Abstract
Purpose
Chronic osteomyelitis and infected nonunion are relatively rare conditions in pediatric patients and are more frequently seen in developing countries. Although relatively rare, they are medically and surgically challenging. Here we report a novel surgical technique used to manage five patients with chronic osteomyelitis of long bones.
Methods
Five skeletally immature patients with chronic osteomyelitis and infected nonunion of the long bones were treated surgically between 2010 and 2014 by a combination of resection of necrotic infected bone, debridement of surrounding soft tissue, and application of antibiotic-laden cement spacer inducing periosteal membrane before final bone reconstruction. Once inflammatory markers normalized, all the patients were re-operated for cement removal, bone graft substitution, and concomitant osteosynthesis of the affected bone, if needed. All patients underwent MRI, CT scan, and laboratory evaluation prior to surgery. The antibiotic regimen was started empirically and then adjusted according to culture and sensitivity results.
Results
Mean patient age at the time of diagnosis was 11 years (range 4–14), and all patients had at least 2-year follow-up (range 2–5). At last follow-up, clinical and laboratory evaluation had normalized, the bone had healed, and all patients had resumed daily living and sports activities.
Conclusion
Surgical debridement is the standard approach to chronic osteomyelitis. Use of antibiotic-laden cement is recommended to penetrate local infection, with antibiotic therapy playing an adjunctive role. The cement also induces membrane formation that aids bone reconstruction.
Level of evidence
IV.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Though relatively rare in the pediatric age group, chronic osteomyelitis (CO) and infected nonunion are challenging medical and surgical conditions. The long duration of treatment necessitates the cooperation of patients and their families [1–6], and a multidisciplinary approach is naturally required to ensure satisfactory outcomes [2, 3, 6]. The gold standard step in the surgical approach to CO is a sharp debridement of necrotic tissue, abundant lavage, sequestrectomy, and removal of any hardware and/or foreign bodies [5–7]. The use of antibiotic-laden cement (ALC) is recommended to control infection locally, with antibiotic therapy playing an adjunctive role. The cement also induces membrane formation that aids bone reconstruction [5, 8–10]. In particular, the induced membrane technique proposed in 1986 by Masquelet has been shown to be effective in treating bone defects regardless of their magnitude or etiology. The technique can be used in case of initial infection and to control established infection before final bone reconstruction [9–11].
The primary aim of this work was to present a relatively novel surgical approach used to manage five consecutive skeletally immature patients with CO and infected nonunion of the long bones. The patients were treated surgically by a combination of resection of necrotic infected bone, debridement of surrounding soft tissue, and application of ALC spacer inducing periosteal membrane before final bone reconstruction. Secondary aims were to evaluate the outcome and efficacy of treatment with the reported surgical technique in children with CO and infected nonunion, and the possible limitations of the procedure.
Materials and methods
Five skeletally immature patients (four males, one female) with CO and infected nonunion of the long bones were retrospectively reviewed after securing institutional review board approval.
The demographic and clinical data recorded were age, gender, etiology of the infection, anatomic location, side involved, details of surgical procedures, causative agent, and length of follow-up (Table 1).
All patients had a clinical examination and laboratory analyses including white blood cells (WBCs), C-reactive protein (CRP), and sedimentation rate (ESR).
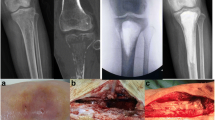
All patients had standard anteroposterior (AP) and lateral radiographs of the affected bone, CT scan, MRI, and a bone scan (BS) to confirm the diagnosis of CO or infected nonunion. In particular, CT scan and MRI were performed to evaluate the extent and anatomy of the lesion (Fig. 1).
