Abstract
The formation of lamellar bone in the soft tissues, where bone normally does not exist, is called myositis ossificans. However, it would be more accurate to describe as myositis ossificans the involvement of skeletal muscles and as ectopic or heterotopic ossification the involvement of soft tissues in general. The lesion is subdivided in genetic and non-genetic or acquired types. Myositis or fibrodysplasia ossificans progressiva is a debilitating rare genetic disorder. Clinical suspicion of the disease in the newborn on the basis of malformed great toes may lead to early clinical diagnosis, confirmatory diagnostic genetic testing and avoidance of iatrogenic harmful procedures. Acquired lesions involve the neurogenic myositis ossificans and the non-neurogenic disorder. The latter is defined either as circumscribed myositis ossificans that is post-traumatic or as idiopathic/pseudomalignant myositis ossificans that is non-traumatic and may be a form fruste of fibrodysplasia ossificans progressiva. Ossification in fibrodysplasia ossificans progressiva is irreversible, unlike other forms of heterotopic ossification. In this retrospective study, a total of 22 children with myositis ossificans treated in a 20-year period were identified and classified. Two patients were diagnosed with myositis/fibrodysplasia ossificans progressiva, one with neurogenic myositis ossificans, one with idiopathic/pseudomalignant myositis ossificans and 18 patients with circumscribed myositis ossificans. The clinical features, imaging and histological findings as well as treatment modalities and complications of myositis ossificans in our patients are presented and discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Myositis ossificans is the process of mature lamellar bone formation in soft tissues, in association with inflammation, caused by traumatic or neurological injury, surgery, burns or diseases. Several other terms have also been used to denote the condition such as ectopic or heterotopic ossification, ossifying fibromyopathy, paraosteoarthropathy and periarticular ossification. Although its pathogenesis is unknown, pathways including a permissive environment, inductive agents and osteoprogenitor cells are required. The differentiation of the mesenchymal progenitor cells into chondroblasts and osteoblasts is induced by the bone morphogenetic proteins (BMPs). This suggests that BMPs may be able to influence both the endochondral bone induction pathway, which is the process by which long bones develop, and direct bone formation. The responding cell population to BMP action includes fibroblasts, mesenchymal and muscle-derived connective tissue cells and many more. In ectopic bone formation, which most likely represents metaplasia of fibroblasts at the site of the lesion, the sequence of events recapitulates the process of bone formation that is observed during embryonic long bone development. Initial clinical presentation is generally characterized by tenderness over the palpable swelling, warmth and erythema, pain on range of motion and increased joint stiffness. After maturation of the lesion, the clinical findings include a limited range of motion and a palpable osseous mass. Treatment may be complex because accurate and early diagnosis is fundamental to a successful outcome [1–17].
Twenty-two children that were admitted for myositis ossificans in a 20-year period were included in the study. They were classified, their clinical, imaging and histological findings were reviewed, and outcome was evaluated.
Materials and methods
A total of 22 children that were admitted with a diagnosis of myositis ossificans and ectopic or heterotopic bone formation between 1984 and 2003 in the Departments of Pediatric Orthopaedics and Pediatric Surgery were identified from the hospital database. There were two patients with myositis/fibrodysplasia ossificans progressiva (Figs. 1, 2), one with neurogenic myositis ossificans (Fig. 3), one with idiopathic/pseudomalignant myositis ossificans (Fig. 4) and 18 patients with circumscribed/post-traumatic myositis ossificans. In the post-traumatic lesions, a single major traumatic insult was recorded in 17 patients, while in one patient with an iliopsoas involvement a history of multiple minor injuries to the hip region was detected. The mean age of the patients at diagnosis was 9 years (range 3–14 years). There were 11 boys and 11 girls. Diagnosis was based on the clinical and imaging findings in 18 patients that were treated conservatively and on the histological findings of the resected lesion in 4 cases (Table 1).
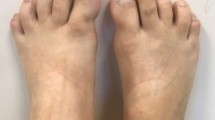
Clinical and radiographic appearance of a 14-month-old boy with severe unilateral hallux valgus deformity and an incomplete preaxial polydactyly. The proximal phalanx is abnormally shaped, and the interphalangeal joint is fixed in valgus. Great toe malformation is the most critical early diagnostic feature of fibrodysplasia ossificans progressiva before the appearance of the preosseous tumor-like swellings
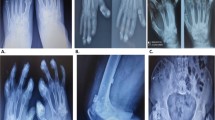
Macroscopic findings of idiopathic myositis ossificans of the chest wall in a 14-year-old boy indicated a well-circumscribed ossified mass adherent to the surrounding muscle. Microscopic examination showed the typical four histologic zones: 1 a central cellular area with extreme variation in the size and shape of cells, 2 an adjacent zone with well-oriented zones of cellular osteoid separated by loose cellular stroma, 3 a more peripheral zone showing new bone formation with osteoblasts and fibrous tissue undergoing trabecular organization and 4 an outmost zone of well-oriented bone encapsulated by fibrous tissue
The patients were followed up for at least 1 year, and the dimensions of the ectopic bone as well as the functional results were assessed. The final follow-up ranged from 1 to 7 years (average 20 months). The clinical, radiological and histological data of these patients were reviewed retrospectively.
Results
Myositis ossificans was localized in the muscle groups of the hip (1 patient), chest wall (1 patient), thigh (4 patients), pelvis (6 patients) and elbow (10 patients). The neurogenic lesion was localized to the hip, while the pseudomalignant to the chest wall. The 4 thigh lesions were localized to the vastus lateralis (2 patients) and to the posteromedial thigh musculature in the genetic disorder (2 patients). No bone injury was diagnosed in the 2 post-traumatic cases with a thigh lesion that followed sport injuries (Fig. 5). The 6 pelvic lesions were localized to the anterior superior iliac spine (1 patient), to the anterior inferior iliac spine (1 patient), to the ischial tuberosity (3 patients) and to the iliopsoas muscle (1 patient). There was no evidence of osseous injury during initial major pelvic trauma on the radiographs of the traumatic lesions localized to the anterior superior and inferior iliac spine (Figs. 6, 7). However, thin flakes of avulsed bone were evident in the 3 lesions involving the ischial tuberosity that followed sport injuries (Fig. 8). The 10 elbow lesions were all localized on the lateral side of the elbow. Eight of the elbow lesions were detected following an injury of the radial head (4 dislocations and 4 fractures of the radial head), while 6 of them were treated surgically and 2 conservatively. Two other elbow lesions presented following a surgically treated supracondylar fracture of the humerus.
Four patients underwent surgical removal of the lesion. The appearance of the lesion was dated 3–4 months (average 3.5 months) prior to surgical removal. Diagnosis was based on the detection of the typical zonal phenomenon at histology. They all showed an uneventful recovery.
Two patients with a post-traumatic lesion localized to the vastus lateralis were treated surgically 4 months after the appearance of the lesion.
The circumscribed myositis ossificans of the iliopsoas muscle in the 13-year-old ballet dancer girl presented with a 3-month history of multiple minor injuries to the region of the left hip. The lesion was surgically removed. She showed an uneventful recovery and was symptom-free with no radiographic evidence of recurrence 4 years postoperatively. This case has been published previously [18].
Idiopathic/pseudomalignant myositis ossificans occurred in the right inferior thoracic wall, anterior to the seventh rib, of a 14-year-old boy. He was referred for a painless lesion that was detected 3 months ago. The lesion was surgically removed.
