 mL/min. However, with intraperitoneal pressure increased in standing posture, we observed reopening of the ASD in 3 out of 18 experiment repetitions. The adjustable gravitational ASDs allow independent opening pressures in horizontal and vertical orientation, but they did not provide constant drainage in upright posture (0.37 ± 0.03 mL/min and 0.26 ± 0.03 mL/min in sitting and standing posture, respectively). Consequently, adaptation to the individual patient is critical. The membrane-controlled ASDs stopped drainage in upright posture. This eliminates the risk of overdrainage, but leads to CSF accumulation up to the volume observed without shunting when the patient is upright.
Conclusions
While all tested ASDs reduced overdrainage, their actual performance will depend on a patient’s specific needs because of the large variation in the way the ASDs influence CSF dynamics: while the flow-regulated shunts provide continuous drainage in upright posture, the gravitational ASDs allow and require additional adaptation, and the membrane-controlled ASDs show robust siphon prevention by a total stop of drainage.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Most commonly, hydrocephalus is treated by surgical placement of a ventriculoperitoneal cerebrospinal fluid (CSF) shunt. About 80% of patients with idiopathic normal pressure hydrocephalus (INPH) improve after shunt insertion [25]. However, up to 10% of INPH patients develop a serious adverse event, such as a subdural hematoma or hygroma, because of overdrainage through the shunt [12].
Flow through current shunt valves are differential pressure driven, and thus a function of intracranial pressure (ICP) and intraperitoneal pressure (IPP), but also dependent on the hydrostatic pressure column in the catheter [3, 18, 19]. A common misconception is that only the hydrostatic pressure column changes with body position, whereas actually all three pressures are influenced by the patient’s posture [17]. In clinic, this creates unpredictable pressure differences, and thus a complex environment, within which the shunt must be able to maintain physiologic ICP and CSF volume. Anti-siphon devices (ASDs) are used to adapt the shunt to these changes in pressures related to posture, especially to avoid overdrainage when sitting or standing. Various manufacturers have tried to solve the problem by using different constructions of the ASD [2]. However, it is not known whether any of these constructions are better than the others, and there is no consensus on the best way to control CSF drainage under postural changes [13].
Bench tests have been used to characterize the opening pressure and resistance of shunts with ASDs under changing valve orientations and distal pressures [1, 6,7,8, 13, 16]. To date, bench test analysis of shunt systems including ASDs has stopped here. However, to understand how ICP will be regulated by the shunt system in upright posture, the next step is to understand the resulting time-dependent shunt flows and how this changes the ICP and craniospinal compliance. In vivo studies have demonstrated changes of equilibrium ICP and pulse pressure amplitude due to posture and for different valve settings [3, 4, 11], with two of them also having included quantitative drainage information [17, 18]. However, CSF volume and drainage rates cannot be measured reliably in vivo, but would be necessary to understand the interaction of shunt and CSF dynamics, especially in upright posture.
In this study, we used a novel in vitro test bench [14] to quantify the influence these three anti-siphon mechanisms have on the CSF dynamic system. The test bench combines a hardware interface for the shunts to be tested with a real-time computational model of the relevant physiology, whereby changes in CSF dynamics due to drainage through the shunt can be emulated.Footnote 1 Therefore, in contrast to previous in vitro test benches, the pressures applied to the tested shunt are not constant, but change dynamically with drainage and posture. This general approach combines advantages of classical in vitro approaches (repeatability) with those of clinical studies (physiological interaction of the shunt with the patient). It allows measuring drainage continuously in different postures and determining clinically relevant parameters, such as CSF volume and ICP. With this, we aim to elucidate how the different ASDs interact with the CSF system and which ASDs can maintain the desired reduction in CSF volume best in upright posture without overdraining.
