Abstract
An accurate preoperative evaluation of the hepatic function and application of portal vein embolization in selected patients have helped improve the safety of major hepatectomy. In planning major hepatectomy, however, several issues remain to be addressed. The first is which cut-off values for serum total bilirubin level and prothrombin time should be used to define post-hepatectomy liver failure. Other issues include what minimum future liver remnant (FLR) volume is required; whether the total liver volume measured using computed tomography or the standard liver volume calculated based on the body surface area should be used to assess the adequacy of the FLR volume; whether there is a discrepancy between the FLR volume and function during the recovery period after portal vein embolization or hepatectomy; and how best the function of a specific FLR can be assessed. Various studies concerning these issues have been reported with controversial results. We should also be aware that different strategies and management are required for different types of liver damage, such as cirrhosis in hepatocellular carcinoma, cholangitis in biliary tract cancer, and chemotherapy-induced hepatic injury.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Major hepatectomy has become a common procedure in patients with large hepatocellular carcinoma (HCC) or cholangiocarcinoma. Over the past decade, in parallel with improvements in systemic chemotherapy for liver tumors, the indications for major hepatectomy have expanded to include metastases in the liver, especially colorectal liver metastases (CRLM). At the same time, the safety of hepatectomy has improved owing to the appropriate preoperative assessment of the liver function and advances in surgical techniques, leading to decreased intraoperative blood loss.
The risk of post-hepatectomy liver failure (PHLF) has fallen over the past decade, but remains high for several types of procedures; a nationwide survey of board-certified training institutions by the Japanese Society of Hepato-Biliary-Pancreatic Surgery showed 90-day mortality rates of 10.3% after left trisectionectomy and 6.7% after hepatopancreatectomy [1]. One of the most extensive types of hepatectomy, associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) [2], which was initially introduced to induce rapid hypertrophy of the future liver remnant (FLR) before second-stage major hepatectomy [3], was still associated with a mortality rate of over 5% in the latest series [4].
Numerous recent studies have reported on strategies for safe major hepatectomy. A historical landmark was the introduction of portal vein embolization (PVE) in the mid-1980s to induce hypertrophy of the FLR [5,6,7]. The introduction of the Makuuchi criteria, which are based on the indocyanine green retention rate at 15 min (ICG-R15) [8], in the mid-1990s facilitated the preoperative assessment of the liver function. Since the 1990s, various methods have been proposed to accelerate FLR hypertrophy and evaluate the function of the FLR before surgery. However, several issues remain unresolved or controversial in the preoperative planning for major hepatectomy, as summarized in Fig. 1 and reviewed in this article.
How should PHLF be defined?
A number of definitions of PHLF have been proposed. Most of them focus on the serum total bilirubin level and prothrombin time. Among the most commonly cited definitions are prothrombin time < 50% and serum bilirubin level > 50 μmol/L (“50–50 criteria”) on the fifth postoperative day, proposed by Balzan et al. [9], and peak postoperative bilirubin level > 7.0 mg/dL (120 μmol/L), proposed by Mullen et al. [10]. Both criteria were shown to predict postoperative liver-related death. However, the bilirubin level > 7.0 mg/dL (120 μmol/L) criterion is simpler, and its use as an index of PHLF seems more feasible because this criterion predicted liver failure-related death with a sensitivity of 93.3% and specificity of 94.3%, while the 50–50 criteria predicted in-hospital death, including mortality not related to liver failure, with a sensitivity of 69.6% and specificity of 98.5% [9, 10].
Many studies have evaluated the preoperative factors that predicted PHLF based on the above definitions. The factors reported to predict PHLF included low immediate postoperative platelet count in patients with HCC or preoperative cholangitis in patients with hilar cholangiocarcinoma [11, 12]. When discussing these studies, it should be noted that not all patients with preoperative factors predictive of PHLF died after hepatectomy. Whether or not hepatectomy should be considered contraindicated in all patients with preoperative factors predictive of PHLF is another question.
The International Study Group of Liver Surgery (ISGLS) proposed a grading system for PHLF (Table 1) based on the literature and expert consensus [13], and this can be applied widely in clinical settings. Although the ISGLS definition of PHLF is based on the postoperative bilirubin level and prothrombin time, the ISGLS grading of severity of PHLF is based on the degree of deviation from normal clinical management. In clinical practice, it is reasonable for grade B and C PHLF to generally be considered clinically relevant PHLF and differentiated from grade A PHLF.
Is pre- or post-operative platelet count helpful as a simple index to predict PHLF?
Several studies have reported that a preoperative platelet count below 10–15 × 104/μL was associated with PHLF or mortality [14,15,16,17,18,19,20,21]. The aspartate aminotransferase (AST)/platelet count ratio index (APRI = AST level [/upper normal limit]/platelet counts [109/L] × 100) initially proposed by Wai et al. as a simple but highly reliable predictor of significant fibrosis and cirrhosis in patients with treatment-naïve chronic hepatitis C [22] and recommended as the preferred noninvasive test for assessing the presence of cirrhosis in the Asian-Pacific Association for the Study of the Liver consensus guidelines [23] has also been reported as a useful predictor of PHLF [24,25,26]. Ichikawa et al. [24] and Mai et al. [26], respectively, reported AST [IU/L]/platelet count [104/μL] > 10 and APRI > 0.55 as predictors of PHLF. Most of these studies evaluated patients with HCC, and one by Ratti et al. evaluated patients with CRLM treated with preoperative chemotherapy (mainly an oxaliplatin-based regimen). The association of thrombocytopenia with PHLF in these patients seems reasonable, as thrombocytopenia indicates portal hypertension that occurs either as a result of liver cirrhosis [27] or oxaliplatin-induced sinusoidal injury [28, 29]. In contrast, in the series by Golriz et al. [20], the most frequent liver disease was cholangiocarcinoma, and HCC accounted only for 15% of the total patients. Other studies showed that an immediate postoperative low platelet count < 10 × 104/μL or postoperative > 40% decrease in the platelet count predicted PHLF or mortality [11, 30, 31]. Margonis et al. evaluated the volume regeneration of remnant liver 2 months after major hepatectomy in 99 patients, and a postoperative platelet count < 15 × 104/μL was associated with a significant reduction in FLR hypertrophy [32]. These findings suggest that platelets may play a role in aiding liver regeneration, and several proteins, such as vascular endothelial grow factor or thrombospondin-1 secreted from platelet α-granules, have been suggested to modulate liver regeneration [33], although the mechanism remains unclear.
What is the minimal safe FLR volume?
Results of systemic volumetry to determine the volume of liver to be resected were first reported in 1997 by Kubota et al. [34], who showed that resection of up to 60% of the nontumorous liver parenchyma could be tolerated in patients with an ICG-R15 of < 10%, and resection of up to 50% of the nontumorous liver parenchyma could be tolerated in patients with an ICG-R15 of 10–20%. Notably, however, there were no deaths in their series, indicating that their criteria of 50% or 60% resection did not reflect the upper limit of safe hepatectomy. Resection of up to 80–90% of the liver was described in 2 case reports published in the 1960s and 1980s [35, 36], but the FLR volume in these reports was only estimated by the surgeons, without an exact volumetric measurement.
Nagasue et al., who were the first to study the regeneration of the FLR using computed tomography (CT), estimated that extended right lobectomy resected 80% of the whole liver [37]. Supporting this estimate, Abdalla et al., in a volumetric study of 102 patients without any hepatobiliary disease, showed that the left liver (segments II, III, and IV) could be very small; in their series, the left liver volume accounted for ≤ 25% of the total liver volume (TLV) in more than 10% of patients [38].
Currently, the consensus regarding the minimal safe FLR volume in patients with a normal liver is approximately 25–30% of the total functional liver volume (TFLV), where the TFLV is the volume of the liver minus the volume occupied by tumor [39,40,41]. At present the University of Texas MD Anderson Cancer Center’s criteria for the standardized liver volume (SLV) of ≥ 20% is the minimal acceptable FLR volume for a safe resection in a normal liver [42].
Should the FLR/TFLV ratio or FLR/SLV ratio be used to assess FLR adequacy?
Another important issue in preoperative planning for major hepatectomy is whether the FLR volume ratio should be estimated using the TFLV or the SLV. While the TFLV is measured directly by CT, the SLV is calculated using the body surface area. Generally, the ratio of the FLR volume to the TFLV is calculated using the formula FLR volume [ml]/(TLV [ml]—tumor volume [ml]). This formula is based on the assumption that the hepatic parenchyma excluding the tumor constitutes the TFLV. This method has been criticized, however, because it may be inaccurate in patients with multiple tumors, may underestimate tumor volume in patients with lesions beyond the resolution of imaging, and may underestimate the liver volume under conditions of compromise due to cholestasis in patients with bile duct tumors [43].
