Abstract
Purpose
Quantitative computed tomography (QCT) is an alternate imaging method to dual X-ray absorptiometry to measure bone mineral density (BMD). One advantage of QCT is that it allows site-specific volumetric BMD (vBMD) measurements in a small region. In this study, we utilized site-specific, endplate vBMD (EP-vBMD) as a potential predictive marker of severe cage subsidence in standalone lateral lumbar interbody fusion (SA-LLIF) patients and conducted a retrospective comparative study between EP-vBMD and trabecular vBMDs (Tb-vBMD) in the vertebrae.
Methods
Patients undergoing SA-LLIF from 2007 to 2016 were retrospectively reviewed. EP-vBMD was defined as the average of the upper and lower endplate volumetric BMDs measured in cortical and trabecular bone included in a 5-mm area of interest beneath the cage contact surfaces. We compared Tb-vBMDs and EP-vBMDs between disk levels that had severe cage subsidence and levels with no severe subsidence.
Results
Both EP-vBMD and Tb-vBMD could be measured in 210 levels of 96 patients. Severe cage subsidence was observed in 58 levels in 38 patients. Median (IQR) Tb-vBMD was 120.5 mg/cm3 (100.8–153.7) in the non-severe subsidence group and 117.9 mg/cm3 (90.6–149.5) in the severe subsidence group (p = 0.393), whereas EP-vBMD was significantly lower in the severe subsidence group than the non-severe subsidence group (non-severe subsidence 257.4 mg/cm3 (216.3–299.4), severe subsidence 233.5 mg/cm3 (193.4–273.3), p = 0.026).
Conclusion
We introduced a novel site-specific vBMD measurement for cage subsidence risk assessment. Our results showed that EP-vBMD was a reproducible measurement and appeared more predictive for severe cage subsidence after SA-LLIF than Tb-vBMD.
Graphic abstract
These slides can be retrieved under Electronic Supplementary Material.

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Lateral lumbar interbody fusion (LLIF) is a relatively new approach and considered less invasive than the traditional anterior approach. Additionally, LLIF can achieve nerve decompression indirectly, without removing posterior elements, for select patients. However, cage subsidence is sometimes seen and may lead to loss of indirect decompression, malalignment, non-union, and eventually revision surgery [1,2,3].
Previous studies showed that osteoporosis is a risk factor for cage subsidence [4,5,6,7]. Currently, dual X-ray absorptiometry (DXA) is the gold standard to measure bone mineral density (BMD) [8]. Computed tomography (CT)-based techniques, such as Hounsfield units (HU) and quantitative computed tomography (QCT), are emerging alternate methods to DXA as assessments of bone strength [9,10,11]. Studies indicated that CT-based BMD measurements allow more accurate measurements than DXA in patients with severe degenerative spine conditions [12]. Additionally, it has been shown that pre-existing CT images for diagnostic or surgical planning purposes can be retrospectively utilized for assessment of bone health [13, 14]. In the majority of spine surgery patients, this alternate assessment for osteoporosis overcomes one of the major drawbacks of X-ray and CT: excessive radiation exposure. The HU assessment is commonly used in previous studies. However, there are substantial differences in HU values depending on the CT model or even tube voltage for the same model [15]. Thus, HU values from one CT machine may not be comparable to those from another machine. Asynchronous phantomless QCT can be applied to CT images by different machine models. This feature is preferable especially for opportunistic screening.
Another advantage of a QCT assessment is that it allows site-specific volumetric BMD (vBMD) measurements in a small region. Recent studies showed that site-specific CT assessments had a higher predictive value for fracture and screw loosening after pedicle screw insertion [16]. Although cage subsidence is a common complication after interbody fusion and osteoporosis is a risk factor, little is known about the association between regional site-specific vBMD values and cage subsidence after spinal interbody fusion. Since the contact surfaces between implant and bone only include the cage and endplates in standalone LLIF (SA-LLIF), it is suitable to assess the effect of site-specific vBMDs on the risk of cage subsidence in this construct type.
In this study, we utilized site-specific, endplate vBMD (EP-vBMD) as a potential predictive marker of severe cage subsidence in SA-LLIF patients and conducted a retrospective comparative study between vertebral EP-vBMD and trabecular vBMDs (Tb-vBMD).