Methods
Three types of state-of-the-art differential pressure shunts with fixed opening pressure and integrated ASD were included in this study. The opening pressures of each shunt type were chosen such that the target ICP in supine position was as close to 10 mmHg as possible. After determining the baseline characteristics of the shunt systems in horizontal and vertical orientation, their influence on CSF dynamics was investigated by simulating a succession of postural changes. As ASDs are often designed under the assumption that the orientation of the valve represents the posture of the patient, we also evaluated the sensitivity of the tested ASDs with respect to their effective position.
Shunts
The CSF shunts tested were: (1) the Codman Hakim fixed pressure valve (medium high range) with integrated flow-regulated SiphonGuard ASD (Codman & Shurtleff, Inc., Raynham, MA, USA); (2) Miethke miniNAV (10 cmH2O opening pressure) combined with the adjustable gravitational Miethke proSA ASD (Christoph Miethke GmbH & Co. KG, Potsdam, Germany); (3) Medtronic Delta fixed pressure valve (performance level 1.5) with integrated membrane-controlled Delta chamber (Medtronic Neurosurgery, Goleta, CA, USA). The opening pressure of the Miethke proSA, the only adjustable ASD in the group, was set to 20 cmH2O (according to the manufacturer’s recommendation for patients above 60 years of age). Three shunts of each of the three types were tested with the corresponding standard catheters. Shunts and catheters were new. The inner diameters of the catheters were 1, 1.2, and 1.3 mm for the Codman, Miethke, and Medtronic valves, respectively. Catheter length was standardized to 20 cm for the proximal catheter and 84 cm for the distal catheter.
Experiments
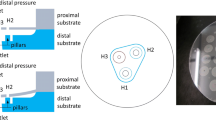
All experiments were conducted using the test bench introduced in [14] and depicted in Fig. 1. The respective shunt’s influence on CSF dynamics was simulated with the mathematical model shown and described in Fig. 2.
The shunt’s proximal and distal catheters are connected to separate reservoirs filled with deionized and deaerated water. The pressures in the reservoirs are dynamically adjusted during each experiment to correspond to expected instantaneous values of intracranial pressure (ICP) and intraperitoneal pressure (IPP), respectively [14]. These values are determined by a real-time computational physiologic model. The instantaneous outflow rate through the tested shunt is continuously measured with an integrated flow probe (SLI-2000, Sensirion AG, Staefa, Switzerland). The valves and catheters are submerged in a closed water bath that guarantees a posture-independent pressure of 10 cmH20 at the location of the valve [1, 14, 16]. The valve is placed at the same level as the pressurized ICP reservoir, which represents the reference location for ICP measurements. The orientations of the valve and catheter are adjusted (as depicted in Fig. 1) to mimic the simulated patient’s posture in terms of trunk orientation α 1 and head orientation α 2. The corresponding lengths l 1 and l 2 are the trunk length (from the IPP reference point to the mid-shoulder) and the distance between the shoulder and the external auditory canal, respectively. Venous HIP denotes the venous system’s hydrostatic indifference point. The whole test bench including the test medium and the shunt is heated to a uniform temperature of 37 ± 1 °C [14]
The CSF pressure-volume relationship implemented in the ICP model is exponential (\( ICP(t)-{p}_v\left({\alpha}_1,{\alpha}_2\right)={p}_1{e}^{k\cdotp {\varDelta V}_{CSF}(t)}+{p}_0 \)) above the relative optimal pressure Δp opt and linear below (ICP(t) − p v (α 1, α 2) = (Δp opt − p 0) · k · (ΔV CSF (t) − V opt ) + Δp opt ) [21]. The dashed line indicates how this relationship shifts from supine to upright posture because of the posture-dependent dural venous pressure p v . CSF volume relative to the volume at baseline (12.5 mmHg ICP) ΔV CSF was calculated from the measured drainage rate through the shunt Q shunt (\( \frac{d}{ d t}\varDelta {V}_{CSF}(t)={Q}_{form}-{Q}_{abs}(t)-{Q}_{shunt}(t) \)) assuming a constant CSF formation rate Q form and ICP-dependent CSF absorption (Q abs (t) = (ICP(t) − p v (α 1, α 2))/R out for ICP(t) ≥ p v (α 1, α 2)). The dural venous pressure p v , which is the reference pressure for the pressure-volume relationship and the CSF absorption, was calculated relative to the pressure in the venous hydrostatic indifference point (HIP) [22]: p v (α 1, α 2) = p v , HIP − ρg(l 2 sin α 2 + (L HIP − l 2) sin α 1). When the jugular veins collapse in upright posture, this posture-related drop in p v is dampened [15, 22]: p v (α 1, α 2) = − ρgL jug sin α 2. The model parameter values are listed in Table 1
Baseline characteristics
The pressure-flow characteristics of each valve were evaluated in horizontal and in vertical position.
