Abstract
Purpose
To quantify the mechanical role of posterior column components in human cervical spine segments.
Methods
Twelve C6-7 segments were subjected to resection of (1) suprasinous/interspinous ligaments (SSL/ISL), (2) ligamenta flavum (LF), (3) facet capsules, and (4) facets. A robot-based testing system performed repeated flexibility testing of flexion–extension (FE), axial rotation (AR), and lateral bending (LB) to 2.5Nm and replayed kinematics from intact flexibility tests for each state. Range-of-motion, stiffness, moment resistance and resultant forces were calculated.
Results
The LF contributes largely to moment resistance, particularly in flexion. Facet joints were primary contributors to AR and LB mechanics. Moment/force responses were more sensitive and precise than kinematic outcomes.
Conclusions
The LF is mechanically important in the cervical spine; its injury could negatively impact load distribution. Damage to facets in a flexion injury could lead to AR or LB hypermobility. Quantifying the contribution of spinal structures to moment resistance is a sensitive, precise process for characterizing structural mechanics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cervical spine injuries are a factor in 3.7 % [1] of all trauma admissions, with distraction-flexion injuries being the most common variant [2]. Clinically, the Cervical Spine Injury Severity Score (CSISS) [3] and Sub-axial Injury Classification (SIC) [4] have been developed to organize and guide treatment, but there is still controversy over how to treat traumatic injuries [5–7]. More generally, neck injuries and associated neck pain [8, 9] are a leading cause of chronic pain and disability [10, 11].
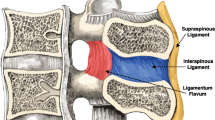
Distraction-flexion (DF) injuries have been shown to vary widely. An MRI study [12] showed only 40 % of clinical unilateral facet injuries having disrupted interspinous ligament (ISL), 60 % having disrupted supraspinous ligament (SSL), and 80 % having a disrupted ligamentum flavum (LF). Grading of injury in the CSISS and SIC schemes does not account of the individual role of posterior/lateral column components despite the wide variation in clinical posterior ligamentous injuries.
Many studies have characterized the role of spinal ligaments in the cervical spine by serial resection with repeated flexibility tests [13–15]. These cervical injury models have shown that the facets and posterior ligamentous complex variously restrict range of motion (ROM) in flexion, extension, lateral bending (LB), and axial rotation (AR). This testing method provides kinematic changes as experimental outcomes. While the change in motion of spinal segments under repeated load control following simulated injury theoretically provides clinically relatable outcomes (i.e. clinical stability scales are based on segmental displacements and rotations), this approach is confounded by changing kinematics after injury and is unable to accurately quantify the mechanical resistance offered by each ligament [16, 17]. Replaying kinematics from the uninjured, intact state following damage to a spinal component allows for the measurement of that structure’s in situ mechanical role [18, 19]. Numerous studies have been performed in other joints, notably in the knee, where robotic testing systems have applied exact intact kinematics in a series of injured or resected joint structures [20, 21] to elucidate the in situ mechanical role of the structures using the principle of superposition [22]. These methods have been applied previously in spine research to characterize the in situ mechanical role of segmental structures in the porcine spine [23], to quantify the mechanical effect of unilateral facetectomy in the human lumbar spine [17], and to calculate the mechanical consequences of combined injuries in sheep lumbar spines [24]. No studies to our knowledge have been performed that examine human cervical ligament and facet injury using replay of intact to elucidate the in situ mechanical role of its component structures.
We performed a serial resection of the posterior column in human C6-7 functional spinal units (FSUs) to simulate different stages of injury and isolate the role of individual structures. The contribution of each posterior column component to loading resistance was obtained via replay of intact kinematics, and changes in ROM and stiffness were determined through repeated flexibility testing. The objectives of the study were to quantify (1) the relative contribution to primary moment and multi-dimensional force resistance of each resected structure for all motions and (2) changes in primary ROM and stiffness for all motions with resection.
