Abstract
Purpose
Cuffed endotracheal tubes (ETTs) have increasingly been used in small children. However, the use of cuffed ETTs in small children is still controversial. The goal of this meta-analysis is to assess the current evidence regarding the postextubation morbidity and tracheal tube (TT) exchange rate of cuffed ETTs compared to uncuffed ETTs in children.
Methods
A systematic literature search in PubMed, Web of Science, Embase, and Cochrane Central Register of Controlled Trials up to November 2014 was conducted to identify randomized controlled trials (RCTs) and prospective cohort studies that compared the use of cuffed and uncuffed ETTs in children. The primary outcome was the incidence of postextubation stridor and the second outcomes were the TT exchange rate, need for re-intubation, and duration of tracheal intubation. All pooled data were estimated using random effects meta-analysis.
Results
Two RCTs and two prospective cohort studies including 3782 patients, in which 1979 patients for cuffed tubes and 1803 patients for uncuffed tubes, were included in our analysis. We found that the use of cuffed ETTs did not significantly increase the incidence of postextubation stridor (RR = 0.88; 95 % CI 0.67–1.16, p = 0.36), and the TT exchange rate was lower in patients receiving cuffed tubes intubation (RR, 0.07; 95 % CI 0.05–0.10, p < 0.00001). The need for re-intubation following planned extubations and duration of tracheal intubation did not differ significantly between the cuffed tube group and the uncuffed tube group.
Conclusions
Our study demonstrates that cuffed ETTs reduce the need for TT exchanges and do not increase the risk for postextubation stridor compared with uncuffed ETTs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Endotracheal intubation is a routinely performed technique in the anesthesia and critical care management in children. For more than 50 years, uncuffed tracheal tubes have been commonly used for intubation in children under 8 years of age [1] because of the anatomy of the pediatric larynx and the fear that the cuff will cause airway mucosal injury, leading to subglottic stenosis [2, 3]. However, there are shortcomings of uncuffed tracheal tubes, such as having a ventilation leak around the tube, which includes unreliable end-tidal carbon dioxide monitoring, wasting, and increasing costs of inhaled anesthetics, increasing pollution of the environment [4], and an increased risk of aspiration [5].
Recently, some clinical studies have shown that cuffed tracheal tubes may be safely used in pediatric airway management. The benefits are that cuffed tubes with more reliable sealing characteristics as well as evidence that cuffed tubes present no increased risk of airway morbidity [6, 7].
Therefore, it is necessary to assess the relative merits of cuffed and uncuffed endotracheal tubes in children. We conducted a meta-analysis to determine whether cuffed or uncuffed endotracheal tubes (ETTs) in children would be associated with postextubation airway morbidity, measured as postextubation stridor in children.
Methods
Literature search strategy
In accordance with recommendations of the Preferred Reporting Items for Systematic Review and Meta-analyses (PRISMA) statement and Cochrane Handbook for Systematic Reviews of Interventions Version 5.1 [8, 9], we searched databases of PubMed, Web of Science, Embase, and Cochrane Central Register of Controlled Trials up to November 2014 without restriction to publication types or languages. Keywords searched were as follows: cuffed/uncuffed/non-cuffed, endotracheal tube/tubes or tracheal tube/tube neonate/newborn/infant/child*/pediatric. The reference lists of all retrieved studies, relevant review articles were also examined. We contacted the authors for additional unpublished data when necessary.
Inclusion and exclusion criteria
Randomized controlled trials (RCTs) and prospective cohort studies were included, if they compared outcomes in children receiving either cuffed or uncuffed ETTs. No restrictions on scenario (operating room or intensive care unit) were applied. Studies that met one of the flowing criteria were excluded: repeated publication, retrospective study, absence of important data, editorials, letters to the editor, case reports, review articles, animal experimental, or studies not written in English.
Data extraction and outcomes of interest
Two independent authors extracted and summarized data from the included studies. All discrepancies were resolved by discussion among authors, with the involvement of the corresponding author if necessary. The following variables were collected from each included study: first author, publication year, study design, patient characteristics, control group, intervention group, cuffed or uncuffed ETT size and insertion depth, cuffed tube type and cuff pressure, the incidence of postextubation stridor, tracheal tubes (TT) exchange rate, need for re-intubation following planned extubations and duration of tracheal intubation.
Quality assessment and statistical analysis
We rated studies for the level of evidence according to the criteria provided by the Centre for Evidence-Based Medicine in Oxford, UK [10].