No invasive procedures were used in 2 genetic, 1 neurogenic and 15 post-traumatic cases. Two patients with myositis ossificans progressiva were diagnosed. The former presented with a unilateral big toe malformation at 14 months of age that was thought to be a congenital lesion, until the age of 5 years that bilateral soft tissue thigh swellings appeared. The latter presented at 8 years of age with the typical form of the disease. No invasive measures were undertaken, and diagnostic biopsies were not suggested. They showed no deterioration of the clinical and radiological findings, but were missed, after 1 year of follow-up.
Neurogenic myositis ossificans was diagnosed in a girl aged 3 years that was struck by an automobile and was admitted to hospital unconscious with a head injury and a right hemiparesis. Pelvic and left femoral fractures were managed with skin traction. Three weeks after the injury, the range of motion of the right hip joint was reduced and movements were painful. Radiographic examination 2 weeks later revealed heterotopic bone formation around the right hip joint. Immobilization was the only treatment offered. One year after injury, the ectopic ossification around the hip had resolved spontaneously, and she was able to walk independently with no restriction. This case has been published previously [19].
Seventeen cases of circumscribed myositis ossificans that were due to a single traumatic injury were classified according to Gilmer and Anderson into three categories on a primarily topographic basis [20]. An extraosseous lesion localized within the substance of a muscle was noted in 2 cases with thigh involvement. A periosteal lesion, which has also been referred in the past as ossifying subperiosteal hematoma or periostoma, was seen in 7 patients (5 with a pelvic and 2 with an elbow involvement). Finally, a parosteal lesion, arising in the immediate proximity of a bone, was diagnosed in 8 lesions following a radial head injury.
No symptomatology was recorded on the final follow-up in the 5 patients with a post-traumatic pelvic lesion that were treated conservatively. Radiographs indicated nonunion of the fracture in all cases with an avulsed ischial tuberosity, extensive healing with exostosis formation in the patient with the fractured anterosuperior spine and extensive new bone formation in the case with the fractured inferior spine. A reduced range of elbow motion was noted in the 10 patients with an elbow lesion. The radiographic follow-up findings in all patients with an elbow lesion showed diminished dimensions and smoother borders in the mature lesions, but complete radiographic regression was not noted in any of the patients. In only one patient, the ectopic bone, localized in the elbow, was complicated by a fracture 1 year after the initial injury (Fig. 9). Regression of the local clinical findings and healing of the fracture of the ectopic bone followed a month of immobilization.
Discussion
Fundamentally, myositis ossificans may be subdivided into genetic and non-genetic or acquired lesions. Myositis or fibrodysplasia ossificans progressiva (also called Munchmeyer’s disease) is an extremely rare and severely disabling genetic disease. It usually appears as a sporadic genetic case, which is not inherited from the parents and arises via a gene mutation, but it may also be due to an autosomal dominant hereditary disorder. The detection of activating mutations in a BMP type 1 receptor ACVR1 (activin type I receptor) in all sporadic and familial cases is a critical milestone in the understanding and treatment of the disease. Children usually appear normal at birth except for malformation of the great toes that is present in all affected individuals. The diagnosis of bilateral hallux valgus on ultrasound may be the first prenatal evidence of the disease. Progressively developing subfascial nodules, commonly located on the posterior neck and back, and heterotopic bone formation of skeletal muscles, tendons, ligaments, fascia and aponeuroses, through an endochondral process, are usually evident within the first 10 years of life. It is the most catastrophic type seen in humans. It leads to progressive immobility, so ‘stone man or woman disease’ is an alternative name of the disease. However, the natural history of the disease is, to a certain extent, different from one patient to another. Atypical forms referring either to the clinical presentation or to the classic defining features of the disease have been described. Prenatal testing is not indicated for general mutation screening; however, it could be considered within a family in which a child had been diagnosed previously. Definitive molecular diagnosis is possible, but correct diagnosis cannot be made if it is not first suspected. Pediatric orthopedic surgeons are often among the first physicians to see a child who has a, usually symmetrical, great toe malformation, a classic, characteristic feature of the disease. Differential diagnosis of the toe malformations includes isolated congenital malformations, brachydactyly, synostosis, symphalangism syndromes and juvenile bunions, while differential diagnosis of the disease includes progressive osseous heteroplasia, osteosarcoma, lymphedema, soft tissue sarcoma, desmoid tumors, aggressive juvenile fibromatosis and acquired heterotopic ossification. There is no efficient treatment of the disease, since biopsies and excision are not recommended because any type of trauma to the deep tissues will provoke further new growths. Intramuscular injections must be avoided, but vaccinations administered by subcutaneous injection and routine venipuncture pose little risk. Preventive oral and dental healthcare measures are essential. Many conservative treatments including retinoids, cyclo-oxygenase-2 inhibitors, leukotriene inhibitors and mast cell stabilizers have been used with no apparent positive results. High-dose glucocorticoids are used in the management of inflammatory flare-ups (painful soft tissue swellings), in addition to analgesics, etidronate and anti-inflammatory drugs. Radiation may be used in adults, but should be avoided in children. Surgery may be indicated only with a focused indication such as to prevent spinal deformity through an arthrodesis. Diagnostic errors occur up to 87% of cases and may cause severe iatrogenic harm, while the mean time to reach diagnosis is 4.1 years. The discovery of the responsible gene and of molecular mechanisms correcting the hyperfunctioning BMPs signaling pathway(s) revealed new long-term approaches to the treatment of the disease and necessitated additional studies of potential medical interventions [21–68].
Diagnosis in the patients with fibrodysplasia ossificans progressiva presented in this report was made after the appearance of the subfascial nodules and ossifications. Prevention of injuries and respiratory infections, avoidance of excessive sunlight exposure, passive physiotherapy and regular follow-up including audiometric evaluation were the only offered approaches.
Neurogenic myositis ossificans has been observed following pathological conditions of the central nervous system, including traumatic, vascular, infectious or neoplastic lesions, associated with paraplegia, Guillain–Barré syndrome, AIDS encephalopathy, poliomyelitis, tetanus, hemiplegia and near-drowning. Its detection following closed head injuries that cause coma was reported in 1968. Since then numerous clinical factors and biochemical markers that are predictive have been identified, although very little is still known about the genetic and bio-molecular element of neurogenic myositis ossificans. The hip is the most commonly affected joint and then the knee, shoulder, elbow and non-joint sites. The features of all presented cases are similar, and extensive new bone is usually associated with fixed contractures. It may result in a variety of complications, including nerve impingement, joint ankylosis, complex regional pain syndrome, osteoporosis and soft tissue infection. The incidence is significantly lower in brain-injured children than in adults and also than spinal cord injury patients. In children, cases with a spontaneous resolution have been described. Salicylates appear to be an effective and safe prophylactic measure in minimizing the occurrence and preventing recurrence of ectopic bone formation following its excision in children. Bisphosphonates, in particular etidronate, and non-steroidal anti-inflammatory drugs have also been used in both the prevention and management of heterotopic ossification. Excision of the ectopic bone allowing correction of the caused deformity together with regaining movement is worthwhile, especially in the presence of returning motor function. The optimal timing for surgery is the neurological recovery, rather than the maturity of the bone [69–103].
The single patient with neurogenic ectopic bone formation included in this review showed spontaneous resolution of the lesion and full recovery within a year.
