Abstract
Primary extragonadal malignant germ cell tumors (EMGCTs) are rare and account for only 2–5% of malignant germ cell tumors. Vascular tumors are uncommon mediastinal masses making up < 1% of all mediastinal masses. We report the case of a 20-year-old man presenting with a germ cell seminoma as well as an atypical hemangioma encapsulated in one mass within the mediastinum. This clinical report focuses on a 20-year-old man who presented with both a germ cell seminoma as well as an atypical hemangioma within one encapsulated mass, a unique case not presented before in literature to our knowledge. The histological analysis is essential in correctly diagnosing a neoplasm. Combining the unique histology of this case with the clinical and scientific knowledge of several specialties allowed the clinicians to successfully develop a treatment for this patient.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Primary extragonadal malignant germ cell tumors (EMGCTs) are rare and account for only 2–5% of malignant germ cell tumors. Vascular tumors are uncommon mediastinal masses making up < 1% of all mediastinal masses (Hirai et al. 2010). The mediastinum is the most common site of EMGCTs, with approximately 50–70% of EMGCTs occurring in the mediastinum (Gao et al. 2015). Mediastinal hemangioma is a vascular neoplasm made up of capillaries or veins that are typically benign (Macchiarini and Ostertag 2004). Hemangiomas of the mediastinum are rare tumors with < 100 having been reported (Cohen et al. 1987).
Case report
A 20-year-old Caucasian male presented with history of fatigue, non-productive cough for 2 months, and a recent travel to Central America. Community-acquired pulmonary tuberculosis was initially considered. A two-view chest X-ray (CXR) was obtained which revealed an anterior mediastinal mass (Fig. 1). A contrast-enhanced computed tomography study of the chest (CCT) was obtained to better characterize the mass. Interestingly, the patient had had a prior chest X-ray 9 months earlier at the same institution for flu-like symptoms to exclude pneumonia. In retrospect, the anterior mediastinal mass was in fact present on the original chest X-ray taken 9 months earlier, but was not appreciated at the time (Fig. 2).
The patient had had a prior chest X-ray 9 months earlier at the same institution for flu-like symptoms to exclude pneumonia. In retrospect, the anterior mediastinal mass was in fact present on the original chest X-ray, but was not appreciated at the time, as it was superimposed upon the “aortic knob,” noted by the aortic “double density sign.” Thus, the mass had grown considerably during this 9-month time frame
CCT demonstrated a rounded, well-circumscribed soft tissue density tumor with considerable central necrosis and prominent vascularity within its peripheral wall, supplied primarily by the left internal mammary artery. There was no direct invasion of the adjacent mediastinal structures. There was mild to moderate mass compression of the main pulmonary artery and the tumor abutted the pericardium and aorta. There was no mediastinal or hilar lymphadenopathy (Fig. 3). Additional imaging studies namely CT scans of the abdomen and pelvis and ultrasound of the testicles were normal. Laboratory work-up (chromogranin A, beta HCG, phosphorous, lactate dehydrogenase, ACTH, am-cortisol, alpha-fetoprotein, and carcinoembryonic antigen) was unremarkable.
The radiologic study shows a rounded, well-circumscribed soft tissue density tumor with considerable central necrosis and prominent vascularity within its peripheral wall, supplied primarily by the left internal mammary artery. While there was no direct invasion of the adjacent mediastinal structures, there was mild to moderate mass compression of the main pulmonary artery, and the tumor abutted the pericardium and aorta. There was no mediastinal or hilar lymphadenopathy
Resection of the mass was performed using left robotic-assisted thoracic surgery with a four-port technique and the patient in a supine position (Stark et al. 2013). The tumor was relatively circumscribed, but there was direct focal invasion into adjacent fibroadipose tissue and mediastinal lymph nodes. The mass, lymph nodes, anterior mediastinal fat, and thymus were exonerated en bloc. Phrenic nerves were uninvolved and left intact. The mass measured 11.0 × 8.0 × 6.5 cm and weighed 260 g on removal.
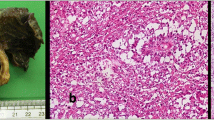
Pathological review demonstrated a germ cell neoplasm representing a seminoma with additional vascular proliferation interpreted as a malignant vasoformative mesenchymal component. Histology demonstrated extensive necrosis (greater than 75% of tumor) with surrounding scar and reactive mesothelial proliferation. There were areas of thrombosis, some organized. Surgical margins were uninvolved and closest margin was two millimeters. Immunostains revealed that tumor cells were positive for OTC4 and CD117 and negative for pancytokeratin and CD45 (Fig. 4).
a The mass contained areas of seminoma with a prominent lymphoid host response (left) adjacent to areas of abnormal vascular proliferation (right). The two components were separated by areas of scar. b High-power image of the seminomatous component shows malignant germ cells with prominent inflammatory host response, typical of seminoma. These cells were positive for OCT 3/4 and CD117 and negative for keratin and CD45 (not shown). c, d Sections of the malignant vascular component show an abnormal vasoformative proliferation with marked cytologic atypia and frequent mitotic figures. Areas of localized thrombosis were also seen within the proliferation. This proliferation was intimately associated with and extended into the scar
The possible utility of two adjuvant cycles of bleomycin, etoposide, and cisplatin (BEP) chemotherapy was discussed but was ultimately not recommended given the negative margins, the vasoformative mesenchymal component, and ability to treat recurrent seminoma with three cycles of BEP with excellent results. The utility of adjuvant radiation therapy was discussed but not recommended given the unclear benefit versus risk treatment. Clinical and imaging surveillance every 6 months was recommended. Four years later, the patient remains without evidence of recurrence.
