Abstract
Background
Clinical management after epidural steroid injections (ESI) of patients with radiculopathy secondary to a cervical disc herniation (CDH) is uncertain. This study aims to determine whether short-term arm pain alleviation following computed tomography-guided ‘indirect’ cervical ESI can predict the 1-month outcome.
Methods
We conducted a prospective observation of 45 consecutive patients at a tertiary radiological department. Study components were visual analog scale arm and neck pain at baseline, 15, 30, and 45 min, 1, 2, and 4 h, on days 1–14, 1 month, and at 1 year. Health-related quality of life and functional impairment were assessed using the short form-12 and Neck Pain and Disability Scale. Patients who reported ≥80 % persisting arm pain, as well as patients who underwent a second injection or an operation within 1 month were defined as ‘non-responders’. Logistic regression was used to analyze the effect size of the relationship between >50 % pain relief at any given study visit and responder status.
Results
Patients experiencing a >50 % pain reduction 4 h after the injection were four times as likely to be responders as those experiencing ≤50 % pain reduction (OR 4.04, 95 % CI 1.10–14.87). The effect was strongest on days 5–6 (OR 18.37, 95 % CI 3.39–99.64) and remained significant until day 14.
Conclusions
The results of this study can guide physicians in managing patients with CDH: a ≤50 % arm pain relief within 1 week after an ‘indirect’ cervical ESI predicts an unfavorable 1-month outcome and suggests that other treatment options may be considered at an earlier point in time.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Epidural steroid injections (ESI) are frequently used in the treatment of radicular pain secondary to a cervical disc herniation (CDH) despite a lack of strong evidence supporting its efficacy [21] and even some reported disappointing results [3]. There is a lack of randomized controlled trials [1, 3, 11, 22] and well-conducted studies [4, 13, 22] on its role as an adjunct to medical therapy preceding surgical treatment. A pivotal discussion point is the underlying work mechanisms, since saline injections were proven to be as equally effective as cortisone [1]. Anti-inflammatory effects [1] and wash-out effects may be responsible for a positive treatment response, which makes designing a placebo-controlled study a challenge. Nevertheless, in the absence of neurological deficits or signs of myelon compression, ESI represents a well-established adjunct to the non-surgical management for patients with radicular arm pain. Whether radiological features identified on cervical magnetic resonance imaging (MRI), such as the location of the CDH and degree of nerve root compression serve as a predictor for the clinical outcome, needs to be studied further since present data is conflicting [13, 18].
An unresolved issue in daily clinical routine is the definition of treatment success or failure. This is especially true for the first days and weeks following ESI. In patients with radiculopathy who report only some positive effect after ESI, the question remains whether or not more time should be invested to see if the patient will finally respond. Cervical radiculopathy can be excruciating and wasted time can add to pain chronification, depression, and extended work leave. Kolstad et al. [10] reported that there was a relatively instant treatment effect in patients who did not require surgery later on. The authors advised surgery to those who did not clearly have a positive effect within a few weeks. In a similar manner, other authors [20, 22] noted that patients with no clear improvement at the first 2-week follow-up would also not experience any alleviation down the road.
The literature on this issue is neither conclusive in terms of methods and details of short-term follow-up, nor are there clear management recommendations so far. Hence, the current study addresses the question of whether and to what extent the short-term pain relief following ESI for CDH can predict the 1-month outcome.
Methods
We prospectively screened all patients between 18 and 70 years of age with cervical radicular pain, with or without findings of radiculopathy, secondary to a single-level CDH (“soft disc compression”; no osseous foraminal stenosis due to osteophytes) who were referred to the Department of Radiology at Cantonal Hospital St. Gallen in Switzerland between August 2013 and March 2015 for study inclusion. Referring physicians were general practitioners, pain physicians, rheumatologists, orthopedic surgeons, and neurosurgeons. In order to be included in our study, MRI had to show a CDH corresponding with clinical symptoms of radiculopathy. All diagnoses and imaging findings were confirmed by A.N., a radiologist with special interest and over 20 years of expertise in ESI, who also performed all of the interventions. Study exclusion criteria were the following: visual analog scale (VAS) arm pain < 20/100 mm, profound motor deficit (British Medical Research Council 0-3), myelopathy, multilevel disc herniation, stenosis, severe scoliosis, discrepancy between imaging findings and symptoms, previous ESI or surgery of the affected segment, uncorrectable bleeding diasthesis, and a significant language barrier.
