Abstract
The antioxidant protective effect of the fruits and peels of Tetrapleura tetraptera were investigated in trona (kaun)-induced oxidative injuries in male albino rats. The peels and fruits of T. tetraptera were air-dried and extracted with ethanol. The concentrated extracts were subjected to FTIR spectroscopy analysis to identify their chemical functional groups. Five groups of six rats each were pretreated with trona (500 mg/kg bw) for 7 days, while a sixth group was administered water only which served as normal control. Four of the pretreated groups were subjected to posttreatment with T. tetraptera extracts, while the fifth served as negative control. At the end of the experiment, their hepatic, kidney, and intestinal tissues were assessed for catalase activity and malondialdehyde (MDA) level. Aspartate transaminase (AST) and alanine transaminase (ALT) levels were analyzed in blood serum. While histopathology was carried out on hepatic tissues. FTIR spectroscopy of the extracts revealed amines, alcohols, carboxylic acids, esters, ethers, aromatics, alkanes, aldehydes, ketones, phenols, and amides as the functional groups. Ingestion of trona caused significant (p < 0.05) increase in all studied biological parameters in all tissues. These were significantly (p < 0.05) reversed to near normal after treatment with both extracts. Histopathology revealed reduction in trona-induced lesions and alterations in hepatic tissue after treatment with the extracts. These results indicate the antioxidant protective effect of T. tetraptera against trona-induced oxidative injury, which can be attributed to the identified functional groups.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Trona, also known as potash (potassium carbonate), is an inorganic compound made up of crude mixture of minerals, salts, and impurities which include sand, clay, and metals such as iron, aluminum, potassium, silicon, magnesium, and titanium (Sodipo 1993). It is a hydrated sequicarbon of sodium carbonate and sodium bicarbonate (Na2CO3*NaHCO3-2H20) (Sodipo 1993). Calcites have been reported to be the major chemical constituent, with hanksite, halite, pirssonite, and borax present in low concentrations (Lu et al. 2005).
Trona is commonly referred to as kaun and kanwa in the southern and northern parts of Nigeria respectively and an indigenous food addictive for tenderizing legumes, vegetables, bone, and meat (Bankole et al. 2015; Okoye et al. 2016). Its folkloric use in the treatment of cough, toothache, stomach ache, diarrhea, and lactation has been reported (Sodipo 1993). However, there are concerns about its toxicity owing to its indiscriminate use (Ajiboye et al. 2015). Ajiboye et al. (2015) also reported that 400 mg/kg of potash may be toxic and could deplete the antioxidant system in rats’ liver and kidney. Bamaiyi and Momoh (2010) established effects of 245 mg/kg of potash on gastrointestinal enzymes and small intestine. However, the trona material safety data sheet states that the oral LD50 for rats is 4090 mg/kg (TATA 2013).
Despite all the reports on the toxic effects of potash, there is dearth of information on the amelioration and attenuation of potash-induced organ damage.
The protective effects of medicinal plants particularly spices against oxidative injuries are well documented (Pai Kotebagilu et al. 2014; Asadi-Samani et al. 2015). These therapeutic effects have been attributed to their phytochemical constituents. Amongst such plants is Tetrapleura tetraptera, naturally distributed in the tropical areas of Africa (Aladesanmi 2007). T. tetraptera is commonly used as spice in the eastern and southern part of Nigeria. Its indigenous names amongst Nigerian tribes include Uyayak (Ibibio), Edeminang (Efik), Oshosho (Igbo), Dawo (Hausa), and Aridan (Yoruba). Its folkloric uses include the treatment of fever, jaundice, leprosy rheumatism, high blood pressure, inflammation, cancer, and diabetes (Atawodi et al. 2014). Its antidiabetic and anti-inflammatory activity has been reported (Ojewole and Adewunmi 2004). Its mineral constituents include potassium, zinc, calcium, phosphorous, and iron (Akin-Idowu et al. 2011). Its phytochemical constituents have been shown to consists of tannins, phenolics, saponins, alkaloids, steroids, and flavonoids (Ojewole and Adewunmi 2004; Akin-Idowu et al. 2011). Secondary metabolites isolated from T. tetraptera fruits include 7-hydroxy-6-methoxy-2H-1-benzopyran-2-one, 3-0-[ß-Dglucopyranosyl-2′-acetamido-2′- deoxy]-oleanolic acid (Aridanin), 3-[(2-acetamido-2-deoxy-ß-D-glucopyranosyl)oxy]-16α-hydroxyolean-12-en-28-oic acid, 3-{[O-ßD-galactopyranosyl-(1 → 4)-(2-acetamido-2-deoxy-ß-D-glucopyranosyl)oxy]}olean-12-en-28-oic, 3-{(O-ß-D-glucopyranosyl-(1 → 6)-(2-acetamido-2-deoxy-ß-Dglucopyranosyl)oxy}olean-12-en-28-oic, and 3-{[O-ß-Dglucopyranosyl-(1 → 6)-ß-D-glucopyranosyl]-oxy}-27-hydroxyolean-12-en-28-oic acid (Adesina et al. 2016).