-
Horizontal position: the ICP reservoir, the valve, and the IPP reservoir were all kept at the same level. The pressure in the ICP reservoir was increased from 0 to 30 mmHg and decreased back to 0 mmHg over 4 min continuously and without pulsations. The pressure in the IPP reservoir was kept constant at 1.8 mmHg, i.e., the IPP value for supine (Table 1) [5].
-
Vertical position: the test bench rotated the valve by 90°, while keeping it at the same level as the ICP reservoir. Both were located 64 cm (l 1 + l 2) above the IPP reservoir, whose pressure was kept at 16.7 mmHg, i.e., the IPP value for sitting [5]. As there is a physiological decrease of ICP in upright position [22], the simulated ICP was varied between −30 and 0 mmHg.
Influences on CSF dynamics
To determine the influence of the shunts on CSF dynamics, ICP was not varied according to a pre-set pattern. Instead, the values of the pressures applied to the shunt by the dynamically pressurized reservoirs were calculated in real time based on the measured shunt flow and a mathematical model of the relevant physiology, which is described in Fig. 2. This model, which describes ICP and IPP as a function of posture and the instantaneous drainage rate through the shunt, was introduced in [14]. ICP pulsatility is included through pulsations of the cranial arterial volume. For the present study, the model was extended to also represent the linear part of the CSF system’s pressure-volume relationship (Fig. 2) [21].
The parameters of the mathematical model (Table 1) were taken from clinical studies with INPH patients [20,21,22]. The physical dimensions of the test bench (l 1 and l 2) corresponded to those of a 50th-percentile American male [23].
Postural changes
The shunts were subjected to a succession of postural changes from supine to sitting to standing and back to supine. Each of the four postures was held for 60 min to reach equilibrium conditions. The respective equilibrium values were then calculated as the arithmetic mean over the last 5 min of each posture. The initial relative CSF volume was 0 mL, corresponding to 12.5 mmHg resting ICP.
Head tilting
Effects of head tilting were investigated by varying the head tilt angle α 2 (Fig. 1) in supine and sitting posture.
-
Supine: After an initial phase of 20 min in supine position (α 1 = α 2 = 0°, IPP = 1.8 mmHg), the head orientation α 2 was linearly increased to 45° within 30 min and then decreased back to 0°. This experiment simulates the effect of an elevated head, e.g., due to thick pillows, in supine position.
-
Sitting: Starting after 20 min initialization in sitting position (α 1 = α 2 = 90°, IPP = 16.7 mmHg), the head orientation was linearly decreased by 45° within 30 min and back to 90°. This experiment simulates sitting upright with inclined head, e.g., while reading.
Signal processing
The measured shunt characteristics were processed using a moving average filter over 1 s, corresponding to 1000 samples.
For the postural changes and head-tilting experiments, the mean values of ICP, V CSF , and Q shunt at time t were calculated as the arithmetic mean of the respective measurement signal over one cardiac cycle. After filtering with a 30-ms moving-average filter, the peak-to-peak pulse amplitudes of these signals were calculated as the difference between the maximum and minimum value during each cardiac cycle.
Valve opening pressure and resistance
The opening and closing pressures are the pressure differences over the shunt that caused an opening or closing of the valve, respectively. The valves were defined to be open when the measured drainage rate was more than 5 mL/h (≈0.08 mL/min). The resistance of a shunt was determined as the average slope of the measured pressure-flow curves for drainage rates between 5 and 50 mL/h (≈0.08–0.83 mL/min).
