Abstract
Purpose
We determined the incidence of and risk factors for clinical adjacent segment pathology (C-ASP) requiring additional surgeries among patients previously treated with one-segment lumbar decompression and fusion surgery.
Methods
We retrospectively analysed 161 consecutive patients who underwent one-segment lumbar decompression and fusion surgery for L4 degenerative spondylolisthesis. Patient age, sex, body mass index (BMI), facet orientation and tropism, laminar inclination angle, spinal canal stenosis ratio [on myelography and magnetic resonance imaging (MRI)], preoperative adjacent segment instability, arthrodesis type, pseudarthrosis, segmental lordosis at L4–5, and the present L4 slip were evaluated by a log-rank test using the Kaplan–Meier method. A multivariate Cox proportional-hazards model was used to analyse all factors found significant by the log-rank test.
Results
Of 161 patients, 22 patients (13.7 %) had additional surgeries at cranial segments located adjacent to the index surgery’s location. Pre-existing canal stenosis ≥47 % at the adjacent segment on myelography, greater facet tropism, and high BMI were significant risk factors for C-ASP. The estimated incidences at 10 years postoperatively for each of these factors were 51.3, 39.6, and 32.5 %, and the risks for C-ASP were 4.9, 3.7, and, 3.1 times higher than their counterparts, respectively. Notably, spinal canal stenosis on myelography, but not on MRI, was found to be a significant risk factor for C-ASP (log-rank test P < 0.0001 and 0.299, respectively).
Conclusions
Pre-existing spinal stenosis, greater facet tropism, and higher BMI significantly increased C-ASP risk. Myelography is a more accurate method for detecting latent spinal canal stenosis as a risk factor for C-ASP.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
During the past few decades, spinal arthrodesis has become a common treatment component for a variety of spinal disorders. However, it alters the biomechanical and kinematic properties of the lumbar spine [1, 2]. Pathological development at mobile segments above or below the site of spinal fusion is known as adjacent segment pathology (ASP). ASP is considered a potential late complication of spinal arthrodesis that requires further surgical treatment. The clinical failure rate of adjacent segments at 5 years after the index spinal fusion surgery has been reported to range from 3 to 32.3 % [3–7].
Several risk factors for ASP have been reported, such as age [4, 5, 7], sex [4], multilevel arthrodesis [3–5], sagittal imbalance [8], the type of arthrodesis [7], facet tropism [9], and laminar inclination [9]. However, few studies have focused on asymptomatic pre-existing spinal stenosis as a risk factor for clinical ASP (C-ASP) that requires additional surgery at an adjacent segment [10]. In fact, when patients demonstrate asymptomatic spinal stenosis adjacent to the fusion segment, there is often controversy as to whether the segment should be included within the surgical site or not.
This study analysed the preoperative prognostic risk factors for C-ASP, and we calculated the survival times of the patients with significant risk factors.
Materials and methods
From January 2000 to December 2006, 204 L4 degenerative spondylolisthesis (DS) patients with radicular pain and/or neurological claudication after unsuccessful conservative treatment underwent either instrumented posterolateral fusion (PLF) or posterior lumbar inter-body fusion (PLIF) at the single level of L4–5. All surgeries were performed using the same procedures at a single institution. Patients with an acute fracture, dislocation, or malignancy were excluded. Informed consent was obtained from all patients. Medical records of all patients were reviewed, and this study was approved by our local ethics committee. Forty-three patients were excluded because of a short follow-up (<2 years) or a lack of preoperative magnetic resonance imaging (MRI) and/or myelography data. The remaining 161 patients with a follow-up period of longer than 2 years were finally selected.
PLF had been performed in 137 patients (85 %) and PLIF in 24 patients (15 %). In all patients undergoing PLF, autogenous cancellous iliac bone was used as a graft. PLIF was performed using a rectangular ceramic cage with morselised local bone from neural decompression in all patients. When patients had spinal stenosis on myelography or MRI at the L3–4 segment, as well as neurological findings (including the presence of patellar tendon reflex and no sensory and motor disturbance associated with the L4 nerve root) and negative findings of L4 nerve root infiltration, we did not include the L3–4 segment in the operation site.
Radiographic evaluation
In all patients, computed tomography (CT), myelography, and MRI were performed within 2 weeks before the index fusion surgery. In this series, no patients required additional surgery at the L5–S1 segment during follow-up, so radiographic evaluations were performed at the L3–4 and L4–5 segments. Standard biplanar anteroposterior, lateral radiography with the lumbosacral spine in neutral, flexion, and extension positions was performed preoperatively, at 24 months after surgery, and at the final follow-up.