Methods
Twelve human C6-7 FSUs (4 female/8 male, 49.7 ± 10.9 years old, see Table 1), which are commonly involved in DF injuries [2], that were without externally visible pathology (e.g. osteophytes or severe disc degeneration) were biomechanically tested. Saline was applied frequently during preparation and testing to prevent desiccation. Four lateral mass screws were inserted into each vertebra for mounting (one in each pedicle and two into the vertebral body). This method of non-destructive fixation rigidly secures vertebrae and allows for facile access to spinal structures for resection [25, 26]. FSUs were then mounted in the robot testing platform as previously described [17].
The robot-based spine testing system consisted of a serial robotic manipulator (Staubli RX90, Staubli Inc., Duncan, SC, USA), an on-board six-axis load cell (UFS Model 90M38A-150, JR3 Inc., Woodland, CA, USA) and custom-built specimen-mounting fixtures (Fig. 1). The robot was controlled quasi-statically via a custom-built, adaptive-displacement algorithm (Matlab 2008R, Mathworks Inc., Natick, MA, USA) [25].
FSUs were subjected to flexion–extension (FE), LB and AR using an end-range moment-target of 2.5 Nm at 1° increments with a 10 N compressive axial preload. Moment targets were based on previous standards, which highlight the mechanical role of ligaments in load support [13, 27]. To simulate the progression of distractive-flexion injury, n = 6 FSUs were sequentially resected in a posterior-to-anterior manner: (1) supraspinous and interspinous ligaments (SSL/ISL), (2) ligamentum flavum (LF), (3) facet capsules, and (4) facets. In a secondary analysis, n = 6 additional specimens were resected in an anterior-to-posterior order—(1) facet capsules, (2) facets, (3) LF, and then (4) SSL/ISL (Fig. 1b)—to determine whether the in situ mechanical roles ascribed to component structures were independent of resection order.
For each motion, FSUs were preconditioned through three cycles of flexibility testing to minimize viscoelastic memory effects, and the six-axis motion of the third cycle was recorded. Adaptive-displacement control (i.e. load control) approximated a flexibility test, as previously described, by minimizing off-axis forces and moments about an updating center-of-rotation at each step in the quasi-static movement to a prescribed moment target [17]. Flexibility testing was repeated for each state, and maximum range-of-motion (ROM) and moment-rotation curve properties including neutral zone width (°), neutral zone stiffness (Nm/°), and elastic zone stiffness (Nm/°), were computed as described elsewhere [28]. This method defines neutral zone stiffness as the mean stiffness within the low-stiffness region of the sigmoidal moment-rotation curve. Neutral zone width is the region of low-stiffness defined by the difference in rotation angles between the inflection points of the sigmoidal curve. Elastic zone stiffness constitutes the slope of the final 10 % of the linear, high-stiffness region. For each state of resection, the testing system also replayed intact motions to measure changes in forces and moments with resection. Intact motion paths were replayed three times (at movement rates an order-of-magnitude faster than quasi-static flexibility tests), and the third path was used for analysis. In resected states, replay of intact motions occurred prior to flexibility testing. In replayed motions, the end-range moment (ERM), the moment magnitude about the primary axis of rotation at the maximum rotation angle, was measured for each state, and normalized differences between states were used to calculate the contribution of resected structures to moment resistance. Additionally, during replayed motions, the difference in component force values (i.e. F Anterior-posterior, F medial-lateral, F superior-inferior) between resection states at the end-range position defined a resultant force vector. This resultant force vector describes the in situ loading supported by resected structures.