Baseline parameters
Prior to ESI we collected patient demographic data, profession according to the international standard classification of occupation [8], work capacity, current use of opioids and symptom duration, as well as baseline intensity of arm and neck pain measured with the VAS (0 to 100 mm). Health-related quality of life (HRQoL) and functional impairment were assessed using the short form (SF)-12 questionnaire and the German version of the Neck Pain and Disability Scale (NPAD) [16]. Based on MRI findings, CDHs were graded as either preforaminal or foraminal (Fig. 1).
‘Indirect’ epidural nerve root injection. The patient is positioned prone with the head rested in a face pillow. A 23G Chiba needle was used as a stylet and advanced through a Terumo Agani® needle (not shown in drawing) in coaxial technique to the lateral aspect of the facet joint under CT guidance. After negative aspiration/flashback, 0.3–0.5 ml of the contrast agent iopamidol diluted with normal saline (1:3) was applied to ensure free spread around the facet joint (white arrow) and along the cervical nerve root. Then, 4 mg of dexamethasone was administered followed by 1 ml of 0.5 % bupivacaine
Injection technique
All injections were performed under computed tomography (CT) guidance (Siemens SOMATOM Emotion®, Munich, Germany) in CT fluoroscopy mode. The ‘indirect’ technique according to Sutter et al. [19] was used in accordance with our standardized protocol. An illustration of the procedure is shown in Fig. 2. The patient was in prone position with the head rested in a face pillow. The correct level was determined with a lateral CT topogram. The posterior entry point was marked, disinfected with isopropyl and propyl alcohol with 2-phenylphenol (Kodan forte®, Schülke & Mayr AG, Zurich, Switzerland), and draped in a sterile manner. Lidocaine (1 %) (Lidocain Streuli®, Streuli Pharma AG, Uznach, Switzerland) was applied as local anesthesia using a Terumo Agani® needle, 18-G, Short Bevel (Zhejiang Kindly Medical Devices Co., Ltd., Zhejiang, China; Shanghai International Holding Corp. GmbH (Europe), Hamburg, Germany). Under CT guidance, a Chiba needle (ECOJEKT 23-G, 15 cm in length, HS Hospital Service S.p.A., Aprilia, Italy) was used as a stylet and advanced through the Terumo Agani® needle in coaxial technique to the lateral aspect of the facet joint. After negative flashback/aspiration, 0.3–0.5 ml of the contrast agent iopamidol 300 mg/ml (Iopamiro 300®, Bracco Suisse SA, Manno, Switzerland) diluted with normal saline (1:3) was applied. This ruled out intravascular needle tip placement and ensured free spread around the facet joint and along the cervical nerve root. Then, 4 mg of dexamethasone (Mephameson®, Mepha Pharma AG, Basel, Switzerland) was administered followed by 1 ml of 0.5 % bupivacaine (Bupivacain Hydrochlorid®, Sintetica S.A., Mendrisio, Switzerland). Throughout the procedure, the dose-length product in mGy*cm was recorded. Afterwards, patients were observed for any side effects in the outpatient department for a minimum of 2 h.
Outcome measurements, endpoints, and definition of responder and non-responder status
In the postinterventional phase, patients continued to have analgetics and physiotherapy as prescribed by their referring physician; there were no changes made to the concomitant therapy by the study protocol. VAS arm and neck pain were recorded ultra-early (15–45 min), early (1–4 h), intermediate (1–7 days), late (8–14 days) and at the 1-month as well as 1-year follow-up in patients who did not reach any of the study endpoints (see below). Functional and HRQoL outcome, including opioid use, work capacity, and satisfaction with ESI, were assessed at 1 month and 1 year as outlined in Supplementary Table 1. A change in the current therapy regimen by means of a second injection or surgery was considered a study endpoint.
Responders and non-responders were determined at the 1-month follow-up. Responders were patients who did not require any repeat ESI or surgery during the study interval and who stated a <80 % pain level compared to their baseline VAS arm pain. Non-responders were patients who had additional ESI or surgery within 1 month (and thereby reached the study endpoint) or who reported persisting arm pain of ≥80 % of their baseline VAS arm pain. Stability of the 1-month responder status was tested by evaluating the long-term outcome up to 1 year.