To the best of our knowledge, we report for the first time the therapeutic potential of the ethanol extract of T. tetraptera fruit and peels against trona-induced oxidative injury in the hepatic, kidney, and intestinal tissues.
Materials and methods
Plant material
Fruits of T. tetraptera were purchased from local farmers at Benin City, Nigeria. They were washed and dehulled. The peels and fruit pulp were air-dried, pulverized, and subjected to ethanolic extraction. The extracts were concentrated in vacuo and stored in airtight containers respectively for further studies.
Fourier transform infrared spectroscopy analysis
The functional groups of the extracts were determined by scanning on FTIR spectrophotometer at room temperature (25–28 °C) at 300–4000 cm−1 spectral range. The functional groups were determined by comparing the peak frequencies to the IR spectroscopy correlation table.
Animals
Thirty-six male albino rats of Wistar strain weighing 180–200 g were used for the present study. They were reared at the Animal House of Bells University of Technology, Ota, Nigeria. The rats were acclimatized for 7 days on normal pelletized mouse chow, and water given ad libitum at room temperature with a 12-h light and dark cycle before the commencement of the experiment. The animals were maintained under the approval of the Animal Ethical Committee, Bells University of Technology, Ota, Nigeria, in accordance with the declaration of Helsinki.
Five groups of six rats each were pretreated with trona (500 mg/kg bw) for 7 days, while a sixth group was administered water only and served as normal control. Four of the pretreated groups were subjected to posttreatment with T. tetraptera extracts for 7 days, while the fifth served as negative control as depicted below:
-
Group 1: Normal control (water only)
-
Group 2: Trona only (500 mg/kg bw)
-
Group 3: Trona (500 mg/kg bw) + T. Tetraptera fruit extract (1000 mg/kg bw)
-
Group 4: Trona (500 mg/kg bw) + T. Tetraptera fruit extract (2000 mg/kg bw)
-
Group 5: Trona (500 mg/kg bw) + T. Tetraptera peel extract (1000 mg/kg bw)
-
Group 6: Trona (500 mg/kg bw) + T. Tetraptera peel extract (2000 mg/kg bw)
All treatments were orally administered via intubation. The rats were fasted overnight at the end of the experiment and sacrificed by cervical dislocation.
The choice of dose for trona was based on trona material safety data sheet (TATA 2013), while that of the extracts was based on previous reports (Effiong et al. 2006).
Preparation of blood serum
Blood was collected from each rat by cardiac puncture in plain bottles and centrifuged at 3000 rpm for 10 min. The serum (supernatant) was transferred into labeled sample bottles and stored at 4 °C until further analysis.
Hepatic enzymes biomarkers
Serum aspartate transaminase (AST) and alanine transaminase (ALT) levels were determined via commercial kits from Randox® Laboratories, UK, according to the manufacturer’s protocol.
Preparation of tissue homogenates
The intestine, kidney, and liver were removed, rinsed in ice-cold 1.15% KCl solution, and weighed. They were homogenized in 20 mM phosphate buffer (pH 6.6) and centrifuged at 10,000 rpm for 15 min at 4 °C. The supernatant was collected and stored at − 20 °C.
Lowry’s method was used in determining the total protein of the tissues, using bovine serum albumin (BSA) as standard (Lowry et al. 1951).
Determination of oxidative stress parameters
The tissues were assayed for malondialdehyde (MDA) level (Chowdhury and Soulsby 2002) and catalase (CAT) activity (Aebi 1983).