ROM at the end-point of flexibility tests and ERM and contribution to moment resistance at end-point of replayed intact motions were calculated for all states in all motions. Neutral zone stiffness and width were computed for FE and LB but not AR data due to its non-sigmoidal shape. Elastic zone stiffness was calculated for all motions. The magnitude of the resultant force vector, after removing the applied compressive force, was compared across states. Repeated-measures ANOVA (SPSS ver. 22.0, IBM, USA) tested significant effects of resection state on ROM, ERM, stiffness measures, and resultant forces (p < 0.05). Tukey HSD post hoc analysis isolated significant differences between resection states. Contributions to moment resistance were compared between orders of resection using a Wilcoxon signed-rank test. All values are presented as mean ± [95 % confidence interval using Student’s distribution] unless otherwise noted.
Results
ROM increased for FE, AR, and LB at each stage of resection (Fig. 2b). The maximum primary moment decreased at the replayed end-range moment (ERM) for all motion paths as well (Table 2; Fig. 2a).
Representative moment-rotation curves for flexion–extension are plotted for a flexibility tests where FSUs were loaded to 2.5 Nm moment targets and changes in rotation angle were measured and b replayed kinematics where intact motion path kinematics are replayed following resection and changes in moment are measured. Data are presented for each state of resection: intact, suprasinous/interspinous ligament (SSL/ISL), ligamentum flavum (LF), facet capsule (CAP), facet surfaces (FACET)
Average ROM and contribution to ERM were normalized to the intact state values during posterior-to-anterior resection (Fig. 3). Generally, ROM increased with resection while primary moments decreased with resection.
Mean (a) range-of-motion (ROM) and b end-point moments are normalized to intact values (intact state set to 1) and shown across resection states—intact, suprasinous/interspinous ligament (SSL/ISL), ligamentum flavum (LF), facet capsule (capsule), facet surfaces (facet)—in flexion, extension (Exten), axial rotation (axial), and lateral bending (lateral). Significant (p < 0.05) differences compared to intact (asterisk), intact and SSL/ISL resection (dagger), and LF resection (double dagger)
Supraspinous/interspinous ligaments
The combined SSL/ISL resection had a non-significant, small effect on total ROM and ERM for all motions except flexion. In flexion, the SSL/ISL contributed to 4.1 % [1.8, 6.4 %] of ERM (p = 0.0056).
Ligamentum flavum
The LF was the largest contributor to flexion ERM, resisting 32.8 % [19.0, 46.5 %] of intact end-point moment (p = 0.0010). Figure 3a shows that combined resection of the LF and SSL/ISL increased flexion ROM (35.8 % [7.0, 64.4 %]), but though not statistically significantly (p = 0.0296 and p = 0.1465). The LF significantly contributed to 15.2 % [7.2, 23.2 %] of LB ERM (p = 0.0053). In extension and AR ERM, the LF non-significantly contributed to 16.0 % [4.4, 27.6 %] and 12.5 % [1.8, 23.2 %], respectively. LF resection did not significantly increase ROM.
Facet capsule
The capsule shared 23.9 % [16.8, 31.1 %] and 22.8 % [11.6, 34.1 %] of the ERM in AR and LB, respectively; both contributions to moment resistance were statistically significant compared to intact and LF-resected states (p = 0.0006–0.0015). Facet resection led to a corresponding 25.6 % [20.3, 31.0 %] increase in AR ROM (p = 0.0003) but only a 7.2 % [−9.6, 24.0 %] increase in LB ROM (p = 0.171). In flexion, the capsule contributed to 14.3 % [2.5, 26.1 %] of ERM (p = 0.0016). Capsule resection had a non-significant effect on extension ROM.
Facet
The facet was the only structure that contributed to a statistically significant increase in AR and LB ROM. Facet resection resulted in a 37.8 % [9.6, 66.0 %] increase in AR ROM and a 22.8 % [7.9, 37.7 %] increase in LB ROM (p = 0.0002 each). It also led to a significant increase in flexion ROM (p = 0.0031) and a large, albeit non-significant, ROM increase (21.1 % [1.4, 40.9 %]) in extension. The facet contributed significantly to moment resistance in all motions. Independent of the capsule, the facet contributed to 18.6 % [3.3, 33.9 %] and 16.8 % [4.8, 28.8 %] of ERM in AR and LB, respectively (p = 0.0005 each). In flexion and extension, the facet contributed 4.0 % [−2.4, 10.4 %] and 10.0 % [3.2, 16.8 %] of the ERM, respectively (p = 0.0031 and p = 0.0645).