Statistical considerations
VAS arm and neck pain levels of responders and non-responders in the ultra-early, early, intermediate, and late follow-up were analyzed. Group differences between responders and non-responders of VAS arm and neck pain were compared using two-sided t tests. The trends of VAS arm and neck pain were plotted in percentages, with the absolute pre-injection VAS set at 100 %. Probability values of <0.1 (+) were considered a tendency, p values of <0.05 (*), <0.005 (**) or <0.001 (***) were considered significant.
Univariable logistic regression was used to assess the effect size of the relationship between >50 % pain relief at any given study visit (15 min to day 14) and responder status to ESI after 1 month. Since there were no baseline group differences in important patient characteristics such as age, gender, BMI, labor status, and opioid use, there was no need to build an additional multivariate model.
The software used for the statistical analysis was Stata v14 (StataCorp LP, College Station, TX, USA) and GraphPad Prism v5.0c (GraphPad Software, Inc. San Diego, CA, USA); the latter was used for drawing Fig. 4a and b.
Ethical considerations and data management
Written informed consent was obtained from all included patients. The study was conducted in accordance with the ethical standards of the Cantonal Ethical Review Board St. Gallen, Switzerland (EKSG 13/061) and with the Helsinki Declaration (1964, amended most recently in 2008) of the World Medical Association and registered under clinicaltrials.gov (Identifier: NCT01945554).
All data were collected by a full-time study nurse (C.L.). In order to conceal patients’ identities, each study participant was given a unique patient number for follow-up questionnaires and data management.
Results
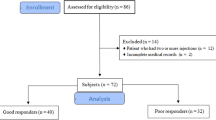
Out of 231 patients undergoing cervical ESI during the recruitment interval, 53 (22.9 %) met the inclusion criteria. Eight patients provided incomplete follow-up data and were thus excluded from analysis. A total of 45 patients built the final study cohort. Of these, 29 patients (64.4 %) were considered responders and 16 (35.6 %) non-responders at the 1-month follow-up. In the non-responder group, ten patients (62.5 %) underwent either surgical treatment (n = 7; 43.8 %) or a second injection (n = 3; 18.8 %) and six patients (37.5 %) had persistent arm pain ≥80 %. A flow chart of included patients is shown in Fig. 3.
Patient baseline data and procedure-related characteristics are shown in Table 1. All parameters were balanced between the two study groups, except for a trend towards a lower percentage of occupation in non-responders. Overall, preforaminal and foraminal CDHs were equally common and equally distributed among the groups. Besides a transient episode of dizziness in one patient and a vasovagal syncope in another (4.4 %), no further complications ascribed to ESI were noted.
Using the definition of responders and non-responders at 1 month as described above, patients with and without a treatment effect were well characterized (Table 2). Except for occupation and short-form SF-12 Mental Component Summary, all pain, functional, and HRQoL metrics 1 month after ESI were significantly in favor of the responder group.
Short-term relief from arm pain and its correlation with 1-month outcome
Figure 4a shows the trend of relative VAS arm pain (in %) until the 1-month follow-up. In general, both responders and non-responders experienced a distinct drop of about 65 % of their baseline pain intensity within the first 2 h after ESI. As early as 45 min after ESI, the study groups could be distinguished. Non-responders still suffered 42 %, whereas responders experienced more relief and stated a remaining 25 % baseline arm pain (p = 0.06). Non-responders had a significantly lower pain reduction after 1 h (42 vs. 23 %, p = 0.01) and benefited significantly less throughout the entire remaining follow-up time. In the intermediate follow-up period, this difference was quite strong at day 3 already, with non-responders experiencing 87 % and responders 33 % of their baseline VAS arm pain (p < 0.01). This difference became most apparent at day 5 (103 vs. 33 %, p < 0.01) and day 6 (93 vs. 34 %, p < 0.01).