In recipients of liver transplants, not TLV but SLV is routinely used to estimate the required graft volume because the liver function of these patients is deteriorated and their liver is usually atrophic or abnormally enlarged. Yamashita et al. recently showed that hepatic atrophy of ≥ 10% after chemotherapy was a predictor of hepatic insufficiency after resection of CRLM [44]. Their results suggested the risk of overestimation of the FLR ratio based on TFLV, especially in patients who receive extensive preoperative chemotherapy for CRLM. Use of the FLR/TFLV ratio after PVE may also produce inaccurate estimates of FLR adequacy if PVE results in greater atrophy of the liver to be resected than hypertrophy of the FLR.
Thus far, only a few studies have compared postoperative outcomes between the two methods of FLR evaluation. Ribero et al. compared the FLR/TFLV and FLR/SLV ratios in 243 patients who underwent major hepatectomy [45]. They reported that TFLV was lower than the SLV in 60% of the patients. In addition, the incidence of PHLF, defined as peak postoperative serum total bilirubin level > 7.0 mg/dL (120 μmol/L), was significantly higher in the patients who underwent hepatectomy without PVE based on a safe FLR/TFLV ratio but deficient FLR/SLV ratio than in the patients who underwent hepatectomy without PVE based on safe FLR/SLV and FLR/TLV ratios (6/27 [22.2%] vs. 8/162 [4.9%], p = 0.001), although the mortality rates in the two groups were similar [45]. Similarly, Kim et al. evaluated the outcomes of right hepatectomy in 74 patients and showed that the incidence of PHLF was significantly higher in the patients with an FLR/SLV ratio < 30% than in patients with an FLR/SLV ratio ≥ 30%, but PHLF ratio did not markedly differ between patients with an FLR/SLV ratio < 40% versus ≥ 40% or the patients with an FLR/TFLV ratio < 40% versus ≥ 40% [46]. These two studies suggested that an evaluation by the FLR/SLV ratio would be more appropriate for predicting the risk of PHLF. In both of these studies, however, the number of patients with a safe FLR/SLV ratio but an insufficient FLR/TFLV ratio was small (5 of 243 patients in the Ribero et al. series, 1 of 74 patients in the Kim et al. series).
Several formulae to calculate the SLV have been reported based on different patient cohorts. Olthof et al. compared the TLV with the SLV calculated according to 16 different formulae for 529 patients in the ALPPS registry, 85% of them Caucasian, and showed that the majority of the formulae produced SLV values substantially higher than the measured TLV [47]. Therefore, whether or not the SLV estimated from the body surface area more appropriately reflects the total liver function than the TLV measured on CT or if a higher value of SLV than TLV results in expanding the indication of PVE to reduce the risk of PHLF remains unclear.
What is the best method for the preoperative assessment of the FLR function?
ICG retention rate
The combination of the FLR/TFLV ratio and ICG-R15 value is commonly used to predict the risk of PHLF, especially in Asian countries. The Makuuchi criteria [8] regulate the degree of hepatic resection based on the ICG-R15 value, and the utility of these criteria were recently validated with a Japanese national clinical database showing a higher incidence of postoperative Clavien–Dindo grade > III morbidities in the patients exceeding the criteria [48].
The ICG-R15 value, however, reflects the function of the TFLV but not that of the FLR, and whether or not a gain of FLR volume as a result of PVE is a true reflection of an improved FLR function remains unclear. Several studies have suggested that the FLR function increases before hypertrophy of volume is apparent on CT [49, 50]. Uesaka et al. measured the biliary ICG concentration from multiple biliary drainage tubes placed for draining the entire liver in 8 patients who underwent right PVE and reported that the ratio of ICG excretion in the FLR to ICG excretion in the whole liver increased 20% on average after PVE, while the FLR/TFLV ratio increased only 8% on average [49].
Pattern of FLR volume regeneration
A number of studies have evaluated the relationship between volume regeneration of the FLR and functional recovery of the FLR. Studies evaluating chronological changes in the remnant liver volume after donor hepatectomy for living donor liver transplant showed a rapid volume increase in the early (within 3 months) postoperative period despite functional recovery taking up to one year for normalization [51, 52]. Maeda et al. recently evaluated the volume change following PVE and also 7 days after extended hepatectomy in 289 patients with perihilar cholangiocarcinoma [53]. They showed a significant negative correlation between the kinetic growth rate (kGR), defined as the change in the FLR/SLV ratio [%] divided by the interval [weeks] [54] after PVE and kGR after hepatectomy. This result makes sense from the viewpoint that remarkable FLR hypertrophy occurred before rather than after surgery and was associated with a reduced incidence of PHLF. These authors also showed that there was no association between the kGR after either PVE or hepatectomy and the incidence of hepatic insufficiency. They concluded that there was a discrepancy between the recovery of the FLR volume and the recovery of the FLR function in the early phase of liver regeneration [53]. Because most patients in their series had hilar cholangiocarcinoma, other factors, such as perioperative cholangitis, might have affected the incidence of PHLF.
Watanabe et al. evaluated the factors that affected hypertrophy of the FLR after PVE and showed that an initial FLR volume ≥ 35% of the TLV, alkaline phosphatase level ≥ 450 IU/dL, and cholinesterase level < 220 ng/dL were independent predictors of insufficient FLR hypertrophy (< 25% increase) [55]. An inverse correlation between the FLR volume and hypertrophic rate was reported in a series of donor hepatectomies that showed faster regeneration after right hepatectomy than after left hepatectomy [52] as well as in an experimental study of the ALPPS model in rats [56]. The ALPPS study also showed that a small FLR volume was associated with increased severity of sinusoidal injury and elevated activation of hepatic progenitor cells after ALPPS. These findings suggested discrepancy between functional recovery and volume regeneration of FLR.
Specific imaging methods proposed to evaluate the FLR function
For the preoperative evaluation of the FLR function, several methods have been reported (Table 2).
Two methods are based on CT. Nagino et al. evaluated the change in the FLR volume and plasma disappearance rate of ICG (kICG) in patients who underwent extended hepatectomy following PVE for biliary cancer; the study included 176 survivors and 17 patients who died due to postoperative complications. The authors showed that FLR hypertrophy did not distinguish between survivors and non-survivors, but the kICG after PVE did, so they proposed a kICG of the FLR ≥ 0.05, with the kICG of the FLR defined by the formula kICG × FLR [ml]/TLV [ml], as a criterion for safe hepatectomy [57]. This cut-off value was validated in their later series [58, 59]. Shindoh et al. proposed evaluating the FLR function using the kGR, defined as the increase in the FLR volume from baseline divided by the length of time in weeks after PVE, and this was shown to be a better predictor of postoperative morbidity and mortality than the preoperative FLR/SLV ratio or FLR hypertrophy rate in a large series of patients [54]. However, in another series, kGR did not correlate with outcomes after ALPPS [60]. In fact, the high incidence of morbidity and mortality suggested that no volumetric measurement predicted outcomes after ALPPS.
Scintigraphy using technetium 99m (99mTc)-labeled galactosyl human serum albumin (GSA), which is mainly available in Japan, was developed on the basis that GSA receptors are expressed exclusively on functional hepatocytes involved in the clearance of glycoproteins containing terminal galactose residues from the circulation [61]. In Europe, however, 99mTc-labeled mebrofenin hepatobiliary scintigraphy is more popular than 99mTc-labeled GSA hepatobiliary scintigraphy. Mebrofenin enters hepatocytes and is excreted into the bile canaliculi unmetabolized; therefore, 99mTc-labeled mebrofenin hepatobiliary scintigraphy measures the kinetic process of the uptake and excretion by hepatocytes [62]. Measuring the uptake rate of these substances using single-photon emission computed tomography permits the estimation of the function of specific regions of the liver, and several studies have shown that the FLR function measured using hepatobiliary scintigraphy was more accurate than the FLR volume for predicting PHLF [63,64,65,66]. Scintigraphy is more expensive than CT, and methods to measure the uptake rate are rather complicated. In addition, the possibility of underestimating the left lobe function and overestimating the right lobe function was reported [67, 68].