Statistics
Where applicable, values are presented as mean ± standard deviation (SD) of the results of the three individual valves tested per shunt model and all experiment repetitions. The shunt characteristics were measured 12 times per valve, and the other two experiments were repeated 6 times per valve. All statistical tests were two-sided non-paired t-tests. Differences were considered significant at a p-value < 0.05.
Results
Baseline characteristics
Flow-regulated ASD
For the shunt with flow-regulated ASD, there were no significant differences between the pressure-flow characteristics measured in horizontal and in vertical orientation (Fig. 3). The valves’ primary pathway closed when the drainage rate exceeded 1.39 ± 0.42 mL/min. With this, the shunt’s resistance increased from 6.2 ± 0.9 mmHg/(mL/min) (see Table 2) to 58.1 ± 7.7 mmHg/(mL/min). The ASD reopened when the flow fell below 0.17 ± 0.02 mL/min, resulting in a large hysteresis of the ASD.
Measured drainage rates Q shunt of the three tested ASD models for quasi-stationary increasing and decreasing ICP in horizontal and vertical orientation plotted as functions of the actual pressure gradient over the shunt Δp shunt . Distal catheter pressure was 1.8 mmHg in horizontal orientation and 16.7 mmHg in vertical orientation, which corresponds to IPP while sitting. Mean flow rates, standard deviation, and significance of the difference between mean flow rates in horizontal and vertical orientation are indicated every 2 mmHg
Gravitational ASD
The opening pressure of the shunts with gravitational ASD increased in vertical orientation (Table 2). However, this increase was substantially lower than the nominal opening pressure of the gravitational unit (20 cmH2O = 14.7 mmHg). Reprogramming of the ASD allowed for changing this, but the measured opening pressures were consistently lower than the adjusted values.
The resistance of the open shunt was approximately constant with a slight increase above ∼1.5 mL/min drainage (Fig. 3). This resistance increase was accompanied by audible vibrations of the valve.
Membrane-controlled ASD
In horizontal orientation, the membrane-controlled shunts had a large hysteresis between opening and closing pressure (Table 2) and a comparably low resistance. In vertical orientation, no drainage was observed for the tested range of ICP values (−30 to 0 mmHg).
Influences on CSF dynamics
In supine position, drainage through the shunts led to a decrease in ICP from the initial 12.5 mmHg to the equilibrium conditions listed in Table 3. Upon sitting up, ICP instantaneously decreased by another 15.1 mmHg with all shunts (Fig. 4).
Flow-regulated ASD
The flow-regulated ASD switched to its high resistance mode immediately after sitting up. However, the drainage rates in sitting posture were still higher than in supine position, leading to a reduction in CSF volume and pulse pressure amplitude (AMP) and a further decrease in ICP (Fig. 4). After standing up, the slight increase in IPP was enough for one of the three tested valves to switch back into the low resistance mode in three out of the six experiment repetitions. This led to a large variability in the resulting equilibrium conditions in standing posture (Table 3).
Gravitational ASD
After sitting up, the lower than expected opening pressure of the gravitational ASD in vertical orientation led to increased drainage and a subsequent decrease in ICP (Table 3). AMP did not decrease further as ICP fell below optimal pressure. As higher IPP was assumed for standing (20 mmHg) compared to sitting posture (16.7 mmHg), the drainage rate decreased after standing up (Table 3).
Membrane-controlled ASD
The lower opening pressure compared to the other two valves led to slightly lower ICP in supine equilibrium (Table 3). In sitting and standing posture, however, the membrane-controlled ASD prevented drainage. This lack of drainage led to an accumulation of CSF up to the initial volume without shunting and caused increased ICP and AMP. Further ICP increase was prevented by the virtual patient’s natural CSF absorption. After lying down, the low resistance of the shunt caused a fast recovery to equilibrium conditions.