Stiffness of neutral/elastic zone and neutral zone width
Elastic zone (EZ) stiffness in AR decreased significantly after resection of facet capsules (p = 0.025) (Fig. 4a), but EZ stiffness was otherwise unaffected by resection. Neutral zone (NZ) stiffness in flexion and extension, although not significant, was affected primarily by SSL/ISL and LF resection (Fig. 4b). The facet and capsule contributed most to NZ stiffness in LB. The anterior column determined the majority of NZ and EZ stiffness for all motions. NZ width increased only with LF resection in flexion and extension (Fig. 4c).
Mean a neutral zone width (°), b neutral zone stiffness (Nm/°), and c elastic zone stiffness (Nm/°) are shown across resection states—intact, suprasinous/interspinous ligament (SSL/ISL), ligamentum flavum (LF), facet capsule (capsule), facet surfaces (Facet)– in flexion, extension (Exten), axial rotation (Axial), and lateral bending (Lateral). Neutral zone properties were not calculated for axial rotation. No effects of resection were significant (p < 0.05)
Resultant forces
Resultant forces at the end-range of replayed intact motions, which describe the magnitudes of the change in the net force vector relative to intact (minimized forces), tended to increase with increasing resection (Fig. 5). The largest increase in resultant forces in flexion, extension, and LB occurred with LF resection, indicating is role in supporting joint forces. Resultant forces showed the largest changes with resection in flexion and extension, and resultant forces varied least and were significantly smaller in LB than other motions.
Mean magnitude of resultant force vector (N) after subtracting the constant, applied compressive force for all motion paths—flexion (Flex), extension (Exten), lateral bending (LB), and axial rotation (AR)—per resection states: intact, suprasinous/interspinous ligament (SSL/ISL), ligamentum flavum (LF), facet capsule (CAP), facet surfaces (FACET)
Resection order
An anterior-to-posterior resection was performed as a secondary analysis to compare moment resistance data of spinal structures to that from posterior-to-anterior resection to determine if resection order influences linear superposition of results in the cervical spine (Fig. 6). In general, the contribution to moment resistance across resected structures and motions were similar between resection orders. However, the contribution of the LF to flexion ERM in the anterior-to-posterior direction was significantly less than in the posterior-to-anterior resection (16.5 [10.4, 22.6 %] and 32.8 [19.5, 46.1 %], respectively). The capsule also contributed more to AR ERM in the posterior-to-anterior direction.
Portion of end-range moment (ERM) supported by each structure—suprasinous/interspinous ligament (SSL/ISL), ligamentum flavum (LF), facet capsule (Capsule), facet surfaces (Facet), and anterior column (Ant. Column) during flexion (Flex), extension (Exten), axial rotation (AR), and lateral bending (LB) subject to (left) posterior-to-anterior resection (n = 6) and (right) anterior-to-posterior resection (n = 6). Anterior Column represents the remaining ERM after facet resection
Discussion
This study examined the mechanical role of posterior structures in the human cervical spine. For the first time, we quantified the contribution of each cervical spine structure to load support. We determined that the LF supports the largest portion of flexion moments in the posterior column. Facet capsules and articulating surfaces support high proportions of AR and lateral bending moments. By comparing contributions to moment resistance in replayed kinematics to ROM assessments in repeated flexibility testing, we found that contributions to moment resistance were generally more precise and sensitive.
