Abstract
Purpose
The goal of this research is to explore the incidence and risk factors of symptomatic spinal epidural hematoma (SSEH) following cervical spine surgery.
Methods
Patients with SSEH from January 2009 to February 2019 were identified as hematoma group. Two control subjects without SSEH were randomly selected for each patient in SSEH group as control group. We collected gender, age, body mass index (BMI), ossification of the posterior ligament (OPLL), comorbidities, anti-platelet or anti-coagulate treatment, coagulation function, segments, instrumental fixation, surgical approach, surgical procedure, duration of surgery and estimated blood loss, which might affect the occurrence of symptomatic epidural hematoma. T-test and Chi-square test were used to univariable test. Multifactor logistic regression analysis was used to investigate the correlation with symptomatic epidural hematoma, furthermore its causes were explored.
Results
Among 18,220 patients, 43 subjects developed SSEH, the incidence was 0.24%. The median time from the end of index surgery to SSEH was 150 min (25 and 75 percentile: 85 min to 290 min). The neurologic function before evacuation by modified Frankel scale is grade B in 5 patients, C in 32 patients, grade D in 6 patients. All patients’ symptoms relieved partially or completely after evacuation. All patients with neurologic deficit worse than grade C pre-evacuation had at least one-grade improvement except for one patient. Multifactor logistic regression revealed OPLL involved segments are significantly correlated to the incidence of postoperative symptomatic epidural hematoma (P < 0.05), with a cut-off value of 1.5 levels.
Conclusion
OPLL involved segments are significantly correlated to the incidence of postoperative symptomatic epidural hematoma.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cervical spine surgery is widely used for the treatment of various pathologies, including degenerative, traumatic, infectious, neoplastic, and iatrogenic conditions. The outcome of cervical spine surgery is generally favorable and can lead to significant improvement in quality of life in well-selected patients [1, 2]. Surgical goals typically include decompression the neural elements, stabilization of unstable segments, and maintenance or restoration of cervical alignment. Even though cervical spine surgery is generally safe, complications can still occur. Some complications, such as mild dysphagia after anterior cervical discectomy and fusion (ACDF) rarely result in any long-term sequelae [3]. On the other hand, rarer complications such as symptomatic spinal epidural hematoma (SSEH), vertebral artery injury, or esophageal injury can lead to devastating consequences, and can cause significant morbidity, even death [4,5,6,7,8,9].
Postoperative spinal epidural hematoma is a common finding on post-operative imaging among patients undergoing spine surgery. Spinal epidural hematoma has been identified by either computed tomography (CT) or magnetic resonance imaging (MRI) in 33% to 100% of the cases undergone lumbar decompression [10,11,12]. Fortunately, most of these cases are asymptomatic and do not require any intervention. However, when SSEH occurs, the consequences can be catastrophic, leading to rapidly progressive neurological deficits, respiratory failure, and even death. Early diagnosis and prompt hematoma evacuation are imperative to optimize neurologic recovery [13, 14], and delayed diagnosis and treatment can lead to irreversible neurological injury [15, 16].
Previous studies have reported the estimated incidence of SSEH ranging from 0.09 to 1.5% [4,5,6,7]. Many of these studies demonstrated a wide range of risk factors for SSEH development. Goldstein et al. [5] reported postoperative use of nonsteroidal anti-inflammatory drugs (NSAIDs) and increased Charlson Comorbidity Index (CCI) as significant predictors of the development of SSEH after posterior cervical surgery. Yin et al. [5] reported hypertension and multilevel cervical procedure with increased bleeding tendency are risk factors for SSEH after cervical spine surgery.
The aim of the current study is to investigate the incidence, risk factors, and clinical outcome of SSEH after cervical spine surgery at a single, tertiary, academic medical center over a 10-year period.
Methods
Design and participants
This study was a retrospective study, approved by the institution ethics review board (IRB00006761-M2020380).
