Abstract
Purpose
Splenectomy is the standard therapy for medically refractory immune thrombocytopenia (ITP). Laparoscopic splenectomy (LS) has gained wide acceptance; however, the long-term outcomes of LS versus open splenectomy (OS) for patients with ITP remain unclear.
Methods
We analyzed, retrospectively, 32 patients who underwent splenectomy, as LS in 22 and OS in 10, for refractory ITP at our institute. Data were evaluated based on the American Society of Hematology 2011 evidence-based practice guidelines for ITP.
Results
Although the operation time was significantly longer in the LS group (p < 0.01), LS was associated with less blood loss (p < 0.01), infrequent blood transfusion during surgery (p < 0.01), quicker resumption of oral intake (p < 0.01), and shorter hospital stay (p < 0.01) than OS. Positive responses, including complete and partial remission, were achieved in 90% of the OS group patients and 77% of the LS group patients. The mean follow-up periods were 183 and 92 months, respectively. Relapse-free survival rates, 15 years after the operation were 63% in the OS group and 94% in the LS group.
Conclusions
LS can provide better short-term results and comparable long-term results to those of OS for ITP.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Immune thrombocytopenia (ITP) is an autoimmune disease characterized by a transient or persistent decrease in the platelet count at peripheral levels and defined by a reduced platelet count <100,000/μL for unknown reasons, which may lead to spontaneous bleeding [1, 2]. The mechanism of ITP was initially considered to be increased platelet destruction by autoantibodies. The concepts have now shifted to more complex mechanisms, including both impaired platelet production and T cell-mediated actions [2].
Corticosteroids are the standard first-line treatment for ITP with platelet counts lower than 30,000/μL [2]. Splenectomy is the standard second-line treatment [3,4,5]. Since laparoscopic splenectomy (LS) was first reported in the early 1990s, this technique has been performed successfully at many medical centers [6,7,8]. LS is associated with less blood loss and fewer complications, leading to shorter hospital stays, more rapid convalescence, and lower costs than open splenectomy (OS) [9,10,11,12,13,14,15,16]. Currently, LS is regarded as the “gold standard” treatment [13]. However, while many papers have reported on the short-term outcomes after LS [15,16,17,18], there are few reports on the long-term outcomes over 10 years [19]. Thus, we investigated the short-term and long-term results of LS in our institution.
Methods
The subjects of this retrospective study were 32 patients who underwent splenectomy for ITP at Oita University Faculty of Medicine between July, 1983 and December, 2016. Between July, 1983 and April, 1993, 10 patients underwent OS and between May, 1993 and December, 2016, 22 patients underwent LS. Patient characteristics and postoperative courses were collected from the patient charts. The indications for splenectomy included no response to medical therapy, side effects of corticosteroids, and a hope of pregnancy. Accessory spleens were diagnosed by abdominal ultrasonography and computed tomography preoperatively. Platelet transfusion or gamma globulin were administered preoperatively to patients with a platelet count of <80,000/μL. The study was approved by the Ethics Committee of Oita University Faculty of Medicine.
Surgical procedures
For OS, patients were placed in the supine position and an abdominal midline incision was made. Accessory spleens were carefully checked and removed prior to mobilization of the spleen. After dissection of the colosplenic, gastrosplenic, and splenophrenic ligaments, the splenic vessels at the splenic hilum were divided manually. Staples were not used in OS. A drainage tube, for postoperative bleeding or pancreatic fistula, was routinely placed in the left upper quadrant abdomen.
For LS, patients were placed in the right lateral decubitus position, and the procedure was performed as described previously [20]. After dissection of the ligaments, the splenic pedicle was divided with a laparoscopic stapler (Endo-GIA™, Medtronic, Dublin, Ireland). The spleen was then placed into a large spectrum retrieval bag (Endo-Catch™, Medtronic) and crushed with the fingers and/or forceps before removal.