Histopathology
Hepatic tissues were sliced to a thickness of 3 mm and arranged in a tissue cassette. They were processed using an automated tissue processor (Leitz 2005 model) according to manufacturer’s protocol.
The processed images were quantitatively analyzed via ImageJ plugin.
Statistical analysis
One-way analysis of variance (ANOVA) was used in analyzing the differences between groups with the aid of Statistical Package for Social Sciences (SPSS) software, SPSS Inc., Chicago, Standard version 10.0.1. Results were expressed as mean ± standard deviation. P values < 0.05 were considered statistically significant.
Results
FTIR spectroscopy of the extracts revealed amines, alcohols, carboxylic acids, esters, ethers, aromatics, alkanes, aldehydes, ketones, phenols, and amides as the functional groups present as depicted in Table 1.
Administration of trona led to significant (p < 0.05) increase in catalase activities in the studied tissues, with the intestinal tissue being the most prominent as depicted in Fig. 1. This was significantly (p < 0.05) reduced to near normal after treatment with T. tetraptera fruit and peel extracts respectively.
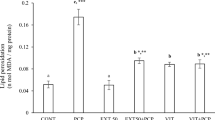
There was significant (p < 0.05) increase in MDA levels on administration of trona, portraying lipid peroxidation in the studied tissues as shown in Fig. 2. These levels were significantly (p < 0.05) suppressed after treating with T. tetraptera fruit and peel extracts to near normal.
An elevated level of serum hepatic biomarkers (ALT and AST) was observed in rats’ serum after administration of trona as revealed in Figs. 3a, b. Treatment with T. tetraptera fruit and peel extracts led to significant (p < 0.05) reductions, except ALT levels of rats (group 6) treated with 2000 mg/kg bw of T. tetraptera peel extract (Fig. 3a).
Histopathology examination revealed injuries in the hepatic tissues of rats following administration of trona as shown in Fig. 4b. This is evident by the increased hepatic lesions/alterations (Fig. 5). These alterations were observed to decrease on treatment with T. tetraptera fruit and peel extracts as revealed in Figs. 4c–f and 5, with 2000 mg/kg bw and 1000 mg/kg bw of the fruit and peel extracts showing the best activities respectively.
Discussion
The influence of oxidative stress in the progression and pathogenesis of most degenerative diseases are well documented (Erukainure et al. 2014; Schieber and Chandel 2014; Pisoschi and Pop 2015). This has been attributed to redox imbalance owing to elevated levels of free radicals that surpasses the body’s endogenous antioxidant system, thereby leading to oxidative damages with cytotoxic consequences (Wang et al. 2014). The hepatic and intestinal tissues are of great importance, as they play a major role in the metabolism of food and make up the gastrointestinal (GI) system. Most food additives exhibit their toxicity when they are being digested. The kidney also functions in the elimination of ingested toxic by-products from the body, thus reducing their toxicity. A compromise in the antioxidant and membrane lipid integrity of these tissues poses a toxic detrimental threat. In this study, the oxidative effect of trona, a common food addictive, was examined in rat models as well as the antioxidant therapeutic effect of T. tetraptera ethanolic extract.
The increased catalase activities in hepatic, renal, and intestinal tissues after ingestion contradict previous reports of depleted activities in trona-induced toxicities (Ajiboye et al. 2015; Imafidon et al. 2016). González-Párraga et al. (2003) reported similar effects in yeast exposed to H2O2. Similar effects were reported in rats exposed to low-lipid diets (Erukainure et al. 2016). Increased catalase activity has been reported in oxidative stress in cigarette smoking-induced oxidative stress in the lungs (Rahman and MacNee 1999). These studies attributed the increased activity as response to oxidative stress. The reduced catalase activities to near normal in the studied tissues after treatment with the extracts thus suggest a protective effect against trona-induced oxidative injuries.