Two authors independently performed the quality assessment. RCTs were assessed by the Cochrane risk of bias tool [11]. Prospective cohort studies were assessed by the modified Newcastle-Ottawa scale [12, 13], which included three factors: patient selection, comparability of the study groups, and assessment of outcome. After discussions to resolve disagreements, a consensus score was arrived for each factor of quality in each article. A full score allocated to each study except RCTs was 9 stars. RCTs and prospective cohort studies achieving a score ≥6 stars were considered to be high quality.
We employed Review Manager 5.2 (Cochrane Collaboration, Oxford, UK) to combine outcomes among studies. For dichotomous variables, we used risk ratio (RR) with 95 % confidence intervals (CIs). For continuous variables, we calculated the weighted mean difference (WMD) with 95 % confidence intervals (CIs). The technique described by Hozo et al. [14] was used to calculate the standard deviations, if studies that showed continuous data as means and range values.
Heterogeneity was assessed using the Chi-square test with significance set at p < 0.10 [15] and I 2 statistic [16], which is significant being set at I 2 > 50 % according to the Cochrane Reviewer’s Handbook. The random-effects model was used for statistical analysis according to DerSimonian and Laird methodology [17] because of wide clinical and methodological variability among the trials. Heterogeneity between studies was evaluated according to the study design (RCT or cohort study). Sensitivity analyses were conducted for high-quality studies. We planned to evaluate potential publication bias with funnel plots. All statistical tests were two-sided.
Results
Evidence synthesis
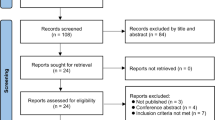
Our initial search yielded 423 studies (264 from PubMed, 35 from Web of Science, and 33 from Cochrane Central Register of Controlled Trials, 91 from Embase). After removing 122 duplicate studies, we evaluated the abstracts of 301 studies. After evaluating the abstract of each study, we excluded 276 studies because 204 studies were irrelevant, 17 were editorials or reviews or surveys, ten were letters, 15 studies were about animals, and 30 studies were case reports. Then we carefully read the full text of the remaining 25 studies and excluded 21 for the following reasons: without a control group in five studies, no interested results in four studies, no full-text available in two studies, non-English in eight studies, about adults in one study and a retrospective study in one study. Four studies including 3782 cases (1979 cases for cuffed tubes and 1803 cases for uncuffed tubes) matched with the inclusion criteria and were included in our analysis (Fig. 1).
Characteristics of eligible studies
The characteristics of included studies are shown in Table 1. In the included studies, there were two RCTs (level of evidence: 2b) [18, 19]; One prospective cohort study compared contemporary series of patients (level of evidence: 2b) [20]; One cohort study failed to carry out a sufficiently long and complete follow-up of patients (level of evidence: 4) [21]. As for scenario, two studies were performed in pediatric intensive care units and two studies were performed in operating rooms.
Methodological quality of included studies
The quality of included studies is shown in Tables 2 and 3. There were only two RCTs using true randomization. The information about allocation concealment was described in just one of the trials [18]. The information about the blinding method of the studies could not be found. Matching criteria were variable between the groups. Only one study [20] described the length of follow-up. The information about handling missing data was only described in two studies [19, 20].
Primary outcomes
Postextubation stridor rate
Ninety-four of 1979 patients (4.75 %) assigned to cuffed ETT intubation and 99 of 1803 patients (5.49 %) assigned to uncuffed ETT intubation developed stridor after extubation and showed no significant difference between the two groups (RR = 0.88; 95 % CI 0.67–1.16, p = 0.36) (Table 4). The result of the overall test for heterogeneity was not statistically significant, and the I 2 was 0 % (no significance of heterogeneity) (Fig. 2a). When stratified by scenario between the 2 groups, the RR for postextubation stridor rate decreased from 0.93 (95 % CI 0.65–1.33, p = 0.70) in the group in the operating room to 0.81 (95 % CI 0.53–1.24, p = 0.33) in the group in the pediatric intensive care unit, but there was still no significant difference between groups.
Secondary outcomes
Tracheal tubes (TT) exchange
Two studies [18, 19] performed in the operating room reported tracheal tubes (TT) exchange events for 2734 included patients. The TT exchange rate was 1.97 % (27 of 1370 patients) in the cuffed tube group and 29.40 % (401 of 1364 patients) in the uncuffed tube group. Reasons for TT exchanges were resistance to pass the tube (12.38 % of TT exchange cases), no air leak at 20 cm H2O (38.08 %), excessive air leak at IPPV (49.07 %) and others (0.47 %). The TT exchange rate was lower in patients receiving cuffed tube intubation (RR = 0.07; 95 % CI 0.05–0.10, p < 0.00001) (Fig. 2b).
