Abstract
Purpose
It is an open question whether cell transplantation can provide safety and effective outcome to spinal cord injury (SCI) patient which has remained controversial for almost 40 years. This study aimed to evaluate the safety and efficacy of cell transplantation in SCI patients.
Method
Studies of the cell transplantation for SCI were retrieved from PubMed, Embase, Medline, Cochrane Library and analyzed quantitative data by Review Manager 5.3.
Results
Twenty-one clinical controlled studies with 973 patients were included. The pooled results suggested that cell transplantation significantly improved ASIA score, ASIA motor score, ASIA sensory score, Barthel Index score, residual urine volume, rehabilitative time of automatic micturition. Furthermore, subgroup analysis indicated that the stem cells exhibited more potent than the non-stem cells in spinal cord repair. Cell transplantation at more than 14 days after injury showed more significant improvements than that within 14 days from injury. The dosage of cell transplantation between 1–5 × 107 and 10–20 × 107 was the potent quantity for the patient with SCI. Intrathecal injection and intravenous + intrathecal injection showed more superior to the other method. The top 5 adverse events were febrile reaction (11.5%), neurologic pain (11.3%), headache (2.6%), neurologic deterioration (2.4%), and rigidity or spasticity (1.6%).
Conclusion
Cell transplantation appears to be a safe therapeutic strategy possessing substantial beneficial effects in the patients with SCI in clinic. Moreover, treating SCI with stem cell, the dosage of cells between 1–5 × 107 and 10–20 × 107, in intermediate or chronic phase, minimally invasive techniques, may bring more advantage to SCI patient.
Graphical abstract
These slides can be retrieved under Electronic Supplementary Material.

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spinal cord injury as a grievous neurological disease leads to paraplegia or tetraplegia with high rate of mortality and disability that mainly due to multifaceted complications including infections in bladder, renal failure, and cardiac and respiratory dysfunctions [1]. Based on the epidemiological survey, the annual incidence of SCI in North America is estimated to be between 24 and 77 per million people, or approximately 12,000 to 20,000 new cases per year. In addition, the total annual cost attributed to SCI in North America is about $14.5 billion, bringing a heavy burden to society in terms of healthcare costs [2, 3]. Those patients who suffer SCI will endure 4 misfortune phases based on pathophysiological classification system: acute (< 48 h), subacute (48 h to 14 days), intermediate (14 days to 6 months), and chronic (> 6 months). It is well known that the neurological impairment caused by primary injury (top 3 leading causes are vehicle accidents, violence, and accidental falls) is ineluctable; therefore, the rationale under current therapeutic strategy on SCI is acute-phase intervention preventing neural tissue from subsequent harmful cascade caused by the second injury and expecting vital impact on long-term function recovery [4].
Methylprednisolone (MPSS) as an initial therapy for SCI has been applied in clinic for more than 30 years. Recommendation developed by AOSpine expert panel in 2017 suggests a 24-h infusion of MPSS be offered to patients within 8 h of acute SCI as a treatment option. Although related corticosteroid clinical trials attract extreme criticisms over wound infection, gastrointestinal hemorrhages, sepsis, pulmonary embolism, severe pneumonia, and even death, MPSS until now is still an expedient strategy in the treatment of SCI [5,6,7]. With the understanding of SCI based on increasing clinical and basic researches, a plenty of key neuroprotective trials about pharmacological therapy (riluzole, magnesium, minocycline, GM-1, fibroblast growth factor, granulocyte colony-stimulating factor, hepatocyte growth factor) [8,9,10] and non-pharmacologic therapy (therapeutic hypothermia, CSF drainage) are underway. Furthermore, in terms of neuroregeneration, the specific clinical result of both drug therapeutics (Rho–ROCK inhibitor, anti-NOGO antibody) and nondrug therapeutics (spinal cord stimulation) are still in pending. To date, there are no standard and efficient treatments confidently available for SCI patients [11, 12].
Cell transplantation as a promising regimen is deemed to bring neural functional benefits following SCI. Candidate cell types including adipose-derived stem cells (ADSCs) [13, 14], bone mesenchymal stem cells (BMSCs), marrow mononuclear cells (MMCs), umbilical cord-derived mesenchymal stem cells (UCMSCs), olfactory ensheathing cells (OECs), autologous incubated macrophage (AIM), schwann cells (SCs), olfactory lamina propria (OLP), and fetal brain-derived neural stem/progenitor cell (FB-DNS/PCs) [15, 16] have an aptitude for providing trophic support, modulating the inflammatory response, regenerating lost neural circuits, and remyelinating denuded axons. Most basic researches and clinical trials also demonstrate that various cell types are generally feasible, but there still remains controversy exists due to significant heterogeneity, leaving us indeterminacy whether cell remedy possesses inherent merits in ameliorating patient’s prognosis. After retrieving and reviewing the literature, especially in systematic review and meta-analysis, there are 11 articles including 3 clinical (1 BMSC, 1 OEC, 1 stem cell) [17, 18] and 8 animal experiments (2 OEC, 1 BMSC, 3 stem cells, 1 SC, 1 NS/PC) [19,20,21,22,23,24,25,26,27]. However, most of the studies centering on a unitary cell type or self-control failure to provide us the comprehensive and detailed understanding of cell therapy, for example, whether different cell lines generate equivalent clinical outcome, what is the optimum time for cellular therapy, how many cells are suitable for the patient. Taken together, we aimed to probe into the issue mentioned above and provide evidence-based guidance for surgeons to make a better clinical decision by a meta-analysis.