The increased MDA levels after ingestion of trona indicate an occurrence of lipid peroxidation. This is consistent with several studies on increased MDA levels in tissues due to induction of oxidative injuries (Noeman et al. 2011; Jain et al. 2015; Li et al. 2015). MDA has been reported to elicit multiple biological and toxicological effects (Long et al. 2009). The significant (p < 0.05) decreased levels after treatment with the extracts indicate an anti-peroxidative effect which can be attributed to the reported phytochemical constituents of the fruits and peels. Similarly, the identified functional groups of the extracts (Fig. 1a, b and Table 1) indicate their antioxidant functions particularly the phenols with reported antioxidant activities (Kähkönen et al. 1999; Balasundram et al. 2006; Palacios et al. 2011). The electron-deficient moieties of N–H bend, N–O symmetric C–C stretch, and C=O stretches portray a potent antioxidant, owing to the ability to attract additional electrons (Harrold and Zavod 2013). The presence of amines may contribute to the anti-peroxidative effect of the extracts, as MDA reacts with amines of tissue proteins to induce oxidative toxicity (Long et al. 2009). The amines present in the extracts can abate such reactions by reacting with MDA, thus protecting the tissue proteins.
Increased serum hepatic biomarkers have been reported in oxidative hepatic injury (Contreras-Zentella and Hernández-Muñoz 2016; Schaffer et al. 2016). The similar effect after ingestion of trona further indicates an occurrence of oxidative injury (Fig. 4a, b). Hepatic leakage of these enzymes has been attributed to inflammation of the liver, leading to disintegration of its plasma membrane (Onyema et al. 2006). This also correlates with the increased MDA level in trona-ingested rats, thus implicating its influence on the increased serum levels of the studied enzyme biomarkers (Figs. 3 and 4b). The reduced levels of these enzymes in rats treated with the extracts indicate a stabilizing effect on the hepatic plasma membrane and restoration of the membrane integrity. This can also be attributed to the synergetic effect of the phytochemical constituents and FTIR-identified chemical functional groups (Fig. 1a, b and Table 1).
The increased hepatic lesions/alterations after ingestion of trona further indicate an occurrence of oxidative hepatic injury which correlates with the high MDA level (Fig. 3a) and increased serum hepatic enzyme biomarkers (Fig. 4a, b). Lesions and alterations in hepatic tissues have been reported in oxidative stress (Richardson et al. 2010; Jarrar and Taib 2012). The reduced levels after treatment with the extracts (Figs. 4c and 5) indicate their protective role against trona-induced oxidative hepatic injury, which correlates with the depleted levels of MDA (Fig. 3a) and serum hepatic enzyme biomarkers (Fig. 4a, b) respectively.
Conclusion
These results indicate the antioxidant protective effect of T. tetraptera against trona-induced oxidative injury. This can be attributed to the identified functional groups, with amine playing an influential role. Thus, the inclusion of this spice in meals is encouraged. Further studies on its molecular mechanism of action are being recommended.
References
Adesina SK, Iwalewa EO, Johnny II (2016) Tetrapleura tetraptera Taub-Ethnopharmacology, chemistry, medicinal and nutritional values—a review. Brit J Pharm Res 12(3):1–22. https://doi.org/10.9734/BJPR/2016/26554
Aebi HE (1983) Methods in enzymatic analysis. Academic, New York
Ajiboye TO, Komolafe YO, Yakubu MT, Ogunbode SM (2015) Effects of trona on the redox status of cellular system of male rats. Toxicol Ind Health 31(2):179–187. https://doi.org/10.1177/0748233712469654
Akin-Idowu PE, Ibitoye DO, Ademoyegun OT, Adeniyi OT (2011) Chemical composition of the dry fruit of Tetrapleura tetraptera and its potential impact on human health. J Herb Spice Med Plant 17(1):52–61. https://doi.org/10.1080/10496475.2011.560087