Materials and methods
Patient population
This study was approved by our institutional review board, and informed consent from each patient was waived because of the retrospective nature of this study. The data of consecutive patients undergoing SA-LLIF from 2007 to 2016 at a single academic institution were retrospectively reviewed. Patients who had a preoperative CT, over 6 months of follow-up, and radiological imaging for cage subsidence assessment (between 6 and 12 months after surgery) were included. We excluded levels with a corpectomy, expandable cage, previous instrumentation at the index levels, previous fracture, planned staged surgery, and poor imaging quality.
All patients underwent SA-LLIF utilizing either the XLIF system (Nuvasive, Inc., San Diego, CA, USA) or the COUGAR system (Depuy Spine Inc., Raynham, MA, USA). All procedures were performed utilizing the mini-open single-incision technique by five fellowship-trained orthopedic spine surgeons with at least 5 years of experience performing LLIF surgeries. In this mini-incision technique, an approximate 3-cm incision was made for a single-level procedure and all steps of the LLIF procedure were performed with direct visualization of the splitting psoas muscle and target disk(s). The details of this technique are published in the literature [17]. Special care was taken to span the apophyseal rings of both end plates during cage insertion.
Trabecular and endplate QCT-vBMD measurements
The calculation of vBMD values was performed on both sides of the LLIF cage using the Mindways QCT Pro Software (Mindways Software, Inc., Austin, TX, USA). The phantomless asynchronously calibrated QCT method that converts HU to vBMD using a calibrated conversion factor specific to each CT model was utilized [18]. All conversion factors were provided by the software manufacturer (Mindways Software, Inc.).
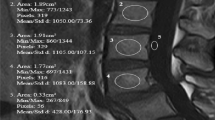
Along with the standard L1/2 vBMD measurement, we measured regional Tb-vBMD that was defined as the average vBMD of the upper and lower vertebral trabecular bones in the same method as the standard L1/2 measurement recommended for osteoporosis screening by 3D QCT (Fig. 1a) [19]. If there were any apparent sclerotic regions, which lead to inaccurate measurement of trabecular vBMD in the vertebra, the regions were excluded from the area of interest (AOI) [20,21,22]. In cases where sclerosis was too diffuse to be excluded, the vertebra was excluded from analysis. All CTs were conducted with the patient in the supine position.
Tb-vBMD and EP-vBMD measurements a Tb-vBMD measurement of L4 vertebra, b EP-vBMD measurement of L4 lower endplate, c EP-vBMD measurement of L5 upper endplate. Upper row: axial images. Middle row: sagittal reconstruction images. Lower row: coronal reconstruction images. Circle and boxes represent for the area of interest
EP-vBMD was defined as the average of the upper and lower endplate volumetric BMDs measured in cortical and trabecular bone included in a 5-mm AOI beneath the cage contact surfaces (Fig. 1b, c). First, the position of the cage was checked with postoperative CT and radiographs. The AOI was set to include as much cage contact surface of each endplate as possible. Special care was taken not to let the AOI exceed the outer cortical margin of the vertebra and endplate to avoid inclusion of any outer- or inter-vertebral spaces. Unlike the Tb-vBMD measurement, all sclerotic regions and endplate defects were included in the AOI for EP-vBMD, if these were located in the cage contact surfaces.
EP-vBMD and Tb-vBMD measurements were taken in a blinded manner by two different researchers who had completed a training session on QCT measurement. Since EP-vBMD was a novel measurement, we also conducted a validation study by two independent raters utilizing 52 randomly selected endplates.
Cage subsidence assessment and comparisons
Cage subsidence was assessed utilizing lumbar spine lateral radiographs and/or CT taken between 6 and 12 months postoperatively. If a patient had multiple radiographs during the period, the most recent radiograph was used for subsidence assessment. If a patient had revision surgery related to subsidence within 6 months after the index surgery, cage subsidence was evaluated using imaging taken immediately before the revision surgery. We used the grading system by Marchi et al. [1] as Grade 0, 0–24% loss of postoperative disk height; Grade I, 25–49%; Grade II, 50–74%; and Grade III, 75–100%. Severe subsidence was defined as Grade II or III subsidence according to Marchi’s original report. Cage subsidence was assessed by three researchers under the supervision of a board-certified orthopedic spine surgeon. For each patient, subsidence grading was conducted by a single examiner. The examiners were blinded to the results of vBMD measurements. Tb-vBMDs and EP-vBMDs were measured as well as other demographic factors between disk levels that had severe cage subsidence and levels with no severe subsidence.