Response and relapse criteria
Responses to surgery were evaluated a month after the operation based on the American Society of Hematology 2011 evidence-based practice guidelines for ITP [21]. Complete response (CR) was defined as a normal platelet count of >100,000/μL and discontinuation of any medication, with no spontaneous bleeding. Partial response (PR) was defined as a rise in the platelet count between 30,000 and 100,000/μL, and at least a twofold increase from the baseline platelet count, without spontaneous bleeding and medication. Non-response (NR) was defined as a platelet count <30,000/μL or an initial rise, but return to a count of <30,000/μL. The need to continue or restart medical therapy, such as steroids, to sustain a normal platelet count was also considered as NR, as was spontaneous bleeding within 30 postoperative days. Relapse in patients with CR and PR was defined as a platelet count <100,000/μL for CR and <30,000/μL for PR, or a less than twofold increase of the platelet count compared to the baseline for a PR. Any occurrence of spontaneous bleeding or the need for medication was also considered as relapse of the ITP.
Statistical analysis
Numerical data were expressed as mean ± standard deviation. Differences between the variables were compared using the Fisher’s exact test or Mann–Whitney test. Kaplan–Meier curves were created and a log-rank test was performed. All statistical analyses were performed using SPSS 23.0 (SPSS Inc., Chicago, IL, USA). p values less than 0.05 were considered significant.
Results
Patient characteristics and short-term results
The two groups were homogeneous for age, sex, and dose of preoperative corticosteroids (Table 1). The LS group had a significantly lower BMI (p < 0.05), a longer interval between diagnosis and surgery (p < 0.05), and more frequent preoperative gamma globulin administration (p < 0.01) than the OS group. One patient from the LS group received Helicobacter pylori (H. pylori) eradication therapy preoperatively. No patient from either group was treated with Rituximab. The platelet counts were equivalent in the two groups on admission, but there were significant differences in the levels just prior to surgery between the groups due to the gamma globulin administration (p < 0.01).
There was no open conversion in the LS group. Although the operation time was significantly longer in the LS group (p < 0.01), there was less blood loss (p < 0.01), less need for blood transfusion during surgery (p < 0.01), quicker resumption of an oral diet (p < 0.01), and a shorter postoperative hospital stay (p < 0.01) than in the OS group (Table 1). Perioperative complications were recognized in 20% (2/10) of the OS group patients versus 5% (1/22) of the LS group patients. Complications in the OS group included pulmonary embolism in one patient and a pneumothorax in one patient, whereas a pancreatic fistula that did not require percutaneous drainage was observed in one patient in the LS group. Postoperative platelet counts at 1 month were 22 × 104/μL in the OS group and 24 × 104/μL in the LS group, respectively. Positive responses, including CR and PR, were achieved in 90% (9/10) of the OS group patients and in 77% (17/22) of the LS group patients, respectively, without a significant difference between the groups. Focusing on the six NR patients in the two groups, the averages of age and time interval between diagnosis and surgery were 32.8 years and 86 months, respectively. There were no significant differences in age between the NR group and the positive response group (32.8 years versus 34.8 years, p = 0.83). Platelet counts on admission in the NR group were lower than those in the positive response group (3.1 × 104/μL versus 5.5 × 104/μL), but there were no significant differences (p = 0.14).
Long-term results
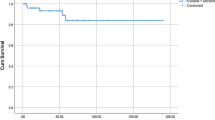
The mean follow-up duration was 183 months for the OS group and 92 months for the LS group. During the follow-up, relapse occurred in three patients from the OS group, 5, 6, and 8 months after surgery, respectively, and in one patient from the LS group, 5 months after surgery. No accessory spleen was found in any of these patients from either group. Kaplan–Meier curves demonstrated 5-, 10-, and 15-year relapse-free survival rates of 63% for all three time points in the OS group, and 94% for all three time points in the LS group (Fig. 1). There were no significant differences between the groups in the relapse-free survival rate, although it was slightly higher in the LS group (p = 0.06).