All patients who underwent cervical spinal surgery at our institute between January 1, 2009, and February 28, 2019, were included in this study. Theatre logbooks were searched for cases who had unexpectedly returned to theater. All patients who were diagnosed with SSEH and underwent evacuation of epidural hematoma were included in the current study. The diagnostic criteria of SSEH include: ①Progressive newly onset of neurologic deficit postoperatively; ②With or without advanced radiological evidence; ③Verified hematoma formation during emergency evacuation; ④Good response to timely surgical evacuation. All SSEH cases were confirmed epidural hematoma formation during evacuation [17]. The following groups of patients were excluded: fractures and/or dislocation of cervical spine, cervical spinal tumor, infectious disease, and upper cervical disease (e.g., atlantoaxial dislocation, atlantoaxial instability). Two control subjects without SSEH were randomly selected for each patient by computer algorithms to increase the power of the study. Each control had undergone a similar cervical spine procedure at the same hospital. (Fig. 1).
Data collection
A detailed review of the medical records for all patients in the SSEH group and control group was carried out. Patient demographics, medical comorbidities, cervical spine diagnosis, surgical procedure details were recorded and analyzed. The neurological status was evaluated using the Frankel scale.
Data analysis
All statistical analysis was performed using SPSS (version 24.0, IBM, Armonk, NY, USA). Continuous variables were summarized with descriptive statistics (mean and standard deviation), and categorical variables were summarized as percentage incidence. T-test was used for continuous variables. Chi-square test was performed for all categorical variables. A p-value of less than 0.05 was considered significant. After filtering with univariable test. We used the Kolmogorov–Smirnov test for determining normality of continuous data. Continuous data that had a normal distribution are expressed as mean ± standard deviation and the independent sample t-test was used for comparison between groups. Continuous data that were not normally distributed are expressed as the median (25th and 75th percentiles), and we used the Mann–Whitney U test for comparison between groups. Multivariable Logistic regression test was performed on potential risk factors to further determine risk factors for SSEH. All tests were two-sided and P < 0.05 was considered statistically significant.
Results
Characteristics of the participants
Between January 1, 2009, and December 31, 2019, a total of 18,220 patients underwent cervical surgery at our institute, 10,514 patients underwent anterior approach, 6,397 patients underwent posterior approach, and 1,166 underwent combined approach. There were 43 patients developed postoperative SSEH, for a total incidence of 0.24%. The mean age for patients in SSEH group was 53.5 years (ranging from 21 to 78 years), and 34 patients were male. The mean BMI (Body mass index) was 25.84 kg·m−2 (ranging from 20.82 to 32.87 kg·m−2). The most common diagnosis for primary surgery is degenerative cervical myelopathy (DCM) (n = 39, 90.7%), followed by cervical radiculopathy (n = 4, 9.3%). The mean pre-operative mJOA score was 12.7 (ranging from 4.5 to 16). The mean pre-operative neck disability index (NDI) score was 5 (ranging from 0 to 21). The most common surgical procedure performed was laminoplasty (n = 25, 58.2%), followed by anterior cervical corpectomy and fusion (ACCF) (n = 8, 18.6%), then ACDF (n = 5, 11.6%), and lastly laminectomy with instrumented fusion (n = 5, 11.6%). The mean levels treated was 4.5 levels (range, 1–7 levels). The mean duration of surgery was 109 min (range, 45–289 min), with an average EBL of 270 ml (range, 10–2000 ml). In our institute, deep drains with mild suction were routinely placed in all patients underwent spine surgery. The drains were removed until discharge was less than 50 ml/24 h. In 37 patients, SSEH developed before the removal of deep drain, in 22 of whom, blood clot could be discovered in the drain. In patients with SSEH developed after removal of the deep drain, the mean time interval from drain removal to SSEH development was 11.9 days (ranging from 1.5 to 34 days).
Within the SSEH group, eleven patients had hypertension, six patients had type 2 diabetes millitus, and 2 patients had coronary artery disease. None of the patients used NSAIDs perioperatively. Seven patients were current smokers, with a mean of 24 pack-year history. (Table 1).
Two patients received anti-platelet therapy pre-operatively, one by aspirin only and one by aspirin combined with clopidogrel. Patients who were on anti-platelet stopped one week before surgery. For patients who received anti-coagulate therapy, warfarin was switched to low molecular weight heparin (LMWH) as a bridging therapy, and then stopped LMWH 12 h before surgery, with a normal international normalized ratio (INR). For patients without risk factor of deep vein thrombosis (DVT) formation, no routinely prophylactic anticoagulant was used post-operatively. One patient developed cerebral infarction 2 days after cervical surgery, after thorough evaluation and consultation from neurologist, aspirin combined with clopidogrel were used as for the treatment of cerebral infarction. 7 days after surgery, the patient was diagnosed with DVT, hence, the patient received LMWH for the treatment of DVT. Unfortunately, 15 days after surgery, the patient suddenly had quadriplegia and confirmed with SSEH formation by MRI, received emergent evacuation.