a AP and LL X-ray of left femur with infected pseudoarthrosis after osteosynthesis with elastic intramedullary nail; b gadolinium injection MRI showing sequestrum and abscess collection; c postoperative X-ray showing antibiotic-laden cement (ALC); d 1-year follow-up X-ray after removal of ALC, substitution with bone graft, and fixation with femoral nail
Surgical technique and follow-up
All patients were treated surgically with a two-step approach. In the first step, the patient was cleared of any hardware and/or foreign bodies, followed by surgical debridement (resection of necrotic and infected bone and surrounding soft tissue), abundant low- and high-pressure lavage, and application of antibiotic-laden cement (ALC). To prepare the ALC, the monomer catalyst (20 mL) was added to methyl methacrylate powder (40.8 g, gentamicin 1 × 0.5 g) and mixed to a ‘dough’-like consistency. The cement was shaped to fill the bone defect, and once it began to stiffen it was placed in the bone. The polymethyl methacrylate spacer (Palacos® R + Gentamicin, Heraeus, Wehrheim, Germany) was placed in the defect to produce a bioactive membrane, which is considered as biochemically and physically mature at 4–8 weeks after spacer placement [9, 10]. Also, as elution of antibiotic occurs from the surface of the cement, a K-wire was used to create multiple small pits over the surface [1]. As the antibiotic diffuses within the surrounding hematoma, which acts as a transporter, drain application is not always recommended. The drain could also form an inroad for germs to a poorly vascularized area. Unless the case needs a drain, such as when risk of bleeding is suspected, the drain can be opened every 6–8 h for 15 min to allow periodic drainage of the wound and let the eluted antibiotic act locally before being periodically drained out. Drain removal should be considered as soon as possible once the drainage level decreases [1]. For our series, drainage was applied as and when needed, and the limb was immobilized in a posterior splint (long-arm cast or long-leg cast) in order to protect it from pathological fractures.
Antibiotic therapy was initially started with an intravenous large-spectrum antibiotic immediately after sample collection. After isolation and identification of bacteria, the antibiotic was changed based on the antibiogram spectrum (Table 2).
The ALC spacer was kept in place at least until normalization of infection markers, in particular WBCs and CRP, and then removed in a secondary intervention. The bone defect was packed with bone substitutes within the induced membrane (β-Tricalcium phosphate), with concomitant osteosynthesis performed as needed. All patients had regular clinical and radiographic follow-up with standard AP and lateral radiographs of the affected bone (Fig. 2).
a AP and LL X-ray of right femur with chronic osteomyelitis; b frontal and axial T1-weight MRI after hardware removal and position of antibiotic-laden cement (ALC); c AP and LL X-ray showing ALC; d 2-year follow-up AP and LL X-ray showing bone remodeling after removal of ALC and substitution with bone graft
Results
The study included a total of five patients (four males, one female) with CO (four cases) and infected nonunion (one case; patient 3) of the long bones. Mean patient age at time of diagnosis was 11 years (range 4–14). Side involved was the right in three cases and the left in two cases (Table 1). All patients had at least 2-year follow-up (range 2–5).
Causative pathogen was methicillin-sensitive Staphylococcus Aureus in four cases and Cronobacter Sakazakii in one case (Table 2).
All patients were treated as per the reported technique. The four patients with CO were surgically treated within 5–18 months from time of initial surgical procedure (Table 1). The bone defect was filled with β-Tricalcium phosphate bone substitute in all cases. Additional internal fixation with a rigid nail (patient 1) or plate and screws (patient 3) was performed in two patients. No cases of pathological fractures were observed (Table 1).
Clinical and laboratory data, antibiotic treatment, causative agent, and length of follow-up are reported in Tables 1 and 2.
Once the inflammatory markers were normalized, intravenous antibiotics were switched to oral regimen according to bacterial sensitivity (Table 2). Mean duration of intravenous antibiotics was 24 days (range 21–28). Mean duration of oral antibiotics was 3.3 months (range 1.5–6).
At last follow-up visit, all patients had resumed daily living and sports activities, inflammatory markers remained within the normal range, and none of the patients showed signs of growth arrest or disturbances based on radiological and clinical assessment. All patients were pain-free at last follow-up, and range of motion of the affected extremity was comparable to the non-operated side in all patients but one (patient 2). The patient, at the 4-year last follow-up, had resumed daily activities and sports, although his right knee showed a relatively limited flexion of 125° compared to 135° on the contralateral side (Fig. 3).
Discussion
CO and infected nonunion in skeletally immature patients are reported to cause several complications and morbidity [4–7], with documented cases of growth disturbance and limb discrepancy, bone and/or soft tissue loss, and amputations [9, 12–15]. Definitive management of CO and infected nonunion requires aggressive surgical debridement and reconstruction followed by antibiotic therapy [3, 6, 7].
All of our patients with CO and infected nonunion were surgically treated by a combination of resection of necrotic infected bone, debridement of surrounding soft tissue, and application of ALC spacer followed by placement of β-tricalcium phosphate bone substitute into the induced membrane (Tables 1, 2).
The technique pioneered by Masquelet involves initial wide debridement and the placement of a spacer to temporarily fill the bony defect [10]. This step is followed—about 4–8 weeks later—by removal of the spacer and placement of bone graft into the induced membrane [9–11, 16]. The membrane is primarily composed of type I collagen, fibroblasts, and blood vessels (inner surface), and it secretes bone morphogenetic protein 2, vascular endothelial growth factor, core-binding factor α1, interleukin-6, collagenase-1, and other growth factors to stimulate bone defect reconstruction. The technique has been shown to promote consolidation of cancellous bone graft within an induced membrane in segmental bone defects of up to 20–25 cm in length [9–11, 16–22].