Discussion
LS has become the standard surgical procedure for the secondary treatment of ITP [6,7,8, 13, 15,16,17,18]. Only a few papers have investigated the long-term outcomes over 10 years after LS. Xu et al. reported that LS achieved good responses for 12 years in China, but they did not compare LS with OS [19]. Previous comparative studies between OS and LS investigated long-term results of less than 10 years [16,17,18]. The present study demonstrated superior short-term and comparable long-term results of over 10 years achieved by LS versus OS. The present study is also the first report of 15-year outcomes after LS.
Previous reports demonstrated that LS is associated with longer operation time but less blood loss than OS [10, 11]. Moreover, because of the small incision and rapid convalescence, LS had fewer major morbidities, less need for postoperative analgesia, and required a shorter postoperative hospital stay than OS [9,10,11,12, 22]. Thus, LS was more cost-effective. Watson et al. demonstrated that the reduction in the postoperative stay after LS led to a 47% cost savings [9] and Friedman et al. reported that hospital costs were $5509 for LS versus $9031 for OS [13].
The positive response rate following splenectomy is reported to be 85–88% [4, 5], but the predictive factors for NR after splenectomy have not been established. Duperier et al. reported that a successful response to splenectomy was significantly related to younger age and a higher preoperative platelet count [23]. The NR patients in the present study tended to have lower platelet counts on admission than the patients with positive responses, but the differences were not significant. Moreover, there were no significant differences in age between the groups. Further investigation is necessary to better understand the factors predictive of a positive response to splenectomy.
The relapse rate after LS is reported to be 28–30% [18, 24]. Decreased platelet production may play a role in recurrence [25, 26], as may a missed accessory spleen [27, 28]. Preoperative computed tomography is useful for the detection of an accessory spleen [29]. Some studies demonstrated that laparoscopy has a good sensitivity and specificity for the detection of an accessory spleen [27, 30]. The combination of an intraoperative laparoscopic search with preoperative computed tomography is thus recommended for the detection of an accessory spleen in LS [31].
In this study, the LS group had significantly more frequent preoperative gamma globulin administrations than the OS group. Although the platelet counts were equivalent in the two groups on admission, they were significantly different just prior to surgery. High-dose gamma globulin therapy for patients with ITP was introduced by Imbach et al. [32]. Intravenous gamma globulin therapy is a safe and fast way to increase platelet counts in patients with acute ITP [33,34,35,36]. Based on clinical trials [37] in the late 1980s, gamma globulin therapy is usually given in Japan. In our institute, gamma globulin therapy before splenectomy for ITP was initiated in 1993. Therefore, few of the early patients who underwent OS received preoperative gamma globulin.
In the past, few drugs were as effective as splenectomy; therefore, splenectomy was generally performed early in patients with medically refractory ITP. H. pylori plays a role in both gastroduodenal disease and ITP [38]. In 2005, a Japanese randomized controlled trial demonstrated that eradication of H. pylori increased platelet counts in patients with ITP [39]. Currently, H. pylori eradication therapy is the first-line of therapy in H. pylori-positive patients with ITP in Japan [40]. Many drugs have been used as second-line or third-line therapy with variable success rates [41,42,43,44,45]. Rituximab—a monoclonal anti-CD20 antibody—demonstrated a 60% response, with 40% reaching a CR [41]. Romiplostim and Eltrombopag both activate the thrombopoietin receptor and increase platelet production [42, 43]. Moreover, the recent treatment algorithm for ITP shows that splenectomy should be performed 1 year after diagnosis [45]. This explains the longer interval between diagnosis and surgery for LS versus OS in this study. In this study, LS resulted in less blood loss, less need for blood transfusions during surgery, quicker resumption of oral intake, and a shorter postoperative hospital stay than OS. The positive response rates were 77 versus 90% in the LS and OS groups, respectively. The 5-, 10-, and 15-year relapse-free survival rates were all 94% in the LS group and 63% in the OS group.