Clinical outcome of SSEH
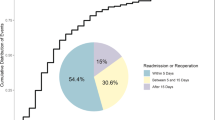
In our study of 43 patients with postoperative SSEH, 23 patients were diagnosed by clinical examination alone and diagnosis was confirmed during surgical intervention. The remaining 20 patients had imaging confirmation of SSEH prior to intervention, with 17 patients diagnosed on MRI (Fig. 2), and three diagnosed on CT. The median time from the onset of symptoms to advanced imaging was 115 min. The median time interval between the end of the index surgery and the time of newly onset of neurological symptoms was 150 min (25th and 75th percentile: 85 min to 290 min). All patients underwent emergent evacuation. The median interval between the diagnosis of hematoma to surgical evacuation was 150 min (range, 15 min–8 days). In 21 patients, the initial self-reported presentation was paraplegia. In other 21 patients, the initial self- reported presentation was hemiplegia. In the remaining one patient, the initial self-reported presentation was quadriplegia.
A 56-year-old male, his clinical diagnose was cervical myelopathy caused by diffusive idiopathic skeleton hyperostosis (DISH), underwent multilevel laminectomy with fusion. He encountered cerebral infarction 2 days after surgery. Hence aspirin combined with clopidogrel were used for the treatment of cerebral infarction. 7 days after surgery, the patient was diagnosed with deep vein thrombosis, and received low molecular weight heparin. 15 days after surgery, the patient had rapid onset of quadriplegia after daily movement. MRI scan was confirmed epidural hematoma formation. An emergent evacuation was arranged immediately. a pre-operative CT scan sagittal plane showed DISH with OPLL and ossification of ligamentum flavum. b pre-operative MRI showed multilevel spinal cord compression with intradural signal change. c MRI after onset of quadriplegia showed acute formation of epidural hematoma (white arrow). d During emergent evacuation, massive blood clot was discovered in epidural space
The neurologic function before evacuation by modified Frankel scale is grade B in 5 patients, C in 32 patients, grade D in 6 patients (partially relieved by changing body position, or deoppilation of the drain). All patients’ symptoms relieved partially or completely after evacuation. All patients with neurologic deficit worse than grade C pre-evacuation had at least one-grade improvement except for one patient. After emergent evacuation, there were two patients in grade C, 34 patients in grade D, and seven patients in grade E. The average follow-up time was 10.2 ± 12.7 months (range: 2 to 58 months). In hematoma group, the pre-operative mJOA score was 12.1 ± 2.8 points, and the mJOA score during last follow-up was 14.7 ± 2.1 points, the average recovery rate was 53.7%. In control group, the pre-operative mJOA score was 13.0 ± 2.6 points, compare to 15.2 ± 1.7 points during last follow-up, the average recovery rate was 46.5%. None of pre-operative mJOA score, mJOA score during last follow-up or recovery rate showed statistically different between two groups using t-test (p = 0.25, 0.29, 0.67).
Risk factors for SSEH development
The data analysis included patients in SSEH group (n = 43) and control group (n = 86).
Using univariant analysis, we found male gender, OPLL involved segment, revision surgery, longer surgical segments, longer surgical duration, higher estimated blood loss (EBL) are the risk factors for SSEH development. Moreover, ACDF had decreased risk of SSEH compared to other cervical procedures (Table 2).
Forward selection stepwise logistic regression analysis revealed one independent risk factor associated with development of SSEH postoperatively: OPLL involved segments (p = 0.007, OR = 1.556) (Table 3). And cut off values calculated through receiver operating characteristic (ROC) curve of surgical segments and OPLL involved segments were 1.5.