Methods and materials
Search strategy
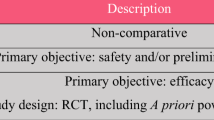
To perform comprehensive retrieval strategy, we systematically searched relevant studies published in the electronic database including PubMed, Embase, Medline, Cochrane Library. Search terms were subjected to the following: “cell,” “spinal cord injury”, and “trial” with the Boolean logic operator “AND,” “NOT,” and “OR”. All studies were published before January 2018 without language and country restriction. Reference cited in the relevant literature and other articles in the meta-analysis were also reviewed (Table 1).
Inclusion and exclusion criteria
The criteria for inclusion article were: (1) clinical trials; (2) containing control group; (3) that all experimental groups were treated with cell transplantation. The criteria for exclusion article were: (1) animal and cell experiment; (2) self-control study; (3) that experimental groups were treated without the cell. Two authors assessed the potentially eligible studies independently based on titles, abstracts, and full texts of the relevant references according to inclusion criteria and exclusion criteria. Any disagreement was discussed and resolved by a third independent author.
Data extraction
Study titles and abstracts were reviewed by two independent investigators to decide if they satisfied the inclusion criteria, and the full text of the included studies was searched for further analysis. The data were extracted by two authors independently using a purpose-designed form: first author and year, region, design, level, number of patients, gender, age, injury, treatment, cell types, cell numbers, duration, follow-up, and outcome. Seven quantitative data were extracted as follows: (1) ASIA score (2) ASIA motion score (3) ASIA sensory score (4) Barthel Index score (5) Residual urine volume (6) Rehabilitative time of automatic micturition. Disagreement between the two reviewers was settled by the third reviewer. If any disagreements existed, a third author was consulted for discussion until consensus was reached.
Quality assessment
The quality of the RCT and CCT studies was independently assessed by the two authors according to a six-item scale recommended by the Cochrane Back Review Group. A: random sequence generation; B: allocation concealment; C: blinding of outcome assessment; D: incomplete outcome data; E: selective reporting; F: other bias. Furthermore, the CCT studies were assessed according to MINORS scale. Every study was assessed by 2 independent researchers and judgment of every item. Any disagreement with respect to eligibility during the extraction was discussed and resolved.
Outcome indictor
In the present meta-analysis, outcome indicators are included: (1) ASIA score (2) ASIA motion score (3) ASIA sensory score (4) Barthel Index score (5) residual urine volume (6) rehabilitative time of automatic micturition (7) cell transplantation techniques (8) adverse events.
Statistical analysis
The risk ratio (RR) and the corresponding 95% confidence interval (CI) were used for the dichotomous outcomes, and the standardized mean difference (SMD) and 95% CI were assessed for the continuous outcomes. The Chi-square test and Higgins’s I2 test were used to evaluate the heterogeneity. A P value < 0.1 for the Chi-square test or I2 values exceeding 50% indicated substantial heterogeneity. A fixed-effect model was used if significant statistical heterogeneity was absent; otherwise, a random-effect model was applied. We used Review Manager Software (RevMan Version 5.3, The Cochrane Collaboration, Copenhagen, Denmark) to conduct the statistical analysis. Subgroup analyses were performed with stratification according to cell types, phase, and the different dosage of cells.
Cell types were performed by dividing research into stem cell and non-stem cell group; the time from injury to cell transplantation is < 14 days, 14–30 days, and > 30 days; the different dosage of cells is < 1 × 107, 1–5 × 107, 10–20 × 107, and > 20 × 107.
Results
Selection of studies
Flow chart for the inclusion of studies is shown in Fig. 1. The literature search initially yielded 6389 relevant trials from PubMed (N = 110), Embase (N = 157), Medline (N = 5260), the Cochrane Library (N = 2), Wanfang (N = 99), CNKI (N = 35), and SinoMed (N = 726). After we reviewed the titles and abstracts of all 6389 articles, 5914 trials were excluded. We continued to refine the 475 candidate studies under provident review. Then, 137 duplicate studies were excluded. 338 candidate studies remain to confirm further. Because of nonconformance with inclusion criteria, 316 articles were excluded. Finally, 22 studies with 964 patients were included (Fig. 1). We recorded the characteristics of the 22 included trials, as well as the details of the clinical outcome measurement (Tables 2 and 3).
Characteristics of the studies included in the meta-analysis
Characteristics of the included studies were given in Tables 2 and 3, including the following item.
Methodological study quality assessment
A summary of methodological domain assessment for each study is detailed in Fig. 2 and Table 4.
ASIA score
Pooled analysis of 15 studies [29,30,31,32,33,34, 38,39,40,41, 44, 46,47,48,49] indicated that cell group improved ASIA score significantly compared with control group. The overall effect of ASIA score was 2.54 (95% CI 1.74–3.71, P < 0.00001); because of low heterogeneity (P = 0.06, I2= 39%), a fixed model was used (Fig. 3). Then, subgroup analysis according to cell types, phase, and the number of the cells was performed to further explore the more valuable details and potential sources of heterogeneity. Although heterogeneity was existed within several subgroups, low-to-moderate heterogeneity within every trial is still considered valid.
In cell-type subgroup analysis, studies were divided into 2 groups, 12 for stem cell and 3 for non-stem cell. The ASIA score of stem cell group was 3.36 (95% CI 2.13–5.28, P < 0.00001) with heterogeneity (P = 0.60, I2= 0%), and non-stem cell group was 0.82 (95% CI 0.39–1.73, P = 0.61) with heterogeneity (P = 0.13, I2 = 51%). Interestingly, the result indicated that cell from different sources showed opposite result, stem cell appeared to be more superior to non-stem cell in meliorating ASIA score (Table 5).