Need for re-intubation following planned extubations
Two studies [19, 20] reported the need for re-intubation rate (0.31 % in the cuffed tube group and 0.44 % in the uncuffed tube group) following planned extubations. Based on the data of the 2434 patients from two studies, the rate of need for re-intubation following planned extubations in the cuffed tube group was not significantly higher than the uncuffed tube group (RR = 0.76; 95 % CI 0.19–3.02; p = 0.7) (Fig. 3a).
Duration of tracheal intubation
Duration of tracheal intubation was found in three studies [19–21]. We found the duration of tracheal intubation showed no significant difference between the cuffed tube group and the uncuffed tube group (WMD = 3.31 h, 95 % CI −9.86 to 16.49, p = 0.62) (Fig. 3b).
Subgroup analysis
Subgroup analysis of RCTs
Two RCTs contributed to the analysis [18, 19]. As for the postextubation stridor rate, there was no significant difference between the two groups (4.07 % compared with 4.35 %; RR = 0.93; 95 % CI 0.65–1.33; p = 0.70) (Fig. 2a). As for the need for re-intubation rate (0.17 % compared with 0.10 %; RR = 1.75; 95 % CI 0.16–19.30; p = 0.65) (Fig. 3a), there was also no significant difference between the two groups, except for two studies that showed that TT exchange rate was lower in cuffed tube group than in the cuffed tube group (1.97 % compared with 29.40 %; RR = 0.07; 95 % CI 0.05–0.10, p < 0.00001) (Fig. 2b). One study including 2246 patients showed that the duration of tracheal intubation was longer in the cuffed tube group than in the uncuffed tube group (WMD = 0.14 h, 95 % CI 0.03–0.25, p = 0.009) (Fig. 3b).
Subgroup analysis of prospective cohort studies
Two prospective cohort studies were included in this subgroup analysis. There was no significant difference in this subgroup analysis in the postextubation stridor rate (6.59 % compared with 8.32 %; RR = 0.81; 95 % CI 0.53–1.24; p = 0.33) (Fig. 2a), need for re-intubation rate (2.15 % compared with 4.21 %; RR = 0.51; 95 % CI 0.10–2.72; p = 0.43) (Fig. 3a), or duration of tracheal intubation (WMD = 22.90 h, 95 % CI −34.37 to 80.16, p = 0.43) (Fig. 3b) between the two groups.
Sensitivity analysis and publication bias
The sensitivity analysis included two RCTs [18, 19] and one prospective cohort study [20, 21], which scored seven stars on the modified Newcastle-Ottawa scale. The outcomes were no change in the significance (Table 5). The degree of between-study heterogeneity was also no change. There were only four studies included in this meta-analysis, so tests for funnel plot asymmetry were not used for the reason that test power was usually too low to distinguish chance from real asymmetry [11].
Discussion
This meta-analysis of two RCTs and two prospective cohort studies including 3782 children comparing cuffed and uncuffed endotracheal tubes (ETTs) showed that cuffed ETTs may be safely used in children, since no significant difference in the incidence of postextubation stridor was found. Furthermore, cuffed ETTs were associated with lower tube exchange rate and a reliable sealed airway. We found no significant difference in re-intubation rate and duration of tracheal intubation between those patients with cuffed ETTs compared with those with uncuffed ETTs.
The safety of children is always important in ETTs selection. The pooled data of postextubation stridor rate indicated that cuffed ETTs might be safe for children if size was appropriately selected and cuffed pressure was well controlled. There was no significant difference in postextubation stridor rate among patients using cuffed and uncuffed tubes. These findings may represent that the strict selection of an anatomically designed cuffed tube with controlled, tube sizes according to size recommendations and a limited cuff pressure was used in the cuffed tube group [7, 18–21]. The outcome of postextubation stridor rate in both groups was found to be in line with the finding of Ashtekar et al. [22] and would alleviate the fear of many pediatric anesthetists that cuffed ETTs might increase postextubation stridor rates in children [23]. It also showed us that postextubation stridor could occur after tracheal intubation with any type of ETT [24]. The pooled data of need for reintubation following planned extubations and duration of intubation showed no significant difference between the two groups. It demonstrated that cuffed ETTs did not increase the rate of reintubation compared to the uncuffed ETTs and might be safely used for prolonged periods of time without causing postextubation stridor. However, it is important to know that cuffed ETTs with oversized outer tube diameters, not adequately designed cuffs, and without cuff pressure control can increase the risk of airway injury [3, 25].