Receiver operating characteristic (ROC) curve analyses were conducted in both Tb-vBMDs and EP-vBMDs, and their areas under the curve (AUC) were compared to determine diagnostic performance for prediction of severe cage subsidence.
Statistical analysis
The Fisher exact test was used for categorical variables. The Shapiro–Wilk test was conducted to assess the normality of continuous variables. As both Tb-vBMD and EP-vBMD were not distributed normally, the Mann–Whitney test was used for the comparisons of continuous variables. The Spearman’s coefficient was calculated for the correlation analysis between EP- and Tb-vBMDs. p < 0.05 was set as statistical significance. For the validation study of EP-vBMD, the interclass correlation coefficient (ICC) and standard error of measurement (SEM) were also calculated. We defined an ICC > 0.90 as excellent, 0.80–0.90 as good, 0.70–0.80 as acceptable, and ≤ 0.70 as poor. All analyses were conducted in R software (R for 3.1.0 GUI 1.64).
Results
Out of 280 levels of the 130 patients who met the inclusion criteria, both EP-vBMD and Tb-vBMD could be measured in 210 levels of 96 patients and included in the final analysis. The reasons for exclusion included CTs taken with QCT software incompatible machines for 34 levels, previous instrumentation at the index levels for 33 levels, and poor imaging quality for 3 levels. The median (IQR) age was 68.4 (62.1–74.2), and 63.3% of patients were females. Median L1/2 vBMD was 104.9 mg/cm3. According to the criteria of the American College of Radiology regarding QCT spine BMD thresholds at L1/L2 [22] (defined as follows: osteoporosis: vBMD < 80 mg/cm3, osteopenia: 80 ≤ vBMD ≤ 120 mg/cm3, normal: vBMD > 120 mg/cm3), 31.2% of the patients were classified as having osteoporosis (Table 1). Median (range) follow-up was 26 (8–102) months. One patient underwent a posterior fusion surgery within 6 months after the first SA-LLIF surgery because of sagittal imbalance due to severe cage subsidence.
For the validation study, the EP-vBMD ICC was excellent [ICC 0.95, 95% confidential interval (CI) 0.92–0.97]. SEM was 22.3 (95% CI 18.7–27.6). A statistically significant moderate correlation was observed between EP- and Tb-vBMDs (ρ = 0.56, 95% CI 0.46–0.64, p < 0.001).
Severe cage subsidence was observed in 58 levels (27.6%) in 39 patients (Table 2 and Fig. 2). No statistically significant difference in age, sex, BMI, current smoking, and previous lumbar surgery was observed between levels with and without severe subsidence. The number of fused levels demonstrated a positive nonsignificant trend (p = 0.051). Median (IQR) Tb-vBMD was 120.5 mg/cm3 (100.8–153.7) in the non-severe subsidence group and 117.9 mg/cm3 (90.6–149.5) in the severe subsidence group (p = 0.393), whereas EP-vBMD was significantly lower in the severe subsidence group [non-severe subsidence 257.4 mg/cm3 (216.3–299.4), severe subsidence 233.5 mg/cm3 (193.4–273.3), p = 0.026] than the non-severe subsidence group (Table 3 and Fig. 3).
Images of a representative 2-level SA-LLIF case with severe cage subsidence. a A lateral lumbar fluoroscopic image taken immediately after L3/4 and L4/5 SA-LLIF. EP-vBMDs in these levels were 205.4 and 413.2 mg/cm3, respectively. b A lateral lumbar radiograph taken at 6 months postoperatively showing Grade III cage subsidence in L3/4, whereas L4/5 demonstrated no subsidence (Grade 0)
ROC curve analyses demonstrated that AUC (95% CI) of Tb-vBMD was 0.54 (0.45–0.63) and that of EP-vBMD was 0.60 (0.51–0.69) (Fig. 4). Since the use of lateral plate could be a confounder for cage subsidence, we performed the same statistical analyses and excluded levels with a lateral plate as a sensitivity analysis. The results showed the same trends [EP-vBMDs: non-severe subsidence 264.5 mg/cm3 (217.8–301.6), severe subsidence 234.3 mg/cm3 (196.6–275.9), p = 0.034, Tb-vBMD: non-severe subsidence 122.0 mg/cm3 (100.1–158.3), severe subsidence 122.2 mg/cm3 (92.0–150.9), p = 0.437].