Discussion
Primary mediastinal seminomas are uncommon. Vascular tumors of the mediastinum are also very rare making up less than 1% of all mediastinal masses (Hirai et al. 2010). To our knowledge, the synchronous occurrence of these tumors in one encapsulated mass in the anterior mediastinum has not been previously reported. Standard treatment of mediastinal seminoma is with initial combination chemotherapy (BEP) with surgery reserved for refractory or locally recurrent cases. This paradigm was established based on a small number of patients in the 1980s (Lawrence Einhorn, personal communication) and subsequently validated in larger cohorts. The seminal report authored by the international germ cell cancer collaborative group (IGCCCG) of more than 5000 patients with metastatic germ cell cancer treated with cisplatin-based chemotherapy included 41 patients with mediastinal seminoma. The 5-year survival rate of those patients was 85% (the International Germ Cell Consensus Classification: a prognostic factor-based staging system for metastatic germ cell cancers, the International Germ Cell Cancer Collaborative Group 1997). Another report of 635 patients with extragonadal germ cell tumors included 51 patients with mediastinal seminoma. The 5-year survival rate of these patients was 88%. In the same report, in 25 patients with extragonadal seminoma who underwent surgical resection of residual lesions after initial therapy, necrosis was identified in 23, viable seminoma in 1, and mature teratoma in 1 (Bokemeyer et al. 2001).
In our case, monitoring the patient for recurrence instead of treating with BEP chemotherapy spared the patient the side effects of ototoxicity, neurotoxicity, heart and blood vessel problems, and increased risk of secondary non-germ cell cancers (Chevreau et al. 2004 and Haugnes et al. 2014). Four years after resection, the patient has no detectable recurrence.
Conclusion
In summary, we report the first case of concurrent seminoma and malignant vasoformative vascular tumor in the anterior mediastinum treated successfully with minimally invasive surgical resection and deferral/avoidance of radiotherapy and systemic chemotherapy. Less invasive transthoracic surgical techniques are an option and may be as effective as systemic medical chemotherapeutic regimens in certain clinical situations, such as the case above given well-defined radiological margins suggesting an encapsulated tumor with no apparent metastases.
References
Bokemeyer C, Droz JP, Horwich A, Gerl A, Fossa SD, Beyer J, Pont J, Schmoll HJ, Kanz L, Einhorn L, Nichols CR, Hartmann JT (2001) Extragonadal seminoma: an international multicenter analysis of prognostic factors and long term treatment outcome. Cancer 91(7):1394–1401. https://doi.org/10.1002/1097-0142(20010401)91:7<1394::AID-CNCR1144>3.0.CO;2-Y
Chevreau C, Mazerolles C, Soulie M, Gaspard MH, Mourey L, Bujan L, Plante P, Rischmann P, Bachaud JM, Malavaud B (2004) Long-term efficacy of two cycles of BEP regimen in high-risk stage I nonseminomatous testicular germ cell tumors with embryonal carcinoma and/or vascular invasion. Eur Urol 46(2):209–215. https://doi.org/10.1016/j.eururo.2004.03.022
Cohen AJ, Sbaschnig RJ, Hochholzer L, Lough FC, Albus RA (1987) Mediastinal hemangiomas. Ann Thorac Surg 43(6):656–659. https://doi.org/10.1016/s0003-4975(10)60244-5
Gao Y, Jiang J, Liu Q (2015) Extragonadal malignant germ cell tumors: a clinicopathological and immunohistochemical analysis of 48 cases at a single Chinese institution. Int J Clin Exp Pathol 8(5):5650–5657
Haugnes HS, Stephenson AJ & Feldman DR (2014) Beyond stage I germ cell tumors: current status regarding treatment and long-term toxicities. American Society of Clinical Oncology Educational Boook, pp e180-e190
Hirai K, Takeuchi S, Bessho R, Ohaki Y, Koizumi K, Shimizu K (2010) Venous hemangioma of the anterior mediastinum. J Nippon Med Sch 77(2):115–118. https://doi.org/10.1272/jnms.77.115.
International Germ Cell Cancer Collaborative Group (1997) International Germ Cell Consensus Classification: a prognostic factor-based staging system for metastatic germ cell cancers. Int J Clin Oncol 15(2):594–603
Macchiarini P, Ostertag H (2004) Uncommon primary mediastinal tumours. Lancet Oncol 5(2):107–118. https://doi.org/10.1016/s1470-2045(04)01385-3
Stark TA, Gupta A, McCabe VV, Sood M & Leichtle SW (2013) Chapter five: the mediastinum. Essentials of Robotic Surgery
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from the patient included in this study.
Rights and permissions
About this article
Cite this article
Hutchison, D.D., Kudirka, A.A., Posa, A. et al. A novel pathology of a mediastinal neoplasm. Comp Clin Pathol 27, 1103–1106 (2018). https://doi.org/10.1007/s00580-018-2734-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-018-2734-1