The tracheal tube (TT) exchange rate was significantly higher in the cuffed ETT group than the uncuffed ETT group [18, 19]. The chance to find the appropriate ETT at the first attempt was higher for cuffed ETTs than uncuffed ETTs, because cuffed ETTs were selected with a smaller size and the cuffs were inflated as required to fill the gap between the tube and the tracheal wall. Furthermore, it is often difficult to determine the correct size of ETTs [26]. The recommendation for tube size [27] in children is various and the incorrect selection of tube size will result in a high TT exchange rate. The survey of Flynn et al. [28] reported that repeated tube exchanges could cause airway injury, which was associated with the tube tip and up-and-down movement of the tube within the larynx during ventilation [2]. However, although the uncuffed ETT group required more intubation attempts, we did not find more postextubation stridor events in patients using uncuffed ETTs. The different findings may be attributed to the limited number of articles in this paper, which only included two RCTs and two prospective cohort studies. The pooled data indicated that cuffed tubes could have a much higher chance of fitting at first attempt than uncuffed tubes when using appropriately designed cuffed ETTs with a clear concept for cuff pressure control and tube size selection.
Subgroup analysis based on the study design (RCTs or prospective cohort studies) was performed. The results showed that most outcomes were consistent with studies of different designs. A sensitivity analysis was conducted including only high-quality studies. The results were consistent with the finding of the analysis except for the duration of intubation, which was longer in the cuffed ETT group than the uncuffed ETT group. However, there was significant heterogeneity among studies. Because of ethical concerns and placing ETT in different locations, randomized trials on the use of cuffed versus uncuffed ETTs in critically ill children are very difficult to conduct. So it is difficult to reach any definitive conclusion for the limited number of RCTs.
It was significant for the duration of intubation between-study heterogeneity. The significant between-study heterogeneity might be due to the wide clinical and methodological variability among the studies. Although we used a random-effects model to combine the data, the effect of heterogeneity might be reduced but not be abolished.
Our study has strengths because we undertook multiple strategies to identify studies, using predefined criteria to evaluate the methodological quality of the studies. We also applied subgroup and sensitivity analysis to minimize heterogeneity. However, our meta-analysis has several limitations that are worthy of comment. First, the number of the included studies is limited. Only four studies were involved in this analysis including two RCTs and two prospective cohort studies. Random sequence generation and blinding were not adequate, which might increase the risk of bias. Second, as with all meta-analytical techniques, the pooled data were combined from different studies. Clinical heterogeneity may also exist because each included study had its own protocol and definitions. The recommendation for choosing ETTs and depth of intubation were different from hospitals and that could influence the outcomes. Third, evidence of the clinical efficacy of cuffed ETTs in the neonatal setting is still absent, so we could not evaluate the efficacy and safety of cuffed and uncuffed ETTs in neonates. Further studies should be directed toward optimizing ETT standards in the neonatal unit, where the greatest incidence of airway damage may occur. What’s more, some data could not be found directly from articles and we got the data from statistical transformation, which might limit the quality of our conclusion. Additionally, the follow-up period of the studies was short, so we could not evaluate the long-term outcomes of using ETTs. Finally, there were no sufficient studies to detect asymmetry in a funnel plot, so we could not fully exclude publication bias.
In conclusion, according to this meta-analysis, we find that cuffed ETTs may be associated with a lower TT exchange rate. Two kinds of ETTs appear to be equivalent in terms of the postextubation stridor rate, the need for re-intubation following planned extubations and the duration of tracheal intubation.
References
Veyckemans F. New developments in the management of the paediatric airway: cuffed or uncuffed tracheal tubes, laryngeal mask airway, cuffed oropharyngeal airway, tracheostomy and one-lung ventilation devices. Curr Opin Anaesthesiol. 1999;12:315–20.
Flynn PE, Black AE, Mitchell V. The use of cuffed tracheal tubes for paediatric tracheal intubation, a survey of specialist practice in the United Kingdom. Eur J Anaesthesiol. 2008;25:685–8. doi:10.1017/S0265021508003839.
Weiss M, Dullenkopf A, Gysin C, Dillier CM, Gerber AC. Shortcomings of cuffed paediatric tracheal tubes. Br J Anaesth. 2004;92:78–88.
Eschertzhuber S, Salgo B, Schmitz A, Roth W, Frotzler A, Keller CH, Gerber AC, Weiss M. Cuffed endotracheal tubes in children reduce sevoflurane and medical gas consumption and related costs. Acta Anaesthesiol Scand. 2010;54:855–8. doi:10.1111/j.1399-6576.2010.02261.x.
Gopalareddy V, He Z, Soundar S, Bolling L, Shah M, Penfil S, McCloskey JJ, Mehta DI. Assessment of the prevalence of microaspiration by gastric pepsin in the airway of ventilated children. Acta Paediatr. 2008;97:55–60. doi:10.1111/j.1651-2227.2007.00578.x.