Discussion
In this study, we introduced a novel site-specific vBMD measurement by QCT for cage subsidence risk assessment. In terms of the association between vBMDs and cage subsidence, although the diagnostic performance of both vBMD measurements was not high enough to be a single predictor, our results demonstrated that EP-vBMD had a significant association with severe SA-LLIF cage subsidence.
The association between osteoporosis and cage subsidence after lumbar interbody fusion has been widely investigated among patients with posterior/transforaminal lumbar interbody fusion (P/TLIF) [5,6,7]. In their DXA study of one-level PLIF patients, Cho et al. [6] reported that cage subsidence (> 2 mm) was more prevalent among patients with a ≤ −2.5 T-score of spinal BMD compared to patients with ≥ −1.0 T-score. Oh et al. [7] also demonstrated that the mean BMD of patients with over 3-mm cage subsidence was significantly lower than that of patients with subsidence of < 1 mm. Regarding CT-based assessments, Mi et al. [5] measured HU values on CT in their matched-case–control study of 32 patients who underwent L4/5 TLIF with unilateral screw fixation and reported that patients with cage subsidence (not defined clearly) showed significantly lower mean HU values than the control group. Our results demonstrated that there was no statistically significant association between subsidence and Tb-vBMDs, which is used clinically in a similar manner as the BMD measurements of Mi’s study. In addition to the patient population differences, one possible reason for this discrepancy is the varying definitions of cage subsidence. In previous studies, cage subsidence was defined as less than 3-mm cage migration into the endplates, which represents less than 20–30% of disk height. It is questionable whether these small changes are clinically significant. In fact, Cho et al. [6] concluded that cage subsidence did not significantly affect clinical outcomes. We utilized the cage subsidence definition of Marchi et al. Using the same definition, Tempel et al. demonstrated that severe cage subsidence was significantly associated with revision surgery after SA-LLIF in contrast to Cho et al. [3]. Since the minimum clinically important threshold for cage subsidence has not been established, further studies are required to address this issue.
In terms of LLIF studies, Tempel et al. [4] showed that patients with a lower DXA-areal BMD, defined as a T-score < −1.0, had a significantly higher incidence of Grade 2 or 3 cage subsidence. The associations between cage subsidence and Tb-vBMDs or bone status categorized using the standard L1/2-vBMD classifications were not statistically significant in our study. Besides the differences in BMD measurement modalities and patient populations, in which patients undergoing LLIF with posterior fixation were included in Tempel’s study, a selection bias for DXA measurement could also explain the discrepancy in results as the authors mentioned was a limitation. They could only include 24% of their total LLIF population due to the availability of DXA results and cage subsidence occurred more commonly among patients who had DXA scan. In contrast, our study was able to include a higher proportion of patients in an unbiased manner. This might also suggest a potential advantage of QCT measurements, which can be performed retrospectively, over DXA.
With respect to site-specific BMD measurements, although no previous study has measured EP-vBMD, our results are comparable to the results of previous studies investigating the effect of site-specific CT-based bone measurement around pedicle screws [16, 23]. Sakai et al. [16] reported that screw trajectory-specific HU was an independent risk factor for pedicle screw loosening, whereas lumbar BMD by DXA demonstrated no significant association with screw loosening. In their QCT study, Ishikawa et al. [23] reported that the pedicle trajectory vBMD, which was measured by setting the AOI over the pedicle screw trajectory, correlated more strongly to screw insertion torque than trabecular vBMD as well as aBMDs by DXA in the spine and hip. These site-specific CT-based assessments might provide better risk estimations for problems in implant-bone contact surfaces such as cage subsidence or screw loosening. One major challenge of site-specific CT assessments is the reproducibility of measurements because the methodology is not as standardized compared to routine assessments. In Sakai’s report, ICCs of screw trajectory HU assessment were 0.91–0.94. Our result of EP-vBMD demonstrated that the ICC of EP-vBMD measured by two raters was 0.95, which was as high as previously reported Tb-vBMD ICCs (0.94–0.99) [24] and pedicle screw trajectory HU. Our data suggest that EP-vBMD is a reproducible measurement and can be utilized in future studies.