Furthermore, the optimal time of intervention for the patient after SCI is a vital issue and still perplexed surgeon to date. Based on the interval time from injury to cell transplantation, the studies were divided into three groups, two for < 14 days group, 1 for 14–30 days group, and 12 for > 30 days group. The ASIA score of < 14 days group was 0.75 (95% CI 0.35–1.64, P = 0.48) with heterogeneity (P = 0.10, I2 = 63%), 14–30 days group was 3.03 (95% CI 0.89–10.34, P = 0.08), and > 30 days group was 3.47 (95% CI 2.14–5.63, P < 0.00001) with heterogeneity (P = 0.57, I2 = 0%). The result indicated that cell transplantation in the chronic phase is more suitable for the patient with SCI. However, in acute and subacute phase, cell therapy does not exhibit efficiency in treatment for SCI. Although only three studies were classified in the acute and subacute group, it could still provide a referable standard for doctors and researchers. The heterogeneity from the subgroup was not high or severe, and the result can be accepted (Table 5).
Cell number subgroup analysis: 3 for < 1 × 107 group, 5 for 1–5 × 107 group, 3 for 10–20 × 107 group, and 3 for > 20 × 107 group. The cell number subgroup of < 1 × 107 was 1.11 (95% CI 0.60–2.07, P = 0.73) with heterogeneity (P = 0.02, I2 = 74%), 1–5 × 107 group was 4.20 (95% CI 1.87–8.95, P = 0.0002) with heterogeneity (P = 0.24, I2 = 27%), 10–20 × 107 group was 5.61 (95% CI 1.57–19.99, P = 0.008) with heterogeneity (P = 0.57, I2 = 0%), and > 20 × 107 group was 1. 98 (95% CI 0.97–4.06, P = 0.06) with heterogeneity (P = 0.75, I2 = 0%). Notably, different from our previous experience that more cells may present better efficacy, the result showed that appropriate amount of cell numbers between 1–5 × 107 and 10–20 × 107 was preferable quantity for patient with SCI comparing with other two cell number groups (Table 5).
The option of cell transplantation techniques is vital question for surgeon and patient. Subgroup analysis: 6 for posterior laminectomy + intraspinal cord cell injection group, 2 for intrathecal injection + intravenous injection group, and 5 for intrathecal injection group. The transplantation techniques subgroup of posterior laminectomy + intraspinal cord cell injection was 2.59 (95% CI 0.65–10.30, P = 0.18) with heterogeneity (P = 0.02, I2 = 64%), intrathecal injection + intravenous injection subgroup was 1.65 (95% CI 0.73–3.74, P = 0.23) with heterogeneity (P = 0.69, I2 = 0%), and intrathecal injection subgroup was 2.84 (95% CI 1.46–5.50, P = 0.002) with heterogeneity (P = 0.35, I2 = 9%). Notably, different from our previous experience that more cells may present better efficacy, the result showed that appropriate amount of cell numbers between 1–5 × 107 and 10–20 × 107 was preferable quantity for patient with SCI comparing with other two cell number group. The results showed that intrathecal injection appeared to be more superior to other two cell transplantation techniques (Table 5).
ASIA motion score
Pooled analysis of the 18 studies [28, 30,31,32,33, 35,36,37,38,39, 41,42,43,44,45,46,47,48] indicated that cellular treatment group significantly improved ASIA motion score compared with control group with heterogeneity (P < 0.00001, I2 = 76%). The overall change of ASIA motion score was 0.62 (95% CI 0.32–0.92, P < 0.0001) (Fig. 4). The overall effect exhibits that cell therapy improves the motion function comparing with control group. Subgroup analyses were conducted as mentioned above.
Cell-type subgroup analysis: 16 for stem cell and 2 for non-stem cell. The motion score of stem cell group was 0.69 (95% CI 0.38–0.99, P < 0.00001) with heterogeneity (P < 0.00001, I2 = 75%), and non-stem cell group was − 0.28 (95% CI − 0.89 to 0.33, P = 0.37) with heterogeneity (P = 0.31, I2 = 5%). The result consistent with ASIA score demonstrated that stem cell was superior to non-stem cell in motion improvement (Table 5).
Phase subgroup analysis: 2 for < 14 days group, 1 for 14–30 days group, and 15 for > 30 days group. The motion score of < 14 days group was 0.35 (95% CI −1.10 to 1.79, P = 0.64) with heterogeneity (P = 0.0003, I2 = 92%), 14–30 days group was 0.88 (95% CI 0.42–1.35), and > 30 days group was 0.64 (95% CI 0.30–0.98, P = 0.0002) with heterogeneity (P < 0.00001, I2 = 74%). The result showed that cell remedy in intermediate and chronic phase was superior to control group. However, in acute and subacute phase, there was no statistically difference comparing with control group (Table 5).
Cell number subgroup analysis: 4 for < 1 × 107 group, 6 for 1–5 × 107 group, 4 for 10–20 × 107 group, and 3 for > 20 × 107 group. The cell number subgroup of < 1 × 107 was 0.26 (95% CI − 0.36 to 0.88, P = 0.41) with heterogeneity (P = 0.008, I2 = 75%), 1–5 × 107 group was 0.64 (95% CI 0.11–1.16, P = 0.02) with heterogeneity (P = 0.0008, I2 = 76%), 10–20 × 107 group was 0.87 (95% CI 0.53–1.21, P < 0.00001) with heterogeneity (P = 0.32, I2 = 14%), and > 20 × 107 was 0.15 (95% CI − 0.22 to 0.52, P = 0.44) with heterogeneity (P = 0.57, I2 = 0%). The result identical to ASIA score showed that the interval of cell number between 1–5 × 107 and 10–20 × 107 demonstrated a better efficacy in treating SCI comparing with < 1 × 107 and > 20 × 107 group (Table 5).