This study was limited by the fact that it was retrospective with a small number of patients. Moreover, the treatment times differed between the OS and LS groups. Larger prospective studies of LS examining long-term outcomes over 10 years are necessary to confirm our results.
In conclusion, LS can provide better short-term results and comparable long-term results to OS for ITP.
References
Rodeghiero F, Stasi R, Gernsheimer T, Michel M, Provan D, Arnold DM, et al. Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: report from an international working group. Blood. 2009;113:2386–93.
Provan D, Stasi R, Newland AC, Blanchette VS, Bolton-Maggs P, Bussel JB, et al. International consensus report on the investigation and management of primary immune thrombocytopenia. Blood. 2010;115:168–86.
Chater C, Terriou L, Duhamel A, Launay D, Chambon JP, Pruvot FR, et al. Reemergence of splenectomy for ITP second-line treatment? Ann Surg. 2016;264:772–7.
Kojouri K, Vesely SK, Terrell DR, George JN. Splenectomy for adult patients with idiopathic thrombocytopenic purpura: a systematic review to assess long-term platelet count responses, prediction of response, and surgical complications. Blood. 2004;104:2623–34.
Vianelli N, Galli M, de Vivo A, Intermesoli T, Giannini B, Mazzucconi MG, et al. Efficacy and safety of splenectomy in immune thrombocytopenic purpura: long-term results of 402 cases. Haematologica. 2005;90:72–7.
Lefor AT, Melvin WS, Bailey RW, Flowers JL. Laparoscopic splenectomy in the management of immune thrombocytopenia purpura. Surgery. 1993;114:613–8.
Gigot JF, Healy ML, Ferrant A, Michaux JL, Njinou B, Kestens PJ. Laparoscopic splenectomy for idiopathic thrombocytopenic purpura. Br J Surg. 1994;81:1171–2.
Hashizume M, Sugimachi K, Kitano S, Shimada M, Baba H, Ueno K, et al. Laparoscopic splenectomy. Am J Surg. 1994;167:611–4.
Watson DI, Coventry BJ, Chin T, Gill PG, Malycha P. Laparoscopic versus open splenectomy for immune thrombocytopenic purpura. Surgery. 1997;121:18–22.
Lozano-Salazar RR, Herrera MF, Vargas-Vorackova F, Lopez-Karpovitch X. Laparoscopic versus open splenectomy for immune thrombocytopenic purpura. Am J Surg. 1998;176:366–9.
Tanoue K, Hashizume M, Morita M, Migoh S, Tsugawa K, Yagi S, et al. Results of laparoscopic splenectomy for immune thrombocytopenic purpura. Am J Surg. 1999;177:222–6.
Winslow ER, Brunt LM. Perioperative outcomes of laparoscopic versus open splenectomy: a meta-analysis with an emphasis on complications. Surgery. 2003;134:647–53 (discussion 54–5).
Friedman RL, Fallas MJ, Carroll BJ, Hiatt JR, Phillips EH. Laparoscopic splenectomy for ITP. The gold standard. Surg Endosc. 1996;10:991–5.
Sampath S, Meneghetti AT, MacFarlane JK, Nguyen NH, Benny WB, Panton ON. An 18-year review of open and laparoscopic splenectomy for idiopathic thrombocytopenic purpura. Am J Surg. 2007;193:580–3 (discussion 3–4).
Wu JM, Lai IR, Yuan RH, Yu SC. Laparoscopic splenectomy for idiopathic thrombocytopenic purpura. Am J Surg. 2004;187:720–3.
Qu Y, Xu J, Jiao C, Cheng Z, Ren S. Long-term outcomes of laparoscopic splenectomy versus open splenectomy for idiopathic thrombocytopenic purpura. Int Surg. 2014;99:286–90.