Aladesanmi AJ (2007) Review-Tetrapleura tetraptera: molluscicidal activity and chemical constituents. Afr J Tradit Complement Altern Med 4(1):23–36
Asadi-Samani M, Kafash-Farkhad N, Azimi N, Fasihi A, Alinia-Ahandani E, Rafieian-Kopaei M (2015) Medicinal plants with hepatoprotective activity in Iranian folk medicine. Asian Pac J Trop Biomed 5(2):146–157. https://doi.org/10.1016/S2221-1691(15)30159-3
Atawodi SE-O, Yakubu OE, Liman ML, Iliemene DU (2014) Effect of methanolic extract of Tetrapleura tetraptera (Schum and Thonn) Taub leaves on hyperglycemia and indices of diabetic complications in alloxan–induced diabetic rats. Asian Pac J Trop Biomed 4(4):272–278. https://doi.org/10.12980/APJTB.4.2014C73
Balasundram N, Sundram K, Samman S (2006) Phenolic compounds in plants and agri-industrial by-products: antioxidant activity, occurrence, and potential uses. Food Chem 99(1):191–203
Bamaiyi O, Momoh S (2010) Effects of kanwa on gastrointestinal phosphatase. Inter J Pharm Sci Nanotech 3(3):1147–1152
Bankole JK, Ngokere AA, Ajibade OM, Igunbor CM, Eloka CCV (2015) Degenerating effects of potash (Kaun-K2co3) on the kidney: unabated continental challenge to human health in Nigeria. Ann Biol Res 6(3):12–18
Chowdhury P, Soulsby M (2002) Lipid peroxidation in rat brain is increased by simulated weightlessness and decreased by a soy-protein diet. Ann Clin Lab Sci 32(2):188–192
Contreras-Zentella ML, Hernández-Muñoz R (2016) Is liver enzyme release really associated with cell necrosis induced by oxidant stress? Oxidative Med Cell Longev 2016:3529149
Effiong G, Udoh I, Essien E, Ajibola D, Archibong K (2006) Effect of aqueous extract of Tetrapleura tetraptera on excision wounds in albino rats. Int Res J Plant Sci 5(4):57–60
Erukainure O, Ajiboye J, Davis F, Obabire K, Okoro E, Adenekan S, Adegbola M, Awogbemi B, Odjobo B, Zaruwa M (2016) Effect of soy oil, orange (Citrus sinensis) peel oil and their blends on total phospholipid, lipid peroxidation, and antioxidant defense system in brain tissues of normo rats. Grasas Aceites 67(1):113
Erukainure OL, Ebuehi OA, Adeboyejo FO, Aliyu M, Elemo GN (2014) Modulatory effect of fibre-enriched cake on alloxan-induced diabetic toxicity in rat brain tissues. Toxicol Rep 1:445–449. https://doi.org/10.1016/j.toxrep.2014.07.010
González-Párraga P, Hernández JA, Argüelles JC (2003) Role of antioxidant enzymatic defences against oxidative stress (H2O2) and the acquisition of oxidative tolerance in Candida albicans. Yeast 20(14):1161–1169. https://doi.org/10.1002/yea.1029
Harrold MW, Zavod RM (2013) Basic concepts in medicinal chemistry. American Society of Health-System Pharmacists
Imafidon K, Egberanmwen I, Omoregie I (2016) Toxicological and biochemical investigations in rats administered “kaun”(trona) a natural food additive used in Nigeria. J Nutr Intermed Metab 6:22–25. https://doi.org/10.1016/j.jnim.2016.05.003
Jain S, Nair A, Shrivastava C (2015) Evaluation of oxidative stress marker malondialdehyde level in the cord blood of newborn infants. Int J Sci Study 3:73–76
Jarrar BM, Taib NT (2012) Histological and histochemical alterations in the liver induced by lead chronic toxicity. Saudi J Biol Sci 19(2):203–210. https://doi.org/10.1016/j.sjbs.2011.12.005
Kähkönen MP, Hopia AI, Vuorela HJ, Rauha J-P, Pihlaja K, Kujala TS, Heinonen M (1999) Antioxidant activity of plant extracts containing phenolic compounds. J Agric Food Chem 47(10):3954–3962. https://doi.org/10.1021/jf990146l
Li S, Tan H-Y, Wang N, Zhang Z-J, Lao L, Wong C-W, Feng Y (2015) The role of oxidative stress and antioxidants in liver diseases. Int J Mol Sci 16(11):26087–26124. https://doi.org/10.3390/ijms161125942
Long J, Liu C, Sun L, Gao H, Liu J (2009) Neuronal mitochondrial toxicity of malondialdehyde: inhibitory effects on respiratory function and enzyme activities in rat brain mitochondria. Neurochem Res 34(4):786–794