In cell transplantation techniques subgroup analysis, the studies were divided into 3 groups, 4 for posterior laminectomy + intraspinal cord cell injection, 5 for intravenous injection + intrathecal injection, and 7 for intrathecal injection. The Motion score of posterior laminectomy + intraspinal cord cell injection group was 0.59 (95% CI − 0.31 to 1.49, P = 0. 20) with heterogeneity (P = 0.002, I2 = 80%), intravenous injection + intrathecal injection group was 0.87 (95% CI 0.14–1.60, P = 0.02) with heterogeneity (P = 0.0003, I2 = 81%), and intrathecal injection group was 0.46 (95% CI 0.06–0.86, P = 0.02) with heterogeneity (P < 0.006, I2 = 67%). The overall results showed similar to the results of motion that intrathecal injection and intravenous injection + intrathecal injection exhibited more favorable outcomes than posterior laminectomy + intraspinal cord cell injection (Table 5).
ASIA sensory score
Pooled analysis of the 18 studies [28, 30,31,32,33, 35,36,37,38,39, 41,42,43,44,45,46,47,48] indicated that cell transplantation group significantly improved ASIA sensory score compared with control group with heterogeneity (P < 0.00001, I2 = 81%). The overall change of ASIA sensory score was 0.77 (95% CI 0.42–1.12, P < 0.0001) (Fig. 5). The overall effect exhibited that cellular therapy was able to improve the sensory function of patients with SCI. Subgroup analysis was also performed.
In cell-type subgroup analysis, the studies were divided into 2 groups, 16 for stem cell and 2 for non-stem cell. The sensory score of stem cell group was 0.87 (95% CI 0.54–1.19, P < 0.00001) with heterogeneity (P < 0.00001, I2= 77%), and non-stem cell group was -0.30 (95% CI − 1.51 to 0.90, P = 0.62) with heterogeneity (P = 0.15, I2 = 52%). The result also indicated that stem cell is superior to non-stem cell in sensory improvement similar to ASIA score and motion score (Table 5).
Phase subgroup analysis: 2 for < 14 days group, 1 for 14–30 days group, and 15 for > 30 days group. The sensory score of < 14 days group was 0.41 (95% CI −1.80 to 2.62, P = 0.72) with heterogeneity (P < 0.00001, I2 = 97%), 14–30 days group was 1.15 (95% CI 0.67 to 1.63, P < 0.00001), and > 30 days group was 0.79 (95% CI 0.44–1.13, P < 0.00001) with heterogeneity (P < 0.00001, I2 = 75%). The result showed that cell transplantation in intermediate or chronic phase was apt at treating SCI. However, control group was non-inferior to cell therapy in acute and subacute phase (Table 5).
Cell number subgroup analysis: 4 for < 1 × 107 group, 6 for 1–5 × 107 group, 4 for 10–20 × 107 group, and 3 for > 20 × 107 group. The cell number subgroup of < 1 × 107 was 0.39 (95% CI − 0.48 to 1.27, P = 0.38) with heterogeneity (P < 0.0001, I2 = 87%), 1–5 × 107 group was 0.71 (95% CI 0.22–1.19, P = 0.004) with heterogeneity (P = 0.004, I2 = 71%), 10–20 × 107 group was 1.62 (95% CI 1.18–2.07, P < 0.00001) with heterogeneity (P = 0.18, I2 = 38%), and > 20 × 107 was 0.08 (95% CI − 0.29–0.45, P = 0.65) with heterogeneity (P = 0.93, I2 = 0%). The result showed that the interval of cell number between 1–5 × 107 and 10–20 × 107 was superior quantity for treating SCI comparing with < 1 × 107 and > 20 × 107 group; this result was equal to ASIA score and motion score (Table 5).
In cell transplantation techniques subgroup analysis, the studies were divided into 3 groups, 4 for posterior laminectomy + intraspinal cord cell injection, 5 for intravenous injection + intrathecal injection, and 7 for intrathecal injection. The sensation score of posterior laminectomy + intraspinal cord cell injection group was 0.67 (95% CI − 0.59 to 1.93, P = 0. 3) with heterogeneity (P < 0.00001, I2 = 89%), intravenous injection + intrathecal injection group was 0.75 (95% CI 0.27–1.23, P = 0.002) with heterogeneity (P = 0.05, I2 = 59%), and intrathecal injection group was 0.92 (95% CI 0.24–1.61, P = 0.008) with heterogeneity (P < 0.0001, I2 = 87%). The overall results showed that intrathecal injection and intravenous injection + intrathecal injection exhibited more favorable outcomes than posterior laminectomy + intraspinal cord cell injection (Table 5).
Barthel Index
Barthel Index was widely used for assessing the activities of daily living. The overall effect of the 8 studies [28, 30, 31, 36, 37, 41, 43, 48] showed that cell therapy group significantly improved Barthel Index score compared with control group with heterogeneity (P = 0.31, I2 = 15%). The overall change of Barthel Index score was 0.88 (95% CI 0.67–1.09, P < 0.00001) (Fig. 6). The result showed that cell therapy recuperated activity of daily living compared with control group. Potential sources of heterogeneity were explored by subgroup analysis in terms of cell types, phase, and the number of the cell.
Cell-type subgroup analysis: all 8 for stem cell. The Barthel Index score of stem cell group was 0.88 (95% CI 0.67–1.09, P < 0.00001) with heterogeneity (P = 0.31, I2= 15%). The result showed that stem cell is able to improve the activity of daily living (Table 5).
In the phase subgroup analysis, only > 30 days group was classified because of lacking acute- and subacute-phase researches. The Barthel Index score of > 30 days group was 0.88 (95% CI 0.67–1.09, P < 0.00001) with heterogeneity (P = 0.31, I2 = 15%). The result showed that cell treatment in intermediate and chronic phase may be a right opportunity in treating SCI. (Table 5).