Vecchio R, Marchese S, Intagliata E, Swehli E, Ferla F, Cacciola E. Long-term results after splenectomy in adult idiopathic thrombocytopenic purpura: comparison between open and laparoscopic procedures. J Laparoendosc Adv Surg Tech A. 2013;23:192–8.
Rijcken E, Mees ST, Bisping G, Krueger K, Bruewer M, Senninger N, et al. Laparoscopic splenectomy for medically refractory immune thrombocytopenia (ITP): a retrospective cohort study on longtime response predicting factors based on consensus criteria. Int J Surg. 2014;12:1428–33.
Xu T, Li N, Jin F, Wu K, Ye Z. Predictive factors of idiopathic thrombocytopenic purpura and long-term survival in Chinese adults undergoing laparoscopic splenectomy. Surg Laparosc Endosc Percutan Tech. 2016;26:397–400.
Ohta M, Nishizaki T, Matsumoto T, Shimabukuro R, Sasaki A, Shibata K, et al. Analysis of risk factors for massive intraoperative bleeding during laparoscopic splenectomy. J Hepatobiliary Pancreat Surg. 2005;12:433–7.
Neunert C, Lim W, Crowther M, Cohen A, Solberg L Jr, Crowther MA. The American Society of Hematology 2011 evidence-based practice guideline for immune thrombocytopenia. Blood. 2011;117:4190–207.
Tsiotos G, Schlinkert RT. Laparoscopic splenectomy for immune thrombocytopenic purpura. Arch Surg. 1997;132:642–6.
Duperier T, Brody F, Felsher J, Walsh RM, Rosen M, Ponsky J. Predictive factors for successful laparoscopic splenectomy in patients with immune thrombocytopenic purpura. Arch Surg. 2004;139:61–6.
Mikhael J, Northridge K, Lindquist K, Kessler C, Deuson R, Danese M. Short-term and long-term failure of laparoscopic splenectomy in adult immune thrombocytopenic purpura patients: a systematic review. Am J Hematol. 2009;84:743–8.
Chang M, Nakagawa PA, Williams SA, Schwartz MR, Imfeld KL, Buzby JS, et al. Immune thrombocytopenic purpura (ITP) plasma and purified ITP monoclonal autoantibodies inhibit megakaryocytopoiesis in vitro. Blood. 2003;102:887–95.
McMillan R, Wang L, Tomer A, Nichol J, Pistillo J. Suppression of in vitro megakaryocyte production by antiplatelet autoantibodies from adult patients with chronic ITP. Blood. 2004;103:1364–9.
Koshenkov VP, Pahuja AK, Nemeth ZH, Abkin A, Carter MS. Identification of accessory spleens during laparoscopic splenectomy is superior to preoperative computed tomography for detection of accessory spleens. JSLS. 2012;16:387–91.
Budzynski A, Bobrzynski A, Sacha T, Skotnicki A. Laparoscopic removal of retroperitoneal accessory spleen in patient with relapsing idiopathic thrombocytopenic purpura 30 years after classical splenectomy. Surg Endosc. 2002;16:1636.
Napoli A, Catalano C, Silecchia G, Fabiano P, Fraioli F, Pediconi F, et al. Laparoscopic splenectomy: multi-detector row CT for preoperative evaluation. Radiology. 2004;232:361–7.
Quah C, Ayiomamitis GD, Shah A, Ammori BJ. Computed tomography to detect accessory spleens before laparoscopic splenectomy: is it necessary? Surg Endosc. 2011;25:261–5.
Habermalz B, Sauerland S, Decker G, Delaitre B, Gigot JF, Leandros E, et al. Laparoscopic splenectomy: the clinical practice guidelines of the European Association for Endoscopic Surgery (EAES). Surg Endosc. 2008;22:821–48.
Imbach P, Barandun S, d’Apuzzo V, Baumgartner C, Hirt A, Morell A, et al. High-dose intravenous gammaglobulin for idiopathic thrombocytopenic purpura in childhood. Lancet. 1981;1:1228–31.