Non-neurogenic acquired lesions may be post-traumatic or non-traumatic. Traumatic or circumscribed myositis ossificans is also defined as myo-osteosis, extraosseous localized non-neoplastic bone, fibrositis ossificans or hematoma ossificans. It is related to evident and direct trauma in the form of a single severe injury, recurrent episodes of minor trauma and overuse injuries or sustained irritation. In the absence of any trauma or evidence of any systemic disease, the lesion is called non-traumatic or pseudomalignant. It may also be diagnosed following severe burns (the incidence in children is higher than in adults), muscular bleeding due to hemophilia and orthopaedic operations. In young infants with non-accidental trauma, the battered child syndrome should be evaluated. The lesion usually occurs in the large muscle groups of the thigh and upper limb of young men. The brachialis, the quadriceps femoris and the thigh adductor muscles are the most common sites of involvement. The soleus muscle is usually involved in ballerinas. The classical complaint is of a localized painful swelling with limitation of motion of the adjacent joint. Physical examination reveals a firm tender mass in the soft tissues, but calcific densities will become apparent in the radiographs about 4 weeks later. The lesion may be mistaken for a malignant bone or soft tissue tumor or infection, such as osteomyelitis or soft tissue abscess. The site of the lesion on a primary topographic basis may be periosteal, parosteal and extraskeletal. The imaging detection of a lucent zone between the lesion and the underlying bone, the presence of an intact cortex, the location adjacent to the shaft of a bone and the densest calcification in the periphery of the lesion are valuable radiographic findings in the differential diagnosis from bone malignancy. After a period of active growth lasting about 10 weeks, the mass becomes painless and stagnant or regresses spontaneously. This may be apparent on serial radiographs. The zonal phenomenon is an important histological diagnostic criterion of the immature lesion. However, fine-needle biopsy may not procure enough histologic specimens to demonstrate the four zones, and a single specimen taken from the center of the lesion may strongly resemble the findings of osteosarcoma. In addition, the lack of invasion of the adjacent tissues and the inclusion of viable muscle fibers in the lesion are helpful in the histological differential diagnosis from extraskeletal, parosteal or synovial sarcomas. Surgical removal should not be attempted before complete maturation of the lesion. Bone scanning has been attempted as a means of evaluating the activity of the lesion prior to surgery. Surgical removal of the calcified lesion is not necessary in children, unless it is painful or interferes with motion [104–180].
In this report, full clinical recovery with complete radiographic resorption of the ectopic bone was noted in the case with neurogenic myositis ossificans. The four patients (1 with an idiopathic and 3 with a post-traumatic lesion) that were treated operatively were also symptom-free at follow-up. An asymptomatic nonunion complicated 3 cases with a fractured ischial tuberosity and an exostosis complicated a fractured anterosuperior spine. An altered range of joint motion complicated all cases with an elbow ectopic bone formation. None of these cases showed complete clinical regression, since the clinical findings were not improved after the maturation of the lesion. Fracture of the ectopic bone was recorded in only one patient with an elbow lesion.
References
Chalmers J, Gray DH, Rush J (1975) Observations on the induction of bone in soft tissues. J Bone Joint Surg Br 57(1):36–45
Urist MR, Nakagawa M, Nakata N, Nogami H (1978) Experimental myositis ossificans: cartilage and bone formation in muscle in response to a diffusible bone matrix-derived morphogen. Arch Pathol Lab Med 102(6):312–316
Puzas JE, Miller MD, Rosier RN (1989) Pathologic bone formation. Clin Orthop Relat Res 245:269–281
Garland DE (1991) A clinical perspective on common forms of acquired heterotopic ossification. Clin Orthop Relat Res 263:13–29
Hirano H, Urist MR (1991) Induced bone development in transplants of fresh human pseudomalignant heterotopic ossification tissue in athymic nude mice. Clin Orthop Relat Res 263:113–120
Reddi AH, Cunningham NS (1993) Initiation and promotion of bone differentiation by bone morphogenetic proteins. J Bone Miner Res 8(Suppl 2):S499–S502
Kaplan FS, Gannon FH, Hahn GV, Wollner N, Prauner R (1998) Pseudomalignant heterotopic ossification. Clin Orthop Relat Res 346:134–140
Bosch P, Musgrave D, Ghivizzani S, Latterman C, Day CS, Huard J (2000) The efficiency of muscle-derived cell-mediated bone formation. Cell Transpl 9(4):463–470
Sykaras N, Opperman LA (2003) Bone morphogenetic proteins (BMPs): how do they function and what can they offer the clinician? J Oral Sci 45(2):57–73
Kaplan FS, Glaser DL, Hebela N, Shore EM (2004) Heterotopic ossification. J Am Acad Orthop Surg 12(2):116–125
Pape HC, Marsh S, Morley JR, Krettek C, Giannoudis PV (2004) Current concepts in the development of heterotopic ossification. J Bone Joint Surg Br 86(6):783–787
Vanden Bossche L, Vanderstraeten G (2005) Heterotopic ossification: a review. J Rehabil Med 37(3):129–136
McCarthy EF, Sundaram M (2005) Heterotopic ossification: a review. Skelet Radiol 34(10):609–619
Elefteriou F (2005) Neuronal signaling and the regulation of bone remodeling. Cell Mol Life Sci 62(19–20):2339–2349
Isaacson BM, Brown AA, Brunker LB, Higgins TF, Bloebaum RD (2011) Clarifying the structure and bone mineral content of heterotopic ossification. J Surg Res 167(2):e163-e170. doi:10.1016/j.jss.2010.12.047
Mavrogenis AF, Soucacos PN, Papagelopoulos PJ (2011) Heterotopic ossification revisited. Orthopedics 34(3):177. doi:10.3928/01477447-20110124-08
Walczak BE, Johnson CN, Howe BM (2015) Myositis Ossificans. J Am Acad Orthop Surg 23(10):612–622. doi:10.5435/JAAOS-D-14-00269
Petropoulos AS, Sferopoulos NK (1997) Post-traumatic myositis ossificans of the iliopsoas muscle. Apropos of a case with review of the literature. Rev Chir Orthop Reparatrice Appar Mot 83(8):747–751