Cell number subgroup analysis: 1 for < 1 × 107 group, other 3 groups were categorized in 3 for 1–5 × 107 group, 1 for 10–20 × 107 group, and 2 for > 20 × 107 group. The cell number subgroup of < 1 × 107 group was 0.58 (95% CI 0.13–1.03, P = 0.01), 1–5 × 107 group was 1.04 (95% CI 0.73–1.35, P < 0.00001) with heterogeneity (P = 0.23, I2 = 33%), 10–20 × 107 group was 0.61 (95% CI − 0.21 to 1.43, P = 0.15), and > 20 × 107 was 0.71 (95% CI 0.17–1.24, P = 0.010) with heterogeneity (P = 0.50, I2 = 0%). The result showed that similar to ASIA score, sensation and motion score, 1–5 × 107 group can be an appropriate dosage in treating for SCI. However, opposite results in 10–20 × 107 and > 20 × 107 were likely on account of insufficient data and other bias. More studies on SCI were needed for assessing (Table 5).
Residual urinal
Pooled analysis of the 4 studies [30, 31, 39, 43], all of which were for stem cell, demonstrated that cell therapy group significantly improved residual urinal compared with control group with heterogeneity (P = 0.24, I2 = 29%). The overall change of residual urinal was 1.00 (95% CI 0.61–1.38, P < 0.00001) (Fig. 7). The results indicate that cellular therapy is able to improve bladder function in patients with SCI (Table 5).
Rehabilitative time of automatic micturition
Overall analysis of the 3 studies [30, 31, 33], all of which were for stem cell, indicated that cell transplantation group significantly improved days of the rehabilitative time of automatic micturition compared with control group with heterogeneity (P = 0.73, I2 = 0%). The overall change of days of automatic urine was -0.67 (95%CI − 1.10 to − 0.23, P = 0.002) (Fig. 8). The result exhibited that stem cell remedy improved the rehabilitative time of automatic micturition (Table 5).
Adverse events
We evaluated the adverse event in cell transplantation group including 13 items: mortality, sepsis or wound infection, meningeal irritation, cardiopulmonary events, neurologic pain, neurologic deterioration, tumor, urinary system event, febrile reaction, headache, constipation, rigidity or spasticity, and abdominal distension. The top 5 adverse events were febrile reaction (11.5%), neurologic pain (11.3%), headache (2.6%), neurologic deterioration (2.4%), and rigidity or spasticity (1.6%). The results were given in (Table 6).
Discussion
We have conducted an overall and systematic review of controlled clinical studies on the efficacy and safety of cell transplantation for the patient with SCI. Our results demonstrated that cell transplantation might be a meritorious method for SCI. Remarkably, in subgroup analyses, the stem cell makes a better performance than the non-stem cell in ASIA score, motion, and sensation score. Moreover, stem cell showed more efficacy than control group in Barthel Index, residental urinal, and rehabilitative time of automatic micturition. In the aspect of opportunity for delivering, > 30 day was a preferable stage in treating SCI. Paralleled results were observed in ASIA score, motion, and sensation score, Barthel Index. Than, results in ASIA score, motion, and sensation score convinced us to consider that the interval of cell dosage between 1–5 × 107 and 10–20 × 107 was the potent quantity for the patient with SCI comparing with < 1 × 107 and > 20 × 107 groups, as well as implied that both low and high dosages might gain no benefits for the patient with SCI. Furthermore, although all above results exhibited that intrathecal injection and intravenous injection + intrathecal injection showed more superior to posterior laminectomy + intraspinal cord cell injection, we noticed that the high heterogeneity existed in forest plot. So, a sensitivity analysis was also conducted, in which 1 study at a time was removed and the others analyzed to estimate whether the results could have been affected markedly by a single study. We found some interesting results. When we removed “Lammertse DP et al. 2012,” the results were reversed in posterior laminectomy + intraspinal cord cell injection group and then showed more superior to other 2 techniques (Supplements 13–15). Furthermore, similar reversed results were observed in sensation and motion sensitivity analysis group. We further analyzed the reason why “Lammertse DP et al. 2012” article made total reversed. First, cell-type-autologous incubated macrophage. Second, complete spinal cord injury. Thirdly, the conclusion of Lammertse DP`s research is the results failed to show a significant difference between the autologous incubated macrophages and control groups. Although forest plot exhibited relatively fine trend results in 3 cell transplantation techniques, we were inclined to choose minimally invasive techniques, which based on 3 reasons (1) the overall condition of patients with SCI (2) the patient`s tolerance to posterior laminectomy + intraspinal cord cell injection surgery, and (3) the patient`s capability of recovery after posterior laminectomy + intraspinal cord cell injection surgery. In terms of safety, although there is no standard definition for “safety,” in this article, we considered “safety” as (1) mild symptoms recovered within few days via treatment or self-healing, (2) or very low serious adverse events rate (such as mortality, infection) after receiving cell transplantation (3) or very low adverse events rate in long-term follow-up. Cell transplantation did not occur to serious adverse events; we found that although a higher degree of febrile reaction (11.5%) and neurological pain (11.3%) given in Table 6, above two symptoms were transient and self-healing in a few days and did not increase the meaningful mortality or morbidity. And then other adverse events such as meningeal irritation, headache, constipation, and rigidity or spasticity were also eliminated by symptomatic treatment or self-healing within few days. Only Nirmeen et al. [32] and Lammertse et al. [38] reported serious adverse events including mortality and infection. Notably, there were few adverse events in long-term follow-up exist in the 22 included studies. Top 5 adverse events were febrile reaction, neurologic pain, headache, neurologic deterioration, and rigidity or spasticity.