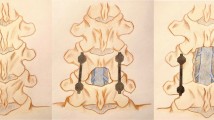
The anteroposterior vertebral slip and intervertebral disc angle were measured on lateral radiographs of the L3–4 and L4–5 taken in the neutral, flexion, and extension positions. To minimise the errors due to different magnifications, the vertebral slip was expressed as a percentage of the caudal vertebral body width (% slip). The ranges of motion (ROM) of the L3–4 and L4–5 segments were defined as the sum of the intervertebral disc angles in the flexion/extension view (Fig. 1). Pseudarthrosis was present if there was no continuity in the PLF fusion mass between the cephalad and caudad transverse processes, no continuity between graft bone and vertebra in PLIF fusion, or if lateral flexion–extension radiographs demonstrated >2° of angular motion or >2 mm of sagittal motion at L4–5 [11].
Plain radiography measurement. a Anterior slip in the flexion position, b posterior slip in the extension position, c vertebral body width. The total percent slip is (a + b)/c × 100. α Intervertebral disc angle in flexion position, β intervertebral disc angle in the extension position. The range of motion is α + β in degrees
The criteria for adjacent segment instability were well-defined spondylolisthesis or dynamic instability with slippage >4 mm and/or an ROM >10° [12]. The laminar inclination angle at L3 was measured as previously described [9] on lateral radiographs (Fig. 2a). Facet orientation and tropism were determined by CT images that were coplanar with the disc and transected the facet joints, as described previously [9]. The sum of the right and left facet angles and the difference between the right and left facet angles were defined as the facet orientation and tropism, respectively (Fig. 2b).
a The laminar inclination angle at L3 was defined as the angle formed by a straight line connecting the base of the superior facet with the base of the inferior facet, and a straight line connecting the midpoints of the anterior and posterior L3 vertebral cortices on lateral radiographs. b Facet orientation and tropism were determined by computed tomography images that were coplanar with the disc and transected the facet joints. The sum of the right and left facet angles and the difference between the right and left facet angles were defined as the facet orientation and tropism, respectively
Myelography measurements
After lumbar puncture and injection of radiographic contrast material into the dural sac under fluoroscopic guidance, the physician moved the patient’s lower back to maximum flexion and extension in the left lateral decubitus position and obtained lateral radiographs in the neutral, flexion, and extension positions under fluoroscopy. The narrowest anteroposterior dural sac diameter at L3–4 was measured on lateral myelography in the neutral, flexion, and extension positions and on the sagittal view of T2-weighted MRI. The dural sac diameter at the midpoint of the L2 vertebral body was also measured. The spinal canal stenosis ratio (SCSR) was calculated as x/y × 100 (Fig. 3).
The narrowest anteroposterior dural sac diameter at L3–4 (x) was measured on lateral myelographs taken in the extension position (a) and on MRI (b) in the same patient. The dural sac diameter at the midpoint of the L2 vertebral body (y) was also measured. The spinal canal stenosis ratio (SCSR) was calculated as x/y × 100
All measurements were performed twice by two independent observers blinded to the patient name and clinical findings using an electronic digitiser (MicroAnalyzer; Japan Poladigital Corp., Tokyo, Japan) with an accuracy of 0.01 mm: measurements were averaged. The inter-observer correlation of all measurement was evaluated by the Pearson’s correlation coefficient test. The kappa statistic was used to assess inter-observer agreement of pseudarthrosis and preoperative instability at L3–4.
Statistical analysis
The final follow-up examination was defined as the last visit. In patients undergoing re-operation at L3–4, the survival period was defined as the interval from the index operation to the second operation due to C-ASP. C-ASP was defined as a condition where an additional surgery at L3–4 was required to treat symptomatic neurological deterioration.
The following prognostic risk factors were examined: age, sex, body mass index (BMI), facet orientation, facet tropism, laminar inclination angle, SCSR by myelography and MRI, preoperative adjacent segment instability, type of fusion, presence of pseudarthrosis, segmental lordosis at L4–5, and the %slip of L4–5 after 2 years postoperatively. Continuous variables were dichotomised to increase the statistical power using the Youden index from the receiver operating characteristic curve (ROC).
A log-rank test was used for univariate analyses using the Kaplan–Meier method, and survival curves for all patients with significant risk factors were constructed to calculate the survival time. A multivariate Cox proportional-hazards model was used to assess all factors demonstrated to be significant by the log-rank test to adjust for confounding factors.