Head tilting
Flow-regulated ASD
With the flow-regulated ASDs, head tilting changed drainage in neither the supine nor sitting posture (Fig. 5).
Gravitational ASD
In supine position, the gravitational ASD kept the drainage constant for head tilt angles of up to approximately 20°. Further elevating the head led to decreasing drainage. A similar pattern was observed in sitting posture, where the drainage started to increase when the head tilt angle deviated substantially from vertical.
Membrane-controlled ASD
Elevating the head from the horizontal orientation rapidly reduced the drainage through the membrane-controlled shunts. In sitting posture, the ASD prevented drainage in any of the tested head tilt angles.
Discussion
With the novel in vitro test bench, we could quantify how the different types of ASDs affect the drainage rate, ICP, and CSF volume under postural changes. This could not have been determined with any previous in vitro test bench. Isolating and measuring the effect of the ASD would be difficult in vivo. With flow-regulated, gravitational, and membrane-controlled ASDs included in the study, all three main ASD functional principles were represented [13].
Generally, sitting up from the supine position leads to an immediate drop in ICP (even without a shunt), a simultaneous increase in IPP, and a hydrostatic pressure column in the vertical catheter, which can cause overdrainage through the shunt [11, 17]. Since the ICP decrease arises from the physiologic change in hydrostatic conditions, it is accompanied by neither a reduction in CSF volume nor a reduction in pulsatility [11, 17]. These expected changes in the shunt’s pressure environment were accurately replicated by the in vitro test bench.
In upright posture, an ASD should maintain drainage without overdrainage despite these changes, i.e., the reduction in CSF volume achieved with a shunt in supine position shall be preserved without risking subdural hemorrhage by further CSF volume reduction.
Comparing the measured equilibrium ICPs in sitting and standing posture (Table 3) with the −20 mmHg that are reached without any means of siphon prevention [14], it becomes evident that all three types of ASDs reduced the effect of siphoning. However, the ASD types each achieved this through a distinct mechanism.
Flow-regulated ASD
Flow-regulated ASDs limit the maximum flow rate through the shunt by closing their main flow path and thereby increasing their resistance. While sitting, the shunts with flow-regulated ASDs provided continuous drainage and the smallest change in CSF volume from the one in supine position. The resulting ICP was therefore close to its desired value in upright posture (−5.1 mmHg) [18, 22]. The flow-regulated ASD achieved this by closing its low resistance pathway during the process of sitting up and keeping the drainage below the rate of CSF production except for a short spike that is necessary to induce the mode switch. However, there was considerable variation in the flow rate that led to this mode switch. Furthermore, in standing posture, the ASD operated close to the opening pressure of its primary flow path, which led to occasional re-opening of this low-resistance path. In general, re-opening or no initial closing of the low resistance path is possible in upright posture when the driving pressure gradient is not large enough. This can be problematic for short patients who have a smaller hydrostatic pressure column driving the drainage in upright posture as well as for obese patients with increased IPP.
Gravitational ASD
In gravitational ASDs, the gravitational force of a counterweight is exploited to vary the ASD’s opening pressure with its orientation. Thus, they aim at increasing the differential pressure over the valve in upright posture. With this opening pressure increase, the gravitational ASDs counteracted siphoning well in standing posture. However, with no change in opening pressure, they struggled to retain the desired CSF volume in sitting posture, where IPP is slightly lower. This shows that even with a programmable gravitational ASD, constant drainage in all postures is not achievable and implies that individual adaptation to the patient is critical as over- as well as underdrainage is possible with a non-adequate ASD setting. It also implies that non-programmable gravitational ASDs may not be advisable as they may be difficult to estimate the required opening pressure before surgery and adaptation is not possible without a shunt revision.