Discussion
SSEH following spinal surgery is a rare event. Previous study had revealed that the incidence ranged from 0.10% to 0.24% in all spine cases [13, 18,19,20,21]. There were several retrospective case series reported the incidence of SSEH following cervical spine surgery. Aono et al. [7] reported 1,376 patients underwent cervical spine surgery, 466 patients underwent anterior procedures, 1 patient developed SSEH (0.21%), 910 patients underwent posterior laminoplasty, 4 SSEH were identified (0.44%). Yin et al. [6] reported 12 out of 2,338 cervical spinal cases developed acute SSEH after cervical spine surgery, with the incidence of 0.5%. In a single institute study, Amiri [17] reported 1,962 patients underwent cervical surgery, the incidence of SSEH was 0.15%. Goldstein et al. [5] identified the rate of SSEH to be 1.5% (8/529) over a 10-year period, this research was restricted to posterior cervical surgery. In a multicenter retrospective cohort during a 7-year period, Schroeder et al. [4] reported the incidence of SSEH following cervical spine surgery of 0.09%.
As one of the largest spine centers in our nation, in our institute, a total of 18,220 patients underwent cervical spine surgery over a 10-year period. There were 10,514 patients underwent anterior procedure, 13 patients developed SSEH (0.12%); 6,397 underwent posterior procedure, 30 of whom developed SSEH (0.47%); 1,166 underwent combined anterior and posterior procedure in one stage, none of them developed SSEH. This study finds that the overall SSEH in the cervical spine is 0.24%. The result is consistent with the previous studies, and our study represents the largest case series over a 10-year period.
Awad et al. [18] had identified preoperative use of non-steroid anti-inflammatory drugs (NSAIDs), Rh-positive blood type, and age more than 60 years old were preoperative risk factors for SSEH formation. Operative levels > five levels, hemoglobin lower than 10 g/dl, and EBL more than 1L were the intraoperative risk factors. Moreover, INR more than 2.0 within the first 48 h was the postoperative risk factor. Amiri et al. [17] reported alcohol greater than 10 units a week, previous spinal surgery, and multilevel procedures were shown to be the risk factors of SSEH. Kou et al. [19] reported multilevel procedures, and the presence of preoperative coagulopathy were the significant risk factors. In a single-institution retrospective review through a 10-year period focused on posterior cervical surgery, Goldstein et al. identified that postoperative usage of NSAIDs and increased CCI were independent risk factors of SSEH.
In univariant analysis, we have identified male, segments involved by OPLL, number of segments, type of procedure, duration of surgery, EBL, and revision surgery were risk factors of SSEH development. Number of segments and EBL had been reported as a risk factor by previous research. Other risk factors were the first time identified as potential risk factors. More extensive surgery, revision surgery and bleeding tendency might contribute to postoperative SSEH formation.
Through multivariate logistic regression analysis, OPLL involved segments was the only independent risk factor of SSEH development postoperatively, other risk factors that we identified might related to or were the result of OPLL involvement. To our knowledge, this is the first study that identified OPLL as an independent risk factor for SSEH development.
OPLL is a pathologic process of lamellar bone deposition at the site of the posterior longitudinal ligament (PLL), more frequently seen in older adults and in males, with a reported male-to-female ration of 2:1 [22, 23]. OPLL is more common in East Asia. In Japan, the prevalence was reported to be 1.9 to 4.3% among the population older than 30 years of age [24,25,26]. In other East Asian countries and regions, the prevalence was 3.6% in Korea [27] and 2.8% in Taiwan [28], which is similar to that of Japan. While the estimated rate among Caucasian population in North America is 0.12%. All previous studies focused on SSEH development after cervical spine surgery were conducted in North America [4, 5]. This might explain the reason for OPLL involved segments being first time identified as an independent risk factor of SSEH development after cervical spine surgery. Previous studies had discovered a bleeding tendency in patients with OPLL than patients with CSM [29, 30], which might lead to postoperative SSEH development. A multi-institutional study in Japan had revealed that patients with an occupying ratio higher than 60% were at 2.4-fold higher risk of intraoperative blood loss greater than 500 g [31]. Kishiya et al. [32] had found that patients with continuous type of ossification show a tendency of bleeding and significant blood loss after surgery compared to DCM patients. Moreover, in a retrospective review of 2,375 anterior cervical spine procedures, to investigate the risk factors involved in the development of anterior cervical retropharyngeal hematoma, O’Neil et al. [33] had identified presence of diffuse idiopathic skeletal hyperostosis (DISH) or OPLL to be risk factors of post-operative anterior retropharyngeal hematoma formation. We assumed that the presence of DISH and OPLL was related to intra-operative and post-operative hemorrhagic complications. There were two main hypotheses to explain this phenomenon. One is ectopic bone formation, like OPLL and diffusive DISH, the ossification initiated through the endochondral pathway. The invasion of blood vessels into cartilaginous template plays an important role in the pathophysiologic endochondral ossification of bone tissue [34, 35]. There is also a difference of angiogenesis-related genes in expression between OPLL patients and DCM patients [34]. The other hypothesis is that the bleeding tendency in OPLL patients is caused by increased intraosseous pressure, which is identical to the epidural venous pressure [35] and abnormal distention of the epidural venous plexus [29]. Thus, more vertebrae involved by OPLL is related to more surgical segments, longer surgical duration, more EBL, surgical technique other than ACDF, and revision surgery.