Comparing with previously published clinical meta-analysis
There are 3 clinical studies on meta-analysis. Li et al. [17] included 24 studies concerning of 594 patients treated with BMSC transplantation and provided that AIS improvement rate was analyzed in favor of BMSCs 6.13 (95% CI, 3.0–12.51; P < 0.001). The application of cell transplantation numbers between 107 and 108 groups seemed to be more beneficial than 106 and 109 (P < 0.05 for all groups). Our results were similar to conclude that stem cell seemed to show more potent efficacy with cell number of 107 and 108 maybe an optimum choice in clinical. Li et al. [18] applied OECs to treat chronic SCI patient; 11 articles that included 1219 patients were selected for review. Total AIS improvement rate, which was 39.0% (95% CI 28.1–51.1%), and also showed improvement in ASIA motor score and ASIA sensation score. We draw the semblable results in the effect, but lower mortality in our research. Fan et al. [19] included 10 studies comprising 377 patients. Three different origins of stem cell including BMCs, MNCs, and UCMSCs that were in treating for SCI were reviewed; the results showed that stem cell significantly improved AIS grading rate 2.95 (95% CI 1.64–5.29, P = 0.0003) and ameliorated lower-limb light touch score 3.43 (95% CI 0.01–6.86, P = 0.05) and lower-limb pinprick score 3.93 (95% CI 0.74–7.12, P = 0.02). However, it did not significantly improve motor score 1.89 (95% CI − 0.25 to 4.03, P = 0.08) and activities of daily living score 1.12 (95% CI − 1.17 to 4.04, P = 0.45). Intriguingly, our research showed that applying stem cell could bring improvement in motion score and activities of daily living score.
Strengths and limitations
To date, this is the first attempt to summarize the efficacy and safety of cell transplantation therapy for SCI. First, different from the previously published clinical meta-analysis, our up-to-date article retrieval yielded 21 eligible clinical controlled studies. It provided more high-level literature from origin and generated more credible results by evidence-based medicine analysis. Second, we also compared cell types to explore which cell lines bring more merits to the patient and provide researchers and surgeons an overall view of cellular therapy on SCI. Finally, we obtained more detailed information by conducting subgroup analyses to make further understanding in treatment for SCI.
This systematic review had some limitations. First, although we included studies with the controlled group, there are 8 studies without randomized method; this may lower the strength of evidence in our research. Second, some trials did not provide the detailed processes of cell transplantation, cell detection, and cell state, and we did not pursue these data to complete the analysis of the trials. Finally, socio-ethics factors, regional healthcare policy, and the medical condition might influence the outcome of cell transplantation.
Conclusions
Our systematic review with meta-analyses concluded that cell transplantation appears to be safe and has substantial beneficial effects in patients with SCI. Moreover, stem cell remains predominant in the treatment of SCI and provide more efficacy than other cells. Notably, treating SCI with stem cell, the dosage of cells between 1–5 × 107 and 10–20 × 107, in intermediate or chronic phase, may bring more advantage to SCI patients.
Change history
23 June 2020
Zhao-he and Sun-qingling are the co-first authors for this manuscript in the initial submission. Because of author���s negligence and fault, this information was not shown clearly in the originally published article.
Abbreviations
- ADSCs:
-
Adipose-derived stem cells
- BMSCs:
-
Bone mesenchymal stem cell
- MMCs:
-
Marrow mononuclear cells
- UCMSCs:
-
Umbilical cord-derived mesenchymal stem cells
- OECs:
-
Olfactory ensheathing cells
- AIM:
-
Autologous incubated macrophage
- SCs:
-
Schwann cells
- OLP:
-
Olfactory lamina propria
- GM-CSF:
-
Granulocyte-macrophage colony-stimulating factor
- FB-DNS/PCs:
-
Fetal brain-derived neural stem/progenitor cell
- CNS:
-
Central nervous system
References
Silva NA, Sousa N, Reis RL (2014) From basics to clinical: a comprehensive review on spinal cord injury. Prog Neurobiol 114:25–57
Devivo MJ (2012) Epidemiology of traumatic spinal cord injury: trends and future implications. Spinal Cord 50:365–372
Lee BB, Cripps RA, Fitzharris M et al (2011) The global map for traumatic spinal cord injury epidemiology: update, global incidence rate. Spinal Cord 52(2):110–116
Hurlbert RJ, Hadley MN, Walters BC et al (2015) Pharmacological therapy for acute spinal cord injury. Neurosurgery 76:S71–S83
Miller SM (2008) Methylprednisolone in acute spinal cord injury: a tarnished standard. J Neurosurg Anesthesiol 20(2):140–142
Breslin K, Agrawal D (2012) The use of methylprednisolone in acute spinal cord injury: a review of the evidence, controversies, and recommendations. Pediatr Emerg Care 28(11):1238–1245
Ahuja CS, Nori S, Tetreault L et al (2017) Traumatic spinal cord injury-repair and regeneration. Neurosurgery 80(3S):S9–S22
Grossman RG, Fehlings MG, Frankowski RF et al (2014) A prospective, multicenter, phase I matched-comparison group trial of safety, pharmacokinetics, and preliminary efficacy of riluzole in patients with traumatic spinal cord injury. J Neurotrauma 31(3):239–255