Due to the retrospective nature of this study and relatively small sample size, our study contains several limitations. First, we focused on cage subsidence in relatively short time period and consequently do not have clinical information about the long-term consequences of cage subsidence or patient-reported outcomes, including symptoms potentially associated with cage subsidence. Studies with larger sample sizes and longer follow-up are warranted to draw a more definitive conclusion. Additionally, our patient population was relatively heterogeneous with regard to surgical indication despite patients undergoing the same surgical procedure. The clinical course of osteoporosis varies based on patient factors such as biological sex and certain comorbidities which can affect the relationship between EP-vBMD and Tb-vBMD. Moreover, because of the limited availability of preoperative DXAs among our patients, direct comparisons to DXA-aBMD were not possible. The AUC of EP-vBMD demonstrated a low diagnostic accuracy as a single predictor. This clearly indicates that cage subsidence is multifactorial and BMD is only one contributor. Other potential contributors for cage subsidence, such as endplate injury during cage insertion [25] and bone qualitative markers other than BMD, were not analyzed. Lastly, we did not evaluate any preventive measures for severe cage subsidence, such as additional posterior screw fixation. These factors should be addressed in future studies.
In summary, we introduced a novel site-specific vBMD measurement for cage subsidence risk assessment. Our results showed that EP-vBMD was a reproducible measurement and appeared more predictive for severe cage subsidence after SA-LLIF than Tb-vBMD. Along with other parameters, EP-vBMD can be utilized as part of a comprehensive risk assessment for implant-related complications after lumbar fusion surgery.
References
Marchi L, Abdala N, Oliveira L, Amaral R, Coutinho E, Pimenta L (2013) Radiographic and clinical evaluation of cage subsidence after stand-alone lateral interbody fusion. J Neurosurg Spine 19:110–118. https://doi.org/10.3171/2013.4.SPINE12319
Le TV, Baaj AA, Dakwar E, Burkett CJ, Murray G, Smith DA, Uribe JS (2012) Subsidence of polyetheretherketone intervertebral cages in minimally invasive lateral retroperitoneal transpsoas lumbar interbody fusion. Spine 37:1268–1273. https://doi.org/10.1097/BRS.0b013e3182458b2f
Tempel ZJ, McDowell MM, Panczykowski DM, Gandhoke GS, Hamilton DK, Okonkwo DO, Kanter AS (2018) Graft subsidence as a predictor of revision surgery following stand-alone lateral lumbar interbody fusion. J Neurosurg Spine 28:50–56. https://doi.org/10.3171/2017.5.SPINE16427
Tempel ZJ, Gandhoke GS, Okonkwo DO, Kanter AS (2015) Impaired bone mineral density as a predictor of graft subsidence following minimally invasive transpsoas lateral lumbar interbody fusion. Eur Spine J Off Publ Eur Spine Soc Eur Spin Deformity Soc Eur Sect Cerv Spine Res Soc 24(Suppl 3):414–419. https://doi.org/10.1007/s00586-015-3844-y
Mi J, Li K, Zhao X, Zhao CQ, Li H, Zhao J (2017) Vertebral body Hounsfield units are associated with cage subsidence after transforaminal lumbar interbody fusion with unilateral pedicle screw fixation. Clin Spine Surg 30:E1130–e1136. https://doi.org/10.1097/bsd.0000000000000490
Cho JH, Hwang CJ, Kim H, Joo YS, Lee DH, Lee CS (2018) Effect of osteoporosis on the clinical and radiological outcomes following one-level posterior lumbar interbody fusion. J Orthop Sci Off J Jpn Orthop Assoc 23:870–877. https://doi.org/10.1016/j.jos.2018.06.009
Oh KW, Lee JH, Lee JH, Lee DY, Shim HJ (2017) The correlation between cage subsidence, bone mineral density, and clinical results in posterior lumbar interbody fusion. Clin Spine Surg 30:E683–E689. https://doi.org/10.1097/BSD.0000000000000315
Expert Panel on Musculoskeletal I, Ward RJ, Roberts CC, Bencardino JT, Arnold E, Baccei SJ, Cassidy RC, Chang EY, Fox MG, Greenspan BS, Gyftopoulos S, Hochman MG, Mintz DN, Newman JS, Reitman C, Rosenberg ZS, Shah NA, Small KM, Weissman BN (2017) ACR appropriateness criteria((R)) osteoporosis and bone mineral density. J Am Coll Radiol 14:S189–S202. https://doi.org/10.1016/j.jacr.2017.02.018