Statistical analyses were performed using the JMP 10 statistical software package (SAS Institute Inc. Cary, NC). A value of P < 0.05 was considered to be statistically significant.
Results
There were 56 males and 105 females. The mean age at index surgery was 65.4 years (range 40–87 years). The average follow-up period was 77.3 months (range 24–183 months). The follow-up rate was 78.9 %. Among the 161 patients, 22 (13.7 %) underwent subsequent procedures at cranial segments adjacent to the L4–5 segment; five patients underwent decompression surgery with arthrodesis and 17 underwent decompression surgery alone. After the additional surgery, all patients show improved neurological symptoms. The mean duration between the index surgery and the additional surgery was 75.9 months (range 24–141 months).
The inter-observer correlation is shown in Table 1. The kappa coefficient for pseudarthrosis rated between observers was 0.82 (P < 0.0001) and that of preoperative instability at L3–4 was 0.89 (P < 0.0001).
Patients with a BMI ≥ 25 kg/m2 had a significantly lower survival rate than their counterparts in a univariate analysis (log-rank test: P = 0.0497). The incidence of C-ASP in patients with a BMI ≥ 25 kg/m2 was estimated to be 32.5 % at 10 years. Conversely, the incidence of C-ASP in patients with a BMI < 25 kg/m2 was lower, at 21.1 % at 10 years. The median survival time for patients with a BMI ≥ 25 kg/m2 was 141 months (Fig. 4).
Patients with facet tropism ≥11° demonstrated a lower survival rate than their counterparts (log-rank test: P = 0.0178). The incidence of C-ASP among patients with facet tropism ≥11° was 39.6 %, while for facet tropism <11° it was 19.4 % at 10 years after the initial operation (Fig. 5).
Regarding SCSR, patients with an SCSR ≥47 % on myelography in the extension position showed a significantly lower survival rate than their counterparts (P < 0.0001). In these patients, the prevalence of C-ASP requiring reoperation was 51.3 % at 10 years, whereas it was 11.4 % in patients with an SCSR <47 %. The median survival time for patients with an SCSR ≥47 % was 110 months (Fig. 6).
Interestingly, SCSR determined by MRI was not a significant risk factor (log-rank test: P = 0.2990). Since a factor with a higher discrimination ability makes an ROC curve closer to the top left corner, the area under the ROC curve (AUC) is used to indicate the sensitivity and specificity of each factor. We compared the AUC values of MRI and myelography in the extension positions. In this analysis, the AUC of the SCSR determined by myelography was significantly higher than that of the SCSR determined by MRI (Fig. 7). In fact, 13.2 % of patients who exhibited SCSR on MRI <50 % had an SCSR ≥50 % on myelography in the extension position.
Other potential risk factors, such as age, sex, facet orientation, laminar inclination angle, preoperative adjacent segment instability, type of fusion, pseudarthrosis, segmental lordosis, the %slip, and SCSR on myelography in the neutral and flexion positions, were not statistically significant (Table 2). A multivariate Cox proportional-hazards model revealed a BMI ≥ 25 kg/m2, facet tropism ≥11°, and SCSR ≥47 % on myelography to be significant risk factors, and patients with these factors had 3.1-, 3.7-, and 4.9-fold higher risks of adjacent segment reoperation than their counterparts, respectively (Table 3).
Discussion
The definition of C-ASP has often been reported as adjacent segment pathology, manifesting radiculopathy, neurogenic intermittent claudication, back pain, or a combination of any of these [13], and the need for additional surgeries [3–5, 8–10] on the index fusion segments. Park et al. [3] reported the incidence of C-ASP to range from 5.2 to 18.5 %. The term ‘degeneration’ itself suggests a time-dependent phenomenon. Therefore, the survival function estimated by the Kaplan–Meier method and the multivariate Cox regression model are good ways to analyse the development of ASP as a late complication of spinal arthrodesis.