A simpler method of evaluating the FLR function is magnetic resonance imaging with intravenous injection of gadoxetic acid contrast medium, which is transported into hepatocytes. The increase in the signal intensity in the FLR in the hepatobiliary phase compared to the unenhanced phase might be an indicator of the FLR function that could predict the risk of PHLF [69, 70]. Graaf et al. proposed using the 99mTc-labeled mebrofenin uptake rate in the FLR [%/min] divided by the body surface area [m2] ≤ 2.69%/min/m2 as a predictor of PHLF [65]. This predictor was recently validated in a multi-institutional study from the ALPPS registry [71], showing that an uptake rate ≤ 2.7% min/m2 and daily kGR ≤ 4.1% were independent predictors of ISGLS grade B/C PHLF.
Should PVE or ALPPS be used to induce hypertrophy of the FLR?
Another key question is whether PVE or ALPPS is superior for ensuring an adequate FLR for two-stage hepatectomy. Initially PVE was performed mostly in patients with hilar cholangiocarcinoma [5]. As the indications for hepatectomy for CRLM expanded to include even advanced bilobar disease, the use of major hepatectomy and, in parallel, the use of PVE increased remarkably [72, 73]. Furthermore, the development of chemotherapy contributed to the improvement of the prognosis after hepatectomy [74].
Jaeck et al. proposed two-stage hepatectomy for bilobar multiple CRLM, consisting of wedge resection of tumors in the FLR followed by PVE and second-stage major hepatectomy, in order to minimize the risk of both liver failure after major hepatectomy and rapid tumor growth after PVE [75]. In this series, the waiting periods between first hepatectomy and PVE and between PVE and second hepatectomy were 5–109 days and 4–6 weeks, respectively. Given these findings, ALPPS seems superior to PVE, as with ALPPS, FLR hypertrophy was confirmed after a median interval of 9 days (range 5–28) following right portal vein ligation and in situ hepatic parenchymal dissection in the first operation [3].
A randomized controlled trial from a Scandinavian group that compared resection rates between two-stage hepatectomy with PVE and ALPPS showed higher resection rates with ALPPS than PVE (44/48 [92%] vs. 28/49 [57%], p < 0.0001) [76]. In that series, the mean kGR [54] during the first 7 days was 12.3% in the PVE group and 35.4% in the ALPPS group [76]. Interestingly, such remarkable hypertrophy has also been shown even without actual liver parenchymal partition. Another randomized controlled trial compared PVE and liver partition with portal vein ligation using radiofrequency for virtual liver parenchymal partition (RALPP) [77] and showed a significantly more rapid increase in the FLR volume after the virtual liver parenchymal partition (mean increase of 80.7% after a median of 20 days after RALPP vs. 18.4% after a median of 35 days after PVE, p < 0.001) [78]. Other modified approaches with only partial parenchymal dissection or with tourniquet placement without splitting induced a 60–70% increase in the FLR volume in 7–14 days [79,80,81].
Although several experimental studies with animal models of ALPPS have suggested that cytokines might be associated with enhanced FLR hypertrophy [82,83,84], how much discrepancy exists between the recovery of the FLR volume and that of the FLR function after ALPPS has not been fully elucidated [85, 86].
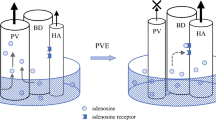
Considering the rather high rate of complications of ALPPS, including biliary fistula after the first-stage operation and PHLF after second-stage hepatectomy, routine application of ALPPS for extensive disease is not yet warranted; PVE, a less-invasive method for inducing FLR hypertrophy, cannot be replaced with ALPPS. The use of spherical microspheres as embolization material or additional embolization of segment IV for extended right hepatectomy may enhance PVE-induced FLR hypertrophy [87]. The median hypertrophy rate of segments II and III after embolization of the right portal vein and segment IV branches exceeded 50% [88, 89]. The combination of PVE with transarterial chemoembolization is also effective, especially in patients with HCC [90, 91]. Furthermore, liver venous deprivation combining PVE with ipsilateral hepatic vein embolization, first reported by Nagino et al. [92] and recently increasingly frequently reported [93,94,95], shows promise. In these studies, although the interval until hepatectomy was 3 to 4 weeks, the degree of hypertrophy [96] or kGR was higher after liver venous deprivation than after PVE alone, and there were few procedure-related or postoperative severe complications. Rapid functional recovery was also confirmed using hepatobiliary scintigraphy [97]. LVD may therefore be a new and safe approach for optimizing liver regeneration before major hepatectomy [98].
What issues need to be considered in planning for major hepatectomy for different types of disease?
Major hepatectomy is indicated for various types of liver tumor, and the background liver damage and approach to surgery, especially whether or not the extrahepatic bile duct is resected, differ by type of disease. These factors influence the degree of liver regeneration. For example, several studies evaluating the liver function by 99mTc-mebrofenin hepatobiliary scintigraphy have shown that the liver function in patients with perihilar cholangiocarcinoma was similar to that in patients with HCC and worse than that in patients with benign liver tumor [99, 100]. Therefore, management should be tailored to the type of disease.
HCC
HCC generally develops in livers damaged by chronic hepatitis or cirrhosis. The ICG test has been a popular method of estimating the degree of liver damage, although it is mostly performed in Asian countries. In the Barcelona Clinic Liver Cancer staging system [101], which is mainly used in Europe, hepatectomy is not indicated in patients with HCC with portal hypertension, defined as a hepatic venous pressure gradient ≥ 10 mmHg [102]. This seems reasonable, as portal hypertension resulting in splenomegaly, thrombocytopenia, or varicose veins is more common in patients with HCC than in those with other hepatic tumors. However, routinely measuring the hepatic venous pressure is not practical. A platelet count-based index such as the APRI is a simple and feasible predictor of liver fibrosis [22], and its validity as a predictor of postoperative outcomes in patients with HCC has been reported [103, 104]. Another study by Navarro et al. showed that a platelet count < 140 × 109/L and FLR/body weight ratio < 0.55% were independent predictors of clinically relevant PHLF in patients with HCC, but an ICR-R15 > 11% and FLR/TLV ratio < 35% were not [21].
Yamashita et al. compared the outcomes after PVE between patients with HCC, patients with biliary tract cancer, and patients with CRLM and showed that subsequent major hepatectomy was achieved most frequently in the patients with HCC (HCC, 64/70 [91%]; biliary tract cancer, 133/172 [77%]; CRLM, 59/77 [77%]; p = 0.029). However, while disease progression was the most frequent reason for deciding against hepatectomy in the patients with biliary tract cancer (79%) and CRLM (83%), it was cited as the reason for only 33% of dropout cases among patients with HCC [105]. These results suggest that the rather slow growth of HCC is associated with a long waiting period after PVE.
Chan et al. compared the outcomes of PVE and ALPPS in patients with HCC, most of whom had HCC related to hepatitis B virus infection. The rates of hypertrophy of the FLR and resectability were both significantly higher in the ALPPS group than in the PVE group, and the postoperative morbidity and mortality rates were similar in the PVE and ALPPS groups [4]. Notably, in their series, the median increment in the FLR volume after the first operation of ALPPS was 48%, which was smaller than the figures from other studies, mainly of patients with CRLM.
We should be aware that HCC is associated with a higher risk of insufficient hypertrophy after PVE than other types of cancer affecting the liver.
Biliary tract cancer
Major hepatectomy for biliary tract cancer is mainly indicated in patients with perihilar cholangiocarcinoma. Management of obstructive jaundice and cholangitis is required both before and after hepatectomy because the procedure usually is accompanied by extrahepatic bile duct resection (EBR). Takagi et al. showed that the regeneration rate of the FLR during the early postoperative period was lower after major hepatectomy with EBR than after major hepatectomy without EBR both in rat models [106] and in a study of 244 human patients [107]. These authors also showed that EBR was an independent predictor of PHLF among the patients who underwent right hepatectomy [107]. Although several studies have shown similar incidences of morbidity and mortality in patients who underwent major hepatectomy with and without preoperative biliary drainage for jaundice, the study populations of those reports were small (range 47–71), and the mortality rate tended to be higher among patients without biliary drainage than among those with drainage [108,109,110,111]. A European multi-institutional study of 366 patients who underwent right or left hepatectomy with EBR revealed that preoperative biliary drainage was associated with decreased mortality in the patients with right hepatectomy, but increased mortality in those with left hepatectomy [112]. Such paradoxical results might be explained by the findings that both preoperative jaundice and cholangitis as well as small FLR were predictors of PHLF [12, 113].