Change in visual analog scale (VAS) pain (in %; group means and standard errors) after ‘indirect’ cervical epidural injections (ESI) in n = 29 and n = 16 patients that were considered responders and non-responders at 1 month, respectively. X-axis Time after ESI and ultra-early (15–45 min), early (1–4 h), intermediate (day 1 to 7) and late (day 8 to 14) period. Y-axis Change in VAS pain (in %). + = p < 0.1; * = p < 0.05; ** p < 0.005; *** = p < 0.001. a Irradiating arm pain. Note that patients considered to be responders at 1-month generally experienced a pain reduction of >50 %, whereas pain relief in non-responders was ≤50 %. Only 45 min after the intervention in the ultra-early phase, the graphs diverge significantly with responders experiencing a steeper dip in pain intensity. The difference of the two graphs is greatest at days 5 and 6 at the end of the intermediate phase. b Axial neck pain. Note that there was no significant difference between the groups in relative VAS neck pain during the early stage after ESI. The difference in relative VAS neck pain became significant in the intermediate phase at day 3
Effect size of the relationship 50 % VAS arm pain relief and 1-month responder status
The data presented in Fig. 4a suggest that 1-month responders generally experience relief of > 50 % from their baseline VAS arm pain, whereas the same is not true for non-responders. Therefore, we analyzed this assertion in more detail. Table 3 shows the effect sizes of the relationship between >50 % pain reduction at any given study visit and the 1-month responder status, calculated using a univariate logistic regression model. Note that as early as 4 h post-ESI, patients experiencing >50 % pain relief from baseline VAS arm pain were four times as likely to be 1-month responders as those experiencing 50 % or less pain relief (OR 4.04, 95 % CI 1.10–14.87, p = 0.04). This relationship was less marked at day 1, but regained statistical significance from day 3 onwards until the final follow-up. At days 5 and 6, patients experiencing a >50 % pain reduction were 18 times as likely to be responders as those experiencing 50 % or less pain reduction (OR 18.37, 95 % CI 3.39–99.64, p < 0.01).
Short-term relief from neck pain and its correlation with 1-month outcome
As patients with CDH frequently also report neck pain, we plotted the evolution of relative VAS neck pain that was present in n = 37 in Fig. 4b. Both responders and non-responders experienced an initial drop of about 65 % of their baseline VAS neck pain within the first 2 h. The effect of ESI on neck pain was similar in responders and non-responders until day 3 (intermediate phase), when a significant difference in relative VAS neck pain was observed (remaining pain, 59 vs. 33 %, p = 0.03). The effect was strongest on day 4 (60 vs. 29 %, %, p < 0.01) but fluctuated in the following days and weeks. At the 1-month follow-up, non-responders reported significantly more remaining VAS neck pain (70 vs. 22 %, p < 0.01).
1-year responder status
At the 1-year follow-up, 28 patients were considered treatment responders, and 17 (37.8 %) non-responders (n = 7 repeat ESI; n = 8 surgery; n = 2 persisting arm pain of ≥80 % of baseline VAS arm pain). Of the 28 1-year responders, 23 had been responders at the 1-month follow-up (82.1 %) and 11 out of 17 1-year non-responders had been non-responders at the 1-month follow-up (64.7 %). Pain, disability, and HRQoL outcomes of those patients not reaching one of the study endpoints are summarized in Table 4. Note that none of the patients used opioids for pain medication at the 1-year follow-up.
Discussion
This study provides detailed prospective data on ultra-early and early outcomes of CT-guided ESI for cervical radicular pain. The results clearly indicate that 1-month responders of cervical ESI can already be distinguished from non-responders at an early point in time as the evolution of irradiating pain is significantly different in both groups. This information helps to guide patient management following ESI into the right direction. Up to now, random guessing of the response to treatment was based on the individual physician’s experience.
To the best of our knowledge, only two studies [4, 22] assessed the ultra-early pain outcome after cervical ESI. While Desai et al. reported a positive correlation between any reduction of pain score at 15 min and pain reduction at 1 month [4], Wald et al. only found a strong correlation between the response at 2-week and 2-month outcomes [22]. In neither of these studies was the evolution of pain reduction recorded over the following hours and days. Our data confirm an overall pain reduction of about 65 % for both neck and arm pain within the first 15 min, but does not allow for discrimination between responders and non-responders at this early point in time. Since pain perception can vary considerably, ultra-early and early pain development should not be overemphasized. According to our findings, responders can be discriminated from non-responders with high certainty by their VAS arm pain about a week after ESI (OR 18.37, 95 % CI 3.39–99.64, p < 0.01, Fig. 4a). An easy-to-remember rule of thumb for clinicians managing patients with CDH can be deducted from this: in patients who experience a >50 % pain relief within 1 week after cervical ESI, pursuit of conservative management is justified, as further positive response is very likely. Translating this finding into clinical practice, we propose to establish a follow-up visit 1 week after ESI in order to evaluate the pain response. Alternative treatment options in case of absence of 50 % pain relief could be discussed.