Link TM, Lang TF (2014) Axial QCT: clinical applications and new developments. J Clin Densitom 17:438–448. https://doi.org/10.1016/j.jocd.2014.04.119
Guglielmi G, Floriani I, Torri V, Li J, van Kuijk C, Genant HK, Lang TF (2005) Effect of spinal degenerative changes on volumetric bone mineral density of the central skeleton as measured by quantitative computed tomography. Acta Radiol (Stockholm, Sweden: 1987) 46:269–275
Zaidi Q, Danisa OA, Cheng W (2019) Measurement techniques and utility of hounsfield unit values for assessment of bone quality prior to spinal instrumentation: a review of current literature. Spine 44:E239–E244. https://doi.org/10.1097/BRS.0000000000002813
Choi MK, Kim SM, Lim JK (2016) Diagnostic efficacy of Hounsfield units in spine CT for the assessment of real bone mineral density of degenerative spine: correlation study between T-scores determined by DEXA scan and Hounsfield units from CT. Acta Neurochir 158:1421–1427. https://doi.org/10.1007/s00701-016-2821-5
Perrier-Cornet J, Omorou AY, Fauny M, Loeuille D, Chary-Valckenaere I (2019) Opportunistic screening for osteoporosis using thoraco-abdomino-pelvic CT-scan assessing the vertebral density in rheumatoid arthritis patients. Osteoporos Int J established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 30:1215–1222. https://doi.org/10.1007/s00198-019-04931-w
Mao SS, Luo Y, Fischer H, Buodff MJ, Li D (2016) Routine coronary calcium scan can precisely measure vertebral bone density without a quantitative calibration phantom. J Comput Assist Tomogr 40:126–130. https://doi.org/10.1097/rct.0000000000000330
Garner HW, Paturzo MM, Gaudier G, Pickhardt PJ, Wessell DE (2017) Variation in attenuation in L1 trabecular bone at different tube voltages: caution is warranted when screening for osteoporosis with the use of opportunistic CT. AJR Am J Roentgenol 208:165–170. https://doi.org/10.2214/ajr.16.16744
Sakai Y, Takenaka S, Matsuo Y, Fujiwara H, Honda H, Makino T, Kaito T (2018) Hounsfield unit of screw trajectory as a predictor of pedicle screw loosening after single level lumbar interbody fusion. J Orthop Sci Off J Jpn Orthop Assoc 23:734–738. https://doi.org/10.1016/j.jos.2018.04.006
Salzmann SN, Fantini GA, Okano I, Sama AA, Hughes AP, Girardi FP (2019) Mini-open access for lateral lumbar interbody fusion: indications, technique, and outcomes. JBJS Essent Surg Tech 9:e37. https://doi.org/10.2106/jbjs.St.19.00013
Brown JK, Timm W, Bodeen G, Chason A, Perry M, Vernacchia F, DeJournett R (2017) Asynchronously calibrated quantitative bone densitometry. J Clin Densitom 20:216–225. https://doi.org/10.1016/j.jocd.2015.11.001
Shepherd JA, Schousboe JT, Broy SB, Engelke K, Leslie WD (2015) Executive summary of the 2015 ISCD position development conference on advanced measures from DXA and QCT: fracture prediction beyond BMD. J Clin Densitom 18:274–286. https://doi.org/10.1016/j.jocd.2015.06.013
Salzmann SN, Shirahata T, Yang J, Miller CO, Carlson BB, Rentenberger C, Carrino JA, Shue J, Sama AA, Cammisa FP, Girardi FP, Hughes AP (2019) Regional bone mineral density differences measured by quantitative computed tomography: Does the standard clinically used L1-L2 average correlate with the entire lumbosacral spine? Spine J Off J N Am Spine Soc 19:695–702. https://doi.org/10.1016/j.spinee.2018.10.007
Okano I, Salzmann SN, Jones C, Ortiz Miller C, Shirahata T, Rentenberger C, Shue J, Carrino JA, Sama AA, Cammisa FP, Girardi FP, Hughes AP (2019) The impact of degenerative disc disease on regional volumetric bone mineral density (vBMD) measured by quantitative computed tomography. Spine J Off J N Am Spine Soc. https://doi.org/10.1016/j.spinee.2019.02.017
American College of Radiologist (2018) ACR–SPR–SSR practice parameter for the performance of musculoskeletal quantitative computed tomography (QCT). https://www.acr.org/-/media/ACR/Files/Practice-Parameters/QCT.pdf. Accessed 14 Nov 2018