Ribero et al. reported that preoperative cholangitis was associated with an increased risk of hepatic insufficiency after major hepatectomy for hilar cholangiocarcinoma, especially in the patients with FLR < 30% [12]. They also showed that cholangitis occurred more frequently in the patients with biliary drainage than in those without. However, their results showed that preoperative total bilirubin level > 3 mg/dL was an independent predictor of hepatic insufficiency, suggesting the importance of managing jaundice with efforts to minimize biliary drainage–induced complications [12]. Watanabe et al. proposed an age ≥ 69 years old as another risk factor for postoperative severe complications in patients with biliary tract cancer [114]. In this series, the rate of hypertrophy of the FLR after PVE was similar between the older and younger patients, and the incidences of postoperative hyperbilirubinemia and severe complications were higher in the elderly group only in the subgroup of patients with preoperative FLR/TLV ratio < 45%. Therefore, the authors proposed a minimum safe preoperative FLR/TLV ratio of 45% in patients ≥ 69 years old.
Colorectal liver metastases
Over the past few decades, hepatectomy has become a standard strategy for obtaining a long-term survival in patients with CRLM. Advances in chemotherapy, especially the introduction of drugs such as oxaliplatin, bevacizumab, and cetuximab, seem to have contributed to the increased chance of curability and a long-term survival [74].
As preoperative chemotherapy in patients with CRLM has become standard, especially in patients with extensive disease, chemotherapy-induced hepatic injury has been increasingly recognized. The most common manifestations are sinusoidal injury, mainly related to oxaliplatin, and steatosis, mainly related to irinotecan [115, 116]. Chemotherapy-induced hepatic injury has been reported to increase the risk of postoperative morbidity and mortality [117, 118]. Bevacizumab, a monoclonal antibody to vascular endothelial growth factor, has been shown to impair hepatic regeneration in murine models [119, 120], and 6 to 8 weeks, corresponding to approximately two half-lives, is usually recommended as an interval between bevacizumab and hepatectomy [121]. In contrast, the influence of bevacizumab on FLR regeneration after PVE or portal vein ligation in human studies has been controversial [122, 123]. More recent studies have shown no adverse influence of bevacizumab on FLR regeneration after hepatectomy [124] or even enhanced regeneration, especially with bevacizumab in combination with oxaliplatin [125]. In fact, it has been reported that bevacizumab may have the paradoxical effect of suppressing the induction of sinusoidal injury by oxaliplatin [126,127,128], and bevacizumab may inhibit splenomegaly and thrombocytopenia [29]. Importantly, not only specific drugs or regimens, but also their cumulative dose or duration of therapy are related to an increased risk of PHLF [128]. As mentioned in the section on the FLR/TFLV ratio versus the FLR/SLV ratio earlier in this article, hepatic atrophy after extensive chemotherapy may be useful as a simple predictor of PHLF [44].
Other concerns in preoperative planning for major hepatectomy for CRLM include whether or not chemotherapy-induced hepatic injury is reversible and whether or not tumor progression during the waiting period after PVE [129,130,131] affects the long-term outcomes. Takamoto et al. reported that ICG-R15 values recover after at least 2 weeks following the cessation of chemotherapy [132]. Omichi et al. reported that even in patients with hepatic atrophy after chemotherapy, the risk of PHLF was not elevated if sufficient FLR hypertrophy, with a kGR ≥ 2%/week, was achieved following PVE [133]. Regarding tumor progression after PVE, Simoneau et al. showed that the disease-free survival was worse in patients with tumor progression after PVE than in those with stable disease, but the overall survival was similar between the two groups. The authors also showed that tumor progression during chemotherapy was the only predictor of tumor progression after PVE in a multivariate analysis [134]. A recent prospective study and a recent propensity score-matched comparison study suggested that tumor progression after PVE did not affect the patients’ survival [135, 136]. As mentioned above, ALPPS, which is still associated with a high incidence of morbidity, has not been established as a standard strategy, and PVE remains a safer strategy than ALPPS for increasing opportunities for extended hepatectomy in cases of advanced disease.
Conclusions
Advances in accurate methods of preoperatively evaluating the FLR function have improved the safety of major hepatectomy. Various techniques can induce FLR regeneration effectively while taking into account disease-specific comorbidities, such as cirrhosis in patients with HCC, cholangitis in patients with biliary tract cancer, and chemotherapy-induced hepatic injury in patients with CRLM. In this review, we mainly focused on preoperative factors. We should be aware, however, that intraoperative factors, such as an increased amount of blood loss or red blood cell transfusion, were also associated with PHLF [53, 137,138,139]. Control of blood loss was achieved through advances in surgical practices [140,141,142] and efforts to decrease central venous pressure during hepatectomy [143,144,145]. Novel techniques and advances are being evaluated, and these efforts are expected to further reduce the incidence of PHLF.
References
Otsubo T, Kobayashi S, Sano K, Misawa T, Ota T, Katagiri S, et al. Safety-related outcomes of the Japanese Society of Hepato-Biliary-Pancreatic Surgery board certification system for expert surgeons. J Hepatobil Pancreat Sci. 2017;24:252–61.
de Santibanes E, Clavien PA. Playing Play-Doh to prevent postoperative liver failure: the “ALPPS” approach. Ann Surg. 2012;255:415–7.
Schnitzbauer AA, Lang SA, Goessmann H, Nadalin S, Baumgart J, Farkas SA, et al. Right portal vein ligation combined with in situ splitting induces rapid left lateral liver lobe hypertrophy enabling 2-staged extended right hepatic resection in small-for-size settings. Ann Surg. 2012;255:405–14.
Chan A, Zhang WY, Chok K, Dai J, Ji R, Kwan C, et al. ALPPS versus portal vein embolization for hepatitis-related hepatocellular carcinoma: a changing paradigm in modulation of future liver remnant before major hepatectomy. Ann Surg. 2019. https://doi.org/10.1097/SLA.0000000000003433.
Makuuchi M, Takayasu K, Takuma T, Yamazaki S, Hasegawa H, Nishiura S, et al. Preoperative transcatheter embolization of the portal venous branch for patients receiving extended lobectomy due to the bile duct carcinoma. J Jpn Soc Clin Surg. 1984;45:14–20(in Japanese).
Kinoshita H, Sakai K, Hirohashi K, Igawa S, Yamasaki O, Kubo S. Preoperative portal vein embolization for hepatocellular carcinoma. World J Surg. 1986;10:803–8.
Makuuchi M, Thai BL, Takayasu K, Takayama T, Kosuge T, Gunven P, et al. Preoperative portal embolization to increase safety of major hepatectomy for hilar bile duct carcinoma: a preliminary report. Surgery. 1990;107:521–7.
Miyagawa S, Makuuchi M, Kawasaki S, Kakazu T. Criteria for safe hepatic resection. Am J Surg. 1995;169:589–94.
Balzan S, Belghiti J, Farges O, Ogata S, Sauvanet A, Delefosse D, et al. The “50-50 criteria” on postoperative day 5: an accurate predictor of liver failure and death after hepatectomy. Ann Surg. 2005;242:824–8 discussion 8–9.
Mullen JT, Ribero D, Reddy SK, Donadon M, Zorzi D, Gautam S, et al. Hepatic insufficiency and mortality in 1,059 noncirrhotic patients undergoing major hepatectomy. J Am Coll Surg. 2007;204:854–62 discussion 62–64.
Wang HQ, Yang J, Yang JY, Wang WT, Yan LN. Low immediate postoperative platelet count is associated with hepatic insufficiency after hepatectomy. World J Gastroenterol. 2014;20:11871–7.
Ribero D, Zimmitti G, Aloia TA, Shindoh J, Fabio F, Amisano M, et al. Preoperative cholangitis and future liver remnant volume determine the risk of liver failure in patients undergoing resection for hilar cholangiocarcinoma. J Am Coll Surg. 2016;223:87–97.
Rahbari NN, Garden OJ, Padbury R, Brooke-Smith M, Crawford M, Adam R, et al. Posthepatectomy liver failure: a definition and grading by the International Study Group of Liver Surgery (ISGLS). Surgery. 2011;149:713–24.
Kaneko K, Shirai Y, Wakai T, Yokoyama N, Akazawa K, Hatakeyama K. Low preoperative platelet counts predict a high mortality after partial hepatectomy in patients with hepatocellular carcinoma. World J Gastroenterol. 2005;11:5888–92.
Rahbari NN, Reissfelder C, Koch M, Elbers H, Striebel F, Buchler MW, et al. The predictive value of postoperative clinical risk scores for outcome after hepatic resection: a validation analysis in 807 patients. Ann Surg Oncol. 2011;18:3640–9.
Yang T, Zhang J, Lu JH, Yang GS, Wu MC, Yu WF. Risk factors influencing postoperative outcomes of major hepatic resection of hepatocellular carcinoma for patients with underlying liver diseases. World J Surg. 2011;35:2073–82.