This study represents the largest clinical series focused on SSEH development after cervical spine surgery, and the first study to identify OPLL involved segments as the independent risk factor of SSEH. However, the study still has limitations. The retrospective nature of this study makes it inevitable for selection and reporting bias. And all surgeries were performed in the same institution, the individual surgeon and institution-based risks cannot be eliminated. In addition, some patients used herbal medicine preoperatively would have interference with the results.
Conclusion
The current study is the largest series to date to analyze the incidence of SSEH following cervical spine surgery. The incidence of SSEH following cervical spine surgery is 0.24%. Male, segments involved by OPLL, surgical segments, procedure, duration of surgery, EBL, and revision surgery were related to SSEH. Further, OPLL involved segments is an independent risk factor of SSEH development postoperatively, and the cut-off value is 1.5 segments. For patients with OPLL involved more than 1.5 segments, spine surgeons should pay increased attention to hemostasis, and monitor patients more closely for new onset of neurological deficits after surgery, due to their increased risk of developing of SSEH.
Ethics approval
This study is compliance with ethical standards and approved by Research Ethics Committee.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Woods BI, Hilibrand AS (2015) Cervical radiculopathy: epidemiology, etiology, diagnosis, and treatment. J Spinal Disord Tech 28:E251-259. https://doi.org/10.1097/BSD.0000000000000284
Shamji MF, Massicotte EM, Traynelis VC, Norvell DC, Hermsmeyer JT, Fehlings MG (2013) Comparison of anterior surgical options for the treatment of multilevel cervical spondylotic myelopathy: a systematic review. Spine Phila Pa. https://doi.org/10.1097/BRS.0b013e3182a7eb27
Joaquim AF, Murar J, Savage JW, Patel AA (2014) Dysphagia after anterior cervical spine surgery: a systematic review of potential preventative measures. Spine J 14:2246–2260. https://doi.org/10.1016/j.spinee.2014.03.030
Schroeder GD, Hilibrand AS, Arnold PM, Fish DE, Wang JC, Gum JL, Smith ZA, Hsu WK, Gokaslan ZL, Isaacs RE, Kanter AS, Mroz TE, Nassr A, Sasso RC, Fehlings MG, Buser Z, Bydon M, Cha PI, Chatterjee D, Gee EL, Lord EL, Mayer EN, McBride OJ, Nguyen EC, Roe AK, Tortolani PJ, Stroh DA, Yanez MY, Riew KD (2017) Epidural hematoma following cervical spine surgery. Global Spine J 7:120S-126S. https://doi.org/10.1177/2192568216687754
Goldstein CL, Bains I, Hurlbert RJ (2015) Symptomatic spinal epidural hematoma after posterior cervical surgery: incidence and risk factors. Spine J 15:1179–1187. https://doi.org/10.1016/j.spinee.2013.11.043
Yin G, Ni B (2014) Acute postoperative cervical spinal epidural hematoma. Acta Orthop Traumatol Turc 48:437–442. https://doi.org/10.3944/AOTT.2014.13.0133
Aono H, Ohwada T, Hosono N, Tobimatsu H, Ariga K, Fuji T, Iwasaki M (2011) Incidence of postoperative symptomatic epidural hematoma in spinal decompression surgery. J Neurosurg Spine 15:202–205. https://doi.org/10.3171/2011.3.SPINE10716
Guan Q, Chen L, Long Y, Xiang Z (2017) Iatrogenic vertebral artery injury during anterior cervical spine surgery: a systematic review. World Neurosurg 106:715–722. https://doi.org/10.1016/j.wneu.2017.07.027