Not much to our surprise, neck pain response to ESI fluctuated, and was less conclusive as a prognosis factor. The local anesthetizing effect of bupivacaine on nociceptive nerve endings of the cervical facet joints and paravertebral soft tissue might account for the observed immediate pain relief. Secondary progressive relaxation of neck muscles as arm pain improved throughout time led to a consecutive reduction of VAS neck pain.
Optionally, the injection can be repeated in patients who experience a clinically meaningful but transient positive response. In several previous reports [6, 9, 10, 13, 14, 17, 20] regarding the efficacy of ESI, repeat injections were part of the study design. In the present study, repeat injections were considered an exclusion criterion because patients opting for repeat injection represent a selected patient population. While they may have responded transiently and significantly to the first injection or not at all, definitive responders did not undergo a repeat injection. Furthermore, the impact of a repeat injection may differ from the first injection (booster effect). Whether repeat injections offer a valuable salvage treatment remains to be proven. There is little evidence in the literature: apart from our own experience [2] with repeat cervical ESI leading to an equally good clinical short-term response as first ESI, only one study found that after the first injection had failed, in seven patients with repeat injections, three (42.8 %) experienced significant pain relief [20]. Long-term follow-up of our data might shed more light on this aspect. Looking at the present results in the context of an often long waiting list for elective spine surgery, for patients with little or no treatment effect 1 week after ESI, surgical treatment could be scheduled. The indication should always take into account age, labor status, co-morbidities, as well as disease-specific factors, presence of new neurological deficits/myelopathy (red flags) and the patient’s personal preference.
Several authors [6, 10, 13, 14] suggested that ESI is a surgery-sparing salvage treatment, but did not provide control groups. In those studies, we see a selection bias in surgical candidates willing to try ESI before surgery. The overall 1-month surgery rate in the current study was 15.5 % (17.8 % at 1 year). This is higher than for example the 1-month rate reported in Cohen’s study (5.5 %, [3]) and is probably due to the fact that there are shorter waiting times for elective spine surgery in Switzerland. Symptom duration before ESI was not associated with treatment success—an observation of the current study that is in line with Lee et al. [13] and in contrast to previous reports [14].
Although we recorded no major complications in the past [2] and in the current study, it should be kept in mind that neurological complications after cervical injections have been reported and caution is advised [15]. A detailed anatomic account of the variable vascular anastomoses near and within the cervical intervertebral foramina was given by Huntoon [7]. We adapted a novel ‘indirect’ cervical nerve root injection technique described by Sutter et al. [19] similar to a posterolateral facet joint injection, which is equally efficient as the transforaminal technique and presumably safer. Similar efforts to come up with an innovating ‘indirect’ technique through the ipsilateral facet joint were made by Kelekis et al. [9]. We also apply non-particulate dexamethasone, which has been demonstrated to be as efficient as, for example, particulate triamcinolone in cervical transforaminal ESI [5, 12, 17].
Arm pain assessment using the 100-mm VAS is common practice in many clinical settings. Keeping a pain-level diary based on VAS is easy to do and very practical. The VAS is time-saving for both the patient (when completing) and for the physician (when evaluating) the forms and has a much lower language barrier than questionnaires.
Strengths and weaknesses
‘Indirect’ cervical ESI was performed in accordance with a standard protocol under CT guidance. As a single, experienced radiologist performed all interventions, biases arising from differences in expertise or technique could be eliminated. We chose to study the effect of short-term pain relief on the 1-month follow-up because the patient’s condition at this point in time can be best attributed to the direct effects of the injection, as opposed to a later point in time (e.g., 3 or 6 months post-ESI) when the natural course of the disease with possible CDH recurrences might skew the results. Our data suggest that the majority of patients deemed responders or non-responders at the 1-month follow-up remained so at the 1-year follow-up as well. To validate our findings, we will report long-term follow-up results from 3, 6, 12, and up to 24 months as soon as their collection has been completed, which will also shed more light on the change in work capacity and tapering off of medication, for example. We further acknowledge confounding factors such as different analgesics’ regimen and concomitant physiotherapy, which were not controlled for. The effect sizes of the present findings with 45 patients were sufficiently large to describe significant differences. However, a larger patient sample could have helped to make more stable point estimates, as some of the confidence intervals were large. Strict exclusion criteria resulted in inclusion of less than a quarter of the screened patients. For example, patients with VAS arm pain <20/100 mm and with concomitant relevant osteophytic foraminal stenosis or with previous surgery—both of which are known to have a negative impact on the response rate to ESI [12]—were not included. Besides the study endpoints ‘second injection’ or ‘surgery’, the definition of non-response as VAS arm pain ≥80 % of the baseline pain intensity was set relatively high. We acknowledge that the reported results might be valid for a selected cohort of patients only. Due to this selection bias and the small sample size, we advise the reader to interpret the study results carefully. The deducted conclusions should be understood as possible guidance for patient management, but not as a universal consensus. Future studies validating our findings will be needed. We did not use a control group because we consider sham injections unethical and acknowledge the well-known problems of performing a true placebo injection.