Regardless of the use of spinal arthrodesis, the clinical course of patients with severe spinal stenosis often deteriorates over time during conservative treatments [14]. This indicates that pre-existing spinal stenosis in itself may be a significant risk factor for C-ASP. Cho et al. [10] reported a significant relationship between pre-existing spinal stenosis and C-ASP. However, they did not indicate a cutoff point for spinal stenosis that may increase the likelihood of C-ASP. In the current study, patients with an SCSR ≥47 % on myelography in the extension position exhibited a 4.87-fold higher risk of adjacent segment reoperation than their counterparts. Interestingly, spinal stenosis demonstrated by MRI was not a significant factor. We attempted to change the cutoff point for SCSR on MRI from 35 to 60 %; however, the MRI findings were not a significant factor. Moreover, the AUC of SCSR on myelography in the extension position (0.7528) was significantly larger than that on MRI (0.5768) (Chi squared; P = 0.0004). These results suggest that myelography has a significantly higher sensitivity and specificity to detect not only latent spinal canal stenosis, but also the risk of ASP requiring additional surgery.
While these results indicate that pre-existing severe stenosis can be a significant risk factor for C-ASP, this does not lead directly to a recommendation for performing laminectomy during the index surgery, since performing laminectomy adjacent to a fusion segment demonstrated a significant association with ASP [5, 15–17]. Imagama et al. [15] recommended that the adjacent segment with asymptomatic spinal stenosis should not be subjected to a concomitant decompression from the viewpoint of preventing ASP. We therefore recommend that surgeons should have thorough discussions with patients to determine whether a concomitant operation at adjacent segments with asymptomatic stenosis should be performed.
Facet tropism is defined as asymmetry in the facet joint that causes an abnormal rotation of the spinal segment, which increases the mechanical stress on the disc and could lead to lumbar degeneration or disc herniation [18, 19]. Several studies have reported greater facet tropism to have a significant relationship with C-ASP [9]. In the current study, patients with facet tropism ≥11° had a 3.74-fold higher risk of adjacent segment reoperation compared to their counterparts. We hypothesised that facet tropism may affect the rotational stability, which accelerates the thickening of the ligamentum flavum, thus resulting in spinal canal stenosis. However, some authors have reported no association between facet tropism and the occurrence of C-ASP [10]. Further studies are required to clarify this issue.
It remains controversial as to whether an association exists between BMI and ASP. Some studies reported no association between BMI and radiographic ASP [20]; however, Cho et al. [10] reported BMI to be a significant risk factor for C-ASP, and Liuke et al. [21] reported that a BMI ≥ 25 kg/m2 increased the risk of lumbar disc degeneration on MRI. It is assumed that being overweight might cause disc degeneration [22], resulting in earlier ASP over the long term. In this series, a BMI ≥ 25 kg/m2 was identified as a significant risk factor for C-ASP, and patients with this factor had a 3.12-fold higher risk of needing adjacent segment reoperation than their counterparts. Patients with high BMI appear to have a higher risk of C-ASP; however, a large prospective study is needed to confirm this finding.
In this study, other factors were not significant risk factors for C-ASP. The association between the conditions of fused segments and the occurrence of ASP also remains controversial. Since almost all past studies were retrospective analyses that contained potential bias, a randomised prospective study will be necessary to resolve these issues.
There are several possible limitations associated with this study. First, it was a retrospective study. Second, the predictors derived were not prospectively validated in an independent population. Third, the sample size was relatively small. Finally, whole spinal radiographs were not routinely taken for DS patients who underwent one-segment spinal fusion and decompression and, therefore, a whole spinal radiographic analysis was not possible in this study. Despite these limitations, the factors identified in this study may assist both surgeons and patients when making decisions about whether or not to include an adjacent segment at the time of index fusion surgery.
In conclusion, a BMI ≥ 25 kg/m2, facet tropism ≥11°, and pre-existing stenosis ≥47 % demonstrated on myelography in the extension position were found to be important risk factors for C-ASP requiring a second operation. Careful consideration of the type and extent of surgery is therefore necessary when these risk factors are present.