Sferopoulos NK, Anagnostopoulos D (1997) Ectopic bone formation in a child with a head injury: complete regression after immobilisation. Int Orthop 21(6):412–414
Gilmer WS, Anderson LD (1959) Reactions of soft somatic tissue which may progress to bone formation: circumscribed (traumatic) myositis ossificans. South Med J 52:1432–1448
Rogers JG, Geho WB (1979) Fibrodysplasia ossificans progressiva. A survey of forty-two cases. J Bone Joint Surg Am 61:909–914
Connor JM, Evans DA (1982) Fibrodysplasia ossificans progressiva. The clinical features and natural history of 34 patients. J Bone Joint Surg Br 64(1):76–83
Cohen RB, Hahn GV, Tabas JA, Peeper J, Levitz CL, Sando A, Sando N, Zasloff M, Kaplan FS (1993) The natural history of heterotopic ossification in patients who have fibrodysplasia ossificans progressiva. A study of forty-four patients. J Bone Joint Surg Am 75(2):215–219
Kaplan FS, Tabas JA, Gannon FH, Finkel G, Hahn GV, Zasloff MA (1993) The histopathology of fibrodysplasia ossificans progressiva. An endochondral process. J Bone Joint Surg Am 75(2):220–230
Shah PB, Zasloff MA, Drummond D, Kaplan FS (1994) Spinal deformity in patients who have fibrodysplasia ossificans progressiva. J Bone Joint Surg Am 76(10):1442–1450
Cottalorda J, Jouve JL, Bollini G, Brehier P, Daoud A, Muracciole P, Toth C (1995) Munchmeyer’s disease in children. Rev Chir Orthop Reparatrice Appar Mot 81(1):74–77
Lanchoney TF, Cohen RB, Rocke DM, Zasloff MA, Kaplan FS (1995) Permanent heterotopic ossification at the injection site after diphtheria-tetanus-pertussis immunizations in children who have fibrodysplasia ossificans progressiva. J Pediatr 126(5):762–764
Moriatis JM, Gannon FH, Shore EM, Bilker W, Zasloff MA, Kaplan FS (1997) Limb swelling in patients who have fibrodysplasia ossificans progressiva. Clin Orthop Relat Res 336:247–253
Smith R (1998) Fibrodysplasia (myositis) ossificans progressiva. Clinical lessons from a rare disease. Clin Orthop Relat Res 346:7–14
Delatycki M, Rogers JG (1998) The genetics of fibrodysplasia ossificans progressiva. Clin Orthop Relat Res 346:15–18
Brantus JF, Meunier PJ (1998) Effects of intravenous etidronate and oral corticosteroids in fibrodysplasia ossificans progressiva. Clin Orthop Relat Res 346:117–120
Kitterman JA, Kantanie S, Rocke DM, Kaplan FS (2005) Iatrogenic harm caused by diagnostic errors in fibrodysplasia ossificans progressiva. Pediatrics 116(5):e654-e661
Shore EM, Xu M, Feldman GJ, Fenstermacher DA, Cho TJ, Choi IH, Connor JM, Delai P, Glaser DL, LeMerrer M, Morhart R, Rogers JG, Smith R, Triffitt JT, Urtizberea JA, Zasloff M, Brown MA, Kaplan FS (2006) A recurrent mutation in the BMP type I receptor ACVR1 causes inherited and sporadic fibrodysplasia ossificans progressiva. Nat Genet 38(5):525–527
Benetos IS, Mavrogenis AF, Themistocleous GS, Kanellopoulos AD, Papagelopoulos PJ, Soucacos PN (2006) Optimal treatment of fibrodysplasia ossificans progressiva with surgical excision of heterotopic bone, indomethacin, and irradiation. J Surg Orthop Adv 15(2):99–104
Kaplan FS, Glaser DL, Pignolo RJ, Shore EM (2007) A new era for fibrodysplasia ossificans progressiva: a druggable target for the second skeleton. Expert Opin Biol Ther 7(5):705–712
Kaplan FS, Le Merrer M, Glaser DL, Pignolo RJ, Goldsby RE, Kitterman JA, Groppe J, Shore EM (2008) Fibrodysplasia ossificans progressiva. Best Pract Res Clin Rheumatol 22(1):191–205. doi:10.1016/j.berh.2007.11.007
Deirmengian GK, Hebela NM, O’Connell M, Glaser DL, Shore EM, Kaplan FS (2008) Proximal tibial osteochondromas in patients with fibrodysplasia ossificans progressiva. J Bone Joint Surg Am 90(2):366–374. doi:10.2106/JBJS.G.00774
Rachkidi R, Ghanem I, Dagher F, Kharrat K (2008) Fibrodysplasia ossificans progressiva: orthopedic pitfalls and controversies. Arch Pediatr 15(3):286–290. doi:10.1016/j.arcped.2008.01.007
Kaplan FS, Groppe J, Shore EM (2008) When one skeleton is enough: approaches and strategies for the treatment of fibrodysplasia ossificans progressiva (FOP). Drug Discov Today Ther Strateg 5(4):255–262
Kaplan FS, Xu M, Glaser DL, Collins F, Connor M, Kitterman J, Sillence D, Zackai E, Ravitsky V, Zasloff M, Ganguly A, Shore EM (2008) Early diagnosis of fibrodysplasia ossificans progressiva. Pediatrics 121(5):e1295-e1300. doi:10.1542/peds.2007-1980
Lounev VY, Ramachandran R, Wosczyna MN, Yamamoto M, Maidment AD, Shore EM, Glaser DL, Goldhamer DJ, Kaplan FS (2009) Identification of progenitor cells that contribute to heterotopic skeletogenesis. J Bone Joint Surg Am 91(3):652–663. doi:10.2106/JBJS.H.01177
Nakashima Y, Haga N, Kitoh H, Kamizono J, Tozawa K, Katagiri T, Susami T, Fukushi J, Iwamoto Y (2010) Deformity of the great toe in fibrodysplasia ossificans progressiva. J Orthop Sci 15(6):804–809. doi:10.1007/s00776-010-1542-5
Kartal-Kaess M, Shore EM, Xu M, Schwering L, Uhl M, Korinthenberg R, Niemeyer C, Kaplan FS, Lauten M (2010) Fibrodysplasia ossificans progressiva (FOP): watch the great toes! Eur J Pediatr 169(11):1417–1421. doi:10.1007/s00431-010-1232-5
Piram M, Le Merrer M, Bughin V, De Prost Y, Fraitag S, Bodemer C (2011) Scalp nodules as a presenting sign of fibrodysplasia ossificans progressiva: a register-based study. J Am Acad Dermatol 64(1):97–101. doi:10.1016/j.jaad.2010.04.025
Trigui M, Ayadi K, Zribi M, Triki Z, Keskes H (2011) Fibrodysplasia ossificans progressiva: diagnosis and surgical management. Acta Orthop Belg 77(2):139–144
Pignolo RJ, Shore EM, Kaplan FS (2011) Fibrodysplasia ossificans progressiva: clinical and genetic aspects. Orphanet J Rare Dis 6:80. doi:10.1186/1750-1172-6-80
Dua SG, Shah S, Purandare NC, Arora B, Rangarajan V (2011) Fibrodysplasia ossificans progressiva detected on FDG PET/CT. Clin Nucl Med 36(10):952–954. doi:10.1097/RLU.0b013e31821a28b1
Shore EM (2012) Fibrodysplasia ossificans progressiva: a human genetic disorder of extraskeletal bone formation, or-how does one tissue become another? Wiley Interdiscip Rev Dev Biol 1(1):153–165
Whyte MP, Wenkert D, Demertzis JL, DiCarlo EF, Westenberg E, Mumm S (2012) Fibrodysplasia ossificans progressiva: middle-age onset of heterotopic ossification from a unique missense mutation (c.974G > C, p. G325A) in ACVR1. J Bone Miner Res 27(3):729–737. doi:10.1002/jbmr.1473