Conclusions
CT-guided ‘indirect’ cervical ESI for radiculopathy secondary to CDH leads to a favorable 1-month outcome in about two-thirds of patients. Responders and non-responders can be identified as early as hours and days following the injection through the course of their arm pain. A follow-up period of about 1 week after ESI may be helpful in managing patients: those reporting less than half of their baseline VAS arm pain at the end of the first week (day 6) post-ESI are 18 times more likely (positive predictive value 0.91) to be responders at 1 month. Additional treatment options could be discussed for patients who do not experience pain relief of more than 50 % within the first week because 1-month treatment success appears less likely.
References
Anderberg L, Annertz M, Persson L, Brandt L, Saveland H (2007) Transforaminal steroid injections for the treatment of cervical radiculopathy: a prospective and randomised study. Eur Spine J 16:321–328
Brandle K, Stienen MN, Neff A, Hildebrandt G, Joswig H (2016) Ten-day response to CT-guided spinal infiltration therapy in more than a thousand patients. J Neurol Surg A Cent Eur Neurosurg 77:181–194
Cohen SP, Hayek S, Semenov Y, Pasquina PF, White RL, Veizi E, Huang JH, Kurihara C, Zhao Z, Guthmiller KB, Griffith SR, Verdun AV, Giampetro DM, Vorobeychik Y (2014) Epidural steroid injections, conservative treatment, or combination treatment for cervical radicular pain: a multicenter, randomized, comparative-effectiveness study. Anesthesiology 121:1045–1055
Desai A, Saha S, Sharma N, Huckerby L, Houghton R (2014) The short- and medium-term effectiveness of CT-guided selective cervical nerve root injection for pain and disability. Skelet Radiol 43:973–978
Dreyfuss P, Baker R, Bogduk N (2006) Comparative effectiveness of cervical transforaminal injections with particulate and nonparticulate corticosteroid preparations for cervical radicular pain. Pain Med 7:237–242
Eun SS, Chang WS, Bae SJ, Lee SH, Lee DY (2010) Computed tomography fluoroscopy-guided selective nerve root block for acute cervical disc herniation. J Korean Neurosurg Soc 48:419–422
Huntoon MA (2005) Anatomy of the cervical intervertebral foramina: vulnerable arteries and ischemic neurologic injuries after transforaminal epidural injections. Pain 117:104–111
International Standard Classification of Occupations (ISCO). http://www.ilo.org/public/english/bureau/stat/isco/. Accessed 15 Sept 2016
Kelekis A, Filippiadis DK, Velonakis G, Martin JB, Oikonomopoulos N, Brountzos E, Kelekis N (2014) Fluoroscopically guided infiltration of the cervical nerve root: an indirect approach through the ipsilateral facet joint. Pain Physician 17:291–296
Kolstad F, Leivseth G, Nygaard OP (2005) Transforaminal steroid injections in the treatment of cervical radiculopathy. A prospective outcome study. Acta Neurochir (Wien) 147:1065–1070, discussion 1070
Lee JH, Lee SH (2011) Comparison of clinical effectiveness of cervical transforaminal steroid injection according to different radiological guidances (C-arm fluoroscopy vs. computed tomography fluoroscopy). Spine J 11:416–423
Lee JW, Park KW, Chung SK, Yeom JS, Kim KJ, Kim HJ, Kang HS (2009) Cervical transforaminal epidural steroid injection for the management of cervical radiculopathy: a comparative study of particulate versus non-particulate steroids. Skelet Radiol 38:1077–1082
Lee SH, Kim KT, Kim DH, Lee BJ, Son ES, Kwack YH (2012) Clinical outcomes of cervical radiculopathy following epidural steroid injection: a prospective study with follow-up for more than 2 years. Spine (Phila Pa 1976) 37:1041–1047
Lin EL, Lieu V, Halevi L, Shamie AN, Wang JC (2006) Cervical epidural steroid injections for symptomatic disc herniations. J Spinal Disord Tech 19:183–186
Rathmell JP, Benzon HT, Dreyfuss P, Huntoon M, Wallace M, Baker R, Riew KD, Rosenquist RW, Aprill C, Rost NS, Buvanendran A, Kreiner DS, Bogduk N, Fourney DR, Fraifeld E, Horn S, Stone J, Vorenkamp K, Lawler G, Summers J, Kloth D, O’Brien D Jr, Tutton S (2015) Safeguards to prevent neurologic complications after epidural steroid injections: consensus opinions from a multidisciplinary working group and national organizations. Anesthesiology 122:974–984