Streijger F, Lee JH, Manouchehri N et al (2016) The evaluation of magnesium chloride within a polyethylene glycol formulation in a porcine model of acute spinal cord injury. J Neurotrauma 33(24):2202–2216
Sledge J, Mahadevappa K, Stacey P et al (2016) A speculative pharmaceutical cocktail to treat spinal cord injury. Am J Phys Med Rehabil 95(7):e108–e110
Thuret S, Moon LD, Gage FH (2006) Therapeutic interventions after spinal cord injury. Nat Rev Neurosci 7(8):628–643
Raspa A, Pugliese R, Maleki M et al (2016) Recent therapeutic approaches for spinal cord injury. Biotechnol Bioeng 113(2):253–259
Marino G, Rosso F, Ferdinando P et al (2012) Growth and endothelial differentiation of adipose stem cells on polycaprolactone. J Biomed Mater Res A 100(3):543–548
Barbarisi M, Marino G, Armenia E (2015) Use of polycaprolactone (PCL) as scaffolds for the regeneration of nerve tissue. J Biomed Mater Res A 103(5):1755–1760
Sahni V, Kessler JA (2010) Stem cell therapies for spinal cord injury. Nat Rev Neurol 6(7):363–372
Ruff CA, Wilcox JT, Fehlings MG (2012) Cell-based transplantation strategies to promote plasticity following spinal cord injury. Exp Neurol 235(1):78–90
Li XC, Zhong CF, Deng GB et al (2015) Efficacy and safety of bone marrow-derived cell transplantation for spinal cord injury: a systematic review and meta-analysis of clinical trials. Clin Transpl 29(9):786–795
Li L, Adnan H, Xu B (2015) Effects of transplantation of olfactory ensheathing cells in chronic spinal cord injury: a systematic review and meta-analysis. Eur Spine J 24(5):919–930
Fan X, Wang JZ, Lin XM et al (2017) Stem cell transplantation for spinal cord injury: a meta-analysis of treatment effectiveness and safety. Neural Regen Res 12(5):815–825
Liu J, Chen P, Wang Q et al (2014) Meta analysis of olfactory ensheathing cell transplantation promoting functional recovery of motor nerves in rats with complete spinal cord transection. Neural Regen Res 9(20):1850–1858
Oliveri RS, Bello S, Biering-Sørensen F (2014) Mesenchymal stem cells improve locomotor recovery in traumatic spinal cord injury: systematic review with meta-analyses of rat models. Neurobiol Dis 62:338–353
Kim JH, Shim SR, Doo SW et al (2015) Bladder recovery by stem cell based cell therapy in the bladder dysfunction induced by spinal cord injury: systematic review and meta-analysis. PLoS ONE 10(3):e0113491
Yang L, Ge Y, Tang J (2015) Schwann cells transplantation improves locomotor recovery in rat models with spinal cord injury: a systematic review and meta-analysis. Cell Physiol Biochem 37(6):2171–2182
Yousefifard M, Rahimi-Movaghar V, Nasirinezhad F et al (2016) Neural stem/progenitor cell transplantation for spinal cord injury treatment; A systematic review and meta-analysis. Neuroscience 322:377–397
Chen X, Xue B, Li Y et al (2017) Meta-analysis of stem cell transplantation for reflex hypersensitivity after spinal cord injury. Neuroscience 363:66–75
Watzlawick R, Rind J, Sena ES et al (2016) Olfactory ensheathing cell transplantation in experimental spinal cord injury: effect size and reporting bias of 62 experimental treatments: a systematic review and meta-analysis. PLoS Biol 14(5):e1002468
Antonic A, Sena ES, Lees JS et al (2013) Stem cell transplantation in traumatic spinal cord injury: a systematic review and meta-analysis of animal studies. PLoS Biol 11(12):e1001738
Chernykh ER, Stupak VV, Muradov GM et al (2007) Application of autologous bone marrow stem cells in the therapy of spinal cord injury patients. Bull Exp Biol Med 143(4):543–547
Yoon SH, Shim YS, Park YH et al (2007) Complete spinal cord injury treatment using autologous bone marrow cell transplantation and bone marrow stimulation with granulocyte macrophage-colony stimulating factor: phase I/II clinical trial. Stem Cells 25(8):2066–2073
Xie ZW, Cui GX, Li YZ et al (2007) Curative effect of autologous mesenchymal stem cell transplantation on spinal cord injury. J Clin Rehabil Tissue Eng Res 11(5):1277–1279
Cui GX, Song CZ, Li YZ et al (2009) Clinical study of autologous marrow mononuclear cells transplantation in patients with spinal cord injury. Chin J Rehabil Med 24(13):309–312
Nirmeen A, Gabr H, Hamdy S et al (2010) Case control series of intrathecal autologous bone marrow mesenchymal stem cell therapy for chronic spinal cord injury. Neurorehabil Neural Re 24(8):702–708
Fang ML, Wang MX, Wang YQ et al (2011) Clinical study of autologous mesenchymal stem cell transplantation on spinal cord injury. J Qiqihar Univ Med 32(4):2064–2066
Karamouzian S, Nematollahi-Mahani SN, Nakhaee N et al (2012) Clinical safety and primary efficacy of bone marrow mesenchymal cell transplantation in subacute spinal cord injured patients. Clin Neurol Neurosurg 114(7):935–939
Xiao YL, Zhu JX, Li ZM et al (2012) The therapeutic effects of two different ways of autalogous bone marrow mesenchymal stem cells transplantation for treating early spinal cord injury. Chin J Postgrad Med 35(9):24–28
Guo GH, Shen LF, Li Z (2012) Clinical studies of umbilical cord blood mesenchymal stem cells transplantation on spinal cord injury. Chin J Pract Med 39(9):58–60
Zhang XB, Li JT, Zhao HT et al (2012) Umbilical cord blood mesenchymal stem cells transplantation on spinal cord injury in clinical study. Asia Pac Tradit Med 8:116–117