References
Axelsson P, Johnsson R, Strömqvist B (1997) The spondylolytic vertebra and its adjacent segment. Mobility measured before and after posterolateral fusion. Spine (Phila Pa 1976) 22(4):414–417
Bastian L, Lange U, Knop C et al (2001) Evaluation of the mobility of adjacent segments after posterior thoracolumbar fixation: a biomechanical study. Eur Spine J 10(4):295–300
Ghiselli G, Wang JC, Bhatia NN et al (2004) Adjacent segment degeneration in the lumbar spine. J Bone Joint Surg Am 86-A(7):1497–1503
Ahn DK, Park HS, Choi DJ et al (2010) Survival and prognostic analysis of adjacent segments after spinal fusion. Clin Orthop Surg 2(3):140–147
Sears WR, Sergides IG, Kazemi N et al (2011) Incidence and prevalence of surgery at segments adjacent to a previous posterior lumbar arthrodesis. Spine J 11(1):11–20
Celestre PC, Montgomery SR, Kupperman AI et al (2014) Lumbar clinical adjacent segment pathology: predilection for proximal levels. Spine (Phila Pa 1976) 39(2):172–176
Lee JC, Kim Y, Soh JW, Shin BJ (2014) Risk factors of adjacent segment disease requiring surgery after lumbar spinal fusion: comparison of posterior lumbar interbody fusion and posterolateral fusion. Spine (Phila Pa 1976) 39(5):E339–E445
Kumar MN, Baklanov A, Chopin D (2001) Correlation between sagittal plane changes and adjacent segment degeneration following lumbar spine fusion. Eur Spine J 10(4):314–319
Okuda S, Oda T, Miyauchi A et al (2008) Lamina horizontalization and facet tropism as the risk factors for adjacent segment degeneration after PLIF. Spine (Phila Pa 1976) 33(25):2754–2758
Cho TK, Lim JH, Kim SH et al (2013) Preoperative Predictable Factors for the Occurrence of Adjacent Segment Degeneration Requiring Second Operation after Spinal Fusion at Isolated L4–L5 Level. J Neurol Surg A Cent Eur Neurosurg [Epub ahead of print]
Kornblum MB, Fischgrund JS, Herkowitz HN et al (2004) Degenerative lumbar spondylolisthesis with spinal stenosis: a prospective long-term study comparing fusion and pseudarthrosis. Spine (Phila Pa 1976) 29(7):726–733
Chen WJ, Lai PL, Niu CC et al (2001) Surgical treatment of adjacent instability after lumbar spine fusion. Spine (Phila Pa 1976) 26(22):E519–E524
Liao JC, Chen WJ, Chen LH et al (2011) Surgical outcomes of degenerative spondylolisthesis with L5–S1 disc degeneration: comparison between lumbar floating fusion and lumbosacral fusion at a minimum 5-year follow-up. Spine (Phila Pa 1976) 36(19):1600–1607
Minamide A, Yoshida M, Maio K (2013) The natural clinical course of lumbar spinal stenosis: a longitudinal cohort study over a minimum of 10 years. J Orthop Sci 18(5):693–698
Imagama S, Kawakami N, Kanemura T et al (2013) Radiographic adjacent segment degeneration at five years after L4/5 posterior lumbar interbody fusion with pedicle screw instrumentation: evaluation by computed tomography and annual screening with magnetic resonance imaging. J Spinal Disord Tech [Epub ahead of print]
Miyagi M, Ikeda O, Ohtori S et al (2013) Additional decompression at adjacent segments leads to adjacent segment degeneration after PLIF. Eur Spine J 22(8):1877–1883
Lai PL, Chen LH, Niu CC et al (2004) Relation between laminectomy and development of adjacent segment instability after lumbar fusion with pedicle fixation. Spine (Phila Pa 1976) 29(22):2527–2532
Noren R, Trafimow J, Andersson GB et al (1991) The role of facet joint tropism and facet angle in disc degeneration. Spine (Phila Pa 1976) 16(5):530–532
Karacan I, Aydin T, Sahin Z et al (2004) Facet angles in lumbar disc herniation: their relation to anthropometric features. Spine (Phila Pa 1976) 29(10):1132–1136
Ha KY, Son JM, Im JH et al (2013) Risk factors for adjacent segment degeneration after surgical correction of degenerative lumbar scoliosis. Indian J Orthop 47(4):346–351
Liuke M, Solovieva S, Lamminen A et al (2005) Disc degeneration of the lumbar spine in relation to overweight. Int J Obes (Lond) 29(8):903–908
Weiler C, Lopez-Ramos M, Mayer HM et al (2011) Histological analysis of surgical lumbar intervertebral disc tissue provides evidence for an association between disc degeneration and increased body mass index. BMC Res Notes 4:497
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No funds were received in support of this work. No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this manuscript.
Ethical standards
The study was approved by the local ethics committee.
Additional information
The manuscript does not contain information about medical device(s)/drug(s).
Rights and permissions
About this article
Cite this article
Yugué, I., Okada, S., Masuda, M. et al. Risk factors for adjacent segment pathology requiring additional surgery after single-level spinal fusion: impact of pre-existing spinal stenosis demonstrated by preoperative myelography. Eur Spine J 25, 1542–1549 (2016). https://doi.org/10.1007/s00586-015-4185-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-015-4185-6