Membrane-controlled ASD
In membrane-controlled ASDs, a membrane distal to the actual valve decouples the valve opening from the IPP and the hydrostatic pressure column in the distal catheter. This membrane acts as a trap that only opens when the proximal valve pressure exceeds the surrounding subcutaneous pressure. In this study, we saw no shunt flow in the upright position for the physiologic ICPs up to 0 mmHg. This could be expected, as opening of the membrane-controlled ASD would require positive proximal valve pressure to subcutaneous pressure gradients [8, 11]. Thus, the positive subcutaneous pressure (10 cmH2O was assumed and applied in the water bath surrounding the valve) prevented the shunts from opening in upright position. It can be expected that the membrane-controlled ASD prevents drainage in upright posture for any physiological ICP [11]. This decouples the CSF dynamics and ICP from changes in IPP, but leads to CSF accumulation in upright posture. However, this type of robust siphon prevention may even be desirable, because it eliminates the risk of overdrainage without the need for adjustments. Similar characteristics can be achieved with gravitational ASDs by deliberately choosing a higher ASD opening pressure. It has been suggested previously that the target ICP of membrane-controlled ASDs in upright posture could also be decreased through a caudally shifted valve placement [24]. However, implantation at the chest would be required to achieve the physiological value of −5.1 mmHg in upright posture.
Conclusions
Flow-regulated, gravitational, and membrane-controlled ASDs can all reduce overdrainage. However, the effects of these ASDs on CSF dynamics vary greatly, and not every device type is suitable for every patient: For the simulated INPH patient investigated in this study, the flow-regulated ASD best maintained the desired drainage rate in upright posture, but it was prone to overdrainage for slightly increased intraperitoneal pressure. The adjustable gravitational ASD allows for individual adaptation of the drainage rate in upright posture, but precise adjustment to the height and intraperitoneal pressure of the patient is required and varying intraperitoneal pressure led to deviations from the desired drainage rate. The tested membrane-controlled shunts did not drain at all in upright posture, leading to a robust siphon prevention, but without continuous drainage.
Notes
A video illustrating this experimental setup can be found at http://ieeexplore.ieee.org/ielx7/10/7384657/7160680/tbme-schmiddaners-2457681-mm.zip?tp=&arnumber=7160680
References
Arnell K, Koskinen L-OD, Malm J, Eklund A (2009) Evaluation of strata NSC and Codman Hakim adjustable cerebrospinal fluid shunts and their corresponding antisiphon devices. J Neurosurg Pediatr 3(3):166–172
Aschoff A, Kremer P, Benesch C, Fruh K, Klank A, Kunze S (1995) Overdrainage and shunt technology, a critical comparison of programmable, hydrostatic and variable-resistance valves and flow-reducing devices. Childs Nerv Syst 11(4):193–202
Bergsneider M, Yang I, Hu X, McArthur DL, Cook SW, Boscardin WJ (2004) Relationship between valve opening pressure, body position, and intracranial pressure in normal pressure hydrocephalus: paradigm for selection of programmable valve pressure setting. Neurosurgery 55(4):851–859
Chapman PH, Cosman ER, Arnold MA (1990) The relationship between ventricular fluid pressure and body position in normal subjects and subjects with shunts: a telemetric study. Neurosurgery 26(2):181–189
Cobb WS, Burns JM, Kercher KW, Matthews BD, James Norton H, Todd Heniford B (2005) Normal intraabdominal pressure in healthy adults. J Surg Res 129(2):231–235
Czosnyka Z, Czosnyka M, Richards HK, Pickard JD (1998) Posture-related overdrainage: comparison of the performance of 10 hydrocephalus shunts in vitro. Neurosurgery 42(2):327–333
Czosnyka Z, Czosnyka M, Pickard JD (1999) Hydrodynamic performance of a new siphon preventing device: the SiphonGuard. J Neurol Neurosurg Psychiatry 66(3):408–410