Morales-Piga A, Bachiller-Corral J, Trujillo-Tiebas MJ, Villaverde-Hueso A, Gamir-Gamir ML, Alonso-Ferreira V, Vázquez-Díaz M, Posada de la Paz M, Ayuso-García C (2012) Fibrodysplasia ossificans progressiva in Spain: epidemiological, clinical, and genetic aspects. Bone 51(4):748–755. doi:10.1016/j.bone.2012.07.002
Hammond P, Suttie M, Hennekam RC, Allanson J, Shore EM, Kaplan FS (2012) The face signature of fibrodysplasia ossificans progressiva. Am J Med Genet A 158A(6):1368–1380. doi:10.1002/ajmg.a.35346
Zan X, Wang J, You C (2012) The danger of biopsy in fibrodysplasia ossificans progressiva. Arch Dis Child 97(9):785–786. doi:10.1136/archdischild-2012-301696
Kitterman JA, Strober JB, Kan L, Rocke DM, Cali A, Peeper J, Snow J, Delai PL, Morhart R, Pignolo RJ, Shore EM, Kaplan FS (2012) Neurological symptoms in individuals with fibrodysplasia ossificans progressiva. J Neurol 259(12):2636–2643. doi:10.1007/s00415-012-6562-y
Dhamangaonkar AC, Tawari AA, Goregaonkar AB (2013) Fibrodysplasia ossificans progressiva: a case report. J Orthop Surg (Hong Kong) 21(3):383–386
Pignolo RJ, Shore EM, Kaplan FS (2013) Fibrodysplasia ossificans progressiva: diagnosis, management, and therapeutic horizons. Pediatr Endocrinol Rev 10(Suppl 2):437–448
Kaplan FS, Pignolo RJ, Shore EM (2013) From mysteries to medicines: drug development for fibrodysplasia ossificans progressive. Expert Opin Orphan Drugs 1(8):637–649
Giacopelli F, Cappato S, Tonachini L, Mura M, Di Lascio S, Fornasari D, Ravazzolo R, Bocciardi R (2013) Identification and characterization of regulatory elements in the promoter of ACVR1, the gene mutated in Fibrodysplasia Ossificans Progressiva. Orphanet J Rare Dis 8:145. doi:10.1186/1750-1172-8-145
Al-Salmi I, Raniga S, Hadidi AA (2014) Fibrodysplasia ossificans progressiva-radiological findings: a case report. Oman Med J 29(5):368–370. doi:10.5001/omj.2014.97
Hüning I, Gillessen-Kaesbach G (2014) Fibrodysplasia ossificans progressiva: clinical course, genetic mutations and genotype–phenotype correlation. Mol Syndromol 5(5):201–211. doi:10.1159/000365770
Maftei C, Rypens F, Thiffault I, Dubé J, Laberge AM, Lemyre E (2015) Fibrodysplasia ossificans progressiva: bilateral hallux valgus on ultrasound a clue for the first prenatal diagnosis for this condition—clinical report and review of the literature. Prenat Diagn 35(3):305–307. doi:10.1002/pd.4518
Morales-Piga A, Bachiller-Corral J, González-Herranz P, Medrano-SanIldelfonso M, Olmedo-Garzón J, Sánchez-Duffhues G (2015) Osteochondromas in fibrodysplasia ossificans progressiva: a widespread trait with a streaking but overlooked appearance when arising at femoral bone end. Rheumatol Int 35(10):1759–1767. doi:10.1007/s00296-015-3301-6
Bertamino M, Severino M, Schiaffino MC, Garrè ML, Bocciardi R, Ravazzolo R, Rossi A, Di Rocco M (2015) New insights into central nervous system involvement in FOP: case report and review of the literature. Am J Med Genet A 167A(11):2817–2821. doi:10.1002/ajmg.a.37271
Saleh M, Commandeur J, Bocciardi R, Kinabo G, Hamel B (2015) Fibrodysplasia ossificans progressiva with minor unilateral hallux anomaly in a sporadic case from Northern Tanzania with the common ACVR1c.617G > A mutation. Pan Afr Med J 22:299. doi:10.11604/pamj.2015.22.299.8032 (eCollection 2015)
Kaplan FS, Pignolo RJ, Shore EM (2016) Granting immunity to FOP and catching heterotopic ossification in the Act. Semin Cell Dev Biol 49:30–36. doi:10.1016/j.semcdb.2015.12.013
Al Kaissi A, Kenis V, Ben Ghachem M, Hofstaetter J, Grill F, Ganger R, Kircher SG (2016) The diversity of the clinical phenotypes in patients with fibrodysplasia ossificans progressiva. J Clin Med Res 8(3):246–253. doi:10.14740/jocmr2465w
Agarwal S, Loder S, Brownley C, Cholok D, Mangiavini L, Li J, Breuler C, Sung HH, Li S, Ranganathan K, Peterson J, Tompkins R, Herndon D, Xiao W, Jumlongras D, Olsen BR, Davis TA, Mishina Y, Schipani E, Levi B (2016) Inhibition of Hif1α prevents both trauma-induced and genetic heterotopic ossification. Proc Natl Acad Sci USA 113(3):E338–E347. doi:10.1073/pnas.1515397113
Pignolo RJ, Bedford-Gay C, Liljesthröm M, Durbin-Johnson BP, Shore EM, Rocke DM, Kaplan FS (2016) The natural history of flare-ups in fibrodysplasia ossificans progressiva (FOP): a comprehensive global assessment. J Bone Miner Res 31(3):650–656. doi:10.1002/jbmr.2728
Cappato S, Tonachini L, Giacopelli F, Tirone M, Galietta LJ, Sormani M, Giovenzana A, Spinelli AE, Canciani B, Brunelli S, Ravazzolo R, Bocciardi R (2016) High-throughput screening for modulators of ACVR1 transcription: discovery of potential therapeutics for fibrodysplasia ossificans progressiva. Dis Model Mech 9(6):685–696. doi:10.1242/dmm.023929
Lorber J (1953) Ectopic ossification in tuberculous meningitis. Arch Dis Child 28(138):98–103
Roberts PH (1968) Heterotopic ossification complicating paralysis of intracranial origin. J Bone Joint Surg Am 50:70–77
Hoffer MM, Garrett A, Brink J, Perry J, Hale W, Nickel VL (1971) The orthopaedic management of brain-injured children. J Bone Joint Surg Am 53(3):567–577
Larson JM, Michalski JP, Collacott EA, Eltorai D, McCombs CC, Madorsky JB (1981) Increased prevalence of HLA-B27 in patients with ectopic ossification following traumatic spinal cord injury. Rheumatol Rehabil 20(4):193–197
Mital MA, Garber JE, Stinson JT (1987) Ectopic bone formation in children and adolescents with head injuries: its management. J Pediatr Orthop 7(1):83–90
Spencer RF (1987) The effect of head injury on fracture healing. A quantitative assessment. J Bone Joint Surg Br 69(4):525–528
Drane WE, Tipler BM (1987) Heterotopic ossification (myositis ossificans) in acquired immune deficiency syndrome. Detection by gallium scintigraphy. Clin Nucl Med 12(6):433–435
Garland DE, Shimoyama ST, Lugo C, Barras D, Gilgoff I (1989) Spinal cord insults and heterotopic ossification in the pediatric population. Clin Orthop Relat Res 245:303–310
Hurvitz EA, Mandac BR, Davidoff G, Johnson JH, Nelson VS (1992) Risk factors for heterotopic ossification in children and adolescents with severe traumatic brain injury. Arch Phys Med Rehabil 73(5):459–462
Heindl UT, Laub MC (1996) Outcome of persistent vegetative state following hypoxic or traumatic brain injury in children and adolescents. Neuropediatrics 27(2):94–100