Bhardwaj N. Pediatric cuffed endotracheal tubes. J Anaesthesiol Clin Pharmacol. 2013;29:13–8. doi:10.4103/0970-9185.105786.
Weiss M, Gerber AC. Safe use of cuffed tracheal tubes in children. Anasthesiol Intensivmed Notfallmed Schmerzther. 2012;47:232–7. doi:10.1055/s-0032-1310411.
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6:e1000100. doi:10.1371/journal.pmed.1000100.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151(264–269):W64.
Phillips B BCSD (2009) Levels of evidence and grades of recommendation. Oxford Centre for Evidence-based Medicine Web site. http://www.cebm.net/index.aspx?o=1025.
Higgins JGS. Cochrane handbook for systematic reviews of interventions. New York: Wiley; 2008.
Wells G SBOC (2014) The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa Hospital Research Institute Web site. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed Aug 6 2014.
Fan X, Lin T, Xu K, Yin Z, Huang H, Dong W, Huang J. Laparoendoscopic single-site nephrectomy compared with conventional laparoscopic nephrectomy: a systematic review and meta-analysis of comparative studies. Eur Urol. 2012;62:601–12. doi:10.1016/j.eururo.2012.05.055.
Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5:13. doi:10.1186/1471-2288-5-13.
Hedges LV, Pigott TD. The power of statistical tests in meta-analysis. Psychol Methods. 2001;6:203–17.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–58. doi:10.1002/sim.1186.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88.
Khine HH, Corddry DH, Kettrick RG, Martin TM, McCloskey JJ, Rose JB, Theroux MC, Zagnoev M. Comparison of cuffed and uncuffed endotracheal tubes in young children during general anesthesia. Anesthesiology. 1997;86(627–631):27A.
Weiss M, Dullenkopf A, Fischer JE, Keller C, Gerber AC. Prospective randomized controlled multi-centre trial of cuffed or uncuffed endotracheal tubes in small children. Br J Anaesth. 2009;103:867–73. doi:10.1093/bja/aep290.
Deakers TW, Reynolds G, Stretton M, Newth CJ. Cuffed endotracheal tubes in pediatric intensive care. J Pediatr. 1994;125:57–62.
Newth CJ, Rachman B, Patel N, Hammer J. The use of cuffed versus uncuffed endotracheal tubes in pediatric intensive care. J Pediatr. 2004;144:333–7. doi:10.1016/j.jpeds.2003.12.018.
Ashtekar CS, Wardhaugh A. Do cuffed endotracheal tubes increase the risk of airway mucosal injury and post-extubation stridor in children? Arch Dis Child. 2005;90:1198–9. doi:10.1136/adc.2005.077651.
Orliaguet GA, Renaud E, Lejay M, Meyer PG, Schmautz E, Telion C, Carli PA. Postal survey of cuffed or uncuffed tracheal tubes used for paediatric tracheal intubation. Paediatr Anaesth. 2001;11:277–81.
Litman RS, Maxwell LG. Cuffed versus uncuffed endotracheal tubes in pediatric anesthesia: the debate should finally end. Anesthesiology. 2013;118:500–1. doi:10.1097/ALN.0b013e318282cc8f.
Silva MJ, Aparicio J, Mota T, Spratley J, Ribeiro A. Ischemic subglottic damage following a short-time intubation. Eur J Emerg Med. 2008;15:351–3. doi:10.1097/MEJ.0b013e3282fc9c5d.
Schramm C, Knop J, Jensen K, Plaschke K. Role of ultrasound compared to age-related formulas for uncuffed endotracheal intubation in a pediatric population. Paediatr Anaesth. 2012;22:781–6. doi:10.1111/j.1460-9592.2012.03889.x.
von Rettberg M, Thil E, Genzwurker H, Gernoth C, Hinkelbein J. Endotracheal tubes in pediatric patients. Published formulas to estimate the optimal size. Anaesthesist. 2011;60:334–42. doi:10.1007/s00101-010-1756-0.
Lonnqvist PA. Cuffed or uncuffed tracheal tubes during anaesthesia in infants and small children: time to put the eternal discussion to rest? Br J Anaesth. 2009;103:783–5. doi:10.1093/bja/aep330.
Acknowledgments
This research work was carried out without funding.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Shi, F., Xiao, Y., Xiong, W. et al. Cuffed versus uncuffed endotracheal tubes in children: a meta-analysis. J Anesth 30, 3–11 (2016). https://doi.org/10.1007/s00540-015-2062-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-015-2062-4