Czosnyka Z, Czosnyka M, Richards HK, Pickard JD (2002) Laboratory testing of hydrocephalus shunts—conclusion of the UK shunt evaluation programme. Acta Neurochir 144(6):525–538
Czosnyka M, Czosnyka Z, Momjian S, Pickard JD (2004) Cerebrospinal fluid dynamics. Physiol Meas 25(5):R51–R76
Czosnyka Z, Cieslicki K, Czosnyka M, Pickard JD (2005) Hydrocephalus shunts and waves of intracranial pressure. Med Biol Eng Comput 43(1):71–77
Farahmand D, Qvarlander S, Malm J, Wikkelso C, Eklund A, Tisell M (2014) Intracranial pressure in hydrocephalus: impact of shunt adjustments and body positions. J Neurol Neurosurg Psychiatry 86(2):222–228
Freimann FB, Sprung C (2012) Shunting with gravitational valves—can adjustments end the era of revisions for overdrainage-related events? Clinical article. J Neurosurg 117(6):1197–1204
Freimann FB, Kimura T, Stockhammer F, Schulz M, Rohde V, Thomale UW (2014) In vitro performance and principles of anti-siphoning devices. Acta Neurochir 156(11):2191–2199
Gehlen M, Kurtcuoglu V, Schmid Daners M (2016) Patient specific hardware-in-the-loop testing of cerebrospinal fluid shunt systems. IEEE Trans Biomed Eng 63(2):348–358
Gehlen M, Kurtcuoglu V, Schmid Daners M (2017) Is posture-related craniospinal compliance shift caused by jugular vein collapse? A theoretical analysis. Fluids and Barriers of the CNS 14(1)
Hassan M, Higashi S, Yamashita J (1996) Risks in using siphon-reducing devices in adult patients with normal-pressure hydrocephalus: bench test investigations with delta valves. J Neurosurg 84(4):634–641
Kajimoto Y, Ohta T, Miyake H, Matsukawa M, Ogawa D, Nagao K, Kuroiwa T (2000) Posture-related changes in the pressure environment of the ventriculoperitoneal shunt system. J Neurosurg 93(4):614–617
Miyake H, Ohta T, Kajimoto Y, Nagao K (2000) New concept for the pressure setting of a programmable pressure valve and measurement of in vivo shunt flow performed using microflow meter. J Neurosurg 92(1):181–187
Portnoy HD, Schulte RR, Fox JL, Croissant PD, Tripp L (1973) Anti-siphon and reversible occlusion valves for shunting in hydrocephalus and preventing post-shunt subdural hematomas. J Neurosurg 38(6):729–738
Qvarlander S (2013) Analysis of ICP pulsatility and CSF dynamics: the pulsatility curve and effects of postural changes, with implications for idiopathic normal pressure hydrocephalus. PhD thesis
Qvarlander S, Lundkvist B, Koskinen L-OD, Malm J, Eklund A (2013) Pulsatility in CSF dynamics: pathophysiology of idiopathic normal pressure hydrocephalus. J Neurol Neurosurg Psychiatry 84(7):735–741
Qvarlander S, Sundstrom N, Malm J, Eklund A (2013) Postural effects on intracranial pressure: modeling and clinical evaluation. J Appl Physiol 115(10):1474–1480
United States. National Aeronautics and Space Administration (1995) Man-systems integration standards. Number Bd. 3 in NASA-STD. National Aeronautics and Space Administration
Watson DA (1994) The delta valve: a physiologic shunt system. Childs Nerv Syst 10(4):224–230
Williams MA, Malm J (2016) Diagnosis and treatment of idiopathic normal pressure hydrocephalus. Continuum Lifelong Learning Neurol 22(2):579–599
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no personal or institutional conflict of interest.
Funding
The Swiss Academy of Engineering Sciences provided financial support through the grant 2014–080, the 3R Research Foundation provided financial support through the grant 140–14, and the Swiss National Science Foundation provided financial support through NCCR Kidney.CH. The tested shunts were provided by the respective manufacturers.
The sponsors had no role in the design or conduct of this research.
Rights and permissions
About this article
Cite this article
Gehlen, M., Eklund, A., Kurtcuoglu, V. et al. Comparison of anti-siphon devices—how do they affect CSF dynamics in supine and upright posture?. Acta Neurochir 159, 1389–1397 (2017). https://doi.org/10.1007/s00701-017-3249-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-017-3249-2