Few studies have assessed the outcome of adult patients with diaphyseal septic bone loss managed with the induced membrane technique.
Woon et al. (2 patients) [17], Scholz et al. (13 patients) [18], and Wang et al. (32 patients) [19] reported satisfactory clinical and radiological outcome. The mean radiographic time to bone union was between 4 and 6 months, regardless of the length of the bone defect. All authors concluded the Masquelet technique can be used successfully to manage diaphyseal septic bone loss in adults. However, they all stress out the technique must be combined with stable fixation, as instability may lead to deformity or nonunion [17–19].
More recently, Xie et al. [20] reported the early effects of induced membrane technique for the reconstruction of large CO defects in limbs of 23 adult patients. Authors concluded the combination of through debridement with induced membrane technique provides satisfactory clinical outcome and, most importantly, shortens the length of treatment and lowers the rate of complications [20].
On the other hand, Marias and Ferreira (7 patients) [21] and Spigel et al. (25 patients) [22] have managed large septic (CO) bone defects with bone transport through an induced membrane. However, despite the high consolidation rate (97 % overall), the number of complications was high. The authors pointed out that the high morbidity may be related to the complexity of the surgical procedure and the severity of cases [21, 22].
The induced membrane technique has shown to be a viable option for the treatment of segmental bony defects but has not been specifically studied in the treatment of CO and infected nonunion in skeletally immature patients [5, 8–11, 16–22].
In our group of skeletally immature patients, the induced periosteal membrane was already formed when the ALC spacer was removed (at an average 4 weeks from initial debridement) and inflammatory markers had normalized. This finding suggests children may be able to generate a periosteal membrane faster than adults. The membrane appeared to prevent graft resorption and promote revascularization and consolidation of new bone. Moreover, the thickened periosteum, which is typical of skeletally immature patients, played an important role in contributing toward β-tricalcium phosphate bone substitute integration and bone healing [23].
In our series, ALC was prepared during surgery. The cement was shaped to fill the bone defect, and once it began to stiffen, it was placed in the bone. In order to increase local availability, small pits were made with a K-wire to increase surface area [1]. Moreover, the medullary canal was always reamed.
The ALC spacer induced periosteal membrane formation and provided local antibiotic treatment by delivering a high concentration of antibiotic to the infected zone with less systemic toxicity, as described by Bucholz et al. [24, 25]. As the antibiotic diffuses in the surrounding hematoma, which acts as a transporter, drain application is not recommended [1]. However, although our patients were young and healthy, with normal renal function, caution is warranted in elderly patients or in cases of renal failure [12, 26–28].
Local antibiotic delivery can be successfully obtained with ALC beads, as reported by Whalig and Bucholz [29], Cho et al. [30], and Walenkamp et al. [31]. However, beads offer no mechanical support to the bone and only suboptimal membrane formation and can be difficult to remove as they are enveloped within the surrounding fibrous tissue. The other disadvantage is the challenge of preparing small-enough oval-ended beads for feeding on steel wire to facilitate removal, in the short time window before the cement hardens [13, 23, 24, 30–33].
Our relatively novel approach is shown to be safe, with eradication of the infection, consolidation of bone, and good functional outcome in skeletally immature patients with CO and infected nonunion of the long bones.
Paley et al. [8] and Bar-On et al. [5] reported two series of skeletally immature patients with CO treated surgically by debridement and lavage, reaming of the intramedullary canal, and insertion of gentamicin-impregnated ALC rods into the canal. The primary objectives of using the intramedullary ALC rods were to provide mechanical support to the bone and local antibiotic delivery. The technique was successful in eradicating the infection, although the authors reported two cases of pathological fracture (one case per series) [5, 8].
More recently, Mansour and Ghanem [11] reported preliminary results on the two-step induced membrane technique in eight children. Outcome was good in all but two patients, and the results showed the feasibility of the technique in children. However, it is difficult to compare their technique here as their patients carried heterogeneous diagnoses: congenital pseudoarthrosis (four cases), tumor (three cases) and CO (one case). Mansour and Ghanem identified chemotherapy, suboptimal fixation, and axial malalignment as risk factors for failure. Compared to Mansour and Ghanem’s work, our group of patients was more homogeneous in terms of diagnosis. None of our patients had chemotherapy, the extremity was protected with a plaster cast when long bones were involved, and no pathological fractures occurred in our case series [11].