Lammertse DP, Jones LA, Charlifue SB et al (2012) Autologous incubated macrophage therapy in acute, complete spinal cord injury: results of the phase 2 randomized controlled multicenter trial. Spinal Cord 50(9):661–671
Dai GH, Liu X, Zhang Z et al (2013) Transplantation of autologous bone marrow mesenchymal stem cells in the treatment of complete and chronic cervical spinal cord injury. Brain Res 1533:73–79
Tabakow P, Jarmundowicz W, Czapiga B et al (2013) Transplantation of autologous olfactory ensheathing cells in complete human spinal cord injury. Cell Transpl 22(9):1591–1612
Guo ZS, Qin BY, Dai RX et al (2014) Bone marrow mesenchymal stem cells in the treatment of spinal cord injury. Chin J Exp Surg 32(7):2605–2607
Xiao YL, Li ZM, Zhu JX et al (2014) Efficacy observation of autologous bone marrow-derived mesenchymal stem cell therapy on early spinal cord injury. Chin J Biomed Eng 20(1):7–11
Cheng HB, Liu X, Hua R et al (2014) Clinical observation of umbilical cord mesenchymal stem cell transplantation in treatment for sequelae of thoracolumbar spinal cord injury. J Transl Med 12:253
El-Kheir WA, Gabr H, Awad MR et al (2014) Autologous bone marrow-derived cell therapy combined with physical therapy induces functional improvement in chronic spinal cord injury patients. Cell Transpl 23(6):729–745
Chen L, Huang H, Xi H et al (2014) A prospective randomized double-blind clinical trial using a combination of olfactory ensheathing cells and Schwann cells for the treatment of chronic complete spinal cord injuries. Cell Transpl 23(1):S35–S44
Shin JC, Kim KN, Yoo J et al (2015) Clinical trial of human fetal brain-derived neural stem/progenitor cell transplantation in patients with traumatic cervical spinal cord injury. Neural Plast 2015:630932
Zhang Z, Dai GH, Liu XB et al (2015) Umbilical cord mesenchymal stem cell transplantation for spinal cord injury. J Chin Pract Diagn Ther 29(3):478–480
Zhang T, Tong X, Liu C (2015) Efficacy observation of autologous bone marrow-derived mesenchymal stem cell therapy on chronic spinal cord injury. Word New Inf 15:65–66
Wang S, Lu J, Li YA et al (2016) Autologous olfactory lamina propria transplantation for chronic spinal cord injury: three-year follow-up outcomes from a prospective double-blinded clinical trial. Cell Transpl 25(1):141–157
Acknowledgments
Thank you so much for contributions made by all members of Pro. Xiu-Mei Wang`s group (School of Materials Science and Engineering, Tsinghua University). Thank you so much for contributions made by all members in department of orthopedics III (Dongzhimen Hospital, Beijing University of Chinese Medicine). Thank you so much for contributions made by Dr. Feng-He (China Academy of Chinese Medical Science), Dr. Wan-Jie Gu (Drum Tower, Medical College of Nanjing University), Dr. Yun-Tao Zhao(Aerospace Center Hospital, Peking University).
Funding
This work was funded by the National Twelfth Five-Year Plan for Science and Technology Support (Grant No. 2012BAI18B05).
Author information
Authors and Affiliations
Contributions
HZ, XMW, and XY designed the systematic review. HZ and QLS drafted the protocol, and LJD, YDY, YSG, and DYZ revised the manuscript. YX and HJW will independently screen the potential studies, extract data, assess the risk of bias, and finish data synthesis. JWS and KTY will arbitrate any disagreements during the review. All authors approved the publication of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplement 1
ASIA subgroup stem cell VS. non-stem cell (TIFF 1367 kb)
Supplement 2
ASIA subgroup phase (TIFF 1520 kb)
Supplement 3
ASIA subgroup cell number (TIFF 1630 kb)
Supplement 4
ASIA motion subgroup stem cell VS. non-stem cell (TIFF 1488 kb)
Supplement 5
ASIA motion subgroup phase (TIFF 1618 kb)
Supplement 6
ASIA motion subgroup cell number (TIFF 1723 kb)
Supplement 7
ASIA sensation subgroup stem cell VS. non-stem cell (TIFF 1497 kb)
Supplement 8
ASIA sensation subgroup phase (TIFF 1635 kb)
Supplement 9
ASIA sensation subgroup cell number (TIFF 1742 kb)
Supplement 10
ASIA subgroup cell transplantation techniques (TIFF 1632 kb)
Supplement 11
ASIA motion subgroup cell transplantation techniques (TIFF 2174 kb)
Supplement 12
ASIA sensation subgroup cell transplantation techniques (TIFF 2215 kb)
Supplement 13
Sensitive analysis of cell transplantation techniques in ASIA score (TIFF 1176 kb)
Supplement 14
Sensitive analysis of cell transplantation techniques in ASIA motion (TIFF 1250 kb)
Supplement 15
Sensitive analysis of cell transplantation techniques in ASIA sensation (TIFF 1263 kb)
Supplement 16
Barthal Index subgroup stem cell VS. non-stem cell (TIFF 853 kb)
Supplement 17
Barthal Index subgroup phase (TIFF 855 kb)
Supplement 18
Barthal Index subgroup cell number (TIFF 1211 kb)
Supplement 19
Residental urinal subgroup stem cell VS. non-stem cell (TIFF 615 kb)
Supplement 20
Rehabilitative time of automatic micturition subgroup stem cell VS. non-stem cell (TIFF 678 kb)
Rights and permissions
About this article
Cite this article
Zhao, H., Sun, QL., Duan, LJ. et al. Is cell transplantation a reliable therapeutic strategy for spinal cord injury in clinical practice? A systematic review and meta-analysis from 22 clinical controlled trials. Eur Spine J 28, 1092–1112 (2019). https://doi.org/10.1007/s00586-019-05882-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-019-05882-w