Hung JC, Appleton RE, Abernethy L (1997) Myositis ossificans complicating severe Guillain–Barré syndrome. Dev Med Child Neurol 39(11):775–776
Freebourn TM, Barber DB, Able AC (1999) The treatment of immature heterotopic ossification in spinal cord injury with combination surgery, radiation therapy and NSAID. Spinal Cord 37(1):50–53
Kluger G, Kochs A, Holthausen H (2000) Heterotopic ossification in childhood and adolescence. J Child Neurol 15(6):406–413
van Kuijk AA, Geurts AC, van Kuppevelt HJ (2002) Neurogenic heterotopic ossification in spinal cord injury. Spinal Cord 40(7):313–326
Zidek K, Srinivasan R (2003) Rehabilitation of a child with a spinal cord injury. Semin Pediatr Neurol 10(2):140–150
Chauveau C, Devedjian JC, Blary MC, Delecourt C, Hardouin P, Jeanfils J, Broux O (2004) Gene expression in human osteoblastic cells from normal and heterotopic ossification. Exp Mol Pathol 76(1):37–43
Banovac K, Sherman AL, Estores IM, Banovac F (2004) Prevention and treatment of heterotopic ossification after spinal cord injury. J Spinal Cord Med 27(4):376–382
Estrores IM, Harrington A, Banovac K (2004) C-reactive protein and erythrocyte sedimentation rate in patients with heterotopic ossification after spinal cord injury. J Spinal Cord Med 27(5):434–437
Schmidt OI, Heyde CE, Ertel W, Stahel PF (2005) Closed head injury—an inflammatory disease? Brain Res Brain Res Rev 48(2):388–399
Lerner UH (2006) Deletions of genes encoding calcitonin/alpha-CGRP, amylin and calcitonin receptor have given new and unexpected insights into the function of calcitonin receptors and calcitonin receptor-like receptors in bone. J Musculoskelet Neuronal Interact 6(1):87–95
Balboni TA, Gobezie R, Mamon HJ (2006) Heterotopic ossification: pathophysiology, clinical features, and the role of radiotherapy for prophylaxis. Int J Radiat Oncol Biol Phys 65(5):1289–1299
Wang F, Li Y, Zhu X (2006) Relationship between the central nervous system injury and the heterotopic ossification. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 20(8):854–857
da Paz AC, Carod Artal FJ, Kalil RK (2007) The function of proprioceptors in bone organization: a possible explanation for neurogenic heterotopic ossification in patients with neurological damage. Med Hypotheses 68(1):67–73
Lerner UH, Persson E (2008) Osteotropic effects by the neuropeptides calcitonin gene-related peptide, substance P and vasoactive intestinal peptide. J Musculoskelet Neuronal Interact 8(2):154–165
Kadhim HJ, Duchateau J, Sébire G (2008) Cytokines and brain injury: invited review. J Intensive Care Med 23(4):236–249. doi:10.1177/0885066608318458
Naot D, Cornish J (2008) The role of peptides and receptors of the calcitonin family in the regulation of bone metabolism. Bone 43(5):813–818. doi:10.1016/j.bone.2008.07.003
Toffoli AM, Gautschi OP, Frey SP, Filgueira L, Zellweger R (2008) From brain to bone: evidence for the release of osteogenic humoral factors after traumatic brain injury. Brain Inj 22(7–8):511–518. doi:10.1080/02699050802158235
Cipriano CA, Pill SG, Keenan MA (2009) Heterotopic ossification following traumatic brain injury and spinal cord injury. J Am Acad Orthop Surg 17(11):689–697
Suzuki A, Uemura T, Nakamura H (2010) Control of bone remodeling by nervous system. Neural involvement in fracture healing and bone regeneration. Clin Calcium 20(12):1820–1827. doi:CliCa101218201827
Togari A (2010) Control of bone remodeling by nervous system. Regulation of bone metabolism by peripheral nervous system. Clin Calcium 20(12):1831–1838. doi:CliCa101218311838
Juneja M, Jain R, Mishra D, Gautam VK (2011) Myositis ossificans of bilateral hip joints in a patient with diplegic cerebral palsy. J Clin Neurosci 18(4):580–581. doi:10.1016/j.jocn.2010.07.143
Mavrogenis AF, Guerra G, Staals EL, Bianchi G, Ruggieri P (2012) A classification method for neurogenic heterotopic ossification of the hip. J Orthop Traumatol 13(2):69–78. doi:10.1007/s10195-012-0193-z
Sakellariou VI, Grigoriou E, Mavrogenis AF, Soucacos PN, Papagelopoulos PJ (2012) Heterotopic ossification following traumatic brain injury and spinal cord injury: insight into the etiology and pathophysiology. J Musculoskelet Neuronal Interact 12(4):230–240
Chang SY, Yoo WJ, Park MS, Chung CY, Choi IH, Cho TJ (2013) Slipped capital femoral epiphysis caused by neurogenic heterotopic ossification. J Pediatr Orthop B 22(6):553–556. doi:10.1097/BPB.0b013e3283654d50
Mohanty SS, Rao NN, Dash KK, Nashikkar PS (2015) Postencephalitic bilateral heterotopic ossification of the hip in a pediatric patient. J Pediatr Orthop B 24(4):299–303. doi:10.1097/BPB.0000000000000130
Paterson DC (1970) Myositis ossificans circumscripta. Report of four cases without history of injury. J Bone Joint Surg Br 52(2):296–301
Munster AM, Bruck HM, Johns LA, Von Prince K, Kirkman EM, Remig RL (1972) Heterotopic calcification following burns: a prospective study. J Trauma 12(12):1071–1074
Jackson DW, Feagin JA (1973) Quadriceps contusions in young athletes. Relation of severity of injury to treatment and prognosis. J Bone Joint Surg Am 55(1):95–105
Suzuki Y, Hisada K, Takeda M (1974) Demonstration of myositis ossificans by 99 mTc pyrophosphate bone scanning. Radiology 111(3):663–664
Goldman AB (1976) Myositis ossificans circumscripta: a benign lesion with a malignant differential diagnosis. AJR Am J Roentgenol 126(1):32–40
Ogilvie-Harris DJ, Fornasier VL (1980) Pseudomalignant myositis ossificans: heterotopic new-bone formation without a history of trauma. J Bone Joint Surg Am 62(8):1274–1283
Eckardt JJ, Ivins JC, Perry HO, Unni KK (1981) Osteosarcoma arising in heterotopic ossification of dermatomyositis: case report and review of the literature. Cancer 48(5):1256–1261
Ben-Youssef L, Schmidt TL (1983) Battered child syndrome simulating myositis. J Pediatr Orthop 3(3):392–395
Amendola MA, Glazer GM, Agha FP, Francis IR, Weatherbee L, Martel W (1983) Myositis ossificans circumscripta: computed tomographic diagnosis. Radiology 149(3):775–779
Carlson WO, Klassen RA (1984) Myositis ossificans of the upper extremity: a long-term follow-up. J Pediatr Orthop 4(6):693–696
Merkow SJ, St Clair HS, Goldberg MJ (1985) Myositis ossificans masquerading as sepsis. J Pediatr Orthop 5(5):601–604