Maithel SK, Kneuertz PJ, Kooby DA, Scoggins CR, Weber SM, Martin RC 2nd, et al. Importance of low preoperative platelet count in selecting patients for resection of hepatocellular carcinoma: a multi-institutional analysis. J Am Coll Surg. 2011;212:638–48 discussion 48–50.
Tomimaru Y, Eguchi H, Gotoh K, Kawamoto K, Wada H, Asaoka T, et al. Platelet count is more useful for predicting posthepatectomy liver failure at surgery for hepatocellular carcinoma than indocyanine green clearance test. J Surg Oncol. 2016;113:565–9.
Venkat R, Hannallah JR, Krouse RS, Maegawa FB. Preoperative thrombocytopenia and outcomes of hepatectomy for hepatocellular carcinoma. J Surg Res. 2016;201:498–505.
Golriz M, Ghamarnejad O, Khajeh E, Sabagh M, Mieth M, Hoffmann K, et al. Preoperative Thrombocytopenia May Predict Poor Surgical Outcome after Extended Hepatectomy. Can J Gastroenterol Hepatol. 2018;2018:1275720.
Navarro JG, Yang SJ, Kang I, Choi GH, Han DH, Kim KS, et al. What are the most important predictive factors for clinically relevant posthepatectomy liver failure after right hepatectomy for hepatocellular carcinoma? Ann Surg Treat Res. 2020;98:62–71.
Wai CT, Greenson JK, Fontana RJ, Kalbfleisch JD, Marrero JA, Conjeevaram HS, et al. A simple noninvasive index can predict both significant fibrosis and cirrhosis in patients with chronic hepatitis C. Hepatology. 2003;38:518–26.
Shiha G, Ibrahim A, Helmy A, Sarin SK, Omata M, Kumar A, et al. Asian-Pacific Association for the Study of the Liver (APASL) consensus guidelines on invasive and non-invasive assessment of hepatic fibrosis: a 2016 update. Hepatol Int. 2017;11:1–30.
Ichikawa T, Uenishi T, Takemura S, Oba K, Ogawa M, Kodai S, et al. A simple, noninvasively determined index predicting hepatic failure following liver resection for hepatocellular carcinoma. J Hepatobiliary Pancreat Surg. 2009;16:42–8.
Ratti F, Cipriani F, Catena M, Paganelli M, Aldrighetti L. Liver failure in patients treated with chemotherapy for colorectal liver metastases: Role of chronic disease scores in patients undergoing major liver surgery. A case-matched analysis. Eur J Surg Oncol. 2014;40:1550-6.
Mai RY, Ye JZ, Long ZR, Shi XM, Bai T, Chen J, et al. Preoperative aspartate aminotransferase-to-platelet-ratio index as a predictor of posthepatectomy liver failure for resectable hepatocellular carcinoma. Cancer Manag Res. 2019;11:1401–14.
Ishizawa T, Hasegawa K, Aoki T, Takahashi M, Inoue Y, Sano K, et al. Neither multiple tumors nor portal hypertension are surgical contraindications for hepatocellular carcinoma. Gastroenterology. 2008;134:1908–16.
Soubrane O, Brouquet A, Zalinski S, Terris B, Brezault C, Mallet V, et al. Predicting high grade lesions of sinusoidal obstruction syndrome related to oxaliplatin-based chemotherapy for colorectal liver metastases: correlation with post-hepatectomy outcome. Ann Surg. 2010;251:454–60.
Overman MJ, Ferrarotto R, Raghav K, George B, Qiao W, Machado KK, et al. The Addition of Bevacizumab to Oxaliplatin-Based Chemotherapy: impact Upon Hepatic Sinusoidal Injury and Thrombocytopenia. J Natl Cancer Inst. 2018;110:888–94.
Alkozai EM, Nijsten MW, de Jong KP, de Boer MT, Peeters PM, Slooff MJ, et al. Immediate postoperative low platelet count is associated with delayed liver function recovery after partial liver resection. Ann Surg. 2010;251:300–6.
Takahashi K, Kurokawa T, Oshiro Y, Fukunaga K, Sakashita S, Ohkohchi N. Postoperative decrease in platelet counts is associated with delayed liver function recovery and complications after partial hepatectomy. Tohoku J Exp Med. 2016;239:47–55.
Margonis GA, Amini N, Buettner S, Besharati S, Kim Y, Sobhani F, et al. Impact of early postoperative platelet count on volumetric liver gain and perioperative outcomes after major liver resection. Br J Surg. 2016;103:899–907.
Starlinger P, Haegele S, Offensperger F, Oehlberger L, Pereyra D, Kral JB, et al. The profile of platelet alpha-granule released molecules affects postoperative liver regeneration. Hepatology. 2016;63:1675–88.
Kubota K, Makuuchi M, Kusaka K, Kobayashi T, Miki K, Hasegawa K, et al. Measurement of liver volume and hepatic functional reserve as a guide to decision-making in resectional surgery for hepatic tumors. Hepatology. 1997;26:1176–81.
Monaco AP, Hallgrimsson J, McDermott WV Jr. multiple adenoma (hamartoma) of the liver treated by subtotal (90 percent) resection: morphological and functional studies of regeneration. Ann Surg. 1964;159:513–9.
Starzl TE, Putnam CW, Groth CG, Corman JL, Taubman J. Alopecia, ascites, and incomplete regeneration after 85 to 90 per cent liver resection. Am J Surg. 1975;129:587–90.
Nagasue N, Yukaya H, Ogawa Y, Kohno H, Nakamura T. Human liver regeneration after major hepatic resection. A study of normal liver and livers with chronic hepatitis and cirrhosis. Ann Surg. 1987;206:30–9.
Abdalla EK, Denys A, Chevalier P, Nemr RA, Vauthey JN. Total and segmental liver volume variations: implications for liver surgery. Surgery. 2004;135:404–10.
Shoup M, Gonen M, D’Angelica M, Jarnagin WR, DeMatteo RP, Schwartz LH, et al. Volumetric analysis predicts hepatic dysfunction in patients undergoing major liver resection. J Gastrointest Surg. 2003;7:325–30.
Schindl MJ, Redhead DN, Fearon KC, Garden OJ, Wigmore SJ. The value of residual liver volume as a predictor of hepatic dysfunction and infection after major liver resection. Gut. 2005;54:289–96.
Ferrero A, Vigano L, Polastri R, Muratore A, Eminefendic H, Regge D, et al. Postoperative liver dysfunction and future remnant liver: where is the limit? Results of a prospective study. World J Surg. 2007;31:1643–51.
Kishi Y, Abdalla EK, Chun YS, Zorzi D, Madoff DC, Wallace MJ, et al. Three hundred and one consecutive extended right hepatectomies: evaluation of outcome based on systematic liver volumetry. Ann Surg. 2009;250(4):540–8.
Vauthey JN, Chaoui A, Do KA, Bilimoria MM, Fenstermacher MJ, Charnsangavej C, et al. Standardized measurement of the future liver remnant prior to extended liver resection: methodology and clinical associations. Surgery. 2000;127:512–9.
Yamashita S, Shindoh J, Mizuno T, Chun YS, Conrad C, Aloia TA, et al. Hepatic atrophy following preoperative chemotherapy predicts hepatic insufficiency after resection of colorectal liver metastases. J Hepatol. 2017;67:56–64.
Ribero D, Amisano M, Bertuzzo F, Langella S, Lo Tesoriere R, Ferrero A, et al. Measured versus estimated total liver volume to preoperatively assess the adequacy of the future liver remnant: which method should we use? Ann Surg. 2013;258:801–6.
Kim HJ, Kim CY, Hur YH, Koh YS, Kim JC, Cho CK, et al. Comparison of remnant to total functional liver volume ratio and remnant to standard liver volume ratio as a predictor of postoperative liver function after liver resection. Korean J Hepatobiliary Pancreat Surg. 2013;17:143–51.
Olthof PB, van Dam R, Jovine E, Campos RR, de Santibanes E, Oldhafer K, et al. Accuracy of estimated total liver volume formulas before liver resection. Surgery. 2019;166:247–53.
Kubota K, Aoki T, Kumamaru H, Shiraki T, Miyata H, Seto Y, et al. Use of the National Clinical Database to evaluate the association between preoperative liver function and postoperative complications among patients undergoing hepatectomy. J Hepatobiliary Pancreat Sci. 2019;26:331–40.
Uesaka K, Nimura Y, Nagino M. Changes in hepatic lobar function after right portal vein embolization. An appraisal by biliary indocyanine green excretion. Ann Surg. 1996;223:77–83.
de Graaf W, van Lienden KP, van den Esschert JW, Bennink RJ, van Gulik TM. Increase in future remnant liver function after preoperative portal vein embolization. Br J Surg. 2011;98:825–34.