Scherer M, Blozik E, Himmel W, Laptinskaya D, Kochen MM, Herrmann-Lingen C (2008) Psychometric properties of a German version of the neck pain and disability scale. Eur Spine J 17:922–929
Shakir A, Ma V, Mehta B (2013) Comparison of pain score reduction using triamcinolone vs. dexamethasone in cervical transforaminal epidural steroid injections. Am J Phys Med Rehabil 92:768–775
Strobel K, Pfirrmann CW, Schmid M, Hodler J, Boos N, Zanetti M (2004) Cervical nerve root blocks: indications and role of MR imaging. Radiology 233:87–92
Sutter R, Pfirrmann CW, Zanetti M, Hodler J, Peterson CK (2011) CT-guided cervical nerve root injections: comparing the immediate post-injection anesthetic-related effects of the transforaminal injection with a new indirect technique. Skelet Radiol 40:1603–1608
Vallee JN, Feydy A, Carlier RY, Mutschler C, Mompoint D, Vallee CA (2001) Chronic cervical radiculopathy: lateral-approach periradicular corticosteroid injection. Radiology 218:886–892
Vasudeva V, Chi J (2015) Defining the role of epidural steroid injections in the treatment of radicular pain from degenerative cervical disk disease. Neurosurgery 76:N16–N17
Wald JT, Maus TP, Geske JR, Diehn FE, Kaufmann TJ, Murthy NS, Thielen KR, Watson S (2013) Immediate pain response does not predict long-term outcome of CT-guided cervical transforaminal epidural steroid injections. AJNR Am J Neuroradiol 34:1665–1668
Acknowledgments
The authors thank their study nurse, Cornelia Lüthi, for her outstanding diligence in pursuing follow-up. The authors thank Carolin Hock for proofreading the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for this study. Expenditures (questionnaires, study nurse) were financed by the Department of Neurosurgery at Cantonal Hospital St. Gallen.
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
The study was conducted in accordance with the ethical standards of the Cantonal Ethical Review Board St. Gallen, Switzerland (EKSG 13/061) and with the Helsinki Declaration (1964, amended most recently in 2008) of the World Medical Association.
Informed consent
Written informed consent was obtained from all included patients.
Additional information
Comments
This interesting article addresses one of the many controversies in neurosurgery, namely when to consider surgical intervention after failed epidural steroid injection (ESI) and how to define success/failure in ESI for brachialgia. The authors correctly stated that there is no strong clinical evidence supporting ESI, but despite this, ESI is considered in several neurosurgical centers as the first treatment option for brachialgia in the absence of neurological deficit or impending cord compression.
The issues neurosurgeons face in clinical practice is when to shift to surgical intervention (anterior cervical discectomy or foraminotomy), how to define the success rate of ESI, and above all how counsel patients regarding treatment options. Naturally, a more conservative approach with a better risk profile is favored by patients (and insurance companies!). But when should one stop ESI? After one, two, or three injections? This is very difficult to say when the literature lacks evidence in this regard. The other factor to keep in mind is the possible conversion of simple brachialgia into chronic neuropathic pain (possibly early decompression may avoid this occurrence).
This article (considering the limitation of the study) offers a relatively simple algorithm; “>50 % pain relief at 1 week predicts a poor outcome following ESI and an alternative surgical option should be considered.”
A larger study in the future should confirm or modify this, but at least it is a starting point and the authors should be congratulated for their effort.
Jibril Osman Farah
Liverpool, UK
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
(DOCX 73 kb)
Rights and permissions
About this article
Cite this article
Joswig, H., Neff, A., Ruppert, C. et al. The value of short-term pain relief in predicting the 1-month outcome of ‘indirect’ cervical epidural steroid injections. Acta Neurochir 159, 291–300 (2017). https://doi.org/10.1007/s00701-016-2997-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-016-2997-8