Pazzaglia UE, Beluffi G, Colombo A, Marchi A, Coci A, Ceciliani L (1986) Myositis ossificans in the newborn. A case report. J Bone Joint Surg Am 68(3):456–458
Peck RJ, Metreweli C (1988) Early myositis ossificans: a new echographic sign. Clin Radiol 39(6):586–588
Booth DW, Westers BM (1989) The management of athletes with myositis ossificans traumatica. Can J Sport Sci 14(1):10–16
Spencer JD, Missen GA (1989) Pseudomalignant heterotopic ossification (“myositis ossificans”). Recurrence after excision with subsequent resorption. J Bone Joint Surg Br 71(2):317–319
Heinrich SD, Zembo MM, MacEwen GD (1989) Pseudomalignant myositis ossificans. Orthopedics 12(4):599–602
Schütte HE, van der Heul RO (1990) Pseudomalignant, nonneoplastic osseous soft-tissue tumors of the hand and foot. Radiology 176(1):149–153
Evans EB (1991) Heterotopic bone formation in thermal burns. Clin Orthop Relat Res 263:94–101
Heifetz SA, Galliani CA, DeRosa GP (1992) Myositis (fasciitis) ossificans in an infant. Pediatr Pathol 12(2):223–229
Koch BM, Wu CM, Randolph J, Eng GD (1992) Heterotopic ossification in children with burns: two case reports. Arch Phys Med Rehabil 73(11):1104–1106
Bronfen C, Touzet P, Peuchmaur M, Prieur AM, Rigault P (1993) Non-traumatic myositis ossificans in a child. Review of the literature apropos of a case mimicking malignant tumor. Rev Chir Orthop Reparatrice Appar Mot 79(3):229–234
Bos CF, Eulderink F, Bloem JL (1993) Bilateral pelvitrochanteric heterotopic ossification in a child. A case report. J Bone Joint Surg Am 75(12):1840–1843
Cushner FD, Morwessel RM (1995) Myositis ossificans in children. Orthopedics 18(3):287–291
Nisolle JF, Delaunois L, Trigaux JP (1996) Myositis ossificans of the chest wall. Eur Respir J 9(1):178–179
Adderson EE, Bohnsack JF (1996) Traumatic myositis ossificans simulating soft tissue infection. Pediatr Infect Dis J 15(6):551–553
Jouve JL, Cottalorda J, Bollini G, Scheiner C, Daoud A (1997) Myositis ossificans: report of seven cases in children. J Pediatr Orthop B 6(1):33–41
Howard CB, Porat S, Bar-On E, Nyska M, Segal D (1998) Traumatic myositis ossificans of the quadriceps in infants. J Pediatr Orthop B 7(1):80–82
Traoré O, Yilboudo J, Cissé R, Compaoré TM, Bandré E, Ouiminga RM (1998) Non-traumatic circumscribed myositis ossificans. Apropos of a bilateral localization. Rev Chir Orthop Reparatrice Appar Mot 84(1):79–83
Akgün I, Erdoğan F, Aydingöz O, Kesmezacar H (1998) Myositis ossificans in early childhood. Arthroscopy 14(5):522–526
Kusuma S, Lourie GM, Lins RE (1999) Myositis ossificans of the hand. J Hand Surg Br 24(1):128–130
Turra CS, Gigante C, Perini L, D’Amore ES (1999) Pseudomalignant myositis ossificans in children. Description of a case and review of the literature. Chir Organi Mov 84(1):93–100
Gindele A, Schwamborn D, Tsironis K, Benz-Bohm G (2000) Myositis ossificans traumatica in young children: report of three cases and review of the literature. Pediatr Radiol 30(7):451–459
Schumacher R (2001) Myositis ossificans traumatica in young children. Pediatr Radiol 31(8):606
Kaleli T, Temiz A, Oztürk H (2003) Pseudomalignant myositis ossificans of the wrist causing compression of the ulnar nerve and artery. A case report. Acta Orthop Belg 69(3):289–291
Messina M, Volterrani L, Molinaro F, Nardi N, Amato G (2006) Myositis ossificans in children: description of a clinical case with a rare localization. Minerva Pediatr 58(1):69–72
Inan M, Chan G, Dabney K, Miller F (2006) Heterotopic ossification following hip osteotomies in cerebral palsy: incidence and risk factors. J Pediatr Orthop 26(4):551–556
Khoury J, Jerushalmi J, Loberant N (2006) Tc-99m MIBI/Tc-99m MDP mismatch: a useful scintigraphic finding in differentiating myositis ossificans from malignant tumor of soft tissue. Clin Nucl Med 31(9):547–549
Chadha M, Agarwal A (2007) Myositis ossificans traumatica of the hand. Can J Surg 50(6):E21–E22
Cabello García D, Rodríguez Fernández A, Gómez Río M, Moreno MJ, Rebollo Aguirre AC, Martín Castro A, Llamas Elvira JM (2008) Circumscript myositis ossificans in a 4-year-old boy. Rev Esp Med Nucl 27(5):358–362
Melamed E, Angel D (2008) Myositis ossificans mimicking compartment syndrome of the forearm. Orthopedics 31(12):pii: orthosupersite.com/view.asp?Rid = 32936
Tsailas PG, Babis GC, Nikolopoulos K, Soucacos PN, Korres DS (2009) The effectiveness of two COX-2 inhibitors in the prophylaxis against heterotopic new bone formation: an experimental study in rabbits. J Surg Res 151(1):108–114. doi:10.1016/j.jss.2007.12.804
Mansoor A, Beals RK (2009) Enchondral ossification of muscles of the calf: a case report on a new form of heterotopic ossification. J Pediatr Orthop B 18(2):86–89. doi:10.1097/BPB.0b013e328321cf21
Kokkosis AA, Balsam D, Lee TK, Schreiber ZJ (2009) Pediatric nontraumatic myositis ossificans of the neck. Pediatr Radiol 39(4):409–412. doi:10.1007/s00247-009-1165-1
Mania VM, Kallivokas AG, Malavaki C, Asimakopoulou AP, Kanakis J, Theocharis AD, Klironomos G, Gatzounis G, Mouzaki A, Panagiotopoulos E, Karamanos NK (2009) A comparative biochemical analysis of glycosaminoglycans and proteoglycans in human orthotopic and heterotopic bone. IUBMB Life 61(4):447–452. doi:10.1002/iub.167
Micheli A, Trapani S, Brizzi I, Campanacci D, Resti M, de Martino M (2009) Myositis ossificans circumscripta: a paediatric case and review of the literature. Eur J Pediatr 168(5):523–529. doi:10.1007/s00431-008-0906-8
Sugiura K, Sugiura M, Kurita K, Okagawa T (2009) Heterotopic and idiopathic ossification in the left foot of a child. Eur J Dermatol 19(6):637–639. doi:10.1684/ejd.2009.0772
Sawyer JR, Kapoor M, Gonzales MH, Warner WC Jr, Canale ST, Beaty JH (2009) Heterotopic ossification of the hip after non-accidental injury in a child: case report. J Pediatr Orthop 29(8):865–867. doi:10.1097/BPO.0b013e3181c1e2ce
Tyler P, Saifuddin A (2010) The imaging of myositis ossificans. Semin Musculoskelet Radiol 14(2):201–216. doi:10.1055/s-0030-1253161
Susnjar T, Biocić M, Pogorelić Z (2010) Traumatic heterotopic ossification of the elbow in children—a case report. Acta Chir Belg 110(2):246–249
Ibarra M, Chou PM, Pachman LM, Zhao YD, Boskey AL (2010) Calcification in a case of circumscribed myositis ossificans. J Rheumatol 37(4):876. doi:10.3899/jrheum.090833