Nadalin S, Testa G, Malago M, Beste M, Frilling A, Schroeder T, et al. Volumetric and functional recovery of the liver after right hepatectomy for living donation. Liver Transpl. 2004;10:1024–9.
Aoki T, Imamura H, Matsuyama Y, Kishi Y, Kobayashi T, Sugawara Y, et al. Convergence process of volumetric liver regeneration after living-donor hepatectomy. J Gastrointest Surg. 2011;15:1594–601.
Maeda T, Yokoyama Y, Ebata T, Igami T, Mizuno T, Yamaguchi J, et al. Discrepancy between volume and functional recovery in early phase liver regeneration following extended hepatectomy with extrahepatic bile duct resection. Hepatol Res. 2019;49:1227–35.
Shindoh J, Truty MJ, Aloia TA, Curley SA, Zimmitti G, Huang SY, et al. Kinetic growth rate after portal vein embolization predicts posthepatectomy outcomes: toward zero liver-related mortality in patients with colorectal liver metastases and small future liver remnant. J Am Coll Surg. 2013;216:201–9.
Watanabe N, Yamamoto Y, Sugiura T, Okamura Y, Ito T, Ashida R, et al. A predictive scoring system for insufficient liver hypertrophy after preoperative portal vein embolization. Surgery. 2018;163:1014–9.
Shi JH, Hammarstrom C, Grzyb K, Line PD. Experimental evaluation of liver regeneration patterns and liver function following ALPPS. BJS Open. 2017;1:84–96.
Nagino M, Kamiya J, Nishio H, Ebata T, Arai T, Nimura Y. Two hundred forty consecutive portal vein embolizations before extended hepatectomy for biliary cancer: surgical outcome and long-term follow-up. Ann Surg. 2006;243:364–72.
Yokoyama Y, Nishio H, Ebata T, Igami T, Sugawara G, Nagino M. Value of indocyanine green clearance of the future liver remnant in predicting outcome after resection for biliary cancer. Br J Surg. 2010;97:1260–8.
Yokoyama Y, Ebata T, Igami T, Sugawara G, Mizuno T, Yamaguchi J, et al. The predictive value of indocyanine green clearance in future liver remnant for posthepatectomy liver failure following hepatectomy with extrahepatic bile duct resection. World J Surg. 2016;40:1440–7.
Olthof PB, Tomassini F, Huespe PE, Truant S, Pruvot FR, Troisi RI, et al. Hepatobiliary scintigraphy to evaluate liver function in associating liver partition and portal vein ligation for staged hepatectomy: liver volume overestimates liver function. Surgery. 2017;162:775–83.
Ashwell G, Morell AG. The role of surface carbohydrates in the hepatic recognition and transport of circulating glycoproteins. Adv Enzymol Relat Areas Mol Biol. 1974;41:99–128.
Krishnamurthy S, Krishnamurthy GT. Technetium-99 m-iminodiacetic acid organic anions: review of biokinetics and clinical application in hepatology. Hepatology. 1989;9:139–53.
Kwon AH, Matsui Y, Kaibori M, Ha-Kawa SK. Preoperative regional maximal removal rate of technetium-99 m-galactosyl human serum albumin (GSA-Rmax) is useful for judging the safety of hepatic resection. Surgery. 2006;140:379–86.
Iimuro Y, Kashiwagi T, Yamanaka J, Hirano T, Saito S, Sugimoto T, et al. Preoperative estimation of asialoglycoprotein receptor expression in the remnant liver from CT/99 mTc-GSA SPECT fusion images correlates well with postoperative liver function parameters. J Hepatobiliary Pancreat Sci. 2010;17:673–81.
de Graaf W, van Lienden KP, Dinant S, Roelofs JJ, Busch OR, Gouma DJ, et al. Assessment of future remnant liver function using hepatobiliary scintigraphy in patients undergoing major liver resection. J Gastrointest Surg. 2010;14:369–78.
Chapelle T, Beeck B, Driessen A, Roeyen G, Bracke B, Hartman V, et al. Estimation of the future remnant liver function is a better tool to predict post-hepatectomy liver failure than platelet-based liver scores. Eur J Surg Oncol. 2017;43:2277–84.
Sumiyoshi T, Shima Y, Tokorodani R, Okabayashi T, Kozuki A, Hata Y, et al. CT/99 mTc-GSA SPECT fusion images demonstrate functional differences between the liver lobes. World J Gastroenterol. 2013;19:3217–25.
Morine Y, Enkhbold C, Imura S, Ikemoto T, Iwahashi S, Saito YU, et al. Accurate estimation of functional liver volume using Gd-EOB-DTPA MRI compared to MDCT/(99m)Tc-SPECT fusion imaging. Anticancer Res. 2017;37:5693–700.
Kim DK, Choi JI, Choi MH, Park MY, Lee YJ, Rha SE, et al. Prediction of posthepatectomy liver failure: mri with hepatocyte-specific contrast agent versus indocyanine green clearance test. AJR Am J Roentgenol. 2018;211:580–7.
Asenbaum U, Kaczirek K, Ba-Ssalamah A, Ringl H, Schwarz C, Waneck F, et al. Post-hepatectomy liver failure after major hepatic surgery: not only size matters. Eur Radiol. 2018;28:4748–56.
Tomassini F, D’Asseler Y, Linecker M, Giglio MC, Castro-Benitez C, Truant S, et al. Hepatobiliary scintigraphy and kinetic growth rate predict liver failure after ALPPS: a multi-institutional study. HPB (Oxford). 2020. https://doi.org/10.1016/j.hpb.2020.01.010.
Abdalla EK, Barnett CC, Doherty D, Curley SA, Vauthey JN. Extended hepatectomy in patients with hepatobiliary malignancies with and without preoperative portal vein embolization. Arch Surg. 2002;137:675–80 discussion 80–1.
Hemming AW, Reed AI, Howard RJ, Fujita S, Hochwald SN, Caridi JG, et al. Preoperative portal vein embolization for extended hepatectomy. Ann Surg. 2003;237:686–91 discussion 91–93.
Kopetz S, Chang GJ, Overman MJ, Eng C, Sargent DJ, Larson DW, et al. Improved survival in metastatic colorectal cancer is associated with adoption of hepatic resection and improved chemotherapy. J Clin Oncol. 2009;27:3677–83.
Jaeck D, Oussoultzoglou E, Rosso E, Greget M, Weber JC, Bachellier P. A two-stage hepatectomy procedure combined with portal vein embolization to achieve curative resection for initially unresectable multiple and bilobar colorectal liver metastases. Ann Surg. 2004;240:1037–49 discussion 49–51.
Sandstrom P, Rosok BI, Sparrelid E, Larsen PN, Larsson AL, Lindell G, et al. ALPPS improves resectability compared with conventional two-stage hepatectomy in patients with advanced colorectal liver metastasis: results from a scandinavian multicenter randomized controlled trial (LIGRO Trial). Ann Surg. 2018;267:833–40.
Gall TM, Sodergren MH, Frampton AE, Fan R, Spalding DR, Habib NA, et al. Radio-frequency-assisted Liver Partition with Portal vein ligation (RALPP) for liver regeneration. Ann Surg. 2015;261:e45–6.
Jiao LR, Fajardo Puerta AB, Gall TMH, Sodergren MH, Frampton AE, Pencavel T, et al. Rapid induction of liver regeneration for major hepatectomy (REBIRTH): a randomized controlled trial of portal vein embolisation versus ALPPS assisted with radiofrequency. Cancers (Basel). 2019;11:302. https://doi.org/10.3390/cancers11030302
Robles R, Parrilla P, Lopez-Conesa A, Brusadin R, de la Pena J, Fuster M, et al. Tourniquet modification of the associating liver partition and portal ligation for staged hepatectomy procedure. Br J Surg. 2014;101:1129–34 discussion 34.
Petrowsky H, Gyori G, de Oliveira M, Lesurtel M, Clavien PA. Is partial-ALPPS safer than ALPPS? A single-center experience. Ann Surg. 2015;261:e90–2.
Sakamoto Y, Inagaki F, Omichi K, Ohkura N, Hasegawa K, Kokudo N. Associating liver partial partition and transileocecal portal vein embolization for staged hepatectomy. Ann Surg. 2016;264:e21–2.
Schlegel A, Lesurtel M, Melloul E, Limani P, Tschuor C, Graf R, et al. ALPPS: from human to mice highlighting accelerated and novel mechanisms of liver regeneration. Ann Surg. 2014;260:839–46.