Bussel JB. The use of intravenous gamma-globulin in idiopathic thrombocytopenic purpura. Clin Immunol Immunopathol. 1989;53:S147–55.
Imbach P, Wagner HP, Berchtold W, Gaedicke G, Hirt A, Joller P, et al. Intravenous immunoglobulin versus oral corticosteroids in acute immune thrombocytopenic purpura in childhood. Lancet. 1985;2:464–8.
Fehr J, Hofmann V, Kappeler U. Transient reversal of thrombocytopenia in idiopathic thrombocytopenic purpura by high-dose intravenous gamma globulin. N Engl J Med. 1982;306:1254–8.
Bussel JB, Kimberly RP, Inman RD, Schulman I, Cunningham-Rundles C, Cheung N, et al. Intravenous gammaglobulin treatment of chronic idiopathic thrombocytopenic purpura. Blood. 1983;62:480–6.
Abe T, Matsuda J, Kawasugi K, Yoshimura Y, Kinoshita T, Kazama M. Clinical effect of intravenous immunoglobulin on chronic idiopathic thrombocytopenic purpura. Blut. 1983;47:69–75.
Hamabe A, Omori T, Oyama T, Akamatsu H, Yoshidome K, Tori M, et al. A case of Helicobacter pylori infection complicated with gastric cancer, gastric mucosa-associated lymphoid tissue lymphoma, and idiopathic thrombocytopenic purpura successfully treated with laparoscopy-assisted total gastrectomy and splenectomy. Asian J Endosc Surg. 2011;4:32–5.
Suzuki T, Matsushima M, Masui A, Watanabe K, Takagi A, Ogawa Y, et al. Effect of Helicobacter pylori eradication in patients with chronic idiopathic thrombocytopenic purpura—a randomized controlled trial. Am J Gastroenterol. 2005;100:1265–70.
Fujimura K, Kuwana M, Kurata Y, Imamura M, Harada H, Sakamaki H, et al. Is eradication therapy useful as the first line of treatment in Helicobacter pylori-positive idiopathic thrombocytopenic purpura? Analysis of 207 eradicated chronic ITP cases in Japan. Int J Hematol. 2005;81:162–8.
Arnold DM, Dentali F, Crowther MA, Meyer RM, Cook RJ, Sigouin C, et al. Systematic review: efficacy and safety of rituximab for adults with idiopathic thrombocytopenic purpura. Ann Intern Med. 2007;146:25–33.
Kuter DJ, Bussel JB, Lyons RM, Pullarkat V, Gernsheimer TB, Senecal FM, et al. Efficacy of romiplostim in patients with chronic immune thrombocytopenic purpura: a double-blind randomised controlled trial. Lancet. 2008;371:395–403.
Bussel JB, Cheng G, Saleh MN, Psaila B, Kovaleva L, Meddeb B, et al. Eltrombopag for the treatment of chronic idiopathic thrombocytopenic purpura. N Engl J Med. 2007;357:2237–47.
Palandri F, Polverelli N, Catani L, Sollazzo D, Romano M, Levorato M, et al. The choice of second-line therapy in steroid-resistant immune thrombocytopenia: role of platelet kinetics in a single-centre long-term study. Am J Hematol. 2014;89:1047–50.
Ghanima W, Godeau B, Cines DB, Bussel JB. How I treat immune thrombocytopenia: the choice between splenectomy or a medical therapy as a second-line treatment. Blood. 2012;120:960–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We declare that we have no conflicts of interest and that no sources of funding or material support were provided for this study.
Rights and permissions
About this article
Cite this article
Tada, K., Ohta, M., Saga, K. et al. Long-term outcomes of laparoscopic versus open splenectomy for immune thrombocytopenia. Surg Today 48, 180–185 (2018). https://doi.org/10.1007/s00595-017-1570-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-017-1570-2