The most striking finding was the prominence of the LF in cervical segmental mechanics (32.8 % flexion moment resistance). While the LF has been shown before to be the largest contributor to flexion moment resistance in the porcine lumbar spine (24.7 %) [23], because the LF did not contribute highly to ROM restraint in previous human cervical spine studies [13, 29–31], its contribution to moment resistance was unexpectedly higher in the human cervical spine. The modest effect of LF resection on ROM and NZ properties also underscores its importance in stabilizing FE movements, not just at end range-of-motion, but throughout the motion path. LF resection also increased resultant forces in FE ad LB, reflecting a multidimensional stabilizing role of the LF in these motions that was larger than that of any other resected structure. Moreover, the LF contributed to moment resistance in LB and AR, which has not been studied previously. Its role in these movements is considerable, nearly equaling the contribution of the facet capsule in AR; which has been shown previously to play a dominant role in controlling AR [31]. Considering all of these findings may help to understand the prior observations that LF-preserving operations can reduce postoperative complications [32, 33]. In summary, the mechanical importance of the LF argues for surgical preservation of or compensation for the LF in the cervical spine.
Similar to the LF, the capsules and facets contribute to moment resistance and ROM restraint across all motions. The capsules and facets were predominant contributors to AR and LB support, accounting for 43 and 40 % of moment resistance, respectively. This confirms previous studies that have demonstrated their importance to ROM restraint in these motions [13]. The role of the capsules in flexion moment resistance, 14 %, was remarkably less than that observed in the lumbar spine, 39 % [34]. This may reflect differences in facet anatomy, proximity to the center-of-rotation or possible coupling with the LF in the cervical spine [34]. The lower role in flexion and greater role in AR and LB illustrates an interesting point: although the primary injury mechanism for cervical spines is in the sagittal plane (e.g. distractive-flexion injury), the consequences may be more profound in out-of-plane motions.
The SSL and ISL played a surprisingly small role in segmental mechanics, which was essentially restricted to flexion. In the lumbar spine, Adams et al. observed that the SSL and ISL contributed to 19 % of the flexion moment resistance [34]. In porcine lumbar spines, Gillespie and Dickey found the combined contribution of SSL and ISL to flexion moment resistance was 36 % [23]. Similar testing has not been performed in the human cervical spine, but the small role of the human cervical SSL/ISL we observed is supported by isolated testing of individual cervical ligaments: SSL and ISL were less stiff and exhibited lower peak force and greater elongation than LF and capsular ligaments [35]. These differences in the load sharing role of the SSL/ISL between lumbar and cervical regions may reflect differences in anatomy, location, collagen fiber alignment and ligament composition [36–38].
The anterior column, consisting of the intervertebral disc and longitudinal ligaments, contributed to around half of moment resistance in all motions. This finding highlights the importance of anterior column integrity in resistance to motion in the cervical spine. It also implies that injury or severe degenerative changes in the anterior column could profoundly alter load distribution in the posterior column.
The disparity in results between moment resistance and ROM restraint demonstrates superior precision and sensitivity of moment resistance in evaluating the mechanical role of spinal structures. To translate in vitro findings to evaluations of clinical stability, use of ROM assessments is preferable because clinical evaluations are made based on vertebral rotations and translations [3, 4]. To understand how damaged structures may impact the development of chronic or degenerative conditions, analyzing contributions to moment resistance and in situ force support is more relevant because it quantifies the deficit in load support with the loss of a structure. Resulting alterations in spinal loading among remaining structures may occur in the absence of clinical instability and contribute to mal-adaptive responses that lead to chronic symptoms [39, 40].