Price CE, Guggenheim M, Rode H (2010) Heterotopic para-articular ossification in a child following flame burns. Eur J Pediatr Surg 20(5):351–353. doi:10.1055/s-0030-1249633
Mitchell EJ, Canter J, Norris P, Jenkins J, Morris J (2010) The genetics of heterotopic ossification: insight into the bone remodeling pathway. J Orthop Trauma 24(9):530–533. doi:10.1097/BOT.0b013e3181ed147b
Leung AH, Rybak LD, Rose DJ, Desai P (2010) Myositis ossificans within the intercondylar notch treated arthroscopically. Skeletal Radiol 39(9):927–930. doi:10.1007/s00256-010-0928-y
Sabatier R, Bouvier C, de Pinieux G, Sarran A, Brenot-Rossi I, Pedeutour F, Chetaille B, Viens P, Weiller PJ, Bertucci F (2010) Low-grade extraskeletal osteosarcoma of the chest wall: case report and review of literature. BMC Cancer 10:645. doi:10.1186/1471-2407-10-645
Findlay I, Lakkireddi PR, Gangone R, Marsh G (2010) A case of myositis ossificans in the upper cervical spine of a young child. Spine (Phila Pa 1976) 35(25):E1525-E8. doi:10.1097/BRS.0b013e3181ec066b
Koob M, Durckel J, Dosch JC, Entz-Werle N, Dietemann JL (2010) Intercostal myositis ossificans misdiagnosed as osteosarcoma in a 10-year-old child. Pediatr Radiol 40(Suppl 1):S34–S37. doi:10.1007/s00247-010-1769-5
Muñoz-Mahamud E, Poggio D, Combalia A (2011) Myositis ossificans mimicking parosteal osteosarcoma: a case report and literature review. Acta Orthop Belg 77(2):274–279
Abate M, Salini V, Rimondi E, Errani C, Alberghini M, Mercuri M, Pelotti P (2011) Post-traumatic myositis ossificans: sonographic findings. J Clin Ultrasound 39(3):135–140. doi:10.1002/jcu.20792
Kalenderer O, Bozoglan M, Agus H (2012) Heterotopic ossification in quadratus femoris muscle in a haemophilic patient. Haemophilia 18(1):e13-e14. doi:10.1111/j.1365-2516.2011.02637.x
Pathak AC, Patil AK, Sheth B, Bansal R (2012) Pseudo-acetabulum due to heterotopic ossification in a child with post-traumatic neglected posterior hip dislocation. J Orthop Case Rep 2(4):14–18
Nelson ER, Wong VW, Krebsbach PH, Wang SC, Levi B (2012) Heterotopic ossification following burn injury: the role of stem cells. J Burn Care Res 33(4):463–470. doi:10.1097/BCR.0b013e31825af547
Naranje S, Kancherla R, Kannan A, Malhotra R, Sharma L, Sankineani SR (2012) Extraarticular bony ankylosis in a child with supracondylar fracture of humerus. Chin J Traumatol 15(5):300–302
De Smet L, Degreef I (2012) Myositis ossificans of the hand in a child: case report. J Pediatr Orthop B 21(6):539–541. doi:10.1097/BPB.0b013e3283524bfa
Harmon J, Rabe AJ, Nichol KK, Shiels WE (2012) Precervical myositis ossificans in an infant secondary to child abuse. Pediatr Radiol 42(7):881–885. doi:10.1007/s00247-011-2270-5
Nauth A, Giles E, Potter BK, Nesti LJ, Oʼbrien FP, Bosse MJ, Anglen JO, Mehta S, Ahn J, Miclau T, Schemitsch EH (2012) Heterotopic ossification in orthopaedic trauma. J Orthop Trauma 26(12):684–688. doi:10.1097/BOT.0b013e3182724624
Grenier G, Leblanc E, Faucheux N, Lauzier D, Kloen P, Hamdy RC (2013) BMP-9 expression in human traumatic heterotopic ossification: a case report. Skelet Muscle 3(1):29. doi:10.1186/2044-5040-3-29
Galeano A, Fraison JB, Viala P, Le Blay P, Jorgensen C, Pers YM (2013) Acute inflammatory myalgia: think of myositis ossificans circumscripta. J Rheumatol 40(9):1614–1615. doi:10.3899/jrheum.130040
Peterson JR, De La Rosa S, Eboda O, Cilwa KE, Agarwal S, Buchman SR, Cederna PS, Xi C, Morris MD, Herndon DN, Xiao W, Tompkins RG, Krebsbach PH, Wang SC, Levi B (2014) Treatment of heterotopic ossification through remote ATP hydrolysis. Sci Transl Med 6(255):255ra132. doi:10.1126/scitranslmed.3008810
Convente MR, Wang H, Pignolo RJ, Kaplan FS, Shore EM (2015) The immunological contribution to heterotopic ossification disorders. Curr Osteoporos Rep 13(2):116–124. doi:10.1007/s11914-015-0258-z
Say F, Coşkun S, Bülbül M, Alici Ö (2015) Myositis ossificans on the forearm in a 10-year-old girl. J Pediatr Orthop B 24(3):223–225. doi:10.1097/BPB.0000000000000152
Yamaga K, Kobayashi E, Kubota D, Setsu N, Tanaka Y, Minami Y, Tanzawa Y, Nakatani F, Kawai A, Chuman H (2015) Pediatric myositis ossificans mimicking osteosarcoma. Pediatr Int 57(5):996–999. doi:10.1111/ped.12672
Adebayo ET, Ayuba GI, Ajike SO, Fomete B (2016) Myositis ossificans of the platysma mimicking a malignancy: a case report with review of the literature. J Korean Assoc Oral Maxillofac Surg 42(1):55–59. doi:10.5125/jkaoms.2016.42.1.55
Kraft CT, Agarwal S, Ranganathan K, Wong VW, Loder S, Li J, Delano MJ, Levi B (2016) Trauma-induced heterotopic bone formation and the role of the immune system: a review. J Trauma Acute Care Surg 80(1):156–165. doi:10.1097/TA.0000000000000883
Sun Y, Cai J, Yu S, Chen S, Li F, Fan C (2016) MiR-630 inhibits endothelial–mesenchymal transition by targeting slug in traumatic heterotopic ossification. Sci Rep 6:22729. doi:10.1038/srep22729
Li PF, Lin ZL, Pang ZH (2016) Non-traumatic myositis ossificans circumscripta at elbow joint in a 9-year old child. Chin J Traumatol 19(2):122–124
Wang SI, Choi SM, Park EH, Kim JR (2016) Post-traumatic intra-articular heterotopic ossification of the shoulder joint in a 15-month-old boy. Skeletal Radiol 45(4):559–565. doi:10.1007/s00256-015-2279-1
Agarwal S, Loder S, Cholok D, Li J, Breuler C, Drake J, Brownley C, Peterson J, Li S, Levi B (2016) Surgical excision of heterotopic ossification leads to re-emergence of mesenchymal stem cell populations responsible for recurrence. Stem Cells Transl Med pii:sctm.2015-0365. [Epub ahead of print]
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author certifies that he has no commercial associations (such as consultancies, stock ownership, equity interest and patent/licensing arrangements) that might pose a conflict of interest in connection with the submitted article. The author received no financial support for this study.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Sferopoulos, N.K., Kotakidou, R. & Petropoulos, A.S. Myositis ossificans in children: a review. Eur J Orthop Surg Traumatol 27, 491–502 (2017). https://doi.org/10.1007/s00590-017-1932-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00590-017-1932-x