Garcia-Perez R, Revilla-Nuin B, Martinez CM, Bernabe-Garcia A, BarojaMazo A. ParrillaParicio P (2015) Associated liver partition and portal vein ligation (ALPPS) vs selective portal vein ligation (PVL) for staged hepatectomy in a rat model Similar Regenerative Response? PLoS ONE. 2015;10:e0144096.
Langiewicz M, Schlegel A, Saponara E, Linecker M, Borger P, Graf R, et al. Hedgehog pathway mediates early acceleration of liver regeneration induced by a novel two-staged hepatectomy in mice. J Hepatol. 2017;66:560–70.
Tanaka K, Matsuo K, Murakami T, Kawaguchi D, Hiroshima Y, Koda K, et al. Associating liver partition and portal vein ligation for staged hepatectomy (ALPPS): short-term outcome, functional changes in the future liver remnant, and tumor growth activity. Eur J Surg Oncol. 2015;41:506–12.
Sparrelid E, Jonas E, Tzortzakakis A, Dahlen U, Murquist G, Brismar T, et al. Dynamic evaluation of liver volume and function in associating liver partition and portal vein ligation for staged hepatectomy. J Gastrointest Surg. 2017;21:967–74.
Madoff DC, Abdalla EK, Gupta S, Wu TT, Morris JS, Denys A, et al. Transhepatic ipsilateral right portal vein embolization extended to segment IV: improving hypertrophy and resection outcomes with spherical particles and coils. J Vasc Interv Radiol. 2005;16:215–25.
Kishi Y, Madoff DC, Abdalla EK, Palavecino M, Ribero D, Chun YS, et al. Is embolization of segment 4 portal veins before extended right hepatectomy justified? Surgery. 2008;144:744–51.
Ito J, Komada T, Suzuki K, Matsushima M, Nakatochi M, Kobayashi Y, et al. Evaluation of segment 4 portal vein embolization added to right portal vein for right hepatic trisectionectomy: A retrospective propensity score-matched study. J Hepatobiliary Pancreat Sci. 2020.
Aoki T, Imamura H, Hasegawa K, Matsukura A, Sano K, Sugawara Y, et al. Sequential preoperative arterial and portal venous embolizations in patients with hepatocellular carcinoma. Arch Surg. 2004;139:766–74.
Ogata S, Belghiti J, Farges O, Varma D, Sibert A, Vilgrain V. Sequential arterial and portal vein embolizations before right hepatectomy in patients with cirrhosis and hepatocellular carcinoma. Br J Surg. 2006;93:1091–8.
Nagino M, Yamada T, Kamiya J, Uesaka K, Arai T, Nimura Y. Left hepatic trisegmentectomy with right hepatic vein resection after right hepatic vein embolization. Surgery. 2003;133:580–2.
Hwang S, Lee SG, Ko GY, Kim BS, Sung KB, Kim MH, et al. Sequential preoperative ipsilateral hepatic vein embolization after portal vein embolization to induce further liver regeneration in patients with hepatobiliary malignancy. Ann Surg. 2009;249:608–16.
Panaro F, Giannone F, Riviere B, Sgarbura O, Cusumano C, Deshayes E, et al. Perioperative impact of liver venous deprivation compared with portal venous embolization in patients undergoing right hepatectomy: preliminary results from the pioneer center. Hepatobiliary Surg Nutr. 2019;8:329–37.
Kobayashi K, Yamaguchi T, Denys A, Perron L, Halkic N, Demartines N, et al. Liver venous deprivation compared to portal vein embolization to induce hypertrophy of the future liver remnant before major hepatectomy: a single center experience. Surgery. 2020;167:917–23.
Ribero D, Abdalla EK, Madoff DC, Donadon M, Loyer EM, Vauthey JN. Portal vein embolization before major hepatectomy and its effects on regeneration, resectability and outcome. Br J Surg. 2007;94:1386–94.
Guiu B, Quenet F, Escal L, Bibeau F, Piron L, Rouanet P, et al. Extended liver venous deprivation before major hepatectomy induces marked and very rapid increase in future liver remnant function. Eur Radiol. 2017;27:3343–52.
Vauthey JN. Liver venous deprivation: optimizing liver regeneration with combined inflow and outflow venous occlusion of the liver. Surgery. 2020;167:924-5.
Erdogan D, Heijnen BH, Bennink RJ, Kok M, Dinant S, Straatsburg IH, et al. Preoperative assessment of liver function: a comparison of 99mTc-Mebrofenin scintigraphy with indocyanine green clearance test. Liver Int. 2004;24:117–23.
Chapelle T, Beeck B, Huyghe I, Francque S, Driessen A, Roeyen G, et al. Future remnant liver function estimated by combining liver volumetry on magnetic resonance imaging with total liver function on (99m)Tc-mebrofenin hepatobiliary scintigraphy: can this tool predict post-hepatectomy liver failure? HPB (Oxford). 2016;18:494–503.
Llovet JM, Bru C, Bruix J. Prognosis of hepatocellular carcinoma: the BCLC staging classification. Semin Liver Dis. 1999;19:329–38.
Bruix J, Castells A, Bosch J, Feu F, Fuster J, Garcia-Pagan JC, et al. Surgical resection of hepatocellular carcinoma in cirrhotic patients: prognostic value of preoperative portal pressure. Gastroenterology. 1996;111:1018–22.
Mai RY, Zeng J, Lu HZ, Liang R, Lin Y, Piao XM, et al. Combining aspartate aminotransferase-to-platelet ratio index with future liver remnant to assess preoperative hepatic functional reserve in patients with hepatocellular carcinoma. J Gastrointest Surg. 2020. https://doi.org/10.1007/s11605-020-04575-w.
Kabir T, Syn NL, Tan ZZX, Tan HJ, Yen C, Koh YX, et al. Predictors of post-operative complications after surgical resection of hepatocellular carcinoma and their prognostic effects on outcome and survival: a propensity-score matched and structural equation modelling study. Eur J Surg Oncol. 2020;46:1756–65.
Yamashita S, Sakamoto Y, Yamamoto S, Takemura N, Omichi K, Shinkawa H, et al. Efficacy of preoperative portal vein embolization among patients with hepatocellular carcinoma, biliary tract cancer, and colorectal liver metastases: a comparative study based on single-center experience of 319 cases. Ann Surg Oncol. 2017;24:1557–68.
Takagi T, Yokoyama Y, Kokuryo T, Yamaguchi J, Nagino M. Liver regeneration following experimental major hepatectomy with choledochojejunostomy. Br J Surg. 2015;102:1410–7.
Takagi T, Yokoyama Y, Kokuryo T, Ebata T, Ando M, Nagino M. A clear difference between the outcomes after a major hepatectomy with and without an extrahepatic bile duct resection. World J Surg. 2017;41:508–15.
Hochwald SN, Burke EC, Jarnagin WR, Fong Y, Blumgart LH. Association of preoperative biliary stenting with increased postoperative infectious complications in proximal cholangiocarcinoma. Arch Surg. 1999;134:261–6.
Cherqui D, Benoist S, Malassagne B, Humeres R, Rodriguez V, Fagniez PL. Major liver resection for carcinoma in jaundiced patients without preoperative biliary drainage. Arch Surg. 2000;135:302–8.
Laurent A, Tayar C, Cherqui D. Cholangiocarcinoma: preoperative biliary drainage (Con). HPB (Oxford). 2008;10:126–9.
Ferrero A, Lo Tesoriere R, Vigano L, Caggiano L, Sgotto E, Capussotti L. Preoperative biliary drainage increases infectious complications after hepatectomy for proximal bile duct tumor obstruction. World J Surg. 2009;33:318–25.
Farges O, Regimbeau JM, Fuks D, Le Treut YP, Cherqui D, Bachellier P, et al. Multicentre European study of preoperative biliary drainage for hilar cholangiocarcinoma. Br J Surg. 2013;100:274–83.
Olthof PB, Wiggers JK, Groot Koerkamp B, Coelen RJ, Allen PJ, Besselink MG, et al. Postoperative liver failure risk score: identifying patients with resectable perihilar cholangiocarcinoma who can benefit from portal vein embolization. J Am Coll Surg. 2017;225:387–94.
Watanabe Y, Kuboki S, Shimizu H, Ohtsuka M, Yoshitomi H, Furukawa K, et al. A new proposal of criteria for the future remnant liver volume in older patients undergoing major hepatectomy for biliary tract cancer. Ann Surg. 2018;267:338–45.