Zhong ZM, Jiang JM, Qu DB, Wang J, Li XP, Lu KW, Xu B, Chen JT (2013) Esophageal perforation related to anterior cervical spinal surgery. J Clin Neurosci 20:1402–1405. https://doi.org/10.1016/j.jocn.2012.11.020
Ross JS, Masaryk TJ, Modic MT, Bohlman H, Delamater R, Wilber G (1987) Lumbar spine: postoperative assessment with surface-coil MR imaging. Radiology 164:851–860. https://doi.org/10.1148/radiology.164.3.3615887
Leonardi MA, Zanetti M, Saupe N, Min K (2010) Early postoperative MRI in detecting hematoma and dural compression after lumbar spinal decompression: prospective study of asymptomatic patients in comparison to patients requiring surgical revision. Eur Spine J 19:2216–2222. https://doi.org/10.1007/s00586-010-1483-x
Mirzai H, Eminoglu M, Orguc S (2006) Are drains useful for lumbar disc surgery? A prospective, randomized clinical study. J Spinal Disord Tech 19:171–177. https://doi.org/10.1097/01.bsd.0000190560.20872.a7
Yi S, Yoon DH, Kim KN, Kim SH, Shin HC (2006) Postoperative spinal epidural hematoma: risk factor and clinical outcome. Yonsei Med J 47:326–332. https://doi.org/10.3349/ymj.2006.47.3.326
Kebaish KM, Awad JN (2004) Spinal epidural hematoma causing acute cauda equina syndrome. Neurosurg Focus 16:e1
Hans P, Delleuze PP, Born JD, Bonhomme V (2003) Epidural hematoma after cervical spine surgery. J Neurosurg Anesthesiol 15:282–285
Mukerji N, Todd N (2013) Spinal epidural haematoma; factors influencing outcome. Br J Neurosurg 27:712–717. https://doi.org/10.3109/02688697.2013.793289
Amiri AR, Fouyas IP, Cro S, Casey AT (2013) Postoperative spinal epidural hematoma (SEH): incidence, risk factors, onset, and management. Spine J 13:134–140. https://doi.org/10.1016/j.spinee.2012.10.028
Awad JN, Kebaish KM, Donigan J, Cohen DB, Kostuik JP (2005) Analysis of the risk factors for the development of post-operative spinal epidural haematoma. J Bone Joint Surg Br 87:1248–1252. https://doi.org/10.1302/0301-620X.87B9.16518
Kou J, Fischgrund J, Biddinger A, Herkowitz H (2002) Risk factors for spinal epidural hematoma after spinal surgery. Spine Phila Pa 27(15):1670–1673. https://doi.org/10.1097/00007632-200208010-00016
Uribe J, Moza K, Jimenez O, Green B, Levi AD (2003) Delayed postoperative spinal epidural hematomas. Spine J 3:125–129
Kao FC, Tsai TT, Chen LH, Lai PL, Fu TS, Niu CC, Ho NY, Chen WJ, Chang CJ (2015) Symptomatic epidural hematoma after lumbar decompression surgery. Eur Spine J 24:348–357. https://doi.org/10.1007/s00586-014-3297-8
Matsunaga S, Yamaguchi M, Hayashi K, Sakou T (1999) Genetic analysis of ossification of the posterior longitudinal ligament. Spine 24(10): 937–938 https://doi.org/10.1097/00007632-199905150-00002.
Okamoto K, Kobashi G, Washio M, Sasaki S, Yokoyama T, Miyake Y, Sakamoto N, Ohta K, Inaba Y, Tanaka H, Japan Collaborative Epidemiological Study Group for Evaluation of Ossification of the Posterior Longitudinal Ligament of the Spine R (2004) Dietary habits and risk of ossification of the posterior longitudinal ligaments of the spine (OPLL); findings from a case-control study in Japan. J Bone Miner Metab 22:612–617. https://doi.org/10.1007/s00774-004-0531-1
Taketomi E, Sakou T, Matsunaga S, Yamaguchi M (1992) Family study of a twin with ossification of the posterior longitudinal ligament in the cervical spine. Spine https://doi.org/10.1097/00007632-199203001-00012.