Ishikawa K, Toyone T, Shirahata T, Kudo Y, Matsuoka A, Maruyama H, Hayakawa C, Tani S, Sekimizu M, Tsuchiya K, Eguro T, Oshita Y, Ozawa T, Nakao Y, Sano S, Nagai T, Kanzaki K, Inagaki K (2018) A novel method for the prediction of the pedicle screw stability: regional bone mineral density around the screw. Clin Spine Surg. https://doi.org/10.1097/bsd.0000000000000703
Gerety EL, Hopper MA, Bearcroft PW (2017) The reliability of measuring the density of the L1 vertebral body on CT imaging as a predictor of bone mineral density. Clin Radiol 72:177 e179-177 e115. https://doi.org/10.1016/j.crad.2016.09.022
Satake K, Kanemura T, Nakashima H, Yamaguchi H, Segi N, Ouchida J (2017) Cage subsidence in lateral interbody fusion with transpsoas approach: intraoperative endplate injury or late-onset settling. Spine Surg Relat Res 1:203–210. https://doi.org/10.22603/ssrr.1.2017-0004
Funding
No funds were received in support of this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
IO, CJ, MJR, OCS, CR, and JS report that they have no potential conflict of interest. JAC reports consulting fee from Pfizer, Inc., Covera, IAG, Image Biopsy Lab, and Simplify Medical, membership of scientific advisory board/other office of IAG, outside the submitted work. AAS reports royalties from Ortho Development Corp., stock ownership of Paradigm Spine, LLC, Spinal Kinetics, Inc., Vestia Ventures MiRus Investment, LLC, and Integrity Implants, consulting fee from Clariance, Inc., Kuros Biosciences AG, Ortho Development Corp., DePuy Synthes Products, Inc., Medical Device Business Services, Inc., and 4WEB, Inc., membership of scientific advisory board/other office of Clariance, Inc., Kuros Biosciences AG, DePuy Synthes Products, Inc., and Medical Device Business Services, Inc, research support from Spinal Kinetics, Inc., and MiMedx Group, Inc., fellowship support from AOSpine North America, outside the submitted work. FPC reports royalties from NuVasive, Inc., stock ownership of VBVP VI, LLC (originally Centinel Spine), private investments for Spinal Kinetics, Inc., Ivy Healthcare Capital Partners, LLC, ISPH II, LLC, Vertical Spine, LLC, Bonovo Orthopedics, Inc., Viscogliosi Brothers, LLC, Liventa Bioscience (fka AF Cell Medical), Paradigm Spine, LLC, Tissue Differentiation Intelligence, LLC, Woven Orthopedic Technologies, Orthobond Corporation, and Healthpoint Capital Partners, LP, consulting fees from Vertical Spine, LLC, and 4WEB Medical, membership of scientific advisory board/other office of Spinal Kinetics, Inc., Paradigm Spine, LLC, Woven Orthopedic Technologies, Orthobond Corporation, and Healthpoint Capital Partners, LP, research support from 4WEB Medical, NuVasive, Inc., Mallinckrodt Pharmaceuticals, Pfizer, Inc., Spinal Kinetics, Inc., Centinel Spine, Inc. (fka Raymedica, LLC), Beatrice & Samuel A. Seaver Foundation, Paradigm Spine, LLC, 7D Surgical, Inc., Woven Orthopedic Technologies, and Depuy Synthes, outside the submitted work. FPG reports royalties from NuVasive, Inc., Ortho Development Corp, and Zimmer Biomet Holdings, Inc., stock ownership of Bonovo Orthopedics, Inc., Liventa Bioscience (fka AF Cell Medical), Paradigm Spine, LLC, Tissue Differentiation Intelligence, LLC, Alphatec Holdings, LLC, LANX, Inc., Healthpoint Capital Partners, LP, Centinel Spine, Inc. (fka Raymedica, LLC), and Spinal Kinetics, Inc., consulting fee from DePuy Synthes Spine, NuVasive, Inc., EIT Emerging Implant Technologies, Spineart USA, Inc., and Ethicon, Inc., grants from NuVasive, Inc., outside the submitted work. APH reports research support from NuVasive, Inc., 4WEB Medical, and Pfizer, Inc., outside the submitted work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Okano, I., Jones, C., Salzmann, S.N. et al. Endplate volumetric bone mineral density measured by quantitative computed tomography as a novel predictive measure of severe cage subsidence after standalone lateral lumbar fusion. Eur Spine J 29, 1131–1140 (2020). https://doi.org/10.1007/s00586-020-06348-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-020-06348-0