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Lu W-Y, Zhang T, Zhang D-Y, Li C-H, Wen J-P, Du L-X (2005) A novel bioflocculant produced by Enterobacter aerogenes and its use in defecating the trona suspension. Biochem Engineer J 27(1):1–7. https://doi.org/10.1016/j.bej.2005.04.026
Noeman SA, Hamooda HE, Baalash AA (2011) Biochemical study of oxidative stress markers in the liver, kidney and heart of high fat diet induced obesity in rats. Diabetol Metabc Synd 3(1):17. https://doi.org/10.1186/1758-5996-3-17
Ojewole JA, Adewunmi CO (2004) Anti-inflammatory and hypoglycaemic effects of Tetrapleura tetraptera (Taub) [Fabaceae] fruit aqueous extract in rats. J Ethnopharmacol 95(2):177–182. https://doi.org/10.1016/j.jep.2004.06.026
Okoye JO, Oranefo NO, Okoli AN (2016) Comparative evaluation of the effects of palm bunch ash and trona on the liver of albino rats. Afri J Cell Path 6:21–27
Onyema OO, Farombi EO, Emerole GO, Ukoha AI, Onyeze GO (2006) Effect of vitamin E on monosodium glutamate induced hepatotoxicity and oxidative stress in rats. Ind J Biochem Biophys 43:20–24
Pai Kotebagilu N, Reddy Palvai V, Urooj A (2014) Protective effect of selected medicinal plants against hydrogen peroxide induced oxidative damage on biological substrates. Int J Med Chem DOI 2014:1–7. https://doi.org/10.1155/2014/861084
Palacios I, Lozano M, Moro C, D’arrigo M, Rostagno M, Martínez J, García-Lafuente A, Guillamón E, Villares A (2011) Antioxidant properties of phenolic compounds occurring in edible mushrooms. Food Chem 128(3):674–678. https://doi.org/10.1016/j.foodchem.2011.03.085
Pisoschi AM, Pop A (2015) The role of antioxidants in the chemistry of oxidative stress: a review. Eur J Med Chem 97:55–74. https://doi.org/10.1016/j.ejmech.2015.04.040
Rahman I, MacNee W (1999) Lung glutathione and oxidative stress: implications in cigarette smoke-induced airway disease. Amer J Physiol Lung Cell Mol Physiol 277(6):L1067–L1088. https://doi.org/10.1152/ajplung.1999.277.6.L1067
Richardson N, Gordon A, Muller W, Pletschke B, Whitfield A (2010) The use of liver histopathology, lipid peroxidation and acetylcholinesterase assays as biomarkers of contaminant-induced stress in the Cape stumpnose, Rhabdosargus holubi (Teleostei: Sparidae), from selected South African estuaries. Water SA 36(4):0–0
Schaffer TK, Wohlenberg MF, Medeiros N, Martins JB, Agostini F, Funchal C, Dani C (2016) Evaluation of antioxidant activity of grapevine leaves extracts (Vitis labrusca) in liver of Wistar rats. Anai Acad Bras Ciênc 88(1):187–196. https://doi.org/10.1590/0001-3765201620140658
Schieber M, Chandel NS (2014) ROS function in redox signaling and oxidative stress. Curr Biol 24(10):R453–R462. https://doi.org/10.1016/j.cub.2014.03.034
Sodipo O (1993) How safe is the consumption of trona? Amer J Publ Health 83(8):1181–1181. https://doi.org/10.2105/AJPH.83.8.1181-a
TATA (2013) Trona material safety data sheet. Chemical (Sodium Ash) Partner. [accessed May 5, 2017]. http://www.tatachemicals.com/upload/pdf/Trona_msds.pdf
Wang E, Chen F, Hu X, Yuan Y (2014) Protective effects of apigenin against furan-induced toxicity in mice. Food Funct 5(8):1804–1812. https://doi.org/10.1039/C4FO00038B
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All studies were carried out under the approval and guidelines of the biological ethical committee of the Animal Ethical Committee, Bells University of Technology, Ota, Nigeria, in accordance to the Declaration of Helsinki
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ajiboye, J.A., Erukainure, O.L., Olasehinde, T. et al. Protective potential of Tetrapleura tetraptera against trona (kaun)-induced hepatic injury in rat models. Comp Clin Pathol 27, 627–633 (2018). https://doi.org/10.1007/s00580-018-2639-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-018-2639-z