Inamasu J, Guiot BH, Sachs DC (2006) Ossification of the posterior longitudinal ligament: an update on its biology epidemiology and natural history. Neurosurgery 58:1027–1039 https://doi.org/10.1227/01.NEU.0000215867.87770.73.
Wang PN, Chen SS, Liu HC, Fuh JL, Kuo BI, Wang SJ [1999] Ossification of the posterior longitudinal ligament of the spine A case-control risk factor study. Spine (Phila Pa 1976) 24:142–144 https://doi.org/10.1097/00007632-199901150-00010.
Chiba K, Ogawa Y, Ishii K, Takaishi H, Nakamura M, Maruiwa H, Matsumoto M, Toyama Y (2006) Long-term results of expansive open-door laminoplasty for cervical myelopathy--average 14-year follow-up study. Spine (Phila Pa 1976) 31: 2998–3005 https://doi.org/10.1097/01.brs.0000250307.78987.6b.
Ogawa Y, Chiba K, Matsumoto M, Nakamura M, Takaishi H, Hirabayashi H, Hirabayashi K, Nishiwaki Y, Toyama Y (2005) Long-term results after expansive open-door laminoplasty for the segmental-type of ossification of the posterior longitudinal ligament of the cervical spine: a comparison with nonsegmental-type lesions. J Neurosurg Spine 3:198–204. https://doi.org/10.3171/spi.2005.3.3.0198
Kato S, Chikuda H, Seichi A, Ohtsu H, Kimura A, Toyama Y [2012] Radiographical risk factors for major intraoperative blood loss during laminoplasty in patients with ossification of the posterior longitudinal ligament. Spine 37: E1588–1593 https://doi.org/10.1097/BRS.0b013e3182712b74.
Kishiya M, Furukawa K, Yokoyama T, Kudo H, Ono A, Numasawa T, Wada K, Toh S (2009) Comparison of cardiovascular parameters between patients with ossification of posterior longitudinal ligament and patients with cervical spondylotic myelopathy. J Spinal Disord Tech 22:361–366. https://doi.org/10.1097/BSD.0b013e31817eee55
Yamamoto Y, Furukawa K, Ueyama K, Nakanishi T, Takigawa M, Harata S [2002] Possible roles of CTGF/Hcs24 in the initiation and development of ossification of the posterior longitudinal ligament. Spine (Phila Pa 1976) 27:1852–1857 https://doi.org/10.1097/00007632-200209010-00009.
el Miedany YM, Wassif G, el Baddini M (2000) Diffuse idiopathic skeletal hyperostosis (DISH): is it of vascular aetiology? Clin Exp Rheumatol 18:193–200
O'Neill KR, Neuman B, Peters C, Riew KD [2014] Risk factors for postoperative retropharyngeal hematoma after anterior cervical spine surgery. Spine (Phila Pa 1976) 39:E246–252 https://doi.org/10.1097/BRS.0000000000000139.
Iwasawa T, Iwasaki K, Sawada T, Okada A, Ueyama K, Motomura S, Harata S, Inoue I, Toh S, Furukawa KI (2006) Pathophysiological role of endothelin in ectopic ossification of human spinal ligaments induced by mechanical stress. Calcif Tissue Int 79:422–430. https://doi.org/10.1007/s00223-006-0147-7
Kakiuchi M (2002) Intraoperative blood loss during cervical laminoplasty correlates with the vertebral intraosseous pressure. J Bone Joint Surg Br 84:518–520. https://doi.org/10.1302/0301-620x.84b4.12934
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Consent to participate
Written informed consent was obtained from individual or guardian participants.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xia, T., Zhou, F., Chu, H. et al. Incidence and risk factors of postoperative symptomatic spinal epidural hematoma in cervical spine surgery: a single center, retrospective study of 18,220 patients. Eur Spine J 31, 2753–2760 (2022). https://doi.org/10.1007/s00586-022-07301-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-022-07301-z