Another surgical option is to enrich the infection area by augmenting blood supply using a muscle flap. Bassey et al. [34] and Noang et al. [35] have evaluated the efficacy of muscle flaps in the treatment of CO in children and adolescents (Table 3). Although successful, the technique is not free of complications and reported disadvantages include donor site morbidity, adjacent joint stiffness, and/or long scars [33–35]. Moreover, Bassey et al. [34] reported a relatively high incidence of limb shortening and pathological fractures.
There are limitations to the results analyzed here. This was a retrospective case series study reporting preliminary results on a limited number of patients. Even so, the number of patients included in the study was similar to the average number of cases in other published studies that have investigated outcome of CO and infected nonunion managed with ALC [5, 8]. The patients also formed a heterogeneous group in terms of age, type of fracture, and type of chronic infection. Nevertheless, the results are encouraging and our preliminary findings show the feasibility of using ACL in the treatment of CO and infected nonunion in skeletally immature patients. Further work is now needed to confirm our results.
Conclusion
CO is a relatively uncommon disease in the pediatric population. Once discovered, it should be treated promptly. Debridement, sequestrectomy, then use of ALC spacer inducing periosteal membrane formation followed by placement of β-tricalcium phosphate bone substitute into the induced membrane give satisfactory outcomes in skeletally immature children presenting CO and infected nonunion of the long bones.
References
Samuel S, Ismavel R, Boopalan PR, Matthai T (2010) Practical considerations in the making and use of high-dose antibiotic-loaded bone cement. Acta Orthop Belg 76:543–545
Harik NS, Smeltzer MS (2010) Management of acute hematogenous osteomyelitis in children. Expert Rev Anti Infect Ther 8:175–181
Gutierrez K (2005) Bone and joint infections in children. Pediatr Clin N Am 52:779–794
Catalano-Pons C, Raymond J, Chalumeau M, Armengaud JB, Kalifa G, Gendrel D (2007) Case 2: paediatric chronic osteomyelitis: report of two cases. Case 1 diagnosis: pulmonary TB complicated by pneumomediastinum. Case 2 diagnosis: osteomyelitis caused by actinomyces. Acta Paediatr 96:1849–1852
Bar-On E, Weigl DM, Bor N, Becker T, Katz K, Mercado E, Livni G (2010) Chronic osteomyelitis in children: treatment by intramedullary reaming and antibiotic impregnated cement rods. J Pediatr Orthop 30:508–513
Matzkin EG, Dabbs DN, Fillman RR, Kyono WT, Yandow SM (2005) Chronic osteomyelitis in children: Shriners Hospital Honolulu experience. J Pediatr Orthop B 14:362–366
Auh JS, Binns HJ, Katz BZ (2004) Retrospective assessment of subacute or chronic osteomyelitis in children and young adults. Clin Pediatr 3:549–555
Paley D, Herzenberg JE (2002) Intramedullary infections treated with antibiotic cement rods: preliminary results in nine cases. J Orthop Trauma 16:723–729
Wang X, Wei F, Luo F, Huang K, Xie Z (2015) Induction of granulation tissue for the secretion of growth factors and the promotion of bone defect repair. J Orthop Surg Res 17(10):147
Masquelet AC, Fitoussi F, Bégué T, Muller GP (2000) Reconstruction of the long bones by the induced membrane and spongy autograft. Ann Chir Plast Esthet 45:346–353
Mansour TM, Ghanem IB (2015) Preliminary results of the induced membrane technique for the reconstruction of large bone defects. J Pediatr Orthop [Epub ahead of print]
Hollmig ST, Copley LA, Browne RH, Grande LM, Wilson PL (2007) Deep venous thrombosis associated with osteomyelitis in children. J Bone Joint Surg Am 89:1517–1523
Zalavras CG, Patzakis MJ, Holtom P (2004) Local antibiotic therapy in the treatment of open fractures and osteomyelitis. Clin Orthop Relat Res 427:86–93
Nourse C, Starr M, Munckhof W (2007) Community-acquired methicillin-resistant Staphylococcus aureus causes severe disseminated infection and deep venous thrombosis in children: literature review and recommendations for management. J Paediatr Child Health 43:656–661
Bouchoucha S, Benghachame F, Trifa M, Saied W, Douira W, Nessib MN et al (2010) Deep venous thrombosis associated with acute hematogenous osteomyelitis in children. Orthop Traumatol Surg Res 96:890–893