To confirm that resection order did not violate the principle of superposition (net loading is a linear sum of load support from each structure) in the cervical spine, we performed a secondary analysis in which we compared posterior-to-anterior and anterior-to-posterior sequences. Researchers have assumed order independence in similar testing for the knee [20] and lumbar spine [16, 24], though some researchers have randomized resection order to avoid any possible influence of resection order [21, 41]. Our findings indicate a modest correlation in moment resistance data between the two directions of cutting, which supports application of the principle of linear superposition. However, the LF and facet capsule showed increased moment resistance when they were cut earlier in the resection order. This finding may indicate that there is a memory effect of previous loading history, which is not removed by preconditioning [17]. Physical coupling of ligamentous components may also underlie this observation [23]. This secondary analysis is insufficient to conclusively confirm the application of linear superposition in the subaxial cervical spine, but it does broadly suggest adherence to the principle of superposition. Given the broad agreement of results between sequences and the relevance of the posterior-to-anterior resection sequence to common injury mechanisms, the primary outcomes of the paper focused on the posterior-to-anterior resection data.
Our study is limited by use of a single FSU, C6-7, as the geometry and joint mechanics in the human cervical spine vary greatly along its length. Therefore our results may not apply directly to other cervical levels. Nonetheless, this study selected a commonly injured level, and its focus on a single level allows for measurements of load sharing, which is not possible in multi-level testing. Additionally, we did not grade specimens for degeneration prior to testing, although we did exclude specimens with evident severe degeneration. Variability in degeneration grade is expected to change load distribution within the FSU. Finally, changing between control methods allowed overshoot of moment targets. Adaptive displacement control (i.e. load control) occurred quasistatically nearly an order of magnitude slower than displacement control used in replay of intact motions. The higher loading rate of displacement control in addition to small overstep of the quasi-static path determination because of its fixed 1° step-size led to higher end-range moments. This overshoot corresponded to moment changes in the elastic zone that were small (<10 % of moment targets) in flexion and extension but moderately large (up to 27 %) in LB. Overshoot, which only affects flexibility test outcomes, was consistent across specimens.
This study replayed intact kinematics and repeated flexibility testing to assess the mechanical role of components of the human cervical spine. The importance of the LF in cervical spine mechanics emerged with possible clinical implications. This study confirmed the importance of facet joints in AR and LB loading in the cervical spine. It also experimentally affirmed the application of the principle of linear superposition to determine load sharing in the cervical spine and found that contribution to moment resistance was more precise and sensitive than ROM changes. This work serves as a basis for future investigations into injury, surgical approaches, complex loading scenarios and mathematical modeling in the human cervical spine.
References
Milby AH, Halpern CH, Guo W, Stein SC (2008) Prevalence of cervical spinal injury in trauma. Neurosurg Focus 25(5):E10
Allen BL Jr, Ferguson RL, Lehmann TR, O’Brien RP (1982) A mechanistic classification of closed, indirect fractures and dislocations of the lower cervical spine. Spine (Phila Pa 1976) 7(1):1–27