Rubbia-Brandt L, Audard V, Sartoretti P, Roth AD, Brezault C, Le Charpentier M, et al. Severe hepatic sinusoidal obstruction associated with oxaliplatin-based chemotherapy in patients with metastatic colorectal cancer. Ann Oncol. 2004;15:460–6.
Zorzi D, Laurent A, Pawlik TM, Lauwers GY, Vauthey JN, Abdalla EK. Chemotherapy-associated hepatotoxicity and surgery for colorectal liver metastases. Br J Surg. 2007;94:274–86.
Nakano H, Oussoultzoglou E, Rosso E, Casnedi S, Chenard-Neu MP, Dufour P, et al. Sinusoidal injury increases morbidity after major hepatectomy in patients with colorectal liver metastases receiving preoperative chemotherapy. Ann Surg. 2008;247:118–24.
Vauthey JN, Pawlik TM, Ribero D, Wu TT, Zorzi D, Hoff PM, et al. Chemotherapy regimen predicts steatohepatitis and an increase in 90-day mortality after surgery for hepatic colorectal metastases. J Clin Oncol. 2006;24:2065–72.
Taniguchi E, Sakisaka S, Matsuo K, Tanikawa K, Sata M. Expression and role of vascular endothelial growth factor in liver regeneration after partial hepatectomy in rats. J Histochem Cytochem. 2001;49:121–30.
Van Buren G, Yang AD, Dallas NA, Gray MJ, Lim SJ, Xia L, et al. Effect of molecular therapeutics on liver regeneration in a murine model. J Clin Oncol. 2008;26:1836–42.
Ellis LM, Curley SA, Grothey A. Surgical resection after downsizing of colorectal liver metastasis in the era of bevacizumab. J Clin Oncol. 2005;23:4853–5.
Zorzi D, Chun YS, Madoff DC, Abdalla EK, Vauthey JN. Chemotherapy with bevacizumab does not affect liver regeneration after portal vein embolization in the treatment of colorectal liver metastases. Ann Surg Oncol. 2008;15:2765–72.
Aussilhou B, Dokmak S, Faivre S, Paradis V, Vilgrain V, Belghiti J. Preoperative liver hypertrophy induced by portal flow occlusion before major hepatic resection for colorectal metastases can be impaired by bevacizumab. Ann Surg Oncol. 2009;16:1553–9.
Millet G, Truant S, Leteurtre E, Hebbar M, Zerbib P, Huet G, et al. Volumetric analysis of remnant liver regeneration after major hepatectomy in bevacizumab-treated patients: a case-matched study in 82 patients. Ann Surg. 2012;256:755–61.
Margonis GA, Buettner S, Andreatos N, Sasaki K, Pour MZ, Deshwar A, et al. Preoperative bevacizumab and volumetric recovery after resection of colorectal liver metastases. J Surg Oncol. 2017;116:1150–8.
Ribero D, Wang H, Donadon M, Zorzi D, Thomas MB, Eng C, et al. Bevacizumab improves pathologic response and protects against hepatic injury in patients treated with oxaliplatin-based chemotherapy for colorectal liver metastases. Cancer. 2007;110:2761–7.
Klinger M, Eipeldauer S, Hacker S, Herberger B, Tamandl D, Dorfmeister M, et al. Bevacizumab protects against sinusoidal obstruction syndrome and does not increase response rate in neoadjuvant XELOX/FOLFOX therapy of colorectal cancer liver metastases. Eur J Surg Oncol. 2009;35:515–20.
Kishi Y, Zorzi D, Contreras CM, Maru DM, Kopetz S, Ribero D, et al. Extended preoperative chemotherapy does not improve pathologic response and increases postoperative liver insufficiency after hepatic resection for colorectal liver metastases. Ann Surg Oncol. 2010;17:2870–6.
Elias D, De Baere T, Roche A, Mducreux, Leclere J, Lasser P. During liver regeneration following right portal embolization the growth rate of liver metastases is more rapid than that of the liver parenchyma. Br J Surg. 1999;86:784–8.
Kokudo N, Tada K, Seki M, Ohta H, Azekura K, Ueno M, et al. Proliferative activity of intrahepatic colorectal metastases after preoperative hemihepatic portal vein embolization. Hepatology. 2001;34:267–72.
Hoekstra LT, van Lienden KP, Doets A, Busch OR, Gouma DJ, van Gulik TM. Tumor progression after preoperative portal vein embolization. Ann Surg. 2012;256:812–7.
Takamoto T, Hashimoto T, Sano K, Maruyama Y, Inoue K, Ogata S, et al. Recovery of liver function after the cessation of preoperative chemotherapy for colorectal liver metastasis. Ann Surg Oncol. 2010;17:2747–55.
Omichi K, Yamashita S, Cloyd JM, Shindoh J, Mizuno T, Chun YS, et al. Portal vein embolization reduces postoperative hepatic insufficiency associated with postchemotherapy hepatic atrophy. J Gastrointest Surg. 2018;22:60–7.
Simoneau E, Hassanain M, Shaheen M, Aljiffry M, Molla N, Chaudhury P, et al. Portal vein embolization and its effect on tumour progression for colorectal cancer liver metastases. Br J Surg. 2015;102:1240–9.
Huiskens J, Olthof PB, van der Stok EP, Bais T, van Lienden KP, Moelker A, et al. Does portal vein embolization prior to liver resection influence the oncological outcomes—a propensity score matched comparison. Eur J Surg Oncol. 2018;44:108–14.
Collin Y, Pare A, Belblidia A, Letourneau R, Plasse M, Dagenais M, et al. Portal vein embolization does not affect the long-term survival and risk of cancer recurrence among colorectal liver metastases patients: a prospective cohort study. Int J Surg. 2019;61:42–7.
Kennedy TJ, Yopp A, Qin Y, Zhao B, Guo P, Liu F, et al. Role of preoperative biliary drainage of liver remnant prior to extended liver resection for hilar cholangiocarcinoma. HPB (Oxford). 2009;11:445–51.
Ribeiro HS, Costa WL Jr, Diniz AL, Godoy AL, Herman P, Coudry RA, et al. Extended preoperative chemotherapy, extent of liver resection and blood transfusion are predictive factors of liver failure following resection of colorectal liver metastasis. Eur J Surg Oncol. 2013;39:380–5.
Dumitrascu T, Brasoveanu V, Stroescu C, Ionescu M, Popescu I. Major hepatectomies for perihilar cholangiocarcinoma: predictors for clinically relevant postoperative complications using the International Study Group of Liver Surgery definitions. Asian J Surg. 2016;39:81–9.
Lordan JT, Worthington TR, Quiney N, Fawcett WJ, Karanjia ND. Operative mortality, blood loss and the use of Pringle manoeuvres in 526 consecutive liver resections. Ann R Coll Surg Engl. 2009;91:578–82.
Palavecino M, Kishi Y, Chun YS, Brown DL, Gottumukkala VN, Lichtiger B, et al. Two-surgeon technique of parenchymal transection contributes to reduced transfusion rate in patients undergoing major hepatectomy: analysis of 1,557 consecutive liver resections. Surgery. 2010;147:40–8.
Ichida A, Hasegawa K, Takayama T, Kudo H, Sakamoto Y, Yamazaki S, et al. Randomized clinical trial comparing two vessel-sealing devices with crush clamping during liver transection. Br J Surg. 2016;103:1795–803.
Rahbari NN, Koch M, Zimmermann JB, Elbers H, Bruckner T, Contin P, et al. Infrahepatic inferior vena cava clamping for reduction of central venous pressure and blood loss during hepatic resection: a randomized controlled trial. Ann Surg. 2011;253:1102–10.
Zhu P, Lau WY, Chen YF, Zhang BX, Huang ZY, Zhang ZW, et al. Randomized clinical trial comparing infrahepatic inferior vena cava clamping with low central venous pressure in complex liver resections involving the Pringle manoeuvre. Br J Surg. 2012;99:781–8.
Ueno M, Kawai M, Hayami S, Hirono S, Okada KI, Uchiyama K, et al. Partial clamping of the infrahepatic inferior vena cava for blood loss reduction during anatomic liver resection: a prospective, randomized, controlled trial. Surgery. 2017;161:1502–13.
Acknowledgements
The authors thank Stephanie P Deming from the Department of Scientific Publications, MD Anderson Cancer Center, for editing a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Yoji Kishi and other co-authors have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kishi, Y., Vauthey, JN. Issues to be considered to address the future liver remnant prior to major hepatectomy. Surg Today 51, 472–484 (2021). https://doi.org/10.1007/s00595-020-02088-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-020-02088-2