Karger C, Kishi T, Schneider L, Fitoussi F, Masquelet AC (2012) Treatment of posttraumatic bone defects by the induced membrane technique. Orthop Traumatol Surg Res 98:97–102
Woon CY, Chong KW, Wong MK (2010) Induced membrane A staged technique of bone-grafting for segmental bone loss. J Bone Joint Surg Am 92:196–201
Scholz AO, Gehrmann S, Glombitza M, Kaufman RA, Bostelmann R, Flohe S et al (2015) Reconstruction of septic diaphyseal bone defects with the induced membrane technique. Injury 46(Suppl. 4):S121–S124
Wang X, Luo F, Huang K, Xie Z (2016) Induced membrane technique for the treatment of bone defects due to post-traumatic osteomyelitis. Bone Joint Res 5:101–105
Xie B, Tian J, Jing YF, Zhou DP, Xiang LB (2015) Early effect of induced membrane technique for the reconstruction of chronic osteomyelitis defects in limbs of adult patients. Zhongguo Gu Shang 28:43–47
Marais LC, Ferreira N (2015) Bone transport through an induced membrane in the management of tibial bone defects resulting from chronic osteomyelitis. Strat Traum Limb Recon 10:27–33
Spiegl U, Pätzold R, Friederichs J, Hungerer S, Militz M, Bühren V (2013) Clinical course, complication rate and outcome of segmental resection and distraction osteogenesis after chronic tibial osteitis. Injury 44:1049–1056
Lindaman LM (2001) Bone healing in children. Clin Podiatr Med Surg 18:97–108
Buchholz HW, Engelbrecht H (1970) Depot effects of various antibiotics mixed with Palacos resins. Chirurg 41:511–515
Buchholz HW, Gartmann HD (1972) Infection prevention and surgical management of deep insidious infection in total endoprosthesis. Chirurg 43:446–453
Dovas S, Liakopoulos V, Papatheodorou L, Chronopoulou I, Papavasiliou V, Atmazidis E et al (2008) Acute renal failure after antibiotic-impregnated bone cement treatment of an infected total knee arthroplasty. Clin Nephrol 69:207–212
Van Raaij TM, Visser LE, Vulto AG, Verhaar JA (2002) Acute local failure after local gentamicin treatment of an infected total knee arthroplasty. J Arthroplasty 17:948–950
Curtis JM, Sternhagen V, Batts D (2005) Acute renal failure after placement of tobramycin-impregnated bone cement in an infected total knee arthroplasty. Pharmacotherapy 25:876–880
Wahlig H, Buchholz HW (1972) Experimental and clinical studies on the release of gentamicin from bone cement. Chirurg 43:441–445
Cho SH, Song HR, Koo KH et al (1997) Antibiotic-impregnated cement beads in the treatment of chronic osteomyelitis. Bull Hosp Jt Dis 56:140–144
Walenkamp GH, Kleijn LL, de Leeuw M (1998) Osteomyelitis treated with gentamicin-PMMA beads: 100 patients followed for 1-12 years. Acta Orthop Scand 69:518–522
Springer BD, Lee GC, Osmon D et al (2004) Systemic safety of high-dose antibiotic-loaded cement spacers after resection of an infected total knee arthroplasty. Clin Orthop Relat Res 427:47–51
Cierny G III, Mader JT, Penninck JJ (2003) A clinical staging system for adult osteomyelitis. Clin Orthop Relat Res 414:7–24
Bassey LO, Antia-Obong OE, Antia UE (1995) Sequestrectomy and local muscle flap implantation for chronic osteomyelitis in children and adolescents. East Afr Med J 72:787–790
Hoang NT, Staudenmeier R, Feucht A et al (2009) Effectiveness of free gracilis muscle flaps in the treatment of chronic osteomyelitis with purulent fistulas at the distal third of the tibia in children. J Pediatr Orthop 29:305–311
Acknowledgments
Institutional review board approval: The study was approved by the Clermont-Ferrand University Hospital Review Board.
Funding
None of the authors received financial support for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest in this research.
Ethical approval
All procedures and studies involving human participants were performed in accordance with the ethical standards of the national and/or institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
No patients were involved. This is a retrospective study of patient’s data for which IRB approval was obtained.
Rights and permissions
About this article
Cite this article
Canavese, F., Corradin, M., Khan, A. et al. Successful treatment of chronic osteomyelitis in children with debridement, antibiotic-laden cement spacer and bone graft substitute. Eur J Orthop Surg Traumatol 27, 221–228 (2017). https://doi.org/10.1007/s00590-016-1859-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00590-016-1859-7