Anderson PA, Moore TA, Davis KW, Molinari RW, Resnick DK, Vaccaro AR et al (2007) Cervical spine injury severity score. Assessment of reliability. J Bone Joint Surg Am 89(5):1057–1065
Vaccaro AR, Hulbert RJ, Patel AA, Fisher C, Dvorak M, Lehman RA et al (2007) The subaxial cervical spine injury classification system: a novel approach to recognize the importance of morphology, neurology, and integrity of the disco-ligamentous complex. Spine (Phila Pa 1976) 32(21):2365–2374
Raslan AM, Nemecek AN (2012) Controversies in the surgical management of spinal cord injuries. Neurol Res Int 417834
Henriques T, Olerud C, Bergman A, Jónsson H (2004) Distractive flexion injuries of the subaxial cervical spine treated with anterior plate alone. J Spinal Disord Tech 17(1):1–7
Yadla S, Ratliff JK, Harrop JS (2008) Whiplash: diagnosis, treatment, and associated injuries. Curr Rev Musculoskelet Med 1:65–68
Croft PR, Lewis M, Papageorgiou AC, Thomas E, Jayson MI, Macfarlane GJ et al (2001) Risk factors for neck pain: a longitudinal study in the general population. Pain 93(3):317–325
Nolet PS, Côté P, Cassidy JD, Carroll LJ (2010) The association between a lifetime history of a neck injury in a motor vehicle collision and future neck pain: a population-based cohort study. Eur Spine J 19(6):972–981
Investigation O (2013) The state of US health, 1990–2010: burden of diseases, injuries, and risk factors. JAMA 310(6):591–608
Jull GA, Söderlund A, Stemper BD, Kenardy J, Gross AR, Côté P, et al (2011) Toward optimal early management after whiplash injury to lessen the rate of transition to chronicity. Spine S335–S42
Vaccaro AR, Madigan L, Schweitzer ME, Flanders AE, Hilibrand AS, Albert TJ (2001) Magnetic resonance imaging analysis of soft tissue disruption after flexion-distraction injuries of the subaxial cervical spine. Spine (Phila Pa 1976) 26(17):1866–1872
Richter M, Wilke HJ, Kluger P, Claes L, Puhl W (2009) Load-displacement properties of the normal and injured lower cervical spine in vitro. Eur Spine J 9(2):104–108
Johnson JA, da Costa RC, Bhattacharya S, Goel V, Allen MJ (2011) Kinematic motion patterns of the cranial and caudal canine cervical spine. Vet Surg 40(6):720–727
DeVries NA, Gandhi AA, Fredericks DC, Grosland NM, Smucker JD (2012) Biomechanical analysis of the intact and destabilized sheep cervical spine. Spine (Phila Pa 1976) 37(16):E957–E963
Gillespie KA, Dickey JP (2004) Biomechanical role of lumbar spine ligaments in flexion and extension: determination using a parallel linkage robot and a porcine model. Spine (Phila Pa 1976) 29(11):1208–1216
Gilbertson L, Doehring TC, Kang J (2000) New methods to study lumbar spine biomechanics: delineation of in vitro load-displacement characteristics by using a robotic/UFS testing system with hybrid control. Oper Tech Orthop 10(4):246–253
Woo SL, Debski RE, Wong EK, Yagi M, Tarinelli D (1999) Use of robotic technology for diathrodial joint research. J Sci Med Sport 2:283–297
Fujie H, Livesay GA, Fujita M, Woo SL (1996) Forces and moments in six-DOF at the human knee joint: mathematical description for control. J Biomech 29(12):1577–1585
Li G, Zayontz S, Most E, Defrate LE, Suggs JF, Rubash HE et al (2004) In situ forces of the anterior and posterior cruciate ligaments in high knee flexion : an in vitro investigation. J Orthop Res 22:293–297
Parsons EM, Gee AO, Spiekerman C, Cavanagh PR (2015) The biomechanical function of the anterolateral ligament of the knee. Am J Sports Med Mar 43(3):669–674
Rudy TW, Livesay GA, Woo SL, Fu FH (1996) A combined robotic/universal force sensor approach to determine in situ forces of knee ligaments. J Biomech 29(10):1357–1360
Gillespie KA, Dickey JP (2004) Biomechanical role of lumbar spine ligaments in flexion and extension: determination using a parallel linkage robot and a porcine model. Spine (Phila Pa 1976) 29(11):1208–1216
De Visser H, Rowe C, Pearcy M (2007) A robotic testing facility for the measurement of the mechanics of spinal joints. Proc Inst Mech Eng Part H J Eng Med 221(3):221–227
Bell KM, Hartman RA, Gilbertson LG, Kang JD (2013) In vitro spine testing using a robot-based testing system: comparison of displacement control and “hybrid control”. J Biomech 46(10):1663–1669
Yan Y, Bell KM, Hartman RA, Hu J, Wang W, Kang JD et al (2015) In vitro evaluation of translating and rotating plates using a robot testing system under follower load. Eur Spine J [Epub ahead of print]
Brodke DS, Gollogly S, Mohr RA, Nguyen B, Dailey AT, Bachus KN (2001) Dynamic cervical plates biomechanical evaluation of load sharing and stiffness. Spine 26(12):1324–1329
Smit TH, van Tunen MS, van der Veen AJ, Kingma I, van Dieën JH (2011) Quantifying intervertebral disc mechanics: a new definition of the neutral zone. BMC Musculoskelet Disord 12(1):38 BioMed Central Ltd
Zander T, Rohlmann A, Klöckner C, Bergmann G (2003) Influence of graded facetectomy and laminectomy on spinal biomechanics. Eur Spine J 12(4):427–434
Busscher I, van Dieën JH, Kingma I, van der Veen AJ, Verkerke GJ, Veldhuizen AG (2009) Biomechanical characteristics of different regions of the human spine: an in vitro study on multilevel spinal segments. Spine (Phila Pa 1976) 34(26):2858–2864
Rasoulinejad P, McLachlin SD, Bailey SI, Gurr KR, Bailey CS, Dunning CE (2012) The importance of the posterior osteoligamentous complex to subaxial cervical spine stability in relation to a unilateral facet injury. Spine J 12(7):590–595
Askar Z, Wardlaw D, Choudhary S, Rege A (2003) A ligamentum flavum-preserving approach to the lumbar spinal canal. Spine (Phila Pa 1976) 28(19):E385–E390
Liu J, Ebraheim NA, Sanford CG, Patil V, Haman SP, Ren L et al (2007) Preservation of the spinous process-ligament-muscle complex to prevent kyphotic deformity following laminoplasty. Spine J 7(2):159–164
Adams MA, Hutton WC, Stott JR (1980) The resistance to flexion of the lumbar intervertebral joint. Spine (Phila Pa 1976) 5(3):245–253
Ivancic PC, Coe MP, Ndu AB, Tominaga Y, Carlson EJ, Rubin W et al (2007) Dynamic mechanical properties of intact human cervical spine ligaments. Spine J 7(6):659–665
Yahia LH, Newman N, Rivard CH (1988) Neurohistology of lumbar spine ligaments. Acta Orthop Scand 59(5):508–512
Scapinelli R, Stecco C, Pozzuoli A, Porzionato A, Macchi V, De Caro R (2006) The lumbar interspinous ligaments in humans: anatomical study and review of the literature. Cells Tissues Organs 183(1):1–11
Barros EMK, Rodrigues CJ, Rodrigues NR, Oliveira RP, Barros TE, Rodrigues AJ (2002) Aging of the elastic and collagen fibers in the human cervical interspinous ligaments. Spine J 2(1):57–62
Solomonow M, Zhou BH, Harris M, Lu Y, Baratta RV (1998) The ligamento-muscular stabilizing system of the spine. Spine (Phila Pa 1976) 23(23):2552–2562
Mulholland RC (2008) The myth of lumbar instability: The importance of abnormal loading as a cause of low back pain. Eur Spine J, p 619–25
Sakane M, Livesay GA, Fox RJ, Rudy TW, Runco TJ, Woo SL (1999) Relative contribution of the ACL, MCL, and bony contact to the anterior stability of the knee. Knee Surg Sport Traumatol Arthrosc 7(2):93–97
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded in part by the University of Pittsburgh Medicel Center Rehabilitation Institute Pilot Grant (2015), the Albert B. Ferguson Jr., MD Fund of the Pittsburgh Foundation, and the Department of Orthopaedic Surgery.
Conflict of interest
Dr. Sowa has received research funding from the Orthopaedic Research Education Foundation, the Foundation for PMR, and the Pittsburgh Foundation and is an author for UpToDate.
Rights and permissions
About this article
Cite this article
Hartman, R.A., Tisherman, R.E., Wang, C. et al. Mechanical role of the posterior column components in the cervical spine. Eur Spine J 25, 2129–2138 (2016). https://doi.org/10.1007/s00586-